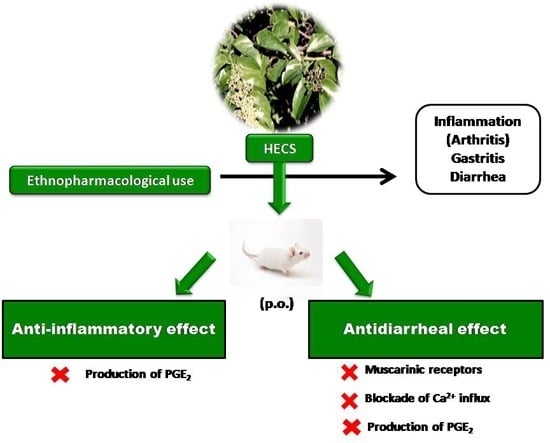
Cissus sicyoides: Pharmacological Mechanisms Involved in the Anti-Inflammatory and Antidiarrheal Activities
Abstract
:1. Introduction
2. Results and Discussion

| Sex | Treatment (p.o.) | Dose (mg/kg) | Liver | Heart | Lung | Kidney | Spleen | Deaths |
|---|---|---|---|---|---|---|---|---|
| ♂ | Vehicle | – | 13.80 ± 0.20 | 3.99 ± 0.07 | 4.30 ± 0.12 | 6.47 ± 0.12 | 3.18 ± 0.16 | 0 |
| HECS | 5000 mg/kg | 14.09 ± 0.17 | 3.97 ± 0.11 | 4.26 ± 0.18 | 6.78 ± 0.19 | 3.10 ± 0.18 | 0 | |
| ♀ | Vehicle | – | 13.90 ± 0.36 | 4.14 ± 0.08 | 5.13 ± 0.20 | 5.99 ± 0.19 | 3.38 ± 0.11 | 0 |
| HECS | 5000 mg/kg | 13.63 ± 0.16 | 4.09 ± 0.09 | 4.81 ± 0.27 | 5.81 ± 0.09 | 3.28 ± 0.13 | 0 |



| Treatment (p.o.) | Dose (mg/kg) | Time to Initial Evacuation (min) | Evacuation Classification | Inhibition (%) | ||
|---|---|---|---|---|---|---|
| Normal | Semi-Solid | Liquid | ||||
| Vehicle | – | 86.88 ± 23.26 | 1.88 ±.0.48 | 2.37 ± 0.26 | 6.00 ± 0.66 | – |
| Loperamide | 10 | 217.90 ± 18.93 *** | 0.00 ± 0.00 * | 0.50 ± 0.26 ** | 0.33 ± 0.34 *** | 89 |
| HECS | 125 | 115.10 ± 10.18 | 1.29 ± 0.64 | 1.00 ± 0.43 | 5.33 ± 0.55 | – |
| HECS | 250 | 173.40 ± 29.43 * | 1.88 ± 0.67 | 1.01 ± 0.26 | 4.28 ± 0.25 * | 54 |
| HECS | 500 | 189.11 ± 24.95 * | 1.75 ± 0.55 | 1.05 ± 0.31 | 2.66 ± 0.67 ** | 67 |
| Treatment (p.o.) | Dose (mg/kg) | Distance Moved by Charcoal | Inhibition (%) | Intestinal Fluid (g) | Inhibition (%) |
|---|---|---|---|---|---|
| Vehicle | – | 1.00 ± 0.04 | – | 1.01 ± 0.06 | – |
| Loperamide | 10 | 0.53 ± 0.04 ** | 47% | 0.56 ± 0.07 *** | 45% |
| HECS | 125 | 0.94 ± 0.03 | – | 0.87 ± 0.08 | – |
| HECS | 250 | 0.89 ± 0.03 | – | 0.77 ± 0.07 | – |
| HECS | 500 | 0.81 ± 0.05 * | 19% | 0.60 ± 0.08 ** | 40% |
| Pretreatment | Treatment | Distance Moved by Charcoal | Inhibition (%) |
|---|---|---|---|
| Saline 0.9% i.p | Vehicle | 0.80 ± 0.03 | – |
| Saline 0.9% i.p | Morphine 2.5 mg/kg, s.c. | 0.59 ± 0.03 ** | 26% |
| Saline 0.9% i.p | HECS 500 mg/kg, p.o. | 0.65 ± 0.03 * | 20% |
| Naloxone 15 mg/kg, i.p | Vehicle | 0.96 ± 0.04 | – |
| Naloxone 15 mg/kg, i.p | Morphine 2.5 mg/kg, s.c. | 0.86 ± 0.02 ns | – |
| Naloxone 15 mg/kg, i.p | HECS 500 mg/kg, p.o. | 0.77 ± 0.04 *** | 20% |
| Saline 0.9% i.p | Vehicle | 0.80 ± 0.02 | – |
| Saline 0.9% i.p | Clonidine 0.1 mg/kg, p.o. | 0.39 ± 0.03 *** | 49% |
| Saline 0.9% i.p | HECS 500 mg/kg, p.o. | 0.65 ± 0.03 ** | 19% |
| Yohimbine 1 mg/kg, i.p | Vehicle | 1.00 ± 0.04 | – |
| Yohimbine 1 mg/kg, i.p | Clonidine 0.1 mg/kg, p.o. | 0.87 ± 0.04 ns | – |
| Yohimbine 1 mg/kg, i.p | HECS 500 mg/kg, p.o. | 0.77 ± 0.03 ** | 22% |



| Induction (Diarrhea) | Treatment | Dose (mg/kg) | Intestinal Fluid (g) | Inhibition (%) |
|---|---|---|---|---|
| PGE2 100 µg/kg, p.o. | Vehicle (p.o.) | – | 0.58 ± 0.03 | – |
| Loperamide (p.o.) | 10 | 0.32 ± 0.03 *** | 46% | |
| HECS (p.o.) | 500 | 0.41 ± 0.03 ** | 29% | |
| l-arginine 600 mg/kg, i.p | Vehicle (p.o.) | – | 0.91 ± 0.05 | – |
| l-NAME (i.p.) | 10 | 0.64 ± 0.04 * | 30% | |
| HECS (p.o.) | 500 | 0.88 ± 0.07 | – |
3. Experimental Section
3.1. Drugs and Chemicals
3.2. Plant Material and Extraction
