Regulation of Human Trophoblast GLUT3 Glucose Transporter by Mammalian Target of Rapamycin Signaling
Abstract
:1. Introduction
2. Results and Discussion
2.1. Validation of mTORC1 Pathway Inhibition
2.2. Effect of mTORC1 Inhibition on JEG-3 Cell Characteristics



2.3. Effects of Rapamycin Treatment on GLUT3 Expression

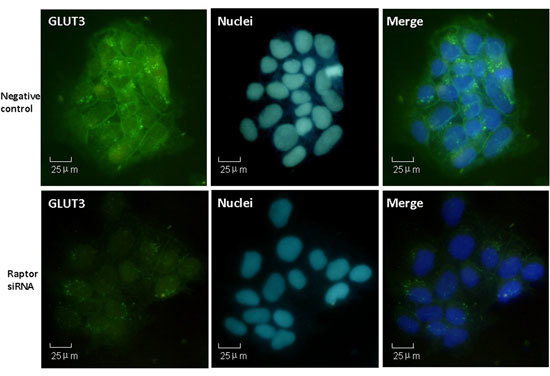
2.4. Effects of siRNA Targeting Raptor on GLUT3 Expression


2.5. Discussion
3. Experimental Section
3.1. Cell Culture
3.2. HCG Measurement
3.3. CCK Assay
3.4. mTORC1 Inhibition Mediated by Rapamycin Treatment
3.5. RNA Interference-Mediated Silencing of mTORC1
3.6. mRNA Isolation and Quantitative Real-Time PCR
3.7. Western Blot Analysis
3.8. Immunofluorescence
3.9. Data Presentation and Statistics
4. Conclusions
Acknowledgments
Author Contributions
Conflict of Interests
References
- Gluckman, P.D.; Hanson, M.A.; Cooper, C.; Thornburg, K.L. Effect of in utero and early-life conditions on adult health and disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Kalhan, S.C.; D’Angelo, L.J.; Savin, S.M.; Adam, P.A. Glucose production in pregnant women at term gestation: Sources of glucose for human fetus. J. Clin. Investig. 1979, 63, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; Heller, D.S.; Zamudio, S.; Illsley, N.P. Glucose transporter 3 (GLUT3) protein expression in human placenta across gestation. Placenta 2011, 32, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Hahn, D.; Blaschitz, A.; Korgun, E.T.; Lang, I.; Desoye, G.; Skofitsch, G.; Dohr, G. From maternal glucose to fetal glycogen: Expression of key regulators in the human placenta. Mol. Hum. Reprod. 2001, 7, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Hay, W.W., Jr.; Molina, R.A.; DiGiacomo, J.E.; Meschia, G. Model of placental glucose consumption and glucose transfer. Am. J. Physiol. 1990, 258, 569–577. [Google Scholar]
- Hay, W.W., Jr. Energy and substrate requirements of the placenta and fetus. Proc. Nutr. Soc. 1991, 50, 321–336. [Google Scholar] [CrossRef] [PubMed]
- Hay, W.W., Jr. Placental-fetal glucose exchange and fetal glucose metabolism. Trans. Am. Clin. Climatol. Assoc. 2006, 117, 321–340. [Google Scholar] [PubMed]
- Illsley, N.P. Placental glucose transport in diabetic pregnancy. Clin. Obstet. Gynecol. 2000, 43, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Gaither, K.; Quraishi, A.N.; Illsley, N.P. Diabetes alters the expression and activity of the human placental GLUT1 glucose transporter. J. Clin. Endocrinol. Metab. 1999, 84, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Jansson, T.; Ekstrand, Y.; Wennergren, M.; Powell, T.L. Placental glucose transport in gestational diabetes mellitus. Am. J. Obstet. Gynecol. 2001, 184, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Illsley, N.P. Glucose transporters in the human placenta. Placenta 2000, 21, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Kamei, Y.; Tsutsumi, O.; Yamakawa, A.; Oka, Y.; aketani, Y.; Imaki, J. Maternal epidermal growth factor deficiency causes fetal hypoglycemia and intrauterine growth retardation in mice: Possible involvement of placental glucose transporter GLUT3 expression. Endocrinology 1999, 140, 4236–4243. [Google Scholar] [CrossRef] [PubMed]
- Kainulainen, H.; Jarvinen, T.; Heinonen, P.K. Placental glucose transporters in fetal intrauterine growth retardation and macrosomia. Gynecol. Obstet. Investig. 1997, 44, 89–92. [Google Scholar] [CrossRef]
- Schmidt, S.; Hommel, A.; Gawlik, V.; Augustin, R.; Junicke, N.; Florian, S.; Richter, M.; Walther, D.J.; Montag, D.; Joost, H.G.; et al. Essential role of glucose transporter GLUT3 for post-implantation embryonic development. J. Endocrinol. 2009, 200, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, A.; McKnight, R.A.; Raychaudhuri, S.; Shin, B.C.; Ma, Z.; Moley, K.; Devaskar, S.U. Glucose transporter isoform-3 mutations cause early pregnancy loss and fetal growth restriction. Am. J. Physiol. Endocrinol. Metab. 2007, 292, 1241–1255. [Google Scholar] [CrossRef] [PubMed]
- Jansson, T.; Aye, I.L.; Goberdhan, D.C. The emerging role of mTORC1 signaling in placental nutrient-sensing. Placenta 2012, 33, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Guan, K.L. Expanding mTOR signaling. Cell Res. 2007, 17, 666–681. [Google Scholar] [CrossRef] [PubMed]
- Foster, K.G.; Fingar, D.C. Mammalian target of rapamycin (mTOR): Conducting the cellular signaling symphony. J. Biol. Chem. 2010, 285, 14071–14077. [Google Scholar] [CrossRef] [PubMed]
