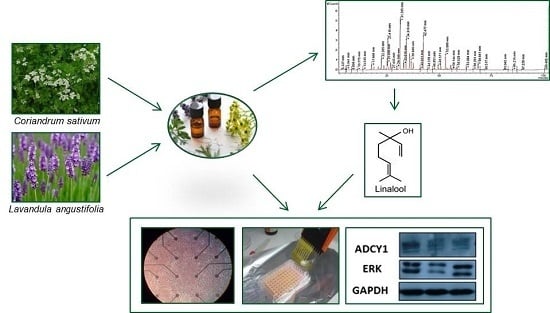
Coriandrum sativum and Lavandula angustifolia Essential Oils: Chemical Composition and Activity on Central Nervous System
Abstract
:1. Introduction
2. Results
2.1. Essential Oil Yields and Composition
2.2. Cytotoxicity of Linalool, L. angustifolia and C. sativum Essential Oils
2.3. ADCY1, ERK: Western Blot Analysis
2.4. Effects on Neuronal Activity
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Isolation of the Volatile Oil
4.3. GC-FID Analysis
4.4. GC/MS Analysis
4.5. Identification of the Essential Oil Components
4.6. Human Neuroblastoma Cell Cultures
4.7. Primary Neuron Cultures
4.8. MTT Bioassay
4.9. Extraction Proteins and Western Blotting
4.10. Data Recordings, Signal Processing and Data Analysis
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| ADCY1 | Adenylyl Cyclase 1 |
| CNS | Central Nervous System |
| ERK | Extracellular signal-Regulated Kinase |
| MEA | Multi Electrode Array |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide |
| OD | Optical Density |
References
- Dobetsberger, C.; Buchbauer, G. Actions of essential oils on the central nervous system: An updated review. Flavour Frag. J. 2011, 26, 300–316. [Google Scholar] [CrossRef]
- Babar, A.; Al-Wabel, N.A.; Shams, S.; Ahmad, A.; Khan, S.A.; Anwar, F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar]
- Nunes, D.S.; Linck, V.M.; Silva, A.L.; Figueiró, M.; Elisabetsky, E. Psychopharmacology of Essential Oils. In Handbook of Essential Oils: Science, Technology, and Applications; Baser, H.C., Buchbauer, G., Eds.; CRC Press: London, UK, 2015; pp. 297–314. [Google Scholar]
- Adorjan, B.; Buchbauer, G. Biological properties of essential oils: An updated review. Flavour Frag. J. 2010, 25, 407–426. [Google Scholar] [CrossRef]
- Mandal, S.; Mandal, M. Coriander (Coriandrum sativum L.) essential oil: Chemistry and biological activity. Asian Pac. J. Trop. Biomed. 2015, 5, 421–428. [Google Scholar] [CrossRef]
- Nadeem, M.; Anjum, F.M.; Khan, M.I.; Tehseen, S.; El-Ghora, A.; Sultan, J.I. Nutritional and medicinal aspects of coriander (Coriandrum sativum L.) A review. Br. Food J. 2013, 115, 743–755. [Google Scholar] [CrossRef]
- Price, S. The Aromatherapy Workbook; Thorsons: London, UK, 1993. [Google Scholar]
- Koulivand, P.H.; Ghadiri, M.K.; Gorji, A. Lavender and the nervous system. Evid. Based Complement. Altern. Med. 2013, 2012, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Mimaki, Y.; Sashida, Y. Anticonvulsive effects of inhaling lavender oil vapour. Biol. Pharm. 1994, 17, 359–360. [Google Scholar] [CrossRef]
- Elisabetsky, E.; Marschner, J.; Souza, D.O. Effects of linalool on glutamatergic system in the rat cerebral cortex. Neurochem. Res. 1995, 20, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Elisabetsky, E.; Silva Brum, L.F.; Souza, D.O. Anticonvulsant properties of linalool in glutamate-related seizure models. Phytomedicine 1999, 6, 107–113. [Google Scholar] [CrossRef]