3.3. Chemical Fingerprint of 70% EtOH by HPLC-PDA-ESI-IT-MS
3.4. Animals
3.5. Acute Toxicity and Hippocratic Screening
3.6. Evaluation of Anti-Inflammatory Activity: Xylene-Induced Ear Edema
3.7. Evaluation of Anti-Inflammatory Activity: Arachidonic Acid-Induced Ear Edema
3.8. Determination of Prostaglandin E2 (PGE2) Level in Arachidonic Acid-Induced Ear Edema
3.9. Antidiarrheal Activity: Castor Oil-Induced Diarrhea
3.10. Castor Oil-Induced Intestinal Fluid Accumulation
3.11. Castor Oil-Induced Intestinal Transit
3.12. Determination of Mechanisms of Action Involved in the Antidiarrheal Effect: Involvement of Opioid Receptors in the Intestinal Transit
3.13. Involvement of Presynaptic α2-Adrenergic Receptors in the Intestinal Transit
3.14. Involvement of Muscarinic Receptors in the Intestinal Transit
3.15. Involvement of NO in the Intestinal Fluid Accumulation
3.16. PGE2-Induced Intestinal Fluid Accumulation
3.17. Phasic Contractions Induced by Carbachol or KCl in Isolated Ileum in Vitro
3.18. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cano, J.H.; Volpato, G. Herbal mixtures in the traditional medicine of Eastern Cuba. J. Ethnopharmacol. 2004, 90, 293–316. [Google Scholar] [CrossRef] [PubMed]
- Viana, G.S.B.; Medeiros, A.C.C.; Lacerda, A.M.R.; Kalyne, L.; Leal, A.M.; Vale, T.G.; Matos, F.J.A. Hypoglycemic and anti-lipemic effects of the aqueous extract from Cissus sicyoides. BMC Pharmacol. 2004, 4, 4–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasconcelos, T.H.C.; Modesto-Filho, J.; Diniz, M.F.F.M.; Santos, H.B.; Aguiar, F.B.; Moreira, P.V.L. Estudo toxicológico pré-clinico agudo com o extrato hidroalcoólico das folhas de Cissus sicyoides L. (Vitaceae). Braz. J. Pharmacogn. 2007, 17, 583–591. [Google Scholar] [CrossRef]
- Silva, M.I.G.; Melo, C.T.V.; Vasconcelos, L.F.; Carvalho, A.M.R.; Sousa, F.C.F. Bioactivity and potential therapeutic benefits of some medicinal plants from the Caatinga (semi-arid) vegetation of Northeast Brazil: A review of the literature. Braz. J. Pharmacogn. 2012, 22, 193–207. [Google Scholar] [CrossRef]
- Beltrame, F.L.; Ferreira, A.G.; Cortez, D.A. Coumarin glycoside from Cissus sicyoides. Nat. Prod. Lett. 2002, 16, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Martínez, M.C.; Pascual Pola, C.N. Medicinal plants used in some rural populations of Oaxaca, Puebla and Veracruz, Mexico. J. Ethnopharmacol. 1992, 35, 229–257. [Google Scholar] [CrossRef]
- Garcia, M.D.; Quílez, A.M.; Sáenz, M.T.; Martínez-Domínguez, M.E.; de la Puerta, R. Anti-inflammatory activity of Agave intermixta Trel. and Cissus sicyoides L., species used in the caribbean traditional medicine. J. Ethnopharmacol. 2000, 71, 395–400. [Google Scholar] [CrossRef]
- Vicentini, V.E.P.; Camparoto, M.L.; Teixeira, R.O.; Mantovani, M.S. Averrhoa carambola L., Syzygium cumini L., Skeels and Cissus sicyoides L.: Medicinal herbal tea effects on vegetal and animal test system. Acta Sci. 2001, 23, 593–598. [Google Scholar]
- Almeida, E.R.; Soares, R.P.F.; Lucena, F.F.R.; Oliveira, J.R.G.; Albuquerque, J.F.C.; Couto, G.B.L. Central antinociceptive effects of Cissus sicyoides on mice. Pharm. Biol. 2006, 44, 304–308. [Google Scholar] [CrossRef]
