MicroRNA-375 Functions as a Tumor-Suppressor Gene in Gastric Cancer by Targeting Recepteur d’Origine Nantais
Abstract
:1. Introduction
2. Results
2.1. miR-375 Directly Targets the 3′-Untranslated Region (3′-UTR) of Recepteur d’Origine Nantais (RON)
2.2. Inverse Association between miR-375 and RON Expression in AGS and MKN-28 Human Gastric Cancer Cells
2.3. miR-375 Inhibits AGS and MKN-28 Human Gastric Cancer Cell Proliferation
2.4. miR-375 Induced G1 Cell Cycle Arrest by Decreasing Cyclin D1 and Cyclin D3 Expression, and Retinoblastoma (Rb) Phosphorylation
2.5. miR-375 Inhibits Migration in AGS and MKN-28 Human Gastric Cancer Cells
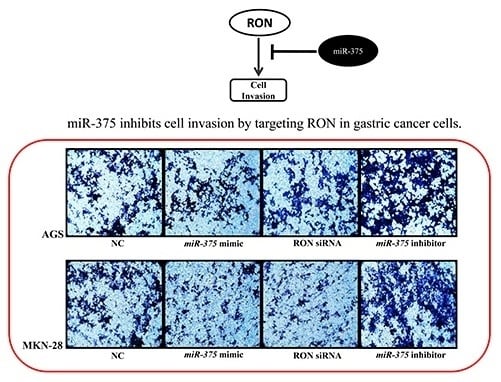
2.6. miR-375 Inhibits Invasion in AGS and MKN-28 Human Gastric Cancer Cells
2.7. Knockdown of RON Reduces Proliferation, Migration, and Invasion in AGS and MKN-28 Human Gastric Cancer Cells
3. Discussion
4. Experimental Section
4.1. Reagents
4.2. Cell Culture
4.3. Cell Transfection
4.4. Luciferase Activity Assay
4.5. RNA Extraction and Quantitative Real Time PCR (qRT-PCR)
4.6. Preparation of Cell Extracts and Western Blot Analysis
4.7. Cell Cycle Analysis
4.8. Cell Proliferation Assay
4.9. In Vitro Wound-Healing Assay
4.10. Matrigel Invasion Assay
4.11. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global cancer statistics, 2002. CA Cancer J. Clin. 2005, 55, 74–108. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.H.; Ronsin, C.; Gesnel, M.C.; Coupey, L.; Skeel, A.; Leonard, E.J.; Breathnach, R. Identification of the RON gene product as the receptor for the human macrophage stimulating protein. Science 1994, 266, 117–119. [Google Scholar] [CrossRef] [PubMed]
- Ronsin, C.; Muscatelli, F.; Mattei, M.; Breathnach, R. A novel putative receptor protein tyrosine kinase of the met family. Oncogene 1993, 8, 1195–1202. [Google Scholar] [PubMed]
- Chen, Y.Q.; Zhou, Y.Q.; Fisher, J.H.; Wang, M.H. Targeted expression of the receptor tyrosine kinase RON in distal lung epithelial cells results in multiple tumor formation: oncogenic potential of RON in vivo. Oncogene 2002, 21, 6382–6386. [Google Scholar] [CrossRef] [PubMed]
- De Bortoli, M.; Di Renzo, M.F.; Costantino, A.; Sismondi, P.; Comoglio, P.M. Overexpression of the RON gene in human breast carcinoma. Oncogene 1998, 16, 2927–2933. [Google Scholar]
- Zhou, Y.Q.; He, C.; Chen, Y.Q.; Wang, D.; Wang, M.H. Altered expression of the RON receptor tyrosine kinase in primary human colorectal adenocarcinomas: Generation of different splicing RON variants and their oncogenic potential. Oncogene 2003, 22, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Logan-Collins, J.; Thomas, R.M.; Yu, P.; Jaquish, D.; Mose, E.; French, R.; Stuart, W.; McClaine, R.; Aronow, B.; Hoffman, R.M. Silencing of RON receptor signaling promotes apoptosis and gemcitabine sensitivity in pancreatic cancers. Cancer Res. 2010, 70, 1130–1140. [Google Scholar] [CrossRef] [PubMed]
- Maggiora, P.; Lorenzato, A.; Fracchioli, S.; Costa, B.; Castagnaro, M.; Arisio, R.; Katsaros, D.; Massobrio, M.; Comoglio, P.M.; di Renzo, M.F. The RON and MET oncogenes are co-expressed in human ovarian carcinomas and cooperate in activating invasiveness. Exp. Cell Res. 2003, 288, 382–389. [Google Scholar] [CrossRef]
- Cheng, H.; Liu, H.; Lin, Y.; Chen, H.H.; Hsu, P.; Chang, T.; Ho, C.; Tzai, T.; Chow, N.-H. Co-expression of RON and MET is a prognostic indicator for patients with transitional-cell carcinoma of the bladder. Br. J. Cancer 2005, 92, 1906–1914. [Google Scholar] [CrossRef] [PubMed]