- Emamghorashi, M.; Heidari-Hamedani, G. Sedative-hypnotic activity of extracts and essential oil of Coriander seeds. Iran. J. Med. Sci. 2006, 31, 22–27. [Google Scholar]
- Sunil, C.; Agastian, P.; Kumarappan, C.; Ignacimuthu, S. In vitro antioxidant, antidiabetic and antilipidemic activities of Symplocos cochinchinensis (Lour.) S. Moore bark. Food Chem. Toxicol. 2012, 50, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, S.N.; Hadian, J.; Ranjbar, H. Essential oil compositions of different accessions of Coriandrum sativum L. from Iran. Nat. Prod. Res. 2010, 24, 1287–1294. [Google Scholar] [CrossRef] [PubMed]
- Souto-Maior, F.M.; Lélis de Carvalho, F.; Soares Lima de Morais, L.S.; Mendonça Netto, S.; Pergentino de Sousa, D.; Nóbrega de Almeida, R. Anxiolytic-like effects of inhaled linalool oxide in experimental mouse anxiety models. Pharmacol. Biochem. Behav. 2011, 100, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Coelho, V.R.; Gianesini, J.; Von Borowski, R.; Mazzardo-Martins, L.; Martins, D.F.; Picada, J.N.; Santos, A.R.; Brum, L.F.; Pereira, P. (−)-Linalool, a naturally occurring monoterpene compound, impairs memory acquisition in the object recognition task, inhibitory avoidance test and habituation to a novel environment in rats. Phytomedicine 2011, 18, 896–901. [Google Scholar] [CrossRef] [PubMed]
- Re, L.; Barocci, S.; Sonnino, S.; Mencarelli, A.; Vivani, C.; Paolucci, G.; Scarpantonio, A.; Rinaldi, L.; Mosca, E. Linalool modifies the nicotinic receptor–ion channel kinetics at the mouse neuromuscular junction. Pharmacol. Res. 2000, 42, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Linck, V.M.; da Silva, A.L.; Figueiró, M.; Piato, A.L.; Herrmann, A.P.; Dupont Birck, F.; Caramão, E.B.; Nunes, D.S.; Moreno, P.R.H.; Elisabetsky, E. Inhaled linalool-induced sedation in mice. Phytomedicine 2009, 16, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Linck, V.M.; da Silva, A.L.; Figueiró, M.; Caramão, E.B.; Moreno, P.R.H.; Elisabetsky, E. Effects of inhaled Linalool in anxiety, social interaction and aggressive behavior in mice. Phytomedicine 2010, 17, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, L.F.S.; Maia, J.G.S.; de Parijós, A.M.; Souza, L.R.Z.; Barata, L.E.S. Linalool from rosewood (Aniba rosaeodora Ducke) oil inhibits adenylate cyclase in the retina, contributing to understanding its biological activity. Phytother. Res. 2012, 26, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Stork, P.J.; Schmitt, J.M. Crosstalk between cAMP and MAP kinase signaling in the regulation of cell proliferation. Trends Cell Biol. 2002, 12, 258–266. [Google Scholar] [CrossRef]
- Grewal, S.S.; York, R.D.; Stork, P.J. Extracellular-signal-regulated kinase signalling in neurons. Curr. Opin. Neurobiol. 1999, 9, 544–553. [Google Scholar] [CrossRef]
