Different Susceptibilities between Apoe- and Ldlr-Deficient Mice to Inflammation-Associated Colorectal Carcinogenesis
Abstract
:1. Introduction
2. Results
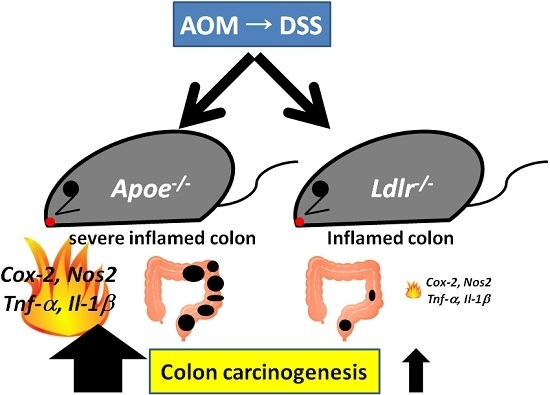
2.1. AOM/DSS-Induced Colorectal Carcinogenesis in the Apoe-Deficient Mice (Experiment 1)
2.2. AOM/DSS-Induced Colorectal Carcinogenesis in the Ldlr-Deficient Mice (Experiment 2)
3. Discussion
4. Materials and Methods
4.1. Animals, Chemicals, and Diets
4.2. Study Design
4.3. Real-Time Quantitative Polymerase Chain Reaction (RT-PCR)
4.4. Clinical Chemistry (Serum Lipid Profiles)
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Colorectal carcinogenesis: Review of human and experimental animal studies. J. Carcinog. 2009, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.S.; Tanaka, T. Interactions of selenium deficiency, vitamin E, polyunsaturated fat, and saturated fat on azoxymethane-induced colon carcinogenesis in male F344 rats. J. Natl. Cancer Inst. 1986, 76, 1157–1162. [Google Scholar] [PubMed]
- Reddy, B.S.; Tanaka, T.; Simi, B. Effect of different levels of dietary trans fat or corn oil on azoxymethane-induced colon carcinogenesis in F344 rats. J. Natl. Cancer Inst. 1985, 75, 791–798. [Google Scholar] [PubMed]
- Shimizu, M.; Kubota, M.; Tanaka, T.; Moriwaki, H. Nutraceutical approach for preventing obesity-related colorectal and liver carcinogenesis. Int. J. Mol. Sci. 2012, 13, 579–595. [Google Scholar] [CrossRef] [PubMed]
- Shirakami, Y.; Shimizu, M.; Kubota, M.; Araki, H.; Tanaka, T.; Moriwaki, H.; Seishima, M. Chemoprevention of colorectal cancer by targeting obesity-related metabolic abnormalities. World J. Gastroenterol. 2014, 20, 8939–8946. [Google Scholar] [PubMed]
- Emmons, K.M.; McBride, C.M.; Puleo, E.; Pollak, K.I.; Marcus, B.H.; Napolitano, M.; Clipp, E.; Onken, J.; Farraye, F.A.; Fletcher, R. Prevalence and predictors of multiple behavioral risk factors for colon cancer. Prev. Med. 2005, 40, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, P.; Jenab, M.; Norat, T.; Moskal, A.; Slimani, N.; Olsen, A.; Tjonneland, A.; Overvad, K.; Jensen, M.K.; Boutron-Ruault, M.C.; et al. Lifetime and baseline alcohol intake and risk of colon and rectal cancers in the European prospective investigation into cancer and nutrition (EPIC). Int. J. Cancer 2007, 121, 2065–2072. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.S. Dietary fat and colon cancer: Animal model studies. Lipids 1992, 27, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Wark, P.A.; van der Kuil, W.; Ploemacher, J.; van Muijen, G.N.; Mulder, C.J.; Weijenberg, M.P.; Kok, F.J.; Kampman, E. Diet, lifestyle and risk of K-ras mutation-positive and -negative colorectal adenomas. Int. J. Cancer 2006, 119, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Radisauskas, R.; Kuzmickiene, I.; Milinaviciene, E.; Everatt, R. Hypertension, serum lipids and cancer risk: A review of epidemiological evidence. Medicina 2016, 52, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Cosnes, J.; Gower-Rousseau, C.; Seksik, P.; Cortot, A. Epidemiology and natural history of inflammatory bowel diseases. Gastroenterology 2011, 140, 1785–1794. [Google Scholar] [CrossRef] [PubMed]
- Molodecky, N.A.; Soon, I.S.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W.; et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012, 142. [Google Scholar] [CrossRef] [PubMed]
