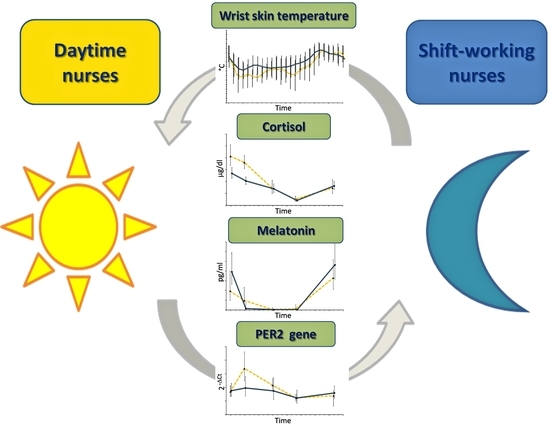
Peripheral Skin Temperature and Circadian Biological Clock in Shift Nurses after a Day off
Abstract
:1. Introduction
2. Results
3. Discussion
4. Experimental Section
4.1. Participants and Sampling
4.2. Peripheral Skin Temperature Measurement
4.3. Cortisol and Melatonin Measurement
4.4. Circadian Expression of PER2 Clock Gene
4.5. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Akhtar, R.A.; Reddy, A.B.; Maywood, E.S.; Clayton, J.D.; King, V.M.; Smith, A.G.; Gant, T.W.; Hastings, M.H.; Kyriacou, C.P. Circadian cycling of the mouse liver transcriptome, as revealed by cDNA microarray, is driven by the suprachiasmatic nucleus. Curr. Biol. 2002, 12, 540–550. [Google Scholar] [CrossRef]
- Archer, S.N.; Laing, E.E.; Moller-Levet, C.S.; van der Veen, D.R.; Bucca, G.; Lazar, A.S.; Santhi, N.; Slak, A.; Kabiljo, R.; von Schantz, M.; et al. Mistimed sleep disrupts circadian regulation of the human transcriptome. Proc. Natl. Acad. Sci. USA 2014, 111, E682–E691. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.H.; McDearmon, E.L.; Panda, S.; Hayes, K.R.; Zhang, J.; Andrews, J.L.; Antoch, M.P.; Walker, J.R.; Esser, K.A.; Hogenesch, J.B.; et al. Circadian and clock-controlled regulation of the mouse transcriptome and cell proliferation. Proc. Natl. Acad. Sci. USA 2007, 104, 3342–3347. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; Antoch, M.P.; Miller, B.H.; Su, A.I.; Schook, A.B.; Straume, M.; Schultz, P.G.; Kay, S.A.; Takahashi, J.S.; Hogenesch, J.B. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell 2002, 109, 307–320. [Google Scholar] [CrossRef]
- Ueda, H.R.; Chen, W.; Adachi, A.; Wakamatsu, H.; Hayashi, S.; Takasugi, T.; Nagano, M.; Nakahama, K.; Suzuki, Y.; Sugano, S.; et al. A transcription factor response element for gene expression during circadian night. Nature 2002, 418, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.G.; Zhu, Y. Electric light, particularly at night, disrupts human circadian rhythmicity: Is that a problem? Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.P.; McHill, A.W.; Birks, B.R.; Griffin, B.R.; Rusterholz, T.; Chinoy, E.D. Entrainment of the human circadian clock to the natural light-dark cycle. Curr. Biol. 2013, 23, 1554–1558. [Google Scholar] [CrossRef] [PubMed]
- Buhr, E.D.; Yoo, S.H.; Takahashi, J.S. Temperature as a universal resetting cue for mammalian circadian oscillators. Science 2010, 330, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Nicolas, A.; Madrid, J.A.; Rol, M.A. Day-night contrast as source of health for the human circadian system. Chronobiol. Int. 2014, 31, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Boudreau, P.; Dumont, G.A.; Boivin, D.B. Circadian adaptation to night shift work influences sleep, performance, mood and the autonomic modulation of the heart. PLoS ONE 2013, 8, e70813. [Google Scholar] [CrossRef] [PubMed]
- Petrov, M.E.; Clark, C.B.; Molzof, H.E.; Johnson, R.L., Jr.; Cropsey, K.L.; Gamble, K.L. Sleep strategies of night-shift nurses on days off: Which ones are most adaptive? Front. Neurol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Costa, G.; Anelli, M.M.; Castellini, G.; Fustinoni, S.; Neri, L. Stress and sleep in nurses employed in “3 × 8” and “2 × 12” fast rotating shift schedules. Chronobiol. Int. 2014, 31, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Arendt, J. Shift work: Coping with the biological clock. Occup. Med. 2010, 60, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Munch, M.; Bromundt, V. Light and chronobiology: Implications for health and disease. Dialogues Clin. Neurosci. 2012, 14, 448–453. [Google Scholar] [PubMed]