- Cavanagh, H.M.A.; Wilkinson, J.M. Biological activities of lavender essential oil. Phytother. Res. 2002, 16, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Woronuk, G.; Demissie, Z.; Rheault, M.; Mahmoud, S. Biosynthesis and therapeutic properties of Lavandula essential oil constituents. Planta Med. 2011, 77, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.S.; Rahman, L.U.; Chanotiya, C.S.; Verma, R.K.; Chauhan, A.; Yadav, A.; Singh, A.; Yadav, A.K. Essential oil composition of Lavandula angustifolia Mill. cultivated in the mid hills of Uttarakhand, India. J. Serb. Chem. Soc. 2010, 75, 343–348. [Google Scholar] [CrossRef]
- Khani, A.; Rahdari, T. Chemical composition and insecticidal activity of essential oil from Coriandrum sativum seeds against Tribolium confusum and Callosobruchus maculatus. ISRN Pharm. 2012, 2012, 1–5. [Google Scholar]
- Shahwar, M.K.; El-Ghorab, A.H.; Anjum, F.M.; Butt, M.S.; Hussain, S.; Nadeem, M. Characterization of coriander (Coriandrum sativum L.) seeds and leaves: Volatile and non-volatile extracts. Int. J. Food Prop. 2012, 15, 736–747. [Google Scholar] [CrossRef]
- Misharina, T.A. Influence of the duration and conditions of storage on the composition of the essential oil from coriander seeds. Appl. Biochem. Microbiol. 2001, 37, 622–628. [Google Scholar] [CrossRef]
- Smallfield, B.M.; van Klink, J.W.; Perry, N.B.; Dodds, K.G. Coriander spice oil: Effects of fruit crushing and distillation time on yield and composition. J. Agric. Food Chem. 2001, 49, 118–123. [Google Scholar] [CrossRef]
- Gil, A.; De La Fuente, E.; Lenardis, A.E.; Pereira, M.N.L.P.; Suarez, S.A.; Bandoni, A.; van Baren, C.; Di Leo Lira, P.; Ghersa, C.M. Coriander essential oil composition from two genotypes grown in different environmental conditions. J. Agric. Food Chem. 2002, 50, 2870–2877. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Alok, U.; Khanna, A. In vitro cytotoxicity of essential oils: A review. Int. J. Res. Pharm. Chem. 2013, 3, 675–681. [Google Scholar]
- Ravizza, R.; Gariboldi, M.B.; Molteni, R.; Monti, E. Linalool, a plant-derived monoterpene alcohol, reverses doxorubicin resistance in human breast adenocarcinoma cells. Oncol. Rep. 2008, 20, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.B.; Wang, S.M.; Li, T.; Yang, Y.Q. Anticancer Activity of Linalool Terpenoid: Apoptosis Induction and Cell Cycle Arrest in Prostate Cancer Cells. Trop. J. Pharm. Res. 2015, 14, 619–625. [Google Scholar] [CrossRef]
- Prashar, A.; Locke, J.C.; Evans, C.S. Cytotoxicity of lavender oil and its major components to human skin cells. Cell Prolif. 2004, 37, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Imelouane, B.; Elbachiri, A.; Whatelet, J.P.; Dubois, J.; Amhamdi, H. Chemical Composition, Cytotoxic and Antioxydant Activity of The Essential Oil of Lavandula dentata. World J. Chem. 2010, 5, 103–110. [Google Scholar]
