Characteristics of Three Thioredoxin Genes and Their Role in Chilling Tolerance of Harvested Banana Fruit
Abstract
:1. Introduction
2. Results
2.1. Cloning of MaTrx6, MaTrx9 and MaTrx12 Genes from Banana Fruit
2.2. Sequence and Phylogenetic Analysis of Banana Fruit Trx Genes
2.3. Subcellular Localization of MaTrx6, MaTrx9 and MaTrx12
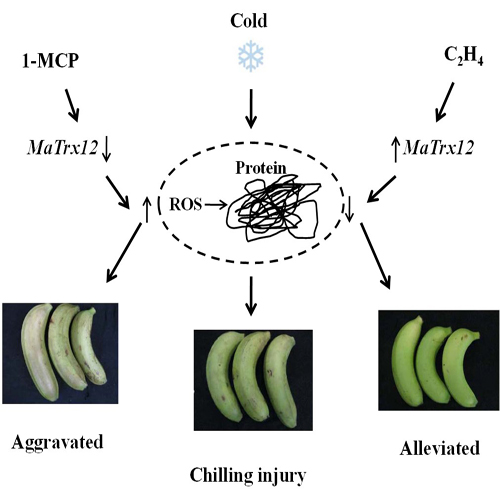
2.4. Effect of Ethylene and 1-MCP Pretreatments on the Development of Chilling Injury in Harvested Banana Fruit
2.5. Expression of MaTrx6, MaTrx9 and MaTrx12 Genes in Ethylene- and 1-MCP-Treated Banana Fruit in Response to Low Temperature Stress
2.6. Effect of MaTrx6, MaTrx9 and MaTrx12 Heterologous Expression on the Growth of a Cytoplasmic Trx-Deficient Yeast Strain under Hydrogen Peroxide Treatment
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Treatments
4.2. Measurement of Chilling Injury (CI) Index
4.3. Color Measurement
4.4. RNA Isolation and cDNA Synthesis
4.5. Amplification of MaTrx Full Length cDNAs
4.6. Sequence and Phylogenetic Analysis
4.7. Real-Time Quantitative PCR (RT-qPCR) Analysis
4.8. Subcellular Localization of MaTrxs
4.9. Heterologous Complementation Analysis
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 1-MCP | 1-Methylcyclopropene |
| CaMV | Cauliflower Mosaic Virus |
| CI | Chilling Injury |
| GFP | Green Fluorescent Protein |
| H2O2 | Hydrogen peroxide |
| iTRAQ | Isobaric Tags for Relative and Absolute Quantitation |
| ORF | Open Reading Frame |
| PEG | Polyethylene Glycol |
| ROS | Reactive Oxygen Species |
| Trx | Thioredoxin |
References
- Singh, S.S.; Devi, S.K.; Ng, T.B. Banana lectin: A brief review. Molecules 2014, 19, 18817–18827. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.H.; Hwang, Y.J. Effect of ethylene treatment on the ripening, polyphenoloxydase activity and water soluble tannin content of Taiwan northern banana at different maturity stages and the stability of banana polyphenoloxydase. Acta Hortic. 1990, 275, 603–610. [Google Scholar] [CrossRef]
- Sisler, E.C.; Serek, M. Inhibitors of ethylene responses in plants at the receptor level: Recent developments. Physiol. Plant 1997, 100, 577–582. [Google Scholar] [CrossRef]
- Jiang, Y.M.; Joyce, D.C.; Macnish, A.J. Response of banana fruit to treatment with 1-methylcyclopropene. Plant Growth Regul. 1999, 28, 77–82. [Google Scholar] [CrossRef]
- Toraskar, M.V.; Modi, V.V. Peroxidase and chilling injury in banana fruit. J. Agric. Food Chem. 1984, 32, 1352–1354. [Google Scholar] [CrossRef]
- Jiang, Y.M.; Joyce, D.C.; Jiang, W.B.; Lu, W.J. Effects of chilling temperatures on ethylene binding by banana fruit. Plant Growth Regul. 2004, 43, 109–115. [Google Scholar] [CrossRef]
- Candan, A.P.; Graell, J.; Larrigaudiere, C. Postharvest quality and chilling injury of plums: Benefits of 1-methylcyclopropene. Span. J. Agric. Res. 2011, 9, 554–564. [Google Scholar] [CrossRef]
- Pesis, E.; Ackerman, M.; Ben-Arie, R.; Feygenberg, O.; Feng, X.Q.; Apelbaum, A.; Goren, R.; Prusky, D. Ethylene involvement in chilling injury symptoms of avocado during cold storage. Postharvest Biol. Technol. 2002, 24, 171–181. [Google Scholar] [CrossRef]
- Dong, Q.L.; Wang, C.R.; Liu, D.D.; Hu, D.G.; Fang, M.J.; You, C.X.; Yao, Y.X.; Hao, Y.J. MdVHA-A encodes an apple subunit A of vacuolar H+-ATPase and enhances drought tolerance in transgenic tobacco seedlings. J. Plant Physiol. 2013, 170, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Argenta, L.; Mattheis, J.P. Interactive effects of 1-MCP and temperature on “Elberta” peach quality. Hortscience 2002, 37, 134–138. [Google Scholar]