- Stevens, R.G. Working against our endogenous circadian clock: Breast cancer and electric lighting in the modern world. Mutat. Res. 2009, 680, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Cermakian, N.; Boivin, D.B. The regulation of central and peripheral circadian clocks in humans. Obes. Rev. 2009, 10 (Suppl. 2), 25–36. [Google Scholar] [CrossRef] [PubMed]
- Romanovsky, A.A. Thermoregulation: Some concepts have changed. Functional architecture of the thermoregulatory system. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 292, R37–R46. [Google Scholar] [CrossRef] [PubMed]
- Tansey, E.A.; Johnson, C.D. Recent advances in thermoregulation. Adv. Physiol. Educ. 2015, 39, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Krauchi, K.; Wirz-Justice, A. Circadian rhythm of heat production, heart rate, and skin and core temperature under unmasking conditions in men. Am. J. Physiol. 1994, 267, R819–R829. [Google Scholar] [PubMed]
- Taylor, N.A.; Tipton, M.J.; Kenny, G.P. Considerations for the measurement of core, skin and mean body temperatures. J. Therm. Biol. 2014, 46, 72–101. [Google Scholar] [CrossRef] [PubMed]
- Krauchi, K. The human sleep-wake cycle reconsidered from a thermoregulatory point of view. Physiol. Behav. 2007, 90, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Van Someren, E.J. More than a marker: Interaction between the circadian regulation of temperature and sleep, age-related changes, and treatment possibilities. Chronobiol. Int. 2000, 17, 313–354. [Google Scholar] [CrossRef] [PubMed]
- Krauchi, K.; Gompper, B.; Hauenstein, D.; Flammer, J.; Pfluger, M.; Studerus, E.; Schotzau, A.; Orgul, S. Diurnal blood pressure variations are associated with changes in distal-proximal skin temperature gradient. Chronobiol. Int. 2012, 29, 1273–1283. [Google Scholar] [CrossRef] [PubMed]
- Raymann, R.J.; Swaab, D.F.; van Someren, E.J. Cutaneous warming promotes sleep onset. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R1589–R1597. [Google Scholar] [CrossRef] [PubMed]
- Krauchi, K.; Wirz-Justice, A. Circadian clues to sleep onset mechanisms. Neuropsychopharmacology 2001, 25, S92–S96. [Google Scholar] [CrossRef]
- Weibel, L.; Brandenberger, G. Disturbances in hormonal profiles of night workers during their usual sleep and work times. J. Biol. Rhythms 1998, 13, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Hucklebridge, F.; Hussain, T.; Evans, P.; Clow, A. The diurnal patterns of the adrenal steroids cortisol and dehydroepiandrosterone (DHEA) in relation to awakening. Psychoneuroendocrinology 2005, 30, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Knutsson, U.; Dahlgren, J.; Marcus, C.; Rosberg, S.; Bronnegard, M.; Stierna, P.; Albertsson-Wikland, K. Circadian cortisol rhythms in healthy boys and girls: Relationship with age, growth, body composition, and pubertal development. J. Clin. Endocrinol. Metab. 1997, 82, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Cameron, A.; Henley, D.; Carrell, R.; Zhou, A.; Clarke, A.; Lightman, S. Temperature-responsive release of cortisol from its binding globulin: A protein thermocouple. J. Clin. Endocrinol. Metab. 2010, 95, 4689–4695. [Google Scholar] [CrossRef] [PubMed]
