Physcomitrella patens Activates Defense Responses against the Pathogen Colletotrichum gloeosporioides
Abstract
:1. Introduction
2. Results and Discussion
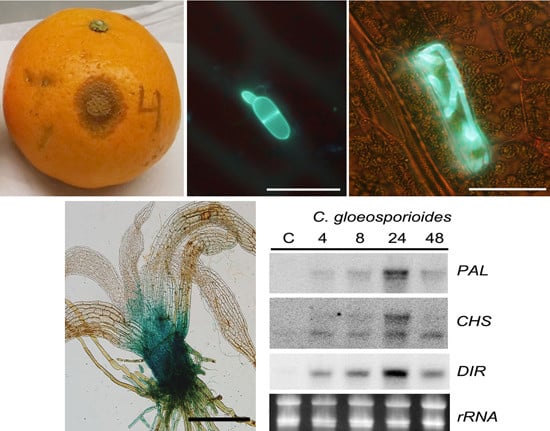
2.1. Colletotrichum gloeosporioides Isolated from Orange Fruits Infects P. patens and Causes Disease Symptoms


2.2. P. patens Activates Cell Wall-Associated Defenses against C. gloeosporioides


2.3. Colletotrichum gloeosporioides Infection Causes Cytoplasmic Shrinkage and Chloroplasts Repositioning


2.4. P. patens Induces Defense Gene Activation in Response to C. gloeosporioides Infection

2.5. P. patens Activates Auxin Signaling in Response to C. gloeosporioides Infection

3. Experimental Section
3.1. Isolation and Identification of Colletotrichum Strain
3.2. Physcomitrella patens and Colletotrichum gloeosporioides Growth Conditions
3.3. Colletotrichum gloeosporioides Inoculation and Staining
3.4. Plant Cell Wall-Associated Defense Responses
3.5. RNA Gel Blot Analysis
3.6. GUS Staining
3.7. NAA Treatment
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ponce de León, I.; Montesano, M. Activation of defense mechanisms against pathogens in mosses and flowering plants. Int. J. Mol. Sci. 2013, 14, 3178–3200. [Google Scholar] [PubMed]
- Morel, J.B.; Dangl, J.L. The hypersensitive response and the induction of cell death in plants. Cell Death Differ. 1997, 4, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Dickman, M.B.; Park, Y.K.; Oltersdorf, T.; Li, W.; Clemente, T.; French, R. Abrogation of disease development in plants expressing animal antiapoptotic genes. Proc. Natl. Acad. Sci. USA 2001, 98, 6957–6962. [Google Scholar] [CrossRef] [PubMed]
- Govrin, E.M.; Levine, A. The hypersensitive response facilitates plant infection by the necrotrophic pathogen Botrytis cinerea. Curr. Biol. 2000, 10, 751–757. [Google Scholar] [CrossRef]
- Glazebrook, J. Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef] [PubMed]
- Robert-Seilaniantz, A.; Grant, M.; Jones, J.D. Hormone crosstalk in plant disease and defense: More than just jasmonate-salicylate antagonism. Annu. Rev. Phytopathol. 2011, 8, 317–343. [Google Scholar] [CrossRef] [PubMed]
- Robert-Seilaniantz, A.; Navarro, L.; Bari, R.; Jones, J.D. Pathological hormone imbalances. Curr. Opin. Plant Biol. 2007, 10, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Agrios, G.N. Plant Pathology, 5th ed.; Elsevier Academic Press: San Diego, CA, USA, 2005; pp. 487–500. [Google Scholar]
- O’Connell, R.J.; Thon, M.R.; Hacquard, S.; Amyotte, S.G.; Kleemann, J.; Torres, M.F.; Damm, U.; Buiate, E.A.; Epstein, L.; Alkan, N.; et al. Lifestyle transitions in plant pathogenic Colletotrichum fungi deciphered by genome and transcriptome analyses. Nat. Genet. 2012, 44, 1060–1065. [Google Scholar] [CrossRef] [PubMed]
- Gan, P.; Ikeda, K.; Irieda, H.; Narusaka, M.; O’Connell, R.J.; Narusaka, Y.; Takano, Y.; Kubo, Y.; Shirasu, K. Comparative genomic and transcriptomic analyses reveal the hemibiotrophic stage shift of Colletotrichum fungi. New Phytol. 2013, 197, 1236–1249. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.D.; Cai, L.; Cannon, P.F.; Crouch, J.A.; Crous, P.W.; Damm, U.; Goodwin, P.H.; Chen, H.; Johnston, P.R.; Jones, E.B.G.; et al. Colletotrichum-names in current use. Fungal Divers. 2009, 39, 147–182. [Google Scholar]
- Timmer, L.W.; Brown, G.E.; Sitko, S.E. The role of Colletotrichum spp. in postharvest anthracnose of citrus and survival of C. acutatum on fruit. Plant Dis. 1998, 82, 415–418. [Google Scholar] [CrossRef]
