Sex-Specific Patterns of Aberrant Brain Function in First-Episode Treatment-Naive Patients with Schizophrenia
Abstract
:1. Introduction
2. Results and Discussion
2.1. Demographic, Clinical and Head-Motion Characteristics
2.2. DC
| Items | FESm (n = 61) | FESf (n = 63) | HCm (n = 50) | HCf (n = 52) | p |
|---|---|---|---|---|---|
| Age | 24.31 (6.57) | 24.62 (6.82) | 24.80 (6.74) | 24.71 (6.98) | 0.982 |
| Education years | 13.13 (2.49) | 12.94 (2.49) | 13.77 (2.91) | 13.40 (2.55) | 0.366 |
| HM-tran (mm) | 0.13 (0.14) | 0.11 (0.13) | 0.11 (0.11) | 0.10 (0.10) | 0.099 |
| HM-rota (degree) | 0.12 (0.12) | 0.11 (0.14) | 0.11 (0.09) | 0.11 (0.11) | 0.649 |
| Total | 87.59 (17.07) | 89.27 (17.04) | – | – | 0.597 |
| Positive | 14.74 (4.40) | 15.77 (4.24) | – | – | 0.200 |
| Negative | 16.58 (7.73) | 14.85 (8.39) | – | – | 0.250 |
| D/C | 9.12 (2.75) | 9.48 (2.96) | – | – | 0.497 |
| Excited | 9.93 (3.85) | 11.32 (4.13) | – | – | 0.063 |
| Depressed | 6.19 (3.08) | 6.12 (2.71) | – | – | 0.887 |
| DUP (months) | 8.19 (8.97) | 5.50 (8.03) | – | – | 0.118 |
| Age of Onset | 23.24 (6.65) | 23.76 (6.64) | – | – | 0.689 |

| Comparisons | Regions | Voxels | t Value * | X, Y, Z |
|---|---|---|---|---|
| Interaction of sex and diagnosis | ||||
| HCm > FESm | Putamen | 22 | 5.24 | 30, −3, −12 |
| HCf > FESf | Middle Frontal Gyrus | 13 | 4.40 | −30, 12, 51 |
| Main effect of diagnosis | ||||
| HC > FES | Putamen | 63 | 4.60 | 36, −15, −6 |
| Inferior Frontal Gyrus | 35 | 3.84 | 36, 3, 30 | |
| Main effect of sex | ||||
| Male > Female | Inferior Frontal Gyrus | 62 | 4.83 | −36, 36, 0 |
| Superior Frontal Gyrus | 25 | 4.29 | 27, 33, 36 | |
| Superior Frontal Gyrus | 35 | 3.89 | −21, 57, −15 | |
| Posterior Cingulate Cortex | 16 | 3.84 | −6, −48, 24 | |
| Female > Male | Inferior Parietal Lobule | 26 | −3.34 | −48, −36, 21 |
| Thalamus | 99 | −3.35 | −9, −33, −3 | |
| Thalamus | 65 | −3.38 | 9, −21, −6 | |
| Insula | 24 | −3.41 | −39, −3, −6 |

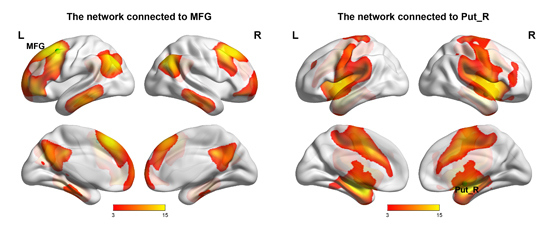
2.3. Networks

2.4. ALFF
| Comparisons | Regions | Voxels | t Value * | X, Y, Z |
|---|---|---|---|---|
| Interaction of sex and diagnosis | ||||
| HCf > FESf | Ventral Medial Prefrontal Cortex | 58 | 4.51 | −6, 30, −24 |
| Main effect of diagnosis | ||||
| HC < FES | Putamen | 315 | 5.81 | 21, 15, −6 |
| Putamen | 363 | 5.37 | −18, 15, −3 | |
| HC > FES | Middle Temporal Gyrus | 52 | 4.84 | 45, −63, 24 |
| Inferior Parietal Lobule | 27 | 4.06 | 48, −48, 42 | |
| Posterior Cingulate Cortex | 38 | 3.99 | 0, −54, 12 | |
| Ventral Medial Prefrontal Cortex | 34 | 3.61 | −3, 40, −21 | |
| Main effect of sex | ||||
| Male > Female | Inferior Parietal Lobule | 3709 | 9.61 | 54, −51, 48 |
| Middle Frontal Gyrus | 257 | 6.06 | −45, 48, 18 | |
| Cerebellar Tonsil | 56 | 5.24 | 12, −42, −51 | |
| Middle Frontal Gyrus | 108 | 5.21 | 48, 48, 15 | |
| Lingual Gyrus | 51 | 4.02 | −15, −102, −9 | |
| Superior Temporal Gyrus | 53 | 5.20 | 57, 15, −6 | |