- Strober, W.; Fuss, I.; Mannon, P. The fundamental basis of inflammatory bowel disease. J. Clin. Investig. 2007, 117, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Inflammation and cancer. In Cancer: Disease Progression and Chemoprevention; Tanaka, T., Ed.; Research Signpost: Trivandrum, Kerala, India, 2007; pp. 27–44. [Google Scholar]
- Tanaka, T.; Kohno, H.; Suzuki, R.; Hata, K.; Sugie, S.; Niho, N.; Sakano, K.; Takahashi, M.; Wakabayashi, K. Dextran sodium sulfate strongly promotes colorectal carcinogenesis in ApcMin/+ mice: Inflammatory stimuli by dextran sodium sulfate results in development of multiple colonic neoplasms. Int. J. Cancer 2006, 118, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Breslow, J.L. Genetic basis of lipoprotein disorders. J. Clin. Investig. 1989, 84, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Dammerman, M.; Breslow, J.L. Genetic basis of lipoprotein disorders. Circulation 1995, 91, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Herz, J.; Willnow, T.E. Lipoprotein and receptor interactions in vivo. Curr. Opin. Lipidol. 1995, 6, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Turley, S.D.; Spady, D.K.; Dietschy, J.M. Role of liver in the synthesis of cholesterol and the clearance of low density lipoproteins in the cynomolgus monkey. J. Lipid Res. 1995, 36, 67–79. [Google Scholar] [PubMed]
- Fong, L.G.; Fujishima, S.E.; Komaromy, M.C.; Pak, Y.K.; Ellsworth, J.L.; Cooper, A.D. Location and regulation of low-density lipoprotein receptors in intestinal epithelium. Am. J. Physiol. 1995, 269, G60–G72. [Google Scholar] [PubMed]
- Soued, M.; Mansbach, C.M., Jr. Chylomicron remnant uptake by enterocytes is receptor dependent. Am. J. Physiol. 1996, 270, G203–G212. [Google Scholar] [PubMed]
- Bertomeu, A.; Ros, E.; Zambon, D.; Vela, M.; Perez-Ayuso, R.M.; Targarona, E.; Trias, M.; Sanllehy, C.; Casals, E.; Ribo, J.M. Apolipoprotein E polymorphism and gallstones. Gastroenterology 1996, 111, 1603–1610. [Google Scholar] [CrossRef]
- Juvonen, T.; Kervinen, K.; Kairaluoma, M.I.; Lajunen, L.H.; Kesaniemi, Y.A. Gallstone cholesterol content is related to apolipoprotein E polymorphism. Gastroenterology 1993, 104, 1806–1813. [Google Scholar] [CrossRef]
- Katan, M.B. Apolipoprotein E isoforms, serum cholesterol, and cancer. Lancet 1986, 1, 507–508. [Google Scholar] [CrossRef]
- Van Erpecum, K.J.; Carey, M.C. Apolipoprotein E4: Another risk factor for cholesterol gallstone formation? Gastroenterology 1996, 111, 1764–1767. [Google Scholar] [CrossRef]
- Van Erpecum, K.J.; van Berge-henegouwen, G.P.; Eckhardt, E.R.; Portincasa, P.; van de Heijning, B.J.; Dallinga-Thie, G.M.; Groen, A.K. Cholesterol crystallization in human gallbladder bile: Relation to gallstone number, bile composition, and apolipoprotein E4 isoform. Hepatology 1998, 27, 1508–1516. [Google Scholar] [CrossRef] [PubMed]
- Berkel, J.; Hombergen, D.A.; Hooymayers, I.E.; Faber, J.A. Cholecystectomy and colon cancer. Am. J. Gastroenterol. 1990, 85, 61–64. [Google Scholar] [PubMed]
- Bach-Ngohou, K.; Ouguerram, K.; Nazih, H.; Maugere, P.; Ripolles-Piquer, B.; Zair, Y.; Frenais, R.; Krempf, M.; Bard, J.M. Apolipoprotein E kinetics: Influence of insulin resistance and type 2 diabetes. Int. J. Obes. Relat. Metab. Disord. 2002, 26, 1451–1458. [Google Scholar] [CrossRef] [PubMed]