- Quilez, A.M.; Saenz, M.T.; Garcia, M.D.; Puerta, R. Phytochemical analysis and anti-allergic study of Agave intermixta Trel. and Cissus sicyoides L. J. Pharm. Pharmacol. 2004, 56, 1185–1189. [Google Scholar] [CrossRef] [PubMed]
- Saenz, M.T.; Garcia, M.D.; Quilez, A.; Ahumanda, M.C. Cytotoxic activity of Agave intermixtya L. (Agavaceae) and Cissus sicyoides L. (Vitaceae). Phytother. Res. 2000, 14, 552–554. [Google Scholar] [CrossRef]
- Beltrame, F.L.; Sartoretto, J.L.; Bazotte, R.B.; Cuman, R.N.; Cortez, D.A.G. Estudo químico e avaliação do potencial antidiabético do Cissus sicyoides L. (Vitaceae). Quím. Nova 2004, 24, 783–785. [Google Scholar] [CrossRef]
- Ferreira, M.P.; Nishijima, C.M.; Seito, L.N.; Dokkedal, A.L.; Lopes-Ferreira, M.; Di Stasi, L.C.; Vilegas, W.; Hiruma-Lima, C.A. Gastroprotective effect of Cissus sicyoides (Vitaceae): Involvement of microcirculation, endogenous sulfhdryls and nitric oxide. J. Ethnopharmacol. 2008, 117, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Matsuda, H.; Hata, H.; Sugawara, K.; Nakamura, S.; Yoshikawa, M. Structures of new flavonoids and benzofuran-type stilbene and degranulation inhibitors of rat basophilic leukemia cells from the Brazilian herbal medicine Cissus sicyoides. Chem. Pharm. Bull. 2009, 57, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Lizama, T.S.; Martinez, M.M.; Pérez, O.C. Contribución al estúdio de Cissus sicyoides L. (béjucoubi). Rev. Cuba. Farm. 2000, 34, 120–124. [Google Scholar]
- Almeida, E.R.; Oliviera, J.R.G.; Lucena, F.F.R.; Soares, R.P.F.; Couto, G.B.L. The action of extract of dry leaves of Cissus sicyoides L. in pregnat rats. Acta Farm. Bonaer. 2006, 25, 421–424. [Google Scholar]
- Mequanint, W.; Makonnen, E.; Urga, K. In vivo anti-inflammatory activities of leaf extracts of Ocimum lamiifolium in mice model. J. Ethnopharmacol. 2011, 134, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Sandor, K.; Helyes, Z.; Elekes, K.; Szolcsányi, J. Involvement of capsaicin-sensitive afferents and the transient receptor potential vanilloid 1 receptor in xylene-induced nocifensive behaviour and inflammation in the mouse. Neurosci. Lett. 2009, 405, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.D.; Vasko, M.R. Cellular mechanisms of neurogenic inflammation. J. Pharmacol. Exp. Ther. 2002, 302, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Adeyemi, O.O.; Yemitan, O.K.; Afolabi, L. Inhibition of chemically induced inflammation and pain by orally and topically administered leaf extract of Manihot esculenta Crantz in rodents. J. Ethnopharmacol. 2008, 119, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, R.A.; Abdul, H.A.; Mohamed, S.; Bakar, F.A. Total phenolic compounds, flavonoids, and radical scavenging activity of 21 selected tropical plants. J. Food Sci. 2010, 75, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Humes, J.L.; Opas, E.E.; Bonney, R.J. Arachidonic Acid Metabolites in Mouse Ear Edema. Advances in Inflammations Research; Raven Press: Nova York, NY, USA, 1986; pp. 57–65. [Google Scholar]