| Insula | 51 | 4.70 | −39, −33, 12 | |
| Female > Male | ParaHippocampal | 105 | 5.08 | 18, −12, −36 |
| Hippocampus | 129 | 4.78 | 33, −33, −9 | |
| Lentiform Nucleus | 731 | 6.41 | −15, 9, −3 | |
| Cerebellum Posterior Lobe | 2966 | 6.49 | 39, −39, −45 |
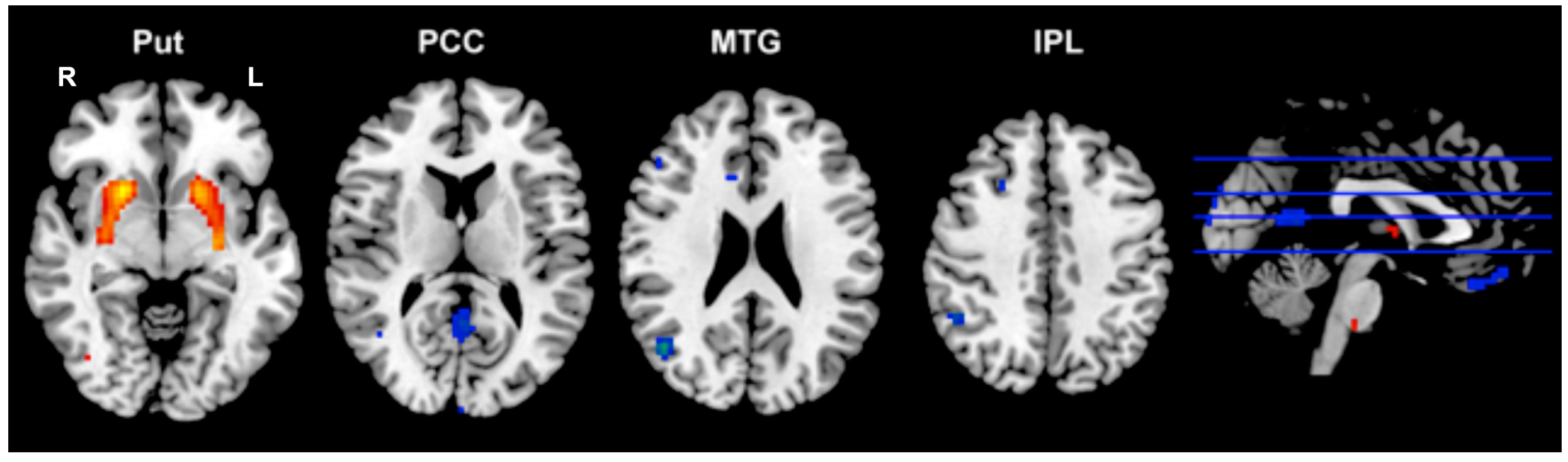

2.5. Correlations of Regional Functional Characteristic with Clinical Profiles
3. Discussion
4. Experimental Section
4.1. Participants
4.2. MRI Data Acquisition
4.3. Image Preprocessing
4.4. DC Calculation
4.5. ALFF Calculation
4.6. Statistical Analysis
4.6.1. Comparison of Demographic and Clinical Data and Head-Motion Parameters
4.6.2. Voxel-Wise Comparison of DC Maps and ALFF Maps
4.7. Correlation Analysis
4.8. Network Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Markham, J.A. Sex steroids and schizophrenia. Rev. Endocr. Metab. Dis. 2012, 13, 187–207. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, S.; Usall, J.; Cobo, J.; Labad, X.; Kulkarni, J. Gender differences in schizophrenia and first-episode psychosis: A comprehensive literature review. Schizophr. Res. Treat. 2012, 2012, 1549–1558. [Google Scholar] [CrossRef] [PubMed]
- Shtasel, D.L.; Gur, R.E.; Gallacher, F.; Heimberg, C.; Gur, R.C. Gender differences in the clinical expression of schizophrenia. Schizophr. Res. 1992, 7, 225–231. [Google Scholar] [CrossRef]
- Robinson, D.; Woerner, M.G.; Alvir, J.M.J.; Bilder, R.; Goldman, R.; Geisler, S.; Koreen, A.; Sheitman, B.; Chakos, M.; Mayerhoff, D. Predictors of relapse following response from a first episode of schizophrenia or schizoaffective disorder. Arch. Gen. Psychiatry 1999, 56, 241. [Google Scholar] [CrossRef] [PubMed]
- Grossman, L.S.; Harrow, M.; Rosen, C.; Faull, R.; Strauss, G.P. Sex differences in schizophrenia and other psychotic disorders: A 20-year longitudinal study of psychosis and recovery. Compr. Psychiatry 2008, 49, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Usall, J.; Suarez, D.; Haro, J.M. Gender differences in response to antipsychotic treatment in outpatients with schizophrenia. Psychiatry Res. 2007, 153, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Grossman, L.; Harrow, M.; Rosen, C.; Faull, R. Sex differences in outcome and recovery for schizophrenia and other psychotic and nonpsychotic disorders. Psychiatr. Serv. 2006, 57, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Gur, R.E.; Petty, R.G.; Turetsky, B.I.; Gur, R.C. Schizophrenia throughout life: Sex differences in severity and profile of symptoms. Schizophr. Res. 1996, 21, 1–12. [Google Scholar] [CrossRef]