- Orchard, T.J.; Eichner, J.; Kuller, L.H.; Becker, D.J.; McCallum, L.M.; Grandits, G.A. Insulin as a predictor of coronary heart disease: Interaction with apolipoprotein E phenotype A report from the Multiple Risk Factor Intervention Trial. Ann. Epidemiol. 1994, 4, 40–45. [Google Scholar] [CrossRef]
- Giovannucci, E. Insulin and colon cancer. Cancer Causes Control 1995, 6, 164–179. [Google Scholar] [CrossRef] [PubMed]
- Kaaks, R. Nutrition, energy balance and colon cancer risk: The role of insulin and insulin-like growth factor-I. IARC Sci. Publ. 2002, 156, 289–293. [Google Scholar] [PubMed]
- Judson, R.; Brain, C.; Dain, B.; Windemuth, A.; Ruano, G.; Reed, C. New and confirmatory evidence of an association between ApoE genotype and baseline C-reactive protein in dyslipidemic individuals. Atherosclerosis 2004, 177, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Peng, D.Q.; Zhao, S.P.; Nie, S.; Li, J. Gene–gene interaction of PPARγ and ApoE affects coronary heart disease risk. Int. J. Cardiol. 2003, 92, 257–263. [Google Scholar] [CrossRef]
- Erlinger, T.P.; Platz, E.A.; Rifai, N.; Helzlsouer, K.J. C-reactive protein and the risk of incident colorectal cancer. JAMA 2004, 291, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. Diet, body weight, and colorectal cancer: A summary of the epidemiologic evidence. J. Womens Health 2003, 12, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Awad, A.B.; Fink, C.S.; Horvath, P.J. Alteration of membrane fatty acid composition and inositol phosphate metabolism in HT-29 human colon cancer cells. Nutr. Cancer 1993, 19, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, M.L.; Neoptolemos, J.P.; Clayton, H.A.; Talbot, I.C.; Bell, P.R. Increased cell membrane arachidonic acid in experimental colorectal tumours. Gut 1991, 32, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Habenicht, A.J.; Salbach, P.; Goerig, M.; Zeh, W.; Janssen-Timmen, U.; Blattner, C.; King, W.C.; Glomset, J.A. The LDL receptor pathway delivers arachidonic acid for eicosanoid formation in cells stimulated by platelet-derived growth factor. Nature 1990, 345, 634–636. [Google Scholar] [CrossRef] [PubMed]
- Habenicht, A.J.; Salbach, P.; Janssen-Timmen, U. LDL receptor-dependent polyunsaturated fatty acid transport and metabolism. Eicosanoids 1992, 5, S29–S31. [Google Scholar] [PubMed]
- Eberhart, C.E.; Coffey, R.J.; Radhika, A.; Giardiello, F.M.; Ferrenbach, S.; DuBois, R.N. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 1994, 107, 1183–1188. [Google Scholar] [CrossRef]
- Kargman, S.L.; O’Neill, G.P.; Vickers, P.J.; Evans, J.F.; Mancini, J.A.; Jothy, S. Expression of prostaglandin G/H synthase-1 and -2 protein in human colon cancer. Cancer Res. 1995, 55, 2556–2559. [Google Scholar] [PubMed]
- Thun, M.J.; Namboodiri, M.M.; Heath, C.W., Jr. Aspirin use and reduced risk of fatal colon cancer. N. Engl. J. Med. 1991, 325, 1593–1596. [Google Scholar] [CrossRef] [PubMed]
- Tjandrawinata, R.R.; Dahiya, R.; Hughes-Fulford, M. Induction of cyclo-oxygenase-2 mRNA by prostaglandin E2 in human prostatic carcinoma cells. Br. J. Cancer 1997, 75, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Tjandrawinata, R.R.; Hughes-Fulford, M. Up-regulation of cyclooxygenase-2 by product-prostaglandin E2. Adv. Exp. Med. Biol. 1997, 407, 163–170. [Google Scholar] [PubMed]