- Crummey, A.; Harper, G.P.; Boyle, E.A.; Mangan, F.R. Inhibition of arachidonic acid-induced ear edema as a model for assessing topical antiinflammatory compounds. Agents Actions 1987, 20, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Ricciotti, E.; Fitzgerald, G.A. Prostaglandins and inflammation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 986–1000. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Shi, D.; Liu, L.; Wang, J.; Xie, X.; Kang, T.; Deng, W. Quercetin suppresses cyclooxygenase-2 expression and angiogenesis through inactivation of P300 signaling. PLoS ONE 2011, 6, e22934. [Google Scholar] [CrossRef] [PubMed]
- Awad, A.B.; Toczek, J.; Fink, C.S. Phytosterols decrease prostaglandin release in cultured 388D/MAD macrophages. Prostaglandins Leukot. Essent. Fatty Acids 2004, 70, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Wenzl, H.H. Diarrhea in chronic inflammatory bowel disease. Gastroenterol. Clin. N. Am. 2012, 41, 651–675. [Google Scholar] [CrossRef] [PubMed]
- Kosek, M.; Bern, C.; Guerrant, R.L. The global burden of diarrheal disease, as estimated from studies published between 1992 and 2000. Bull. World Health Organ. 2003, 81, 197–204. [Google Scholar] [PubMed]
- Power, A.M.; Talley, N.J.; Ford, A.C. Association between constipation and colorectal cancer: Systematic review and meta-analysis of observational studies. Am. J. Gastroenterol. 2013, 108, 894–903. [Google Scholar] [CrossRef] [PubMed]
- Sebai, H.; Jabri, M.A.; Souli, A.; Rtibi, K.; Selmi, S.; Tebourbi, O.; El-Benna, J.; Sakly, M. Antidiarrheal and antioxidant activities of chamomile (Matricaria recutita L.) decoction extract in rats. J. Ethnopharmacol. 2014, 152, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Awouters, F.; Niemegeers, C.J.E.; Lenaerts, F.M.; Janseen, P.A.J. Delay of castor oil diarrhoea in rats: A new way to evaluate inhibitors of prostaglandin synthesis. J. Pharm. Pharmacol. 1978, 30, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Mascolo, N.; Izzo, A.A.; Autore, G.; Barbato, F.; Capasso, F. Nitric oxide and castor oil-induced diarrhea. J. Pharmacol. Exp. Ther. 1994, 268, 291–295. [Google Scholar] [PubMed]
- Akindele, A.J.; Adeyemi, O.O. Evaluation of the antidharrhoeal activity of Byrsocarpus coccineus. J. Ethnopharmacol. 2006, 108, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, F.; Capasso, F.; Capasso, R.; Ascione, V.; Aviello, G.; Longo, L.; Izzo, A.A. Effect of Boswellia serrata on intestinal motility in rodents: Inhibition of diarrhoea without constipation. Br. J. Pharmacol. 2006, 148, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Beard, T.L.; Leslie, J.B.; Nemeth, J. The opioid component of delayed gastrointestinal recovery after bowel resection. J. Gastrointest. Surg. 2011, 15, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Fülöp, K.; Zádori, Z.; Rónai, A.Z.; Gyires, K. Characterization of α2-adrenoceptor subtypes involved in gastric emptying gastric motility and gastric mucosal defense. Eur. J. Pharmacol. 2005, 528, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D.; Galligan, J.J. Function of opioids in the enteric nervous system. Neurogastroenterol. Motil. 2004, 16, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Watterson, K.R.; Ratz, P.H.; Spiegel, S. The role of sphingosine-1-phosphate in smooth muscle contraction. Cell Signal. 