- Aardal, E.; Holm, A.C. Cortisol in saliva—Reference ranges and relation to cortisol in serum. Eur. J. Clin. Chem. Clin. Biochem. 1995, 33, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Burgess, H.J.; Fogg, L.F. Individual differences in the amount and timing of salivary melatonin secretion. PLoS ONE 2008, 3, e3055. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.M.; Belancio, V.P.; Dauchy, R.T.; Xiang, S.; Brimer, S.; Mao, L.; Hauch, A.; Lundberg, P.W.; Summers, W.; Yuan, L.; et al. Melatonin: An inhibitor of breast cancer. Endocr. Relat. Cancer 2015, 22, R183–R204. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.G.; Brainard, G.C.; Blask, D.E.; Lockley, S.W.; Motta, M.E. Breast cancer and circadian disruption from electric lighting in the modern world. CA Cancer J. Clin. 2014, 64, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Bracci, M.; Copertaro, A.; Manzella, N.; Staffolani, S.; Strafella, E.; Nocchi, L.; Barbaresi, M.; Copertaro, B.; Rapisarda, V.; Valentino, M.; et al. Influence of night-shift and napping at work on urinary melatonin, 17-beta-estradiol and clock gene expression in pre-menopausal nurses. J. Biol. Regul. Homeost. Agents 2013, 27, 267–274. [Google Scholar] [PubMed]
- Brown, S.A.; Azzi, A. Peripheral circadian oscillators in mammals. Handb. Exp. Pharmacol. 2013, 217, 45–66. [Google Scholar] [PubMed]
- Buhr, E.D.; Takahashi, J.S. Molecular components of the mammalian circadian clock. Handb. Exp. Pharmacol. 2013, 217, 3–27. [Google Scholar] [PubMed]
- Lowrey, P.L.; Takahashi, J.S. Genetics of circadian rhythms in mammalian model organisms. Adv. Genet. 2011, 74, 175–230. [Google Scholar] [PubMed]
- Reppert, S.M.; Weaver, D.R. Coordination of circadian timing in mammals. Nature 2002, 418, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, J.S. Molecular components of the circadian clock in mammals. Diabetes Obes. Metab. 2015, 17 (Suppl. 1), 6–11. [Google Scholar] [CrossRef] [PubMed]
- Chappuis, S.; Ripperger, J.A.; Schnell, A.; Rando, G.; Jud, C.; Wahli, W.; Albrecht, U. Role of the circadian clock gene Per2 in adaptation to cold temperature. Mol. Metab. 2013, 2, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, N.; Tahara, Y.; Kuriki, D.; Haraguchi, A.; Shibata, S. Warm water bath stimulates phase-shifts of the peripheral circadian clocks in PER2::Luciferase mouse. PLoS ONE 2014, 9, e100272. [Google Scholar] [CrossRef] [PubMed]
- Boivin, D.B.; Boudreau, P. Impacts of shift work on sleep and circadian rhythms. Pathol. Biol. 2014, 62, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Corbalan-Tutau, M.D.; Gomez-Abellan, P.; Madrid, J.A.; Canteras, M.; Ordovas, J.M.; Garaulet, M. Toward a chronobiological characterization of obesity and metabolic syndrome in clinical practice. Clin. Nutr. 2015, 34, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Corbalan-Tutau, M.D.; Madrid, J.A.; Ordovas, J.M.; Smith, C.E.; Nicolas, F.; Garaulet, M. Differences in daily rhythms of wrist temperature between obese and normal-weight women: Associations with metabolic syndrome features. Chronobiol. Int. 2011, 28, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Proper, K.I.; van de Langenberg, D.; Rodenburg, W.; Vermeulen, R.C.; van der Beek, A.J.; van Steeg, H.; van Kerkhof, L.W. The relationship between shift work and metabolic risk factors: A systematic review of longitudinal studies. Am. J. Prev. Med. 2016, 50. [Google Scholar] [CrossRef] [PubMed]