- Caruso, M.G.; Notarnicola, M.; Cavallini, A.; Guerra, V.; Misciagna, G.; di Leo, A. Demonstration of low density lipoprotein receptor in human colonic carcinoma and surrounding mucosa by immunoenzymatic assay. Ital. J. Gastroenterol. 1993, 25, 361–367. [Google Scholar] [PubMed]
- Gueddari, N.; Favre, G.; Hachem, H.; Marek, E.; Le Gaillard, F.; Soula, G. Evidence for up-regulated low density lipoprotein receptor in human lung adenocarcinoma cell line A549. Biochimie 1993, 75, 811–819. [Google Scholar] [CrossRef]
- Lum, D.F.; McQuaid, K.R.; Gilbertson, V.L.; Hughes-Fulford, M. Coordinate up-regulation of low-density lipoprotein receptor and cyclo-oxygenase-2 gene expression in human colorectal cells and in colorectal adenocarcinoma biopsies. Int. J. Cancer 1999, 83, 162–166. [Google Scholar] [CrossRef]
- Tanaka, T.; Kohno, H.; Suzuki, R.; Yamada, Y.; Sugie, S.; Mori, H. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 2003, 94, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Jawien, J. The role of an experimental model of atherosclerosis: ApoE-knockout mice in developing new drugs against atherogenesis. Curr. Pharm. Biotechnol. 2012, 13, 2435–2439. [Google Scholar] [CrossRef] [PubMed]
- Kolovou, G.; Anagnostopoulou, K.; Mikhailidis, D.P.; Cokkinos, D.V. Apolipoprotein E knockout models. Curr. Pharm. Des. 2008, 14, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Hagberg, J.M.; Wilund, K.R.; Ferrell, R.E. APO E gene and gene-environment effects on plasma lipoprotein-lipid levels. Physiol. Genom. 2000, 4, 101–108. [Google Scholar]
- Cedazo-Minguez, A.; Popescu, B.O.; Blanco-Millan, J.M.; Akterin, S.; Pei, J.J.; Winblad, B.; Cowburn, R.F. Apolipoprotein E and β-amyloid (1–42) regulation of glycogen synthase kinase-3β. J. Neurochem. 2003, 87, 1152–1164. [Google Scholar] [CrossRef] [PubMed]
- Grocott, H.P.; Newman, M.F.; El-Moalem, H.; Bainbridge, D.; Butler, A.; Laskowitz, D.T. Apolipoprotein E genotype differentially influences the proinflammatory and anti-inflammatory response to cardiopulmonary bypass. J. Thorac. Cardiovasc. Surg. 2001, 122, 622–623. [Google Scholar] [CrossRef] [PubMed]
- Niemi, M.; Hakkinen, T.; Karttunen, T.J.; Eskelinen, S.; Kervinen, K.; Savolainen, M.J.; Lehtola, J.; Makela, J.; Yla-Herttuala, S.; Kesaniemi, Y.A. Apolipoprotein E and colon cancer. Expression in normal and malignant human intestine and effect on cultured human colonic adenocarcinoma cells. Eur. J. Intern. Med. 2002, 13, 37–43. [Google Scholar] [CrossRef]
- Vogel, T.; Guo, N.H.; Guy, R.; Drezlich, N.; Krutzsch, H.C.; Blake, D.A.; Panet, A.; Roberts, D.D. Apolipoprotein E: A potent inhibitor of endothelial and tumor cell proliferation. J. Cell. Biochem. 1994, 54, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Behrens, J.; von Kries, J.P.; Kuhl, M.; Bruhn, L.; Wedlich, D.; Grosschedl, R.; Birchmeier, W. Functional interaction of β-catenin with the transcription factor LEF-1. Nature 1996, 382, 638–642. [Google Scholar] [CrossRef] [PubMed]
- Molenaar, M.; van de Wetering, M.; Oosterwegel, M.; Peterson-Maduro, J.; Godsave, S.; Korinek, V.; Roose, J.; Destree, O.; Clevers, H. XTcf-3 transcription factor mediates beta-catenin-induced axis formation in Xenopus embryos. Cell 1996, 86, 391–399. [Google Scholar] [CrossRef]
- Pap, M.; Cooper, G.M. Role of glycogen synthase kinase-3 in the phosphatidylinositol 3-Kinase/Akt cell survival pathway. J. Biol. Chem. 1998, 273, 19929–19932. [Google Scholar] [CrossRef] [PubMed]