- Geran, R.I.; Greenberg, N.H.; Macdonald, M.M.; Schumacher, A.M.; Abbott, B.J. Protocols for screening chemical agents and natural products against animal tumours and other biological systems. Cancer Chemother. Rep. 1972, 3, 59–61. [Google Scholar]
- Impey, S.; Obrietan, K.; Storm, D.R. Making new connections: Role of ERK/MAP kinase signaling in neuronal plasticity. Neuron 1999, 23, 11–14. [Google Scholar] [CrossRef]
- Davis, M.I.; Ronesi, J.; Lovinger, D.M. A predominant role for inhibition of the adenylate cyclase/protein kinase A pathway in ERK activation by cannabinoid receptor 1 in N1E-115 neuroblastoma cells. J. Biol. Chem. 2003, 278, 48973–48980. [Google Scholar] [CrossRef] [PubMed]
- Impey, S.; Obrietan, K.; Wong, S.T.; Poser, S.; Yano, S.; Wayman, G.; Storm, D.R. Cross talk between ERK and PKA is required for Ca2+ stimulation of CREB-dependent transcription and ERK nuclear translocation. Neuron 1998, 21, 869–883. [Google Scholar] [CrossRef]
- Council of Europe. European Pharmacopeia, 5th ed.; Council of Europe: Strasbourg Cedex, France, 2004; Volume I, pp. 217–218. [Google Scholar]
- Jennings, W.; Shibamoto, T. Qualitative Analysis of Flavour and Fragrance Volatiles by Glass Capillary Gas Chromatography; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Davies, N.W. Gas chromatographic retention indices of monoterpenes and sesquiterpenes on methyl silicone and Carbowax 20M phases. J. Chromatogr. 1990, 503, 1–24. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Goodner, K.L. Practical retention index models of OV-101, DB-1, DB-5, and DB-Wax for flavor and fragrance compounds. LWT Food Sci. Technol. 2008, 41, 951–958. [Google Scholar] [CrossRef]
- Wiley. The Wiley Registry of Mass Spectral Data, with NIST Spectral Data CD Rom, 7th ed.; John Wiley & Sons: New York, NY, USA, 1998. [Google Scholar]
- Novellino, A.; Scelfo, B.; Palosaari, T.; Price, A.; Sobanski, T.; Shafer, T.J.; Johnstone, A.F.M.; Gross, G.W.; Gramowski, A.; Schroeder, O.; et al. Development of micro-electrode array based tests for neurotoxicity: Assessment of interlaboratory reproducibility with neuroactive chemicals. Front. Neuroeng. 2011, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hogberg, H.T.; Sobanski, T.; Novellino, A.; Whelan, M.; Weiss, D.G.; Bal-Price, A.K. Application of micro-electrode arrays (MEAs) as an emerging technology for developmental neurotoxicity: Evaluation of domoic acid-induced effects in primary cultures of rat cortical neurons. Neurotoxicology 2011, 32, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Picerno, P.; Autore, G.; Marzocco, S.; Meloni, M.; Sanogo, R.; Aquino, R.P. Anti-inflammatory activity of verminoside from Kigelia africana and evaluation of cutaneous irritation in cell cultures and reconstituted human epidermis. J. Nat. Prod. 2005, 68, 1610–1614. [Google Scholar] [CrossRef] [PubMed]