- Fan, X.T.; Mattheis, J.P. Development of apple superficial scald, soft scald, core flush, and greasiness is reduced by MCP. J. Agric. Food Chem. 1999, 47, 3063–3068. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.L.; Li, T.T.; Jiang, G.X.; Xie, W.P.; Chang, S.D.; Jiang, Y.M.; Duan, X.W. 1-Methylcyclopropene reduces chilling injury of harvested okra (Hibiscus esculentus L.) pods. Sci. Hortic. 2012, 141, 42–46. [Google Scholar] [CrossRef]
- Cao, S.F.; Zheng, Y.H.; Wang, K.T.; Rui, H.J.; Tang, S.S. Effect of 1-methylcyclopropene treatment on chilling injury, fatty acid and cell wall polysaccharide composition in loquat fruit. J. Agric. Food Chem. 2009, 57, 8439–8443. [Google Scholar] [CrossRef] [PubMed]
- Collet, J.-F.; Messens, J. Structure, function, and mechanism of thioredoxin proteins. Antioxid. Redox Signal. 2010, 13, 1205–1216. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Holmgren, A. The thioredoxin antioxidant system. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Powis, G.; Mustacich, D.; Coon, A. The role of the redox protein thioredoxin in cell growth and cancer. Free Radic. Biol. Med. 2000, 29, 312–322. [Google Scholar] [CrossRef]
- Stadtman, E.R.; Van Remmen, H.; Richardson, A.; Wehr, N.B.; Levine, R.L. Methionine oxidation and aging. BBA-Proteins Proteom. 2005, 1703, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Lillig, C.H.; Holmgren, A. Thioredoxin and related molecules–from biology to health and disease. Antioxid. Redox Signal. 2007, 9, 25–47. [Google Scholar] [CrossRef] [PubMed]
- Ugarte, N.; Petropoulos, I.; Friguet, B. Oxidized mitochondrial protein degradation and repair in aging and oxidative stress. Antioxid. Redox Signal. 2010, 13, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Meyer, Y.; Belin, C.; Delorme-Hinoux, V.; Reichheld, J.-P.; Riondet, C. Thioredoxin and glutaredoxin systems in plants: Molecular mechanisms, crosstalks, and functional significance. Antioxid. Redox Signal. 2012, 17, 1124–1160. [Google Scholar] [CrossRef] [PubMed]
- Montrichard, F.; Alkhalfioui, F.; Yano, H.; Vensel, W.H.; Hurkman, W.J.; Buchanan, B.B. Thioredoxin targets in plants: The first 30 years. J. Proteom. 2009, 72, 452–474. [Google Scholar] [CrossRef] [PubMed]
- Balmer, Y.; Vensel, W.H.; Tanaka, C.K.; Hurkman, W.J.; Gelhaye, E.; Rouhier, N.; Jacquot, J.P.; Manieri, W.; Schuurmann, P.; Droux, M.; et al. Thioredoxin links redox to the regulation of fundamental processes of plant mitochondria. Proc. Natl. Acad. Sci. USA 2004, 101, 2642–2647. [Google Scholar] [CrossRef] [PubMed]
- Broin, M.; Rey, P. Potato plants lacking the CDSP32 plastidic thioredoxin exhibit overoxidation of the BAS1 2-cysteine peroxiredoxin and increased lipid peroxidation in thylakoids under photooxidative stress. Plant Physiol. 2003, 132, 1335–1343. [Google Scholar] [CrossRef] [PubMed]
- Laloi, C.; Mestres-Ortega, D.; Marco, Y.; Meyer, Y.; Reichheld, J.-P. The Arabidopsis cytosolic thioredoxin h5 gene induction by oxidative stress and its W-box-mediated response to pathogen elicitor. Plant Physiol. 2004, 134, 1006–1016. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.J.; Zhao, B.C.; Ge, W.N.; Zhang, Y.F.; Song, Y.; Sun, D.Y.; Guo, Y. An apoplastic H-type thioredoxin is involved in the stress response through regulation of the apoplastic reactive oxygen species in rice. Plant Physiol. 2011, 157, 1884–1899. [Google Scholar] [CrossRef] [PubMed]