- Cotton, S.; Lambert, M.; Schimmelmann, B.; Foley, D.; Morley, K.; McGorry, P.; Conus, P. Gender differences in premorbid, entry, treatment, and outcome characteristics in a treated epidemiological sample of 661 patients with first episode psychosis. Schizophr. Res. 2009, 114, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.M. Sex, hormones and affective arousal circuitry dysfunction in schizophrenia. Horm. Behav. 2006, 50, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.M.; Seidman, L.J.; O’Brien, L.M.; Horton, N.J.; Kennedy, D.N.; Makris, N.; Caviness, V.S., Jr.; Faraone, S.V.; Tsuang, M.T. Impact of normal sexual dimorphisms on sex differences in structural brain abnormalities in schizophrenia assessed by magnetic resonance imaging. Arch. Gen. Psychiatry 2002, 59, 154. [Google Scholar] [CrossRef] [PubMed]
- Gur, R.E.; Kohler, C.; Turetsky, B.I.; Siegel, S.J.; Kanes, S.J.; Bilker, W.B.; Brennan, A.R.; Gur, R.C. A sexually dimorphic ratio of orbitofrontal to amygdala volume is altered in schizophrenia. Biol. Psychiatry 2004, 55, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Guillem, F.; Mendrek, A.; Lavoie, M.E.; Pampoulova, T.; Stip, E. Sex differences in memory processing in schizophrenia: An event-related potential (erp) study. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, J.A.; Mancini-Marïe, A.; Lakis, N.; Rinaldi, M.; Mendrek., A. Disturbed sexual dimorphism of brain activation during mental rotation in schizophrenia. Schizophr. Res. 2010, 122, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Mendrek, A.; Lakis, N.; Jiménez, J. Associations of sex steroid hormones with cerebral activations during mental rotation in men and women with schizophrenia. Psychoneuroendocrinology 2011, 36, 1422–1426. [Google Scholar] [CrossRef] [PubMed]
- Mendrek, A.; Jiménez, J.; Mancini-Marïe, A.; Fahim, C.; Stip, E. Correlations between sadness-induced cerebral activations and schizophrenia symptoms: An fmri study of sex differences. Eur. Psychiatry 2011, 26, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Glahn, D.C.; Ragland, J.D.; Abramoff, A.; Barrett, J.; Laird, A.R.; Bearden, C.E.; Velligan, D.I. Beyond hypofrontality: A quantitative meta-analysis of functional neuroimaging studies of working memory in schizophrenia. Hum. Brain Mapp. 2005, 25, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Cordes, D.; Haughton, V.M.; Arfanakis, K.; Carew, J.D.; Turski, P.A.; Moritz, C.H.; Quigley, M.A.; Meyerand, M.E. Frequencies contributing to functional connectivity in the cerebral cortex in “resting-state” data. Am. J. Neuroradiol. 2001, 22, 1326–1333. [Google Scholar] [PubMed]
- Bullmore, E.; Sporns, O. Complex brain networks: Graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 2009, 10, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Zalesky, A.; Fornito, A.; Egan, G.F.; Pantelis, C.; Bullmore, E.T. The relationship between regional and inter-regional functional connectivity deficits in schizophrenia. Hum. Brain Mapp. 2012, 33, 2535–2549. [Google Scholar] [CrossRef] [PubMed]
- Lynall, M.-E.; Bassett, D.S.; Kerwin, R.; McKenna, P.J.; Kitzbichler, M.; Muller, U.; Bullmore, E. Functional connectivity and brain networks in schizophrenia. J. Neurosci. 2010, 30, 9477–9487. [Google Scholar] [CrossRef] [PubMed]