- Petrella, A.; Ercolino, S.F.; Festa, M.; Gentilella, A.; Tosco, A.; Conzen, S.D.; Parente, L. Dexamethasone inhibits TRAIL-induced apoptosis of thyroid cancer cells via Bcl-xL induction. Eur. J. Cancer 2006, 42, 3287–3293. [Google Scholar] [CrossRef] [PubMed]




| No. | Compound | L | C | Ki a | Ki b | Identification c |
|---|---|---|---|---|---|---|
| 1 | α-Thujene | - | 0.1 | 909 | 929 | 1,3 |
| 2 | α-Pinene | 0.8 | 5.0 | 922 | 939 | 1,2 |
| 3 | Camphene | 0.6 | 1.0 | 935 | 954 | 1,2 |
| 4 | Thuja-2,4(10)-diene | 0.1 | - | 957 | 960 | 1,2 |
| 5 | Sabinene | - | 0.7 | 961 | 975 | 1,2 |
| 6 | β-Pinene | 0.9 | 0.8 | 980 | 979 | 1,3 |
| 7 | Myrcene | 1.9 | 0.1 | 985 | 990 | 1,2 |
| 8 | α-Phellandrene | 0.2 | - | 991 | 1002 | 1,2 |
| 9 | δ-3-Carene | 0.9 | 0.1 | 1000 | 1008 | 1,2 |
| 10 | α-Terpinene | 0.2 | - | 1000 | 1017 | 1,3 |
| 11 | p-Cymene | 0.4 | 2.8 | 1009 | 1024 | 1,2 |
| 12 | Limonene | 2.1 | 2.6 | 1014 | 1029 | 1,2,3 |
| 13 | 1,8-Cineole | 8.0 | 0.1 | 1017 | 1031 | 1,2 |
| 14 | (Z)-β-Ocimene | 0.6 | 1.1 | 1025 | 1037 | 1,2 |
| 15 | (E)-β-Ocimene | 1.4 | 0.1 | 1036 | 1050 | 1,2 |
| 16 | γ-Terpinene | 0.3 | 2.7 | 1046 | 1059 | 1,2 |
| 17 | cis-sabinene Hydrate | 0.2 | t | 1057 | 1070 | 1,2 |
| 18 | cis-Linalool oxide | 0.1 | 0.7 | 1063 | 1072 | 1,2,3 |
| 19 | Terpinolene | 0.7 | 0.5 | 1076 | 1088 | 1,2,3 |
| 20 | Linalool | 33.1 | 67.8 | 1099 | 1096 | 1,2,3 |
| 21 | (2E)-Heptenyl acetate | 0.2 | - | 1100 | 1113 | 1,2 |
| 22 | Menth-en-1-ol | t | - | 1109 | 1121 | 1,2,3 |
| 23 | allo-Ocymene | 1.3 | - | 1115 | 1132 | 1,2 |
| 24 | trans-Pinocarveol | 0.1 | - | 1125 | 1135 | 1,2 |
| 25 | Camphor | 11.0 | 5.0 | 1135 | 1146 | 1,2,3 |
| 26 | Lavandulol | 0.1 | - | 1153 | 1169 | 1,2 |
| 27 | Borneol | 4.5 | 0.3 | 1155 | 1160 | 1,2 |
| 28 | Pinocarvone | 0.1 | - | 1165 | 1164 | 1,2 |
| 29 | neo-iso-Isopulegol | 2.3 | - | 1166 | 1171 | 1,2 |
| 30 | Terpinen-4-ol | - | 0.3 | 1167 | 1177 | 1,2 |
| 31 | cis-Linalool oxide | t | - | 1172 | 1170 | 1,2,3 |
| 32 | p-Cimen-8-ol | - | 0.1 | 1176 | 1182 | 1,2 |
| 34 | Menthol | 0.2 | - | 1177 | 1171 | 1,2 |
| 35 | α-Terpineol | 1.6 | 0.6 | 1182 | 1188 | 1,2 |
| 36 | Hexyl butanoate | 0.5 | - | 1183 | 1192 | 1,2 |
| 37 | Methyl Chavicol | - | 0.1 | 1188 | 1196 | 1,2 |
| 38 | Safranale | - | t | 1197 | 1196 | 1,2 |
| 39 | n-Decanal | - | t | 1207 | 1201 | 1,2 |
| 40 | Nerol | 0.2 | - | 1215 | 1229 | 1,2 |
| 41 | Citronellol | - | 0.3 | 1217 | 1225 | 1,2 |
| 42 | Hexyl-(2E)-butenoate | 0.3 | - | 1228 | 1242 | 1,2 |
| 43 | Neral | - | 0.1 | 1230 | 1238 | 1,2 |
| 44 | Linalyl acetate | 10.4 | - | 1247 | 1257 | 1,2 |
| 45 | Geraniol | - | 2.0 | 1248 | 1252 | 1,2 |
| 46 | Geranial | - | 0.1 | 1268 | 1267 | 1,2 |
| 47 | Iso-3-Thujanol acetate | 0.1 | - | 1275 | 1270 | 1,2 |
| 48 | Neo-3-Thujanol acetate | 0.1 | - | 1281 | 1276 | 1,2 |
| 50 | α-Terpinen-7-ale | 0.1 | - | 1283 | 1285 | 1,2 |