- Copertaro, A.; Bracci, M.; Barbaresi, M.; Santarelli, L. Assessment of cardiovascular risk in shift healthcare workers. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Te Kulve, M.; Schellen, L.; Schlangen, L.J.; van Marken Lichtenbelt, W.D. The influence of light on thermal responses. Acta Physiol. 2016, 216, 163–185. [Google Scholar] [CrossRef] [PubMed]
- Seron-Ferre, M.; Reynolds, H.; Mendez, N.A.; Mondaca, M.; Valenzuela, F.; Ebensperger, R.; Valenzuela, G.J.; Herrera, E.A.; Llanos, A.J.; Torres-Farfan, C. Impact of maternal melatonin suppression on amount and functionality of brown adipose tissue (BAT) in the newborn sheep. Front. Endocrinol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Bracci, M.; Manzella, N.; Copertaro, A.; Staffolani, S.; Strafella, E.; Barbaresi, M.; Copertaro, B.; Rapisarda, V.; Valentino, M.; Santarelli, L. Rotating-shift nurses after a day off: Peripheral clock gene expression, urinary melatonin, and serum 17-beta-estradiol levels. Scand. J. Work Environ. Health 2014, 40, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.; Mirick, D.K.; Chen, C.; Stanczyk, F.Z. Night shift work and hormone levels in women. Cancer Epidemiol. Biomark. Prev. 2012, 21, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Grundy, A.; Tranmer, J.; Richardson, H.; Graham, C.H.; Aronson, K.J. The influence of light at night exposure on melatonin levels among canadian rotating shift nurses. Cancer Epidemiol. Biomark. Prev. 2011, 20, 2404–2412. [Google Scholar] [CrossRef] [PubMed]
- Marie Hansen, A.; Helene Garde, A.; Hansen, J. Diurnal urinary 6-sulfatoxymelatonin levels among healthy danish nurses during work and leisure time. Chronobiol. Int. 2006, 23, 1203–1215. [Google Scholar] [CrossRef] [PubMed]
- Peplonska, B.; Bukowska, A.; Gromadzinska, J.; Sobala, W.; Reszka, E.; Lie, J.A.; Kjuus, H.; Wasowicz, W. Night shift work characteristics and 6-sulfatoxymelatonin (MT6s) in rotating night shift nurses and midwives. Occup. Environ. Med. 2012, 69, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Schernhammer, E.S.; Rosner, B.; Willett, W.C.; Laden, F.; Colditz, G.A.; Hankinson, S.E. Epidemiology of urinary melatonin in women and its relation to other hormones and night work. Cancer Epidemiol. Biomark. Prev. 2004, 13, 936–943. [Google Scholar]
- Knauth, P.; Rutenfranz, J. Experimental shift work studies of permanent night, and rapidly rotating, shift systems. I. Circadian rhythm of body temperature and re-entrainment at shift change. Int. Arch. Occup. Environ. Health 1976, 37, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Niu, S.F.; Chung, M.H.; Chu, H.; Tsai, J.C.; Lin, C.C.; Liao, Y.M.; Ou, K.L.; O’Brien, A.P.; Chou, K.R. Differences in cortisol profiles and circadian adjustment time between nurses working night shifts and regular day shifts: A prospective longitudinal study. Int. J. Nurs. Stud. 2015, 52, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Ulhoa, M.A.; Marqueze, E.C.; Burgos, L.G.; Moreno, C.R. Shift work and endocrine disorders. Int. J. Endocrinol. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Merkus, S.L.; Holte, K.A.; Huysmans, M.A.; Hansen, A.M.; van de Ven, P.M.; van Mechelen, W.; van der Beek, A.J. Neuroendocrine recovery after 2-week 12-h day and night shifts: An 11-day follow-up. Int. Arch. Occup. Environ. Health 2015, 88, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Adam, E.K.; Kumari, M. Assessing salivary cortisol in large-scale, epidemiological research. Psychoneuroendocrinology 2009, 34, 1423–1436. [Google Scholar] [CrossRef] [PubMed]