- Bassett, D.S.; Nelson, B.G.; Mueller, B.A.; Camchong, J.; Lim, K.O. Altered resting state complexity in schizophrenia. NeuroImage 2012, 59, 2196–2207. [Google Scholar] [CrossRef] [PubMed]
- Rotarska-Jagiela, A.; van de Ven, V.; Oertel-Knöchel, V.; Uhlhaas, P.J.; Vogeley, K.; Linden, D.E. Resting-state functional network correlates of psychotic symptoms in schizophrenia. Schizophr. Res. 2010, 117, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Dai, Z.; Peng, H.; Tan, L.; Ding, Y.; He, Z.; Zhang, Y.; Xia, M.; Li, Z.; Li, W. Overlapping and segregated resting-state functional connectivity in patients with major depressive disorder with and without childhood neglect. Hum. Brain Mapp. 2014, 35, 1154–1166. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L.; Sepulcre, J.; Talukdar, T.; Krienen, F.M.; Liu, H.; Hedden, T.; Andrews-Hanna, J.R.; Sperling, R.A.; Johnson, K.A. Cortical hubs revealed by intrinsic functional connectivity: Mapping, assessment of stability, and relation to alzheimer’s disease. J. Neurosci. 2009, 29, 1860–1873. [Google Scholar] [CrossRef] [PubMed]
- Zuo, X.N.; Ehmke, R.; Mennes, M.; Imperati, D.; Castellanos, F.X.; Sporns, O.; Milham, M.P. Network centrality in the human functional connectome. Cereb. Cortex 2012, 22, 1862–1875. [Google Scholar] [CrossRef] [PubMed]
- Logothetis, N.K.; Pauls, J.; Augath, M.; Trinath, T.; Oeltermann, A. Neurophysiological investigation of the basis of the fmri signal. Nature 2001, 412, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Van den Heuvel, M.P.; Hulshoff Pol, H.E. Exploring the brain network: A review on resting-state fmri functional connectivity. Eur. Neuropsychopharmacol. 2010, 20, 519–534. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Lui, S.; Deng, W.; Li, F.; Li, M.; Huang, X.; Wang, Y.; Li, T.; Sweeney, J.A.; Gong, Q. Anatomical and functional brain abnormalities in drug-naive first-episode schizophrenia. Am. J. Psychiatry 2013, 170, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.-Q.; Lui, S.; Deng, W.; Chan, R.; Wu, Q.-Z.; Jiang, L.-J.; Zhang, J.-R.; Jia, Z.-Y.; Li, X.-L.; Li, F. Localization of cerebral functional deficits in treatment-naive, first-episode schizophrenia using resting-state fMRI. NeuroImage 2010, 49, 2901. [Google Scholar] [CrossRef] [PubMed]
- Lui, S.; Li, T.; Deng, W.; Jiang, L.; Wu, Q.; Tang, H.; Yue, Q.; Huang, X.; Chan, R.C.; Collier, D.A. Short-term effects of antipsychotic treatment on cerebral function in drug-naive first-episode schizophrenia revealed by “resting state” functional magnetic resonance imaging. Arch. Gen. Psychiatry 2010, 67, 783. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.A.; Chen, H.; Mathalon, D.H.; Allen, E.A.; Mayer, A.R.; Abbott, C.C.; Calhoun, V.D.; Bustillo, J. Reliability of the amplitude of low-frequency fluctuations in resting state fmri in chronic schizophrenia. Psychiatry Res. Neuroimaging 2012, 201, 253–255. [Google Scholar] [CrossRef] [PubMed]