- Thobe, M.N.; Gurusamy, D.; Pathrose, P.; Waltz, S.E. The RON receptor tyrosine kinase positively regulates angiogenic chemokine production in prostate cancer cells. Oncogene 2010, 29, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Park, J.H.; Khoi, P.N.; Joo, Y.E.; Do Jung, Y. MSP-induced RON activation upregulates uPAR expression and cell invasiveness via MAPK, AP-1 and NF-κB signals in gastric cancer cells. Carcinogenesis 2011, 32, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Pan, G.; Zheng, C.; Zheng, J.; Yian, L.; Teng, X. Expression of the RON receptor tyrosine kinase and its association with gastric carcinoma versus normal gastric tissues. BMC Cancer 2008, 8, 353. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Chen, M.S.; Lo, Y.-H.; Waltz, S.E.; Wang, J.; Ho, P.C.; Vasiliauskas, J.; Plattner, R.; Wang, Y.-L.; Wang, S.C. The RON receptor tyrosine kinase activates c-Abl to promote cell proliferation through tyrosine phosphorylation of PCNA in breast cancer. Oncogene 2014, 33, 1429–1437. [Google Scholar] [CrossRef] [PubMed]
- Danilkovitch-Miagkova, A.; Angeloni, D.; Skeel, A.; Donley, S.; Lerman, M.; Leonard, E.J. Integrin-mediated RON growth factor receptor phosphorylation requires tyrosine kinase activity of both the receptor and c-Src. J. Biol. Chem. 2000, 275, 14783–14786. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Rajput, A.; Kan, J.L.; Rose, R.; Liu, X.Q.; Kuropatwinski, K.; Hauser, J.; Beko, A.; Dominquez, I.; Sharratt, E.A. Knockdown of RON kinase inhibits mutant phosphatidylinositol 3-kinase and reduces metastasis in human colon carcinoma. J. Biol. Chem. 2009, 284, 10912–10922. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Esquela-Kerscher, A.; Slack, F.J. Oncomirs—microRNAs with a role in cancer. Nat. Rev. Cancer 2006, 6, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-K. Extracellular microRNAs as biomarkers in human disease. Chonnam Med. J. 2015, 51, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Calin, G.A.; Sevignani, C.; Dumitru, C.D.; Hyslop, T.; Noch, E.; Yendamuri, S.; Shimizu, M.; Rattan, S.; Bullrich, F.; Negrini, M.; et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc. Natl. Acad. Sci. USA 2004, 101, 2999–3004. [Google Scholar] [CrossRef] [PubMed]
- Ambros, V. The functions of animal microRNAs. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Lewis, B.P.; Shih, I.H.; Jones-Rhoades, M.W.; Bartel, D.P.; Burge, C.B. Prediction of mammalian microRNA targets. Cell 2003, 115, 787–798. [Google Scholar] [CrossRef]
- Farh, K.K.H.; Grimson, A.; Jan, C.; Lewis, B.P.; Johnston, W.K.; Lim, L.P.; Burge, C.B.; Bartel, D.P. The widespread impact of mammalian MicroRNAs on mRNA repression and evolution. Science 2005, 310, 1817–1821. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Getz, G.; Miska, E.A.; Alvarez-Saavedra, E.; Lamb, J.; Peck, D.; Sweet-Cordero, A.; Ebert, B.L.; Mak, R.H.; Ferrando, A.A. MicroRNA expression profiles classify human cancers. Nature 2005, 435, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Volinia, S.; Calin, G.A.; Liu, C.G.; Ambs, S.; Cimmino, A.; Petrocca, F.; Visone, R.; Iorio, M.; Roldo, C.; Ferracin, M.; et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc. Natl. Acad. Sci. USA 2006, 103, 2257–2261. [Google Scholar] [CrossRef] [PubMed]
- Petrocca, F.; Visone, R.; Onelli, M.R.; Shah, M.H.; Nicoloso, M.S.; de Martino, I.; Iliopoulos, D.; Pilozzi, E.; Liu, C.G.; Negrini, M. E2F1-regulated microRNAs impair TGFβ-dependent cell-cycle arrest and apoptosis in gastric cancer. Cancer Cell 2008, 13, 272–286. [Google Scholar] [CrossRef] [PubMed]