- Kalsbeek, A.; van der Spek, R.; Lei, J.; Endert, E.; Buijs, R.M.; Fliers, E. Circadian rhythms in the hypothalamo-pituitary-adrenal (HPA) axis. Mol. Cell. Endocrinol. 2012, 349, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Balsalobre, A.; Brown, S.A.; Marcacci, L.; Tronche, F.; Kellendonk, C.; Reichardt, H.M.; Schutz, G.; Schibler, U. Resetting of circadian time in peripheral tissues by glucocorticoid signaling. Science 2000, 289, 2344–2347. [Google Scholar] [CrossRef] [PubMed]
- Cuesta, M.; Cermakian, N.; Boivin, D.B. Glucocorticoids entrain molecular clock components in human peripheral cells. FASEB J. 2015, 29, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Hida, A.; Kitamura, S.; Enomoto, M.; Ohsawa, Y.; Katayose, Y.; Nozaki, K.; Moriguchi, Y.; Aritake, S.; Higuchi, S.; et al. Rhythmic expression of circadian clock genes in human leukocytes and beard hair follicle cells. Biochem. Biophys. Res. Commun. 2012, 425, 902–907. [Google Scholar] [CrossRef] [PubMed]
- James, F.O.; Cermakian, N.; Boivin, D.B. Circadian rhythms of melatonin, cortisol, and clock gene expression during simulated night shift work. Sleep 2007, 30, 1427–1436. [Google Scholar] [PubMed]
- Reszka, E.; Peplonska, B.; Wieczorek, E.; Sobala, W.; Bukowska, A.; Gromadzinska, J.; Lie, J.A.; Kjuus, H.; Wasowicz, W. Circadian gene expression in peripheral blood leukocytes of rotating night shift nurses. Scand. J. Work Environ. Health 2013, 39, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Stevens, R.G.; Hoffman, A.E.; Tjonneland, A.; Vogel, U.B.; Zheng, T.; Hansen, J. Epigenetic impact of long-term shiftwork: Pilot evidence from circadian genes and whole-genome methylation analysis. Chronobiol. Int. 2011, 28, 852–861. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, K.; Plomp, R.; Lao, O.; Middleton, B.; Revell, V.L.; Skene, D.J.; Kayser, M. Effect of sleep deprivation on rhythms of clock gene expression and melatonin in humans. Chronobiol. Int. 2013, 30, 901–909. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.G. Circadian disruption and breast cancer: From melatonin to clock genes. Epidemiology 2005, 16, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Johns, M.W. A new method for measuring daytime sleepiness: The Epworth sleepiness scale. Sleep 1991, 14, 540–545. [Google Scholar] [PubMed]
- Vignatelli, L.; Plazzi, G.; Barbato, A.; Ferini-Strambi, L.; Manni, R.; Pompei, F.; D’Alessandro, R. Italian version of the Epworth sleepiness scale: External validity. Neurol. Sci. 2003, 23, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Horne, J.A.; Ostberg, O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int. J. Chronobiol. 1976, 4, 97–110. [Google Scholar] [PubMed]
- Bajaj, A.; Rosner, B.; Lockley, S.W.; Schernhammer, E.S. Validation of a light questionnaire with real-life photopic illuminance measurements: The harvard light exposure assessment questionnaire. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1341–1349. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.H.; Chang, F.M.; Yang, C.C.; Kuo, T.B.; Hsu, N. Sleep quality and morningness-eveningness of shift nurses. J. Clin. Nurs. 2009, 18, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Yazdi, Z.; Sadeghniiat-Haghighi, K.; Javadi, A.R.; Rikhtegar, G. Sleep quality and insomnia in nurses with different circadian chronotypes: Morningness and eveningness orientation. Work 2014, 47, 561–567. [Google Scholar] [PubMed]