- Brown, G.E. Factors affecting postharvest development of Colletotrichum gloeosporioides in citrus fruits. Phytopathology 1975, 65, 404–409. [Google Scholar] [CrossRef]
- Taverner, P. Anthracnose. Packer Newslett. 2003, 71, 1–5. [Google Scholar]
- Brown, G.E.; Barmore, C.R. The effect of ethylene on susceptibility of Robinson tangerines to anthracnose. Phytopathology 1977, 67, 120–123. [Google Scholar] [CrossRef]
- Akita, M.; Lehtonen, M.T.; Koponen, H.; Marttinen, E.M.; Valkonen, J.P. Infection of the Sunagoke moss panels with fungal pathogens hampers sustainable greening in urban environments. Sci. Total Environ. 2011, 409, 3166–3173. [Google Scholar] [CrossRef] [PubMed]
- Ponce de León, I.; Oliver, J.P.; Castro, A.; Gaggero, C.; Bentancor, M.; Vidal, S. Erwinia carotovora elicitors and Botrytis cinerea activate defense responses in Physcomitrella patens. BMC Plant Biol. 2007, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.P.; Castro, A.; Gaggero, C.; Cascón, T.; Schmelz, E.A.; Castresana, C.; Ponce de León, I. Pythium infection activates conserved plant defense responses in mosses. Planta 2009, 230, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Ponce de León, I.; Schmelz, E.A.; Gaggero, C.; Castro, A.; Alvarez, A.; Montesano, M. Physcomitrella patens activates reinforcement of the cell wall, programmed cell death and accumulation of evolutionary conserved defence signals, such as salicylic acid and 12-oxophytodienoic acid, but not jasmonic acid, upon Botrytis cinerea infection. Mol. Plant Pathol. 2012, 13, 960–974. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, M.T.; Akita, M.; Frank, W.; Reski, R.; Valkonen, J.P. Involvement of a class III peroxidase and the mitochondrial protein TSPO in oxidative burst upon treatment of moss plants with a fungal elicitor. Mol. Plant Microbe Interact. 2012, 25, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Machado, L.; Castro, A.; Hamberg, M.; Bannenberg, G.; Gaggero, C.; Castresana, C.; Ponce de León, I. The Physcomitrella patens unique alpha-dioxygenase participates in both developmental processes and defense responses. BMC Plant Biol. 2015, 15, 45. [Google Scholar] [CrossRef] [PubMed]
- Mittag, J.; Šola, I.; Rusak, G.; Ludwig-Müller, J. Physcomitrella patens auxin conjugate synthetase (GH3) double knockout mutants are more resistant to Pythium infection than wild type. J. Plant Physiol. 2015, 183, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Agostini, J.P.; Timmer, L.W.; Mitchell, D.J. Morphological and pathological characteristics of strains of Colletotrichum gloeosporioides from citrus. Phytopathology 1992, 82, 1377–1382. [Google Scholar] [CrossRef]
- Flaishman, M.A.; Hwang, C.H.; Kolattukudy, P.E. Involvement of protein phosphorylation in the induction of appressorium formation in Colletotrichum gloeosporioides by its host surface wax and ethylene. Physiol. Mol. Plant Pathol. 1995, 47, 103–117. [Google Scholar] [CrossRef]
- Kim, Y.K.; Kawano, T.; Li, D.; Kolattukudy, P.E. A mitogen-activated protein kinase kinase required for induction of cytokinesis and appressorium formation by host signals in the conidia of Colletotrichum gloeosporioides. Plant Cell 2000, 12, 1331–1343. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Li, G.; Lin, C.; Shi, T.; Zhai, L.; Chen, Y.; Huang, G. Identifying pathogenicity genes in the rubber tree anthracnose fungus Colletotrichum gloeosporioides through random insertional mutagenesis. Microbiol. Res. 2013, 168, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Mendgen, K.; Struck, C.; Voegele, R.T.; Hahn, M. Biotrophy and rust haustoria. Physiol. Mol. Plant Pathol. 2000, 56, 141–145. [Google Scholar] [CrossRef]