- Kay, S.R.; Flszbein, A.; Opfer, L.A. The positive and negative syndrome scale (panss) for schizophrenia. Schizophr. Bull. 1987, 13, 261. [Google Scholar] [CrossRef] [PubMed]
- Wallwork, R.; Fortgang, R.; Hashimoto, R.; Weinberger, D.; Dickinson, D. Searching for a consensus five-factor model of the positive and negative syndrome scale for schizophrenia. Schizophr. Res. 2012, 137, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L.; Andrews-Hanna, J.R.; Schacter, D.L. The brain’s default network. Ann. N. Y. Acad. Sci. 2008, 1124, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Cauda, F.; D’Agata, F.; Sacco, K.; Duca, S.; Geminiani, G.; Vercelli, A. Functional connectivity of the insula in the resting brain. NeuroImage 2011, 55, 8–23. [Google Scholar] [CrossRef] [PubMed]
- Damoiseaux, J.S.; Rombouts, S.; Barkhof, F.; Scheltens, P.; Stam, C.J.; Smith, S.M.; Beckmann, C.F. Consistent resting-state networks across healthy subjects. Proc. Natl. Acad. Sci. USA 2006, 103, 13848–13853. [Google Scholar] [CrossRef] [PubMed]
- Stone, J.M.; Davis, J.M.; Leucht, S.; Pilowsky, L.S. Cortical dopamine d2/d3 receptors are a common site of action for antipsychotic drugs—An original patient data meta-analysis of the spect and pet in vivo receptor imaging literature. Schizophr. Bull. 2009, 35, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Pettersson-Yeo, W.; Allen, P.; Benetti, S.; McGuire, P.; Mechelli, A. Dysconnectivity in schizophrenia: Where are we now? Neurosci. Biobehav. Rev. 2011, 35, 1110–1124. [Google Scholar] [CrossRef] [PubMed]
- Garrity, A.; Pearlson, G.; McKiernan, K.; Lloyd, D.; Kiehl, K.; Calhoun, V. Aberrant “default mode” functional connectivity in schizophrenia. Am. J. Psychiatry 2007, 164, 450–457. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Deng, W.; Li, M.; Chen, Z.; Jiang, L.; Wang, Q.; Huang, C.; Collier, D.; Gong, Q.; Ma, X. Aberrant intrinsic brain activity and cognitive deficit in first-episode treatment-naive patients with schizophrenia. Psychol. Med. 2012, 1, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sorg, C.; Manoliu, A.; Neufang, S.; Myers, N.; Peters, H.; Schwerthöffer, D.; Scherr, M.; Mühlau, M.; Zimmer, C.; Drzezga, A. Increased intrinsic brain activity in the striatum reflects symptom dimensions in schizophrenia. Schizophr. Bull. 2013, 39, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.A.; Damaraju, E.; van Erp, T.G.; Mathalon, D.H.; Ford, J.M.; Voyvodic, J.; Mueller, B.A.; Belger, A.; Bustillo, J.; McEwen, S. A multi-site resting state fmri study on the amplitude of low frequency fluctuations in schizophrenia. Front. Neurosci. 2013, 7, 137. [Google Scholar] [CrossRef] [PubMed]
- Howes, O.D.; Kapur, S. The dopamine hypothesis of schizophrenia: Version III—The final common pathway. Schizophr. Bull. 2009, 35, 549–562. [Google Scholar] [CrossRef] [PubMed]