- Roos, S.; Jansson, N.; Palmberg, I.; Saljo, K.; Powell, T.L.; Jansson, T. Mammalian target of rapamycin in the human placenta regulates leucine transport and is down-regulated in restricted fetal growth. J. Physiol. 2007, 582, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Yung, H.W.; Calabrese, S.; Hynx, D.; Hemmings, B.A.; Cetin, I.; Charnock-Jones, D.S.; Burton, G.J. Evidence of placental translation inhibition and endoplasmic reticulum stress in the etiology of human intrauterine growth restriction. Am. J. Pathol. 2008, 173, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, S.; Gawlik, V.; Holter, S.M.; Augustin, R.; Scheepers, A.; Behrens, M.; Wurst, W.; Gailus-Durner, V.; Fuchs, H.; Hrabe de Angelis, M.; et al. Deletion of glucose transporter GLUT8 in mice increases locomotor activity. Behav. Genet. 2008, 38, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Ringler, G.E.; Strauss, J.F., III. In vitro systems for the study of human placental endocrine function. Endocr. Rev. 1990, 11, 105–123. [Google Scholar] [CrossRef] [PubMed]
- Ntrivalas, E.; Kwak-Kim, J.; Beaman, K.; Mantouvalos, H.; Gilman-Sachs, A. An in vitro coculture model to study cytokine profiles of natural killer cells during maternal immune cell-trophoblast interactions. J. Soc. Gynecol. Investig. 2006, 13, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Roos, S.; Lagerlof, O.; Wennergren, M.; Powell, T.L.; Jansson, T. Regulation of amino acid transporters by glucose and growth factors in cultured primary human trophoblast cells is mediated by mTOR signaling. Am. J. Physiol. Cell Physiol. 2009, 297, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Dicke, J.M.; Henderson, G.I. Placental amino acid uptake in normal and complicated pregnancies. Am. J. Med. Sci. 1988, 295, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Jansson, T.; Scholtbach, V.; Powell, T.L. Placental transport of leucine and lysine is reduced in intrauterine growth restriction. Pediatr. Res. 1998, 44, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Mahendran, D.; Donnai, P.; Glazier, J.D.; D’Souza, S.W.; Boyd, R.D.; Sibley, C.P. Amino acid (system A) transporter activity in microvillous membrane vesicles from the placentas of appropriate and small for gestational age babies. Pediatr. Res. 1993, 34, 661–665. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, H.D.; Serek, R.; Crane, D.I.; Veveris-Lowe, T.; Parry, A.; Johnson, S.; Leung, K.C.; Ho, K.K.; Bougoussa, M.; Hennen, G.; et al. Placental growth hormone (GH), GH-binding protein, and insulin-like growth factor axis in normal, growth-retarded, and diabetic pregnancies: Correlations with fetal growth. J. Clin. Endocrinol. Metab. 2000, 85, 1143–1150. [Google Scholar] [PubMed]
- Mirlesse, V.; Frankenne, F.; Alsat, E.; Poncelet, M.; Hennen, G.; Evain-Brion, D. Placental growth hormone levels in normal pregnancy and in pregnancies with intrauterine growth retardation. Pediatr. Res. 1993, 34, 439–442. [Google Scholar] [CrossRef] [PubMed]
- Economides, D.L.; Nicolaides, K.H. Blood glucose and oxygen tension levels in small-for-gestational-age fetuses. Am. J. Obstet. Gynecol. 1989, 160, 385–389. [Google Scholar] [CrossRef]
- Rosario, F.J.; Kanai, Y.; Powell, T.L.; Jansson, T. Mammalian target of rapamycin signalling modulates amino acid uptake by regulating transporter cell surface abundance in primary human trophoblast cells. J. Physiol. 2013, 591, 609–625. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, J.; Lu, C.; Wang, J.; Zhang, R.; Qian, X.; Zhu, H. Regulation of Human Trophoblast GLUT3 Glucose Transporter by Mammalian Target of Rapamycin Signaling. Int. J. Mol. Sci. 2015, 16, 13815-13828. https://doi.org/10.3390/ijms160613815
Xu J, Lu C, Wang J, Zhang R, Qian X, Zhu H. Regulation of Human Trophoblast GLUT3 Glucose Transporter by Mammalian Target of Rapamycin Signaling. International Journal of Molecular Sciences. 2015; 16(6):13815-13828. https://doi.org/10.3390/ijms160613815
Chicago/Turabian StyleXu, Jie, Chunmei Lu, Jiao Wang, Ruotong Zhang, Xin Qian, and Hui Zhu. 2015. "Regulation of Human Trophoblast GLUT3 Glucose Transporter by Mammalian Target of Rapamycin Signaling" International Journal of Molecular Sciences 16, no. 6: 13815-13828. https://doi.org/10.3390/ijms160613815
APA StyleXu, J., Lu, C., Wang, J., Zhang, R., Qian, X., & Zhu, H. (2015). Regulation of Human Trophoblast GLUT3 Glucose Transporter by Mammalian Target of Rapamycin Signaling. International Journal of Molecular Sciences, 16(6), 13815-13828. https://doi.org/10.3390/ijms160613815