2005, 17, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Wegener, J.W.; Schulla, V.; Koller, A.; Klugbauer, N.; Feil, R.; Hofmann, J. Control of intestinal motility by the Ca2+ 1.2 l-type calcium in mice. FASEB J. 2006, 20, E566–E573. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, I.J.; Gould, R.J.; Snyder, S.H. Loperamide: Blockade of calcium channels as a mechanism for antidiarrheal effects. J. Pharmacol. Exp. Ther. 1984, 231, 628–632. [Google Scholar] [PubMed]
- Lee, C.W.; Sarna, S.K.; Singaram, C.; Casper, M.A. Ca2+ channel blockade by verapamil inhibits GMCs and diarrhea during small intestinal inflammation. Am. J. Physiol. 1997, 273, G785–B794. [Google Scholar] [PubMed]
- Khan, A.U.; Gilani, A.H. Antispasmodic and bronchodilator activities of Artemisia vulgaris are mediated through dual blockade of muscarinic receptors and calcium influx. J. Ethnopharmacol. 2009, 126, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Unno, T.; Matsuyama, H.; Sakamoto, T.; Uchiyama, M.; Izumi, Y.; Okamoto, H.; Yamada, M.; Wess, J.; Komori, S. M2 and M3 muscarinic receptor-mediated contractions in longitudinal smooth muscle of the ileum studied with receptor knockout mice. Br. J. Pharmacol. 2005, 146, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Gurgel, L.A.; Silva, R.M.; Santos, F.A.; Martins, D.T.O.; Mattos, P.O.; Rao, V.S.N. Studies on the antidiarrhoeal effect of Dragon’s Blood from Croton urucurana. Phytother. Res. 2000, 14, 1–4. [Google Scholar]
- Ward, S.M.; Dalziel, H.H.; Bradley, M.E.; Buxton, I.L.O.; Keef, K.; Westfall, D.P.; Sanders, K.M. Involvement of cyclic GMP in non-adrenergic, non-colinergic inhibitory neurotransmission in dog proximal colon. Br. J. Pharmacol. 1992, 107, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Katsuki, S.; Arnold, W.P.; Murad, F. Effects of sodium nitroprusside, nitroglycerin, and sodium azide on levels of cyclic nucleotides and mechanical activity of various tissues. J. Cyclic Nucleotide Res. 1977, 3, 239–247. [Google Scholar] [PubMed]
- Brasitus, T.A.; Field, M.; Kimberg, V. Intestinal mucosal cyclic GMP: Regulation and relation to ion transport. Am. J. Physiol. 1976, 231, G275–G282. [Google Scholar]
- Di Carlo, G.; Autore, G.; Izzo, A.A.; Maiolino, P.; Mascolo, N.; Viola, P.; Diurno, M.V.; Capasso, F. Inhibition of intestinal motility and secretion by flavonoids in mice and rats: Structure-activity relationships. J. Pharm. Pharmacol. 1993, 45, 1054–1059. [Google Scholar] [CrossRef] [PubMed]
- Fanning, M.J.; Macander, P.; Drzewiecki, G.; Middleton, E. Quercetin inhibits anaphylactic contraction of guinea-pig ileum smooth muscle. Int. Arch. Allergy Immunol. 1983, 71, 371–373. [Google Scholar] [CrossRef]
- Baggio, C.H.; Freitas, C.S.; Rieck, L.; Marques, M.C. Gastroprotective effects of a crude extract of Baccharis illinita DC in rats. Pharmacol. Res. 2003, 47, 93–98. [Google Scholar] [CrossRef]
- Souza Brito, A.R.M. Manual de Ensaios Toxicológicos in Vivo; Editora da Unicamp: Campinas, SP, Brazil, 1994; pp. 15–22. [Google Scholar]