- Lakhssassi, N.; Doblas, V.G.; Rosado, A.; Esteban del Valle, A.; Pose, D.; Jimenez, A.J.; Castillo, A.G.; Valpuesta, V.; Borsani, O.; Botella, M.A. The Arabidopsis tetratricopeptide thioredoxin-like gene family is required for osmotic stress tolerance and male sporogenesis. Plant Physiol. 2012, 158, 1252–1266. [Google Scholar] [CrossRef] [PubMed]
- Martí, M.C.; Florez-Sarasa, I.; Camejo, D.; Ribas-Carbó, M.; Lázaro, J.J.; Sevilla, F.; Jiménez, A. Response of mitochondrial thioredoxin PsTrxo1, antioxidant enzymes, and respiration to salinity in pea (Pisum sativum L.) leaves. J. Exp. Bot. 2011, 62, 3863–3874. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.J.; Ren, H.Y.; Liu, R.X.; Li, B.Y.; Wu, T.Q.; Sun, F.; Liu, H.M.; Wang, X.M.; Dong, H.S. An h-type thioredoxin functions in tobacco defense responses to two species of viruses and an abiotic oxidative stress. Mol. Plant-Microbe Interact. 2010, 23, 1470–1485. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, C.V.; Rey, P. Plant thioredoxins are key actors in the oxidative stress response. Trends Plant Sci. 2006, 11, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Daloso, D.M.; Mueller, K.; Obata, T.; Florian, A.; Tohge, T.; Bottcher, A.; Riondet, C.; Banat, L.; Carrari, F.; Nunes-Nesi, A.; et al. Thioredoxin, a master regulator of the tricarboxylic acid cycle in plant mitochondria. Proc. Natl. Acad. Sci. USA 2015, 112, E1392–E1400. [Google Scholar] [CrossRef] [PubMed]
- Thormaehlen, I.; Meitzel, T.; Groysman, J.; Oechsner, A.B.; von Roepenack-Lahaye, E.; Naranjo, B.; Cejudo, F.J.; Geigenberger, P. Thioredoxin f1 and NADPH-dependent thioredoxin reductase C have overlapping functions in regulating photosynthetic metabolism and plant growth in response to varying light conditions. Plant Physiol. 2015, 169, 1766–1786. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.S.; Kato, H.; Sasaki, K.; Imai, R. A cold-induced thioredoxin h of rice, OsTrx23, negatively regulates kinase activities of OsMPK3 and OsMPK6 in vitro. FEBS Lett. 2009, 583, 2734–2738. [Google Scholar] [CrossRef] [PubMed]
- Rorat, T.; Havaux, M.; Irzykowski, W.; Cuine, S.; Becuwe, N.; Rey, P. PSII-S gene expression, photosynthetic activity and abundance of plastid thioredoxin-related and lipid-associated proteins during chilling stress in Solanum species differing in freezing resistance. Physiol. Plant. 2001, 113, 72–78. [Google Scholar] [CrossRef]
- Yousfi, K.; Weiland, C.M.; Garcia, J.M. Effect of harvesting system and fruit cold storage on virgin olive oil chemical composition and quality of superintensive cultivated ‘Arbequina‘ olives. J. Agric. Food Chem. 2012, 60, 4743–4750. [Google Scholar] [CrossRef] [PubMed]
- Aghdam, M.S.; Bodbodak, S. Physiological and biochemical mechanisms regulating chilling tolerance in fruits and vegetables under postharvest salicylates and jasmonates treatments. Sci. Hortic. 2013, 156, 73–85. [Google Scholar] [CrossRef]
- Wang, Y.S.; Luo, Z.S.; Du, R.X.; Liu, Y.; Ying, T.J.; Mao, L.C. Effect of nitric oxide on antioxidative response and proline metabolism in banana during cold storage. J. Agric. Food Chem. 2013, 61, 8880–8887. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Jian, Q.J.; Jiang, Y.M.; Duan, X.W.; Qu, H.X. Enhanced chilling tolerance of banana fruit treated with malic acid prior to low-temperature storage. Postharvest Biol. Technol. 2016, 111, 209–213. [Google Scholar] [CrossRef]