- Guillin, O.; Abi-Dargham, A.; Laruelle, M. Neurobiology of dopamine in schizophrenia. Int. Rev. Neurobiol. 2007, 78, 1–39. [Google Scholar] [PubMed]
- Schmitt, G.J.E.; Meisenzahl, E.M.; Frodl, T.; La Fougère, C.; Hahn, K.; Möller, H.-J.; Dresel, S. Increase of striatal dopamine transmission in first episode drug-naive schizophrenic patients as demonstrated by [123i] IBZM SPECT. Psychiatry Res. Neuroimaging 2009, 173, 183–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abi-Dargham, A. Do we still believe in the dopamine hypothesis? New data bring new evidence. Int. J. Neuropsychopharmacol. 2004, 7, S1–S5. [Google Scholar] [CrossRef] [PubMed]
- Knable, M.; Egan, M.; Heinz, A.; Gorey, J.; Lee, K.; Coppola, R.; Weinberger, D. Altered dopaminergic function and negative symptoms in drug-free patients with schizophrenia. [123i]-iodobenzamide SPECT study. Br. J. Psychiatry 1997, 171, 574–577. [Google Scholar] [CrossRef] [PubMed]
- Winograd-Gurvich, C.; Fitzgerald, P.; Georgiou-Karistianis, N.; Bradshaw, J.; White, O. Negative symptoms: A review of schizophrenia, melancholic depression and parkinson’s disease. Brain Res. Bull. 2006, 70, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, H.; Happe, F.; Brunswick, N.; Fletcher, P.; Frith, U.; Frith, C. Reading the mind in cartoons and stories: An fmri study of “theory of mind” in verbal and nonverbal tasks. Neuropsychologia 2000, 38, 11–21. [Google Scholar] [CrossRef]
- Lee, K.-H.; Brown, W.; Egleston, P.; Green, R.; Farrow, T.; Hunter, M.; Parks, R.; Wilkinson, I.; Spence, S.; Woodruff, P. A functional magnetic resonance imaging study of social cognition in schizophrenia during an acute episode and after recovery. Am. J. Psychiatry 2006, 163, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Kalbe, E.; Schlegel, M.; Sack, A.T.; Nowak, D.A.; Dafotakis, M.; Bangard, C.; Brand, M.; Shamay-Tsoory, S.; Onur, O.A.; Kessler, J. Dissociating cognitive from affective theory of mind: A TMS study. Cortex 2010, 46, 769–780. [Google Scholar] [CrossRef] [PubMed]
- Torrey, E.F. Schizophrenia and the inferior parietal lobule. Schizophr. Res. 2007, 97, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Abu-Akel, A.; Shamay-Tsoory, S. Neuroanatomical and neurochemical bases of theory of mind. Neuropsychologia 2011, 49, 2971–2984. [Google Scholar] [CrossRef] [PubMed]
- Bora, E.; Pantelis, C. Theory of mind impairments in first-episode psychosis, individuals at ultra-high risk for psychosis and in first-degree relatives of schizophrenia: Systematic review and meta-analysis. Schizophr. Res. 2013, 144, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Irani, F.; Seligman, S.; Kamath, V.; Kohler, C.; Gur, R.C. A meta-analysis of emotion perception and functional outcomes in schizophrenia. Schizophr. Res. 2012, 137, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Sumich, A.; Castro, A.; Anilkumar, A.P.; Zachariah, E.; Kumari, V. Neurophysiological correlates of excitement in schizophrenia. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2013, 46, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Sumich, A.; Anilkumar, A.P.; Kumari, V. Sex specific event-related potential (erp) correlates of depression in schizophrenia. Psychiatr. Danub. 2014, 26, 27–33. [Google Scholar] [PubMed]