| 51 | Thymol | - | 0.1 | 1296 | 1290 | 1,2 |
| 52 | 10-Undecenal | - | t | 1294 | 1299 | 1,2 |
| 53 | p-Cymen-7-ol | t | - | 1297 | 1290 | 1,2 |
| 54 | Terpinyl acetate | 0.1 | - | 1315 | 1317 | 1,2 |
| 55 | Mirtenyl acetate | t | 0.2 | 1315 | 1326 | 1,2,3 |
| 56 | Neryl acetate | - | t | 1346 | 1361 | 1,2 |
| 57 | α-Terpinyl acetate | 0.8 | - | 1355 | 1349 | 1,2 |
| 58 | (E)-2-undecenal | - | 0.1 | 1359 | 1360 | 1,2 |
| 59 | α-Cubebene | 0.2 | - | 1368 | 1,2 | |
| 60 | α-Copaene | 1.4 | - | 1374 | 1376 | 1,2 |
| 61 | β-Cubebene | 0.2 | - | 1378 | 1,2 | |
| 62 | Geranyl acetate | - | 3.7 | 1382 | 1381 | 1,2 |
| 63 | Longifolene | 0.2 | - | 1394 | 1,2 | |
| 64 | (Z)-Caryophyllene | 3.3 | - | 1408 | 1408 | 1,2 |
| 65 | (E)-Caryophyllene | 0.4 | 0.1 | 1413 | 1419 | 1,2,3 |
| 66 | cis-Thujopsene | 0.3 | - | 1424 | 1431 | 1,2 |
| 67 | β-Copaene | 0.3 | - | 1433 | 1432 | 1,2 |
| 68 | α-Guaiene | 0.3 | - | 1442 | 1,2 | |
| 69 | 6,9-guaiadiene | 0.9 | - | 1445 | 1444 | 1,2 |
| 70 | allo-Aromadendrene | 0.3 | - | 1458 | 1466 | 1,2 |
| 71 | cis-Muurola-4(14,5)diene | 0.2 | - | 1468 | 1468 | 1,2 |
| 72 | β-Selinene | 0.3 | - | 1486 | 1490 | 1,2 |
| 73 | δ-Selinene | 0.5 | - | 1490 | 1492 | 1,2 |
| 74 | γ-Cadinene | 0.2 | - | 1511 | 1513 | 1,2 |
| 75 | Caryophyllene oxide | 0.4 | - | 1572 | 1583 | 1,2,3 |
| 76 | epi-α-Cadinol | 0.2 | - | 1629 | 1640 | 1,2 |
| 77 | β-Bisabolol | 1.2 | - | 1672 | 1,2 | |
| Total | 97.3 | 99.3 | ||||
| Oxygenated Monoterpene | 72.9 | 77.8 | ||||
| Monoterpene hydrocarbons | 12.6 | 17.6 | ||||
| Sesquiterpene hydrocarbons | 9.2 | 3.7 | ||||
| Oxygenated sesquiterpenes | 1.6 | - |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caputo, L.; Souza, L.F.; Alloisio, S.; Cornara, L.; De Feo, V. Coriandrum sativum and Lavandula angustifolia Essential Oils: Chemical Composition and Activity on Central Nervous System. Int. J. Mol. Sci. 2016, 17, 1999. https://doi.org/10.3390/ijms17121999
Caputo L, Souza LF, Alloisio S, Cornara L, De Feo V. Coriandrum sativum and Lavandula angustifolia Essential Oils: Chemical Composition and Activity on Central Nervous System. International Journal of Molecular Sciences. 2016; 17(12):1999. https://doi.org/10.3390/ijms17121999
Chicago/Turabian StyleCaputo, Lucia, Lucéia Fátima Souza, Susanna Alloisio, Laura Cornara, and Vincenzo De Feo. 2016. "Coriandrum sativum and Lavandula angustifolia Essential Oils: Chemical Composition and Activity on Central Nervous System" International Journal of Molecular Sciences 17, no. 12: 1999. https://doi.org/10.3390/ijms17121999
APA StyleCaputo, L., Souza, L. F., Alloisio, S., Cornara, L., & De Feo, V. (2016). Coriandrum sativum and Lavandula angustifolia Essential Oils: Chemical Composition and Activity on Central Nervous System. International Journal of Molecular Sciences, 17(12), 1999. https://doi.org/10.3390/ijms17121999