- Aghdam, M.S.; Sevillano, L.; Flores, F.B.; Bodbodak, S. Heat shock proteins as biochemical markers for postharvest chilling stress in fruits and vegetables. Sci. Hortic. 2013, 160, 54–64. [Google Scholar] [CrossRef]
- Gelhaye, E.; Rouhier, N.; Jacquot, J.P. Evidence for a subgroup of thioredoxin h that requires GSH/Grx for its reduction. FEBS Lett. 2003, 555, 443–448. [Google Scholar] [CrossRef]
- Roos, G.; Garcia-Pino, A.; van belle, K.; Brosens, E.; Wahni, K.; Vandenbussche, G.; Wyns, L.; Loris, R.; Messens, J. The conserved active site proline determines the reducing power of Staphylococcus aureus thioredoxin. J. Mol. Biol. 2007, 368, 800–811. [Google Scholar] [CrossRef] [PubMed]
- Dangoor, I.; Peled-Zehavi, H.; Levitan, A.; Pasand, O.; Danon, A. A small family of chloroplast atypical thioredoxins. Plant Physiol. 2009, 149, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
- Shahpiri, A.; Svensson, B.; Finnie, C. From proteomics to structural studies of cytosolic/mitochondrial-type thioredoxin systems in barley seeds. Mol. Plant 2009, 2, 378–389. [Google Scholar] [CrossRef] [PubMed]
- Shankar, R.; Bhattacharjee, A.; Jain, M. Transcriptome analysis in different rice cultivars provides novel insights into desiccation and salinity stress responses. Sci. Rep. 2016, 6, 23719. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Yang, D.H.; Yao, H.; Bai, G.; Zhang, Y.H.; Xiao, B.G. iTRAQ-based quantitative proteomic analysis reveals proteomic changes in leaves of cultivated tobacco (Nicotiana tabacum) in response to drought stress. Biochem. Biophys. Res. Commun. 2016, 469, 768–775. [Google Scholar] [CrossRef] [PubMed]
- Qian, C.L.; He, Z.P.; Zhao, Y.Y.; Mi, H.B.; Chen, X.H.; Mao, L.C. Maturity-dependent chilling tolerance regulated by the antioxidative capacity in postharvest cucumber (Cucumis sativus L.) fruits. J. Sci. Food Agric. 2013, 93, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.L.; Cao, J.K.; Jiang, W.B.; Gu, Y.H.; Zhao, Y.M. Maturity-related chilling tolerance in mango fruit and the antioxidant capacity involved. J. Sci. Food Agric. 2009, 89, 304–309. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, W.; Jiang, Y.; Luo, Y.; Jiang, W.; Joyce, D. Expression of ethylene-related expansin genes in cool-stored ripening banana fruit. Plant Sci. 2006, 170, 962–967. [Google Scholar] [CrossRef]
- Moon, J.C.; Lee, S.M.; Shin, S.Y.; Chae, H.B.; Jung, Y.J.; Jung, H.S.; Lee, K.O.; Lee, J.R.; Lee, S.Y. Overexpression of Arabidopsis NADPH-dependent thioredoxin reductase C (AtNTRC) confers freezing and cold shock tolerance to plants. Biochem. Biophys. Res. Commun. 2015, 463, 1225–1229. [Google Scholar] [CrossRef] [PubMed]
- Müller, E.G.D. Thioredoxin deficiency in yeast prolongs S phase and shortens the G1 interval of the cell cycle. J. Biol. Chem. 1991, 266, 9194–9202. [Google Scholar] [PubMed]
- Traverso, J.A.; Vignols, F.; Cazalis, R.; Serrato, A.J.; Pulido, P.; Sahrawy, M.; Meyer, Y.; Javier Cejudo, F.; Chueca, A. Immunocytochemical localization of Pisum sativum Trxs f and m in non-photosynthetic tissues. J. Exp. Bot. 2008, 59, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Trijueque, J.; de Dios Barajas-Lopez, J.; Chueca, A.; Cazalis, R.; Sahrawy, M.; Jesus Serrato, A. Plastid thioredoxins f and m are related to the developing and salinity response of post-germinating seeds of Pisum sativum. Plant Sci. 2012, 188, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Espin, A.; Locato, V.; Camejo, D.; Schiermeyer, A.; De Gara, L.; Sevilla, F.; Jimenez, A. Over-expression of Trxo1 increases the viability of tobacco by-2 cells under H2O2 treatment. Ann. Bot. 2015, 116, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Mouaheb, N.; Thomas, D.; Verdoucq, L.; Monfort, P.; Meyer, Y. In vivo functional discrimination between plant thioredoxins by heterologous expression in the yeast Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1998, 95, 3312–3317. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Guo, Q.; Li, Q.P.; Ha, Y.M.; Li, X.P.; Chen, W.X. Impact of postharvest nitric oxide treatment on antioxidant enzymes and related genes in banana fruit in response to chilling tolerance. Postharvest Biol. Technol. 2014, 92, 157–163. [Google Scholar] [CrossRef]
- McGuire, R.G. Reporting of objective color measurements. Hortscience 1992, 27, 1254–1255. [Google Scholar]
- Wan, C.Y.; Wilkins, T.A. A modified hot borate method significantly enhances the yield of high-quality RNA from cotton (Gossypium hirsutum L.). Anal. Biochem. 1994, 223, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhong, H.Y.; Kuang, J.F.; Li, J.G.; Lu, W.J.; Chen, J.Y. Validation of reference genes for RT-qPCR studies of gene expression in banana fruit under different experimental conditions. Planta 2011, 234, 377–390. [Google Scholar] [CrossRef] [PubMed]
- Minet, M.; Dufour, M.E.; Lacroute, F. Complementation of Saccharomyces cerevisiae auxotrophic mutants by arabidopsis thaliana cdnas. Plant J. 1992, 2, 417–422. [Google Scholar] [PubMed]
- Ito, H.; Fukuda, Y.; Murata, K.; Kimura, A. Transformation of intact yeast cells treated with alkali cations. J. Bacteriol. 1983, 153, 163–168. [Google Scholar] [PubMed]






| Name | Nucleotide Sequence (5’-3’) |
|---|---|
| Trx-For1 | CGACTTCGCCGCCACNTGGTGYGGNC |
| Trx-For2 | CGTGGAGTTCGTCAAGATCGAYGTNGAYGA |
| MaTrx6-5RACE1 | CGGAGTACTGGGCGGCCATC |
| MaTrx6-5RACE2 | CTCCACGAAGCGGCACGGC |
| MaTrx9-5RACE1 | TGTTCCTCGTGGTTTAGTTG |
| MaTrx9-5RACE2 | ATCCTCAGCCGACTTGATA |
| MaTrx12-5RACE1 | GTTGCTGAGATGTCCCACGA |
| MaTrx12-5RACE2 | GCCACAACTATCCTGCCATC |
| eGFP-MaTrx6-Sal I-F | AGTCGACATGGGTGGTTTCTTCTCCAGCC |
| eGFP-MaTrx6-Sal I-R | AGTCGACGGTGTTGATCTGCTCTTGGATCC |
| eGFP-MaTrx9-Mlu I-F | CGACGCGTATGGCGGATGCTTTGGCTC |
| eGFP-MaTrx9-Mlu I-R | CGACGCGTTCTACCGGCTGCGGC |
| eGFP-MaTrx12-Sal I-F | AGTCGACATGGGAAACTGCATAAGAAAGAATGTGAG |
| eGFP-MaTrx12-Sal I-R | AGTCGACTCGTAGCGGGAGAGTAGCATC |
| MaTrx6-F | AGGAGGTGGGCAGAATCGT |
| MaTrx6-R | TGGTGGCGGTAATACAGACAG |
| MaTrx9-F | TCACCTACACAAGAACGCCT |
| MaTrx9-R | CCACATACATAACCAATAAGCAG |
| MaTrx12-F | TCGTGGGACATCTCAGCAAC |
| MaTrx12-R | CTTTCTTCATCGTAGCGGGA |
| Actin-F | TGGTATGGAAGCCGCTGGTA |
| Actin-R | TCTGCTGGAATGTGCTGAGG |
| pFL61-MaTrx6-Not I-F | TTGCGGCCGCATGGGTGGTTTCTTCTCCAGCC |
| pFL61-MaTrx6-Not I-R | TTGCGGCCGCTTAGGTGTTGATCTGCTCCTGGATCC |
| pFL61-MaTrx9-Not I-F | TTGCGGCCGCATGGCGGATGCTTTGGCTC |
| pFL61-MaTrx9-Not I-R | TTGCGGCCGCTCATCTACCGGCTGCGGCAAAAG |
| pFL61-MaTrx12-Not I-F | TTGCGGCCGCATGGGAAACTGCATAAGAAAG |
| pFL61-MaTrx12-Not I-R | TTGCGGCCGCTCATCGTAGCGGGAGAGTAGC |
| FLDR | CTATTATTTTAGCGTAAAGGATGG |
| FLGA | CTCTTTTTTACAGATCATCAAGG |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, F.; Li, Q.; Yan, H.; Zhang, D.; Jiang, G.; Jiang, Y.; Duan, X. Characteristics of Three Thioredoxin Genes and Their Role in Chilling Tolerance of Harvested Banana Fruit. Int. J. Mol. Sci. 2016, 