- Swingle, K.F.; Reiter, M.J.; Schwartzmiller, D.H. Comparison of croton oil and cantharidin induced inflammations of the mouse ear and their modification by topically applied drugs. Arch. Int. Pharmacodyn. Ther. 1981, 254, 168–176. [Google Scholar] [PubMed]
- Young, J.M.; Spires, D.A.; Bedord, C.J.; Wagner, B.; Ballaron, S.J.; de Young, L.M. The mouse ear inflammatory response to topical arachidonic acid. J. Investig. Dermatol. 1984, 82, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Xian, Y.F.; Lin, Z.X.; Xu, X.Y.; Su, Z.R.; Chen, J.N.; Lai, X.P.; Ip, S.P. Effect of Rhizoma polygonati on 12-O-tetradecanoylphorbol-acetate-induced ear edema in mice. J. Ethnopharmacol. 2012, 142, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Robert, A.; Nezamis, J.E.; Lancaster, C.; Hanchar, A.J.; Klepper, M.S. Enteropooling assay: A test for diarrhoeal produced by prostaglandins. Prostaglandins 1976, 11, 809–814. [Google Scholar] [CrossRef]
- Stickney, J.C.; Northup, D.W. Effect of gastric emptying upon propulsive motility of small intestine of rats. Proc. Soc. Exp. Biol. Med. 1959, 101, 582–583. [Google Scholar] [CrossRef] [PubMed]
- Howd, R.A.; Adamovics, A.; Palekar, A. Naloxone and intestinal motility. Cell. Mol. Life Sci. 1978, 3, 1310–1311. [Google Scholar] [CrossRef]
- Meli, R.; Autore, G.; Di Carlo, G.; Capasso, F. Inhibitory action of quercetin on intestinal transit in mice. Phytother. Res. 1990, 4, 201–201. [Google Scholar] [CrossRef]
- Izzo, A.A.; Mascolo, N.; Di Carlo, G.; Capasso, F. NG-nitro-l-arginine methyl ester modulates intestinal secretion and motility produced by carbachol. Eur. J. Pharmacol. 1994, 271, 31–35. [Google Scholar] [CrossRef]
- Mazzolin, L.P.; Kiguti, L.R.A.; Maia, E.O.; Fernandes, L.T.L.; Rocha, L.R.M.; Vilegas, W.; Pupo, A.S.; di Stasi, L.C.; Hiruma-Lima, C.A. Antidiarrheal and intestinal anti-inflammatory activities of a methanolic extract of Qualea parviflora Mart. in experimental models. J. Ethnopharmacol. 2013, 150, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beserra, F.P.; De Cássia Santos, R.; Périco, L.L.; Rodrigues, V.P.; De Almeida Kiguti, L.R.; Saldanha, L.L.; Pupo, A.S.; Da Rocha, L.R.M.; Dokkedal, A.L.; Vilegas, W.; et al. Cissus sicyoides: Pharmacological Mechanisms Involved in the Anti-Inflammatory and Antidiarrheal Activities. Int. J. Mol. Sci. 2016, 17, 149. https://doi.org/10.3390/ijms17020149
Beserra FP, De Cássia Santos R, Périco LL, Rodrigues VP, De Almeida Kiguti LR, Saldanha LL, Pupo AS, Da Rocha LRM, Dokkedal AL, Vilegas W, et al. Cissus sicyoides: Pharmacological Mechanisms Involved in the Anti-Inflammatory and Antidiarrheal Activities. International Journal of Molecular Sciences. 2016; 17(2):149. https://doi.org/10.3390/ijms17020149
Chicago/Turabian StyleBeserra, Fernando Pereira, Raquel De Cássia Santos, Larissa Lucena Périco, Vinicius Peixoto Rodrigues, Luiz Ricardo De Almeida Kiguti, Luiz Leonardo Saldanha, André Sampaio Pupo, Lúcia Regina Machado Da Rocha, Anne Lígia Dokkedal, Wagner Vilegas, and et al. 2016. "Cissus sicyoides: Pharmacological Mechanisms Involved in the Anti-Inflammatory and Antidiarrheal Activities" International Journal of Molecular Sciences 17, no. 2: 149. https://doi.org/10.3390/ijms17020149