- Perfect, S.E.; Hughes, H.B.; O’Connell, R.J.; Green, J.R. Colletotrichum: A model genus for studies on pathology and fungal-plant interactions. Fungal Genet. Biol. 1999, 27, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Barreto, A.L.H.; Vasconcelos, I.M.; Grangeiro, T.B.; Melo, V.M.M.; Matos, T.E.; Eloy, Y.R.G.; Fernandes, C.F.; Torres, D.C.; Freire-Filho, F.R.; Freire, F.C.O.; et al. Infection process and host defense responses in compatible and incompatible interactions between cowpea (Vigna unguiculata) and Colletotrichum gloeosporioides. Int. J. Plant Sci. 2007, 168, 193–203. [Google Scholar] [CrossRef]
- Rockenbach, M.F.; Boneti, J.I.; Cangahuala-Inocente, G.C.; Gavioli-Nascimento, M.C.A.; Guerra, M.P. Histological and proteomics analysis of apple defense responses to the development of Colletotrichum gloeosporioides on leaves. Physiol. Mol. Plant Pathol. 2015, 89, 97–107. [Google Scholar] [CrossRef]
- Wharton, P.S.; Uribeondo, J.D. The biology of Colletotrichum acutatum. Anales Jard Bot. Madrid 2004, 61, 3–22. [Google Scholar]
- O’Connell, R.; Perfect, S.; Hughes, B.; Carzaniga, R.; Bailey, J.; Green, J. Dissecting the cell biology of Colletotrichum infection processes. In Colletotrichum: Host Specificity, Pathology and Host-Pathogen Interaction; Prusky, D., Freeman, S., Dickman, M.B., Eds.; APS Press: St. Paul, MN, USA, 2000; pp. 57–77. [Google Scholar]
- Latunde-Dada, A.O. Colletotrichum: Tales of forcible entry, stealth, transient confinement and breakout. Mol. Plant Pathol. 2001, 2, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Ogle, H.J.; Gowanlock, D.H.; Irwin, J.A.G. Infection of Stylosanthes guianensis and S. scabra by Colletotrichum gloeosporioides. Phytopathology 1990, 80, 837–842. [Google Scholar] [CrossRef]
- Reski, R. Physcomitrella and Arabidopsis: The David and Goliath of reverse genetics. Trends Plant Sci. 1998, 3, 209–210. [Google Scholar] [CrossRef]
- Kim, K.H.; Yoon, J.B.; Park, H.G.; Park, E.W.; Kim, Y.H. Structural modifications and programmed cell death of chili pepper fruit related to resistance responses to Colletotrichum gloeosporioides infection. Phytopathology 2004, 94, 1295–1304. [Google Scholar] [CrossRef] [PubMed]
- Yakoby, N.; Kobiler, I.; Dinoor, A.; Prusky, D. pH regulation of pectate lyase secreation modulates the attack of Colletotrichum gloeosporioides on avocado fruits. Appl. Environ. Microbiol. 2000, 66, 1026e30. [Google Scholar]
- Alkan, N.; Friedlander, G.; Ment, D.; Prusky, D.; Fluhr, R. Simultaneous transcriptome analysis of Colletotrichum gloeosporioides and tomato fruit pathosystem reveals novel fungal pathogenicity and fruit defense strategies. New Phytol. 2015, 205, 801–815. [Google Scholar] [CrossRef] [PubMed]
- Hiruma, K.; Fukunaga, S.; Bednarek, P.; Pislewska-Bednarek, M.; Watanabe, S.; Narusaka, Y.; Shirasu, K.; Takano, Y. Glutathione and tryptophan metabolism are required for Arabidopsis immunity during the hypersensitive response to hemibiotrophs. Proc. Natl. Acad. Sci. USA 2013, 110, 9589–9594. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, M.T.; Marttinen, E.M.; Akita, M.; Valkonen, J.P.T. Fungi infecting cultivated moss can also cause diseases in crop plants. Ann. Appl. Biol. 2012, 160, 298–307. [Google Scholar] [CrossRef]
- Torres, M.A.; Jones, J.D.; Dangl, J.L. Reactive oxygen species signaling in response to pathogens. Plant Physiol. 2006, 141, 373–378. [Google Scholar] [PubMed]