- Wada, R.; Akiyama, Y.; Hashimoto, Y.; Fukamachi, H.; Yuasa, Y. miR-212 is downregulated and suppresses methyl-CpG-binding protein MeCP2 in human gastric cancer. Int. J. Cancer 2010, 127, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Tang, H.; Lang, Y.; Liu, M.; Li, X. MicroRNA-27a functions as an oncogene in gastric adenocarcinoma by targeting prohibitin. Cancer Lett. 2009, 273, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.M.; Wang, C.S.; Tsai, C.Y.; Huang, H.W.; Chi, H.C.; Lin, Y.H.; Lu, P.H.; Lin, K.H. Potential diagnostic, prognostic and therapeutic targets of MicroRNAs in human gastric cancer. Int. J. Mol. Sci. 2016, 17, 945. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Chang, Y.; Meng, F.; Wang, M.; Xie, Q.; Tang, F.; Li, P.; Song, Y.; Lin, J. MicroRNA-375 targets AEG-1 in hepatocellular carcinoma and suppresses liver cancer cell growth in vitro and in vivo. Oncogene 2012, 31, 3357–3369. [Google Scholar] [CrossRef] [PubMed]
- Ward, A.; Balwierz, A.; Zhang, J.; Küblbeck, M.; Pawitan, Y.; Hielscher, T.; Wiemann, S.; Sahin, Ö. Re-expression of microRNA-375 reverses both tamoxifen resistance and accompanying EMT-like properties in breast cancer. Oncogene 2013, 32, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Mathé, E.A.; Nguyen, G.H.; Bowman, E.D.; Zhao, Y.; Budhu, A.; Schetter, A.J.; Braun, R.; Reimers, M.; Kumamoto, K.; Hughes, D. MicroRNA expression in squamous cell carcinoma and adenocarcinoma of the esophagus: Associations with survival. Clin. Cancer Res. 2009, 15, 6192–6200. [Google Scholar] [CrossRef] [PubMed]
- Avissar, M.; Christensen, B.C.; Kelsey, K.T.; Marsit, C.J. MicroRNA expression ratio is predictive of head and neck squamous cell carcinoma. Clin. Cancer Res. 2009, 15, 2850–2855. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, Y.; Nakada, C.; Noguchi, T.; Tanigawa, M.; Nguyen, L.T.; Uchida, T.; Hijiya, N.; Matsuura, K.; Fujioka, T.; Seto, M.; et al. MicroRNA-375 is downregulated in gastric carcinomas and regulates cell survival by targeting PDK1 and 14-3-3ζ. Cancer Res. 2010, 70, 2339–2349. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Xu, Y.; Zhang, W.; Deng, Y.; Si, M.; Du, Y.; Yao, H.; Liu, X.; Ke, Y.; Si, J.; et al. MiR-375 frequently downregulated in gastric cancer inhibits cell proliferation by targeting JAK2. Cell Res. 2010, 20, 784–793. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.W.; Men, T.; Feng, R.C.; Li, Y.C.; Zhou, D.; Teng, C.B. miR-375 inhibits proliferation of mouse pancreatic progenitor cells by targeting YAP1. Cell. Physiol. Biochem. 2013, 32, 1808–1817. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Li, Y.; Zhou, J.; Xu, J.; Peng, C.; Ye, F.; Shen, Y.; Lu, W.; Wan, X.; Xie, X. miR-375 is down-regulated in squamous cervical cancer and inhibits cell migration and invasion via targeting transcription factor SP1. Am. J. Pathol. 2011, 179, 2580–2588. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, C.; Chintagari, N.R.; Bhaskaran, M.; Weng, T.; Guo, Y.; Xiao, X.; Liu, L. miR-375 regulates rat alveolar epithelial cell trans-differentiation by inhibiting Wnt/β-catenin pathway. Nucleic Acids Res. 2013, 41, 3833–3844. [Google Scholar] [CrossRef] [PubMed]
- TargetStan Release 5.1 Online Software. Available online: http://targetscan.org/ (accessed on 19 January 2016).
- PicTar. Available online: http://pictar.mdc-berlin.de (accessed on 19 January 2016).