- Sarabia, J.A.; Rol, M.A.; Mendiola, P.; Madrid, J.A. Circadian rhythm of wrist temperature in normal-living subjects a candidate of new index of the circadian system. Physiol. Behav. 2008, 95, 570–580. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.D.; Crabtree, D.R.; Bilzon, J.L.; Walsh, N.P. The validity of wireless ibuttons and thermistors for human skin temperature measurement. Physiol. Meas. 2010, 31, 95–114. [Google Scholar] [CrossRef] [PubMed]
- Van Marken Lichtenbelt, W.D.; Daanen, H.A.; Wouters, L.; Fronczek, R.; Raymann, R.J.; Severens, N.M.; van Someren, E.J. Evaluation of wireless determination of skin temperature using ibuttons. Physiol. Behav. 2006, 88, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Hasselberg, M.J.; McMahon, J.; Parker, K. The validity, reliability, and utility of the ibutton(r) for measurement of body temperature circadian rhythms in sleep/wake research. Sleep Med. 2013, 14, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Garde, A.H.; Hansen, A.M. Long-term stability of salivary cortisol. Scand. J. Clin. Lab. Investig. 2005, 65, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Vining, R.F.; McGinley, R.A. The measurement of hormones in saliva: Possibilities and pitfalls. J. Steroid Biochem. 1987, 27, 81–94. [Google Scholar] [CrossRef]
- Daniel, M.; Moore, D.S.; Decker, S.; Belton, L.; DeVellis, B.; Doolen, A.; Campbell, M.K. Associations among education, cortisol rhythm, and BMI in blue-collar women. Obesity 2006, 14, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Dorn, L.D.; Kolko, D.J.; Susman, E.J.; Huang, B.; Stein, H.; Music, E.; Bukstein, O.G. Salivary gonadal and adrenal hormone differences in boys and girls with and without disruptive behavior disorders: Contextual variants. Biol. Psychol. 2009, 81, 31–39. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, E.A.; Di Mascio, P.; Harumi, T.; Spence, D.W.; Moscovitch, A.; Hardeland, R.; Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Measurement of melatonin in body fluids: Standards, protocols and procedures. Childs Nerv. Syst. 2011, 27, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Nowak, R.; McMillen, I.C.; Redman, J.; Short, R.V. The correlation between serum and salivary melatonin concentrations and urinary 6-hydroxymelatonin sulphate excretion rates: Two non-invasive techniques for monitoring human circadian rhythmicity. Clin. Endocrinol. 1987, 27, 445–452. [Google Scholar] [CrossRef]
- Fukuya, H.; Emoto, N.; Nonaka, H.; Yagita, K.; Okamura, H.; Yokoyama, M. Circadian expression of clock genes in human peripheral leukocytes. Biochem. Biophys. Res. Commun. 2007, 354, 924–928. [Google Scholar] [CrossRef] [PubMed]
- Bjarnason, G.A.; Jordan, R.C.; Wood, P.A.; Li, Q.; Lincoln, D.W.; Sothern, R.B.; Hrushesky, W.J.; Ben-David, Y. Circadian expression of clock genes in human oral mucosa and skin: Association with specific cell-cycle phases. Am. J. Pathol. 2001, 158, 1793–1801. [Google Scholar] [CrossRef]
- Boivin, D.B.; James, F.O.; Wu, A.; Cho-Park, P.F.; Xiong, H.; Sun, Z.S. Circadian clock genes oscillate in human peripheral blood mononuclear cells. Blood 2003, 102, 4143–4145. [Google Scholar] [CrossRef] [PubMed]
- Akashi, M.; Soma, H.; Yamamoto, T.; Tsugitomi, A.; Yamashita, S.; Yamamoto, T.; Nishida, E.; Yasuda, A.; Liao, J.K.; Node, K. Noninvasive method for assessing the human circadian clock using hair follicle cells. Proc. Natl. Acad. Sci. USA 2010, 107, 15643–15648. [Google Scholar] [CrossRef] [PubMed]
- Zuther, P.; Gorbey, S.; Lemmer, B. Chronos-Fit 1.06. Available online: http://www.ma.uni-heidelberg.de/inst/phar/lehre/chrono.html (accessed on 14 July 2015).