- Ripolles Piquer, B.; Nazih, H.; Bourreille, A.; Segain, J.P.; Huvelin, J.M.; Galmiche, J.P.; Bard, J.M. Altered lipid, apolipoprotein, and lipoprotein profiles in inflammatory bowel disease: Consequences on the cholesterol efflux capacity of serum using Fu5AH cell system. Metabolism 2006, 55, 980–988. [Google Scholar] [CrossRef] [PubMed]
- De Bont, N.; Netea, M.G.; Demacker, P.N.; Verschueren, I.; Kullberg, B.J.; van Dijk, K.W.; van der Meer, J.W.; Stalenhoef, A.F. Apolipoprotein E knock-out mice are highly susceptible to endotoxemia and Klebsiella pneumoniae infection. J. Lipid Res. 1999, 40, 680–685. [Google Scholar] [CrossRef]
- Netea, M.G.; Demacker, P.N.; Kullberg, B.J.; Boerman, O.C.; Verschueren, I.; Stalenhoef, A.F.; van der Meer, J.W. Low-density lipoprotein receptor-deficient mice are protected against lethal endotoxemia and severe gram-negative infections. J. Clin. Investig. 1996, 97, 1366–1372. [Google Scholar] [CrossRef] [PubMed]
- Laskowitz, D.T.; Lee, D.M.; Schmechel, D.; Staats, H.F. Altered immune responses in apolipoprotein E-deficient mice. J. Lipid Res. 2000, 41, 613–620. [Google Scholar] [PubMed]
- Van Oosten, M.; Rensen, P.C.; van Amersfoort, E.S.; van Eck, M.; van Dam, A.M.; Breve, J.J.; Vogel, T.; Panet, A.; van Berkel, T.J.; Kuiper, J. Apolipoprotein E protects against bacterial lipopolysaccharide-induced lethality. A new therapeutic approach to treat gram-negative sepsis. J. Biol. Chem. 2001, 276, 8820–8824. [Google Scholar] [CrossRef] [PubMed]
- Waterman, M.; Xu, W.; Stempak, J.M.; Milgrom, R.; Bernstein, C.N.; Griffiths, A.M.; Greenberg, G.R.; Steinhart, A.H.; Silverberg, M.S. Distinct and overlapping genetic loci in Crohn’s disease and ulcerative colitis: Correlations with pathogenesis. Inflamm. Bowel Dis. 2011, 17, 1936–1942. [Google Scholar] [CrossRef] [PubMed]
- Baitsch, D.; Bock, H.H.; Engel, T.; Telgmann, R.; Muller-Tidow, C.; Varga, G.; Bot, M.; Herz, J.; Robenek, H.; von Eckardstein, A.; et al. Apolipoprotein E induces antiinflammatory phenotype in macrophages. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1160–1168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, L.M.; Wu, J. Cross-talk between apolipoprotein E and cytokines. Mediat. Inflamm. 2011, 2011, 949072. [Google Scholar] [CrossRef] [PubMed]
- Postigo, J.; Genre, F.; Iglesias, M.; Fernandez-Rey, M.; Buelta, L.; Carlos Rodriguez-Rey, J.; Merino, J.; Merino, R. Exacerbation of type II collagen-induced arthritis in apolipoprotein E-deficient mice in association with the expansion of Th1 and Th17 cells. Arthritis Rheum. 2011, 63, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Animal models of carcinogenesis in inflamed colorectum: Potential use in chemoprevention study. Curr. Drug Targets 2012, 13, 1689–1697. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kochi, T.; Shirakami, Y.; Mori, T.; Kurata, A.; Watanabe, N.; Moriwaki, H.; Shimizu, M. Cimetidine and Clobenpropit Attenuate Inflammation-Associated Colorectal Carcinogenesis in Male ICR Mice. Cancers 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Rath, T.; Billmeier, U.; Waldner, M.J.; Atreya, R.; Neurath, M.F. From physiology to disease and targeted therapy: Interleukin-6 in inflammation and inflammation-associated carcinogenesis. Arch. Toxicol. 2015, 89, 541–554. [Google Scholar] [CrossRef] [PubMed]