- Nomura, H.; Komori, T.; Uemura, S.; Kanda, Y.; Shimotani, K.; Nakai, K.; Furuichi, T.; Takebayashi, K.; Sugimoto, T.; Sano, S.; et al. Chloroplast-mediated activation of plant immune signalling in Arabidopsis. Nat. Commun. 2012, 3, 926. [Google Scholar] [CrossRef] [PubMed]
- Trotta, A.; Rahikainen, M.; Konert, G.; Finazzi, G.; Kangasjärvi, S. Signalling crosstalk in light stress and immune reactions in plants. Philos. Trans. R. Soc. Lond. B 2014, 369, 20130235. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.C.; Verma, D.; Jin, S.; Singh, N.D.; Daniell, H. Release of proteins from intact chloroplasts induced by reactive oxygen species during biotic and abiotic stress. PLoS ONE 2013, 8, e67106. [Google Scholar] [CrossRef] [PubMed]
- Caplan, J.L.; Mamillapalli, P.; Burch-Smith, T.M.; Czymmek, K.; Dinesh-Kumar, S.P. Chloroplastic protein NRIP1 mediates innate immune receptor recognition of a viral effector. Cell 2008, 132, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Caplan, J.L.; Kumar, A.S.; Park, E.; Padmanabhan, M.S.; Hoban, K.; Modla, S.; Czymmek, K.; Dinesh-Kumar, S.P. Chloroplast dtromules function during innate immunity. Dev. Cell 2015, 34, 45–57. [Google Scholar] [CrossRef]
- Freytag, S.; Arabatzis, N.; Hahlbrock, K.; Schmelzer, E. Reversible cytoplasmic rearrangements precede wall apposition, hypersensitive cell death and defense-related gene activation in potato/Phytophthora infestans interactions. Planta 1994, 194, 123–135. [Google Scholar] [CrossRef]
- Wolf, L.; Rizzini, L.; Stracke, R.; Ulm, R.; Rensing, S.A. The molecular and physiological responses of Physcomitrella patens to ultraviolet-B radiation. Plant Physiol. 2010, 153, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Koduri, P.K.; Gordon, G.S.; Barker, E.I.; Colpitts, C.C.; Ashton, N.W.; Suh, D.Y. Genome-wide analysis of the chalcone synthase superfamily genes of Physcomitrella patens. Plant Mol. Biol. 2010, 72, 247–263. [Google Scholar] [CrossRef] [PubMed]
- Tavernier, V.; Cadiou, S.; Pageau, K.; Laugé, R.; Reisdorf-Cren, M.; Langin, T.; Masclaux-Daubresse, C. The plant nitrogen mobilization promoted by Colletotrichum lindemuthianum in Phaseolus leaves depends on fungus pathogenicity. J. Exp. Bot. 2007, 58, 3351–3360. [Google Scholar] [CrossRef] [PubMed]
- Samac, D.A.; Peñuela, S.; Schnurr, J.A.; Hunt, E.N.; Foster-Hartnett, D.; Vandenbosch, K.A.; Gantt, J.S. Expression of coordinately regulated defence response genes and analysis of their role in disease resistance in Medicago truncatula. Mol. Plant Pathol. 2011, 12, 786–798. [Google Scholar] [CrossRef] [PubMed]
- Davin, L.B.; Lewis, N.G. Dirigent proteins and dirigent sites explain the mystery of specificity of radical precursor coupling in lignan and lignin biosynthesis. Plant Physiol. 2000, 123, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.; Riov, J.; Sharon, A. Indole-3-acetic acid biosynthesis in Colletotrichum gloeosporioides f. sp. aeschynomene. Appl. Environ. Microbiol. 1998, 64, 5030–5032. [Google Scholar] [PubMed]
- Maor, R.; Haskin, S.; Levi-Kedmi, H.; Sharon, A. In planta production of indole-3-acetic acid by Colletotrichum gloeosporioides f. sp. aeschynomene. Appl. Environ. Microbiol. 2004, 70, 1852–1854. [Google Scholar] [CrossRef] [PubMed]
- Reineke, G.; Heinze, B.; Schirawski, J.; Buttner, H.; Kahmann, R.; Basse, C.W. Indole-3-acetic acid (IAA) biosynthesis in the smut fungus Ustilago maydis and its relevance for increased IAA levels in infected tissue and host tumor formation. Mol. Plant Pathol. 2008, 9, 339–355. [Google Scholar] [CrossRef] [PubMed]