- First, M.B.; Gibbon, M. User’s Guide for the Structured Clinical Interview for DSM-IV Axis I Disorders: SCID-I Clinician Version; American Psychiatric Pub: Arlington, VA, USA, 1997. [Google Scholar]
- First, M.B.; Gibbon, M.; Williams, J.B. Structured Clinical Interview for DSM-IV Axis I Disorders-non-patient Edition (SCID-I/P); New York State Psychiatric Institute, Biometrics Research: New York, NY, USA, 1996. [Google Scholar]
- Annett, M. A classification of hand preference by association analysis. Br. J. Psychol. 1970, 61, 303–321. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Zang, Y. Dparsf: A matlab toolbox for “pipeline” data analysis of resting-state fMRI. Front. Syst. Neurosci. 2010, 4, 13. [Google Scholar] [CrossRef] [PubMed]
- Song, X.-W.; Dong, Z.-Y.; Long, X.-Y.; Li, S.-F.; Zuo, X.-N.; Zhu, C.-Z.; He, Y.; Yan, C.-G.; Zang, Y.-F. Rest: A toolkit for resting-state functional magnetic resonance imaging data processing. PLoS ONE 2011, 6, e25031. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.; Birn, R.M.; Handwerker, D.A.; Jones, T.B.; Bandettini, P.A. The impact of global signal regression on resting state correlations: Are anti-correlated networks introduced? NeuroImage 2009, 44, 893. [Google Scholar] [CrossRef] [PubMed]
- Statistical Parametric Mapping. Available online: http://www.fil.ion.ucl.ac.uk/spm/ (accessed on 1 July 2014).
- Liao, W.; Chen, H.; Feng, Y.; Mantini, D.; Gentili, C.; Pan, Z.; Ding, J.; Duan, X.; Qiu, C.; Lui, S. Selective aberrant functional connectivity of resting state networks in social anxiety disorder. NeuroImage 2010, 52, 1549–1558. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lei, W.; Li, M.; Deng, W.; Zhou, Y.; Ma, X.; Wang, Q.; Guo, W.; Li, Y.; Jiang, L.; Han, Y.; et al. Sex-Specific Patterns of Aberrant Brain Function in First-Episode Treatment-Naive Patients with Schizophrenia. Int. J. Mol. Sci. 2015, 16, 16125-16143. https://doi.org/10.3390/ijms160716125
Lei W, Li M, Deng W, Zhou Y, Ma X, Wang Q, Guo W, Li Y, Jiang L, Han Y, et al. Sex-Specific Patterns of Aberrant Brain Function in First-Episode Treatment-Naive Patients with Schizophrenia. International Journal of Molecular Sciences. 2015; 16(7):16125-16143. https://doi.org/10.3390/ijms160716125
Chicago/Turabian StyleLei, Wei, Mingli Li, Wei Deng, Yi Zhou, Xiaohong Ma, Qiang Wang, Wanjun Guo, Yinfei Li, Lijun Jiang, Yuanyuan Han, and et al. 2015. "Sex-Specific Patterns of Aberrant Brain Function in First-Episode Treatment-Naive Patients with Schizophrenia" International Journal of Molecular Sciences 16, no. 7: 16125-16143. https://doi.org/10.3390/ijms160716125