- Yudkin, J.S.; Kumari, M.; Humphries, S.E.; Mohamed-Ali, V. Inflammation, obesity, stress and coronary heart disease: Is interleukin-6 the link? Atherosclerosis 2000, 148, 209–214. [Google Scholar] [CrossRef]
- Bozec, D.; Iuga, A.C.; Roda, G.; Dahan, S.; Yeretssian, G. Critical function of the necroptosis adaptor RIPK3 in protecting from intestinal tumorigenesis. Oncotarget 2016. [Google Scholar] [CrossRef] [PubMed]






| Measurements | Apoe-Deficient Mice (n = 20) | WT Mice (n = 21) | Ldlr-Deficient Mice (n = 14) | WT Mice (n = 10) |
|---|---|---|---|---|
| Body weight (g) | 34.1 ± 2.2 a,b | 31.4 ± 3.5 | 30.1 ± 3.1 b | 32.6 ± 1.7 |
| Liver weight (g) | 1.31 ± 0.07 c | 1.14 ± 0.09 | 1.43 ± 0.13 d | 1.60 ± 0.22 |
| % Liver weight (Liver weight/100 g body weight) | 3.77 ± 0.33 | 3.69 ± 0.36 | 4.78 ± 0.28 | 4.91 ± 0.52 |
| Colon length (cm) | 12.35 ± 0.90 | 12.50 ± 0.90 | 10.09 ± 0.62 | 10.4 ± 0.72 |
| Pathological Lesions | Apoe-Deficient Mice (n = 20) | WT Mice (n = 21) | Ldlr-Deficient Mice (n = 14) | WT Mice (n = 10) |
|---|---|---|---|---|
| Mucosal ulcer | 1.25 ± 1.74 a | 0.57 ± 0.98 | 2.71 ± 2.13 | 3.40 ± 1.65 |
| (10/20, 50%) | (7/21, 33%) | (12/14, 86%) | (10/10, 100%) | |
| Dysplastic crypts | 1.05 ± 1.50 | 0.14 ± 0.48 | 0.43 ± 0.65 c | 4.30 ± 2.67 |
| (9/20, 45% b) | (2/21, 10%) | (5/14, 36% b) | (9/10, 90%) | |
| Adenoma (AD) | 1.00 ± 1.08 | 0.62 ± 0.74 | 0.29 ± 0.61 c | 2.10 ± 1.52 |
| (11/20, 55%) | (10/21, 48%) | (3/14, 21% b) | (8/10, 80%) | |
| Adenocarcinoma (ADC) | 2.75 ± 1.48 c | 0.62 ± 0.67 | 0.50 ± 0.94 c | 3.10 ± 2.38 |
| (19/20, 95% d) | (11/21, 52%) | (4/14, 29% e) | (8/10, 80%) | |
| AD + ADC | 3.75 ± −1.83 c | 1.24 ± 0.94 | 0.79 ± 1.31 c | 5.20 ± 3.12 |
| (20/20, 100%) | (17/21, 81%) | (5/14, 36% e) | (8/10, 80%) |
| Measurements | Apoe-Deficient Mice (n = 21) | WT Mice (n = 20) | Ldlr-Deficient Mice (n = 14) | WT Mice (n = 10) |
|---|---|---|---|---|
| Serum cholesterol (mg/dL) | 628 ± 110 a,b | 132 ± 23 | 414 ± 41 b | 131 ± 21 |
| Serum triglycerides (mg/dL) | 106 ± 16 | 85 ± 28 | 230 ± 51 b | 126 ± 21 |
| Serum VLDL (mg/dL) | 403 ± 99 b | 110 ± 17 | 149 ± 4 | 120 ± 4 |
| Serum LDL (mg/dL) | 439 ± 170 b | 49 ± 12 | 287 ± 11 b | 42 ± 5 |
| Serum HDL (mg/dL) | 36 ± 32 | 59 ± 16 | 81 ± 3 | 63 ± 35 |
| Serum glucose (mg/dL) | 147 ± 15 | 175 ± 25 | 181 ± 39 | 184 ± 14 |
| Serum adiponectin (µg/mL) | 12.9 ± 2.3 | 12.0 ± 1.7 | 16.6 ± 2.1 b | 12.1 ± 3.5 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanaka, T.; Oyama, T.; Sugie, S.; Shimizu, M. Different Susceptibilities between Apoe- and Ldlr-Deficient Mice to Inflammation-Associated Colorectal Carcinogenesis. Int. J. Mol. Sci. 2016, 17, 1806. https://doi.org/10.3390/ijms17111806
Tanaka T, Oyama T, Sugie S, Shimizu M. Different Susceptibilities between Apoe- and Ldlr-Deficient Mice to Inflammation-Associated Colorectal Carcinogenesis. International Journal of Molecular Sciences. 2016; 17(11):1806. https://doi.org/10.3390/ijms17111806
Chicago/Turabian StyleTanaka, Takuji, Takeru Oyama, Shigeyuki Sugie, and Masahito Shimizu. 2016. "Different Susceptibilities between Apoe- and Ldlr-Deficient Mice to Inflammation-Associated Colorectal Carcinogenesis" International Journal of Molecular Sciences 17, no. 11: 1806. https://doi.org/10.3390/ijms17111806