- Musgrove, E.A.; Caldon, C.E.; Barraclough, J.; Stone, A.; Sutherland, R.L. Cyclin D as a therapeutic target in cancer. Nat. Rev. Cancer 2011, 11, 558–572. [Google Scholar] [CrossRef] [PubMed]
- Feres, K.; Ischenko, I.; Hayman, M. The RON receptor tyrosine kinase promotes MSP-independent cell spreading and survival in breast epithelial cells. Oncogene 2009, 28, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.T.; Babicky, M.; Jaquish, D.; French, R.; Marayuma, K.; Mose, E.; Niessen, S.; Hoover, H.; Shields, D.; Cheresh, D.; et al. The RON-receptor regulates pancreatic cancer cell migration through phosphorylation-dependent breakdown of the hemidesmosome. Int. J. Cancer 2012, 131, 1744–1754. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Lian, S.; Khoi, P.N.; Yoon, H.J.; Han, J.Y.; Chay, K.O.; Kim, K.K.; Jung, Y.D. Chrysin inhibits cell invasion by inhibition of Recepteur d’origine Nantais via suppressing early growth response-1 and NF-κB transcription factor activities in gastric cancer cells. Int. J. Oncol. 2015, 46, 1835–1843. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.A.; Park, Y.L.; Kim, K.Y.; Myung, E.; Chung, C.Y.; Cho, S.B.; Lee, W.S.; Jung, Y.D.; Kweon, S.S.; Joo, Y.E. RON is associated with tumor progression via the inhibition of apoptosis and cell cycle arrest in human gastric cancer. Pathol. Int. 2012, 62, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Filipowicz, W.; Bhattacharyya, S.N.; Sonenberg, N. Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat. Rev. Genet. 2008, 9, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.; Mendell, J. MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br. J. Cancer 2006, 94, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Croce, C.M. Causes and consequences of microRNA dysregulation in cancer. Nat. Rev. Genet. 2009, 10, 704–714. [Google Scholar] [CrossRef] [PubMed]
- Nasser, M.W.; Datta, J.; Nuovo, G.; Kutay, H.; Motiwala, T.; Majumder, S.; Wang, B.; Suster, S.; Jacob, S.T.; Ghoshal, K. Down-regulation of micro-RNA-1 (miR-1) in lung cancer suppression of tumorigenic property of lung cancer cells and their sensitization to doxorubicin-induced apoptosis by miR-1. J. Biol. Chem. 2008, 283, 33394–33405. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.E.; Park, J.S.; Khoi, P.N.; Joo, Y.E.; Lee, Y.H.; Jung, Y.D. Upregulation of recepteur d’origine nantais tyrosine kinase and cell invasiveness via early growth response-1 in gastric cancer cells. J. Cell. Biochem. 2012, 113, 1217–1223. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Kaldis, P. Cdks, cyclins and CKIs: Roles beyond cell cycle regulation. Development 2013, 140, 3079–3093. [Google Scholar] [CrossRef] [PubMed]
- Malumbres, M.; Barbacid, M. Cell cycle, CDKs and cancer: A changing paradigm. Nat. Rev. Cancer 2009, 9, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, R.A. The retinoblastoma protein and cell cycle control. Cell 1995, 81, 323–330. [Google Scholar] [CrossRef]
- Johnson, M.R.; Wang, K.; Smith, J.B.; Heslin, M.J.; Diasio, R.B. Quantitation of dihydropyrimidine dehydrogenase expression by real-time reverse transcription polymerase chain reaction. Anal. Biochem. 2000, 278, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Lian, S.; Xia, Y.; Ung, T.T.; Khoi, P.N.; Yoon, H.J.; Lee, S.G.; Kim, K.K.; Jung, Y.D. Prostaglandin E2 stimulates urokinase-type plasminogen activator receptor via EP2 receptor-dependent signaling pathways in human AGS gastric cancer cells. Mol. Carcinog. 2016. [Google Scholar] [CrossRef] [PubMed]






© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lian, S.; Park, J.S.; Xia, Y.; Nguyen, T.T.; Joo, Y.E.; Kim, K.K.; Kim, H.K.; Jung, Y.D. MicroRNA-375 Functions as a Tumor-Suppressor Gene in Gastric Cancer by Targeting Recepteur d’Origine Nantais. Int. J. Mol. Sci. 2016, 17, 1633. https://doi.org/10.3390/ijms17101633
Lian S, Park JS, Xia Y, Nguyen TT, Joo YE, Kim KK, Kim HK, Jung YD. MicroRNA-375 Functions as a Tumor-Suppressor Gene in Gastric Cancer by Targeting Recepteur d’Origine Nantais. International Journal of Molecular Sciences. 2016; 17(10):1633. https://doi.org/10.3390/ijms17101633
Chicago/Turabian StyleLian, Sen, Jung Sun Park, Yong Xia, Thi Thinh Nguyen, Young Eun Joo, Kyung Keun Kim, Hark Kyun Kim, and Young Do Jung. 2016. "MicroRNA-375 Functions as a Tumor-Suppressor Gene in Gastric Cancer by Targeting Recepteur d’Origine Nantais" International Journal of Molecular Sciences 17, no. 10: 1633. https://doi.org/10.3390/ijms17101633
APA StyleLian, S., Park, J. S., Xia, Y., Nguyen, T. T., Joo, Y. E., Kim, K. K., Kim, H. K., & Jung, Y. D. (2016). MicroRNA-375 Functions as a Tumor-Suppressor Gene in Gastric Cancer by Targeting Recepteur d’Origine Nantais. International Journal of Molecular Sciences, 17(10), 1633. https://doi.org/10.3390/ijms17101633