- Bierfreund, N.M.; Reski, R.; Decker, E.L. Use of an inducible reporter gene system for the analysis of auxin distribution in the moss Physcomitrella patens. Plant Cell Rep. 2003, 21, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Ludwig-Müller, J.; Jülke, S.; Bierfreund, N.M.; Decker, E.L.; Reski, R. Moss (Physcomitrella patens) GH3 proteins act in auxin homeostasis. New Phytol. 2009, 181, 323–338. [Google Scholar] [CrossRef]
- Menand, B.; Calder, G.; Dolan, L. Both chloronemal and caulonemal cells expand by tip growth in the moss Physcomitrella patens. J. Exp. Bot. 2007, 58, 1843–1849. [Google Scholar] [CrossRef] [PubMed]
- Lahey, K.A.; Yuan, R.; Burns, J.K.; Ueng, P.P.; Timmer, L.W.; Chung, K.R. Induction of phytohormones and differential gene expression in citrus flowers infected by the fungus Colletotrichum acutatum. Mol. Plant Microb. Interact. 2004, 17, 1394–1401. [Google Scholar] [CrossRef] [PubMed]
- Llorente, F.; Muskett, P.; Sánchez-Vallet, A.; López, G.; Ramos, B.; Sánchez-Rodríguez, C.; Jordá, L.; Parker, J.; Molina, A. Repression of the auxin response pathway increases Arabidopsis susceptibility to necrotrophic fungi. Mol. Plant. 2008, 1, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Yan, J.; Li, Y.; Jiang, H.; Sun, J.; Chen, Q.; Li, H.; Chu, J.; Yan, C.; Sun, X.; et al. Arabidopsis thaliana plants differentially modulate auxin biosynthesis and transport during defense responses to the necrotrophic pathogen Alternaria brassicicola. New Phytol. 2012, 195, 872–882. [Google Scholar] [CrossRef] [PubMed]
- Lima, W.G.; Spósito, M.B.; Amorim, L.; Goncalves, F.P.; de Filho, P.A.M. Colletotrichum gloeosporioides, a new causal agent of citrus post-bloom fruit drop. Eur. J. Plant Pathol. 2011, 131, 157–165. [Google Scholar]
- Nishiyama, T.; Fujita, T.; Shin-I, T.; Seki, M.; Nishide, H.; Uchiyama, I.; Kamiya, A.; Carninci, P.; Hayashizaki, Y.; Shinozaki, K.; et al. Comparative genomics of Physcomitrella patens gametophytic transcriptome and Arabidopsis thaliana: Implication for land plant evolution. Proc. Natl. Acad. Sci. USA 2003, 100, 8007–8012. [Google Scholar] [CrossRef]
- Peleman, J.; Boerjan, W.; Engler, G.; Seurinck, J.; Botterman, J.; Alliote, T.; van Montagu, M.; Inzé, D. Strong cellular preference in the expression of a housekeeping gene of Arabidopsis thaliana encoding S-adenosylmethionine synthetase. Plant Cell 1989, 1, 81–93. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, D.G. A new moss genetics: Targeted mutagenesis in Physcomitrella patens. Annu. Rev. Plant Biol. 2002, 53, 477–501. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reboledo, G.; Del Campo, R.; Alvarez, A.; Montesano, M.; Mara, H.; Ponce de León, I. Physcomitrella patens Activates Defense Responses against the Pathogen Colletotrichum gloeosporioides. Int. J. Mol. Sci. 2015, 16, 22280-22298. https://doi.org/10.3390/ijms160922280
Reboledo G, Del Campo R, Alvarez A, Montesano M, Mara H, Ponce de León I. Physcomitrella patens Activates Defense Responses against the Pathogen Colletotrichum gloeosporioides. International Journal of Molecular Sciences. 2015; 16(9):22280-22298. https://doi.org/10.3390/ijms160922280
Chicago/Turabian StyleReboledo, Guillermo, Raquel Del Campo, Alfonso Alvarez, Marcos Montesano, Héctor Mara, and Inés Ponce de León. 2015. "Physcomitrella patens Activates Defense Responses against the Pathogen Colletotrichum gloeosporioides" International Journal of Molecular Sciences 16, no. 9: 22280-22298. https://doi.org/10.3390/ijms160922280