| Parameters | SW Nurses (n = 23) | DT Nurses (n = 25) | p-Value |
|---|---|---|---|
| Age (years) mean ± SD | 38.8 ± 3.9 | 39.2 ± 3.2 | 0.699 |
| Job seniority (years) mean ± SD | 13.6 ± 3.4 | 12.8 ± 4.4 | 0.487 |
| Shift-work seniority (years) mean ± SD | 13.6 ± 3.4 | - | - |
| Night-shift work (nights per month) mean ± SD | 6.0 ± 1.0 | - | - |
| Body Mass Index (kg/m2) mean ± SD | 24.2 ± 4.7 | 25.0 ± 4.3 | 0.541 |
| Smokers (%) | 39.1 | 44.0 | 0.732 |
| Alcohol drinkers (%) | 34.8 | 40.0 | 0.709 |
| Epworth Sleepiness Scale (score) mean ± SD | 6.0 ± 3.7 | 5.9 ± 3.1 | 0.919 |
| Chronotype a (MEQ score) mean ± SD | 54.3 ± 7.8 | 57.2 ± 9.6 | 0.259 |
| Wrist Skin Temperature | SW Nurses (n = 23) | DT Nurses (n = 25) | p-Value |
|---|---|---|---|
| Mesor (°C) mean ± SD | 34.47 ± 0.42 | 33.96 ± 0.62 | <0.001 |
| Maximum (°C) mean ± SD | 36.20 ± 0.45 | 36.31 ± 0.46 | 0.407 |
| Minimum (°C) mean ± SD | 32.85 ± 0.92 | 31.59 ± 0.99 | <0.001 |
| Amplitude (°C) mean ± SD | 0.95 ± 0.44 | 1.23 ± 0.38 | 0.022 |
| Acrophase (h) mean ± SD | 5:05 ± 7:43 | 4:05 ± 5:50 | 0.617 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bracci, M.; Ciarapica, V.; Copertaro, A.; Barbaresi, M.; Manzella, N.; Tomasetti, M.; Gaetani, S.; Monaco, F.; Amati, M.; Valentino, M.; et al. Peripheral Skin Temperature and Circadian Biological Clock in Shift Nurses after a Day off. Int. J. Mol. Sci. 2016, 17, 623. https://doi.org/10.3390/ijms17050623
Bracci M, Ciarapica V, Copertaro A, Barbaresi M, Manzella N, Tomasetti M, Gaetani S, Monaco F, Amati M, Valentino M, et al. Peripheral Skin Temperature and Circadian Biological Clock in Shift Nurses after a Day off. International Journal of Molecular Sciences. 2016; 17(5):623. https://doi.org/10.3390/ijms17050623
Chicago/Turabian StyleBracci, Massimo, Veronica Ciarapica, Alfredo Copertaro, Mariella Barbaresi, Nicola Manzella, Marco Tomasetti, Simona Gaetani, Federica Monaco, Monica Amati, Matteo Valentino, and et al. 2016. "Peripheral Skin Temperature and Circadian Biological Clock in Shift Nurses after a Day off" International Journal of Molecular Sciences 17, no. 5: 623. https://doi.org/10.3390/ijms17050623
APA StyleBracci, M., Ciarapica, V., Copertaro, A., Barbaresi, M., Manzella, N., Tomasetti, M., Gaetani, S., Monaco, F., Amati, M., Valentino, M., Rapisarda, V., & Santarelli, L. (2016). Peripheral Skin Temperature and Circadian Biological Clock in Shift Nurses after a Day off. International Journal of Molecular Sciences, 17(5), 623. https://doi.org/10.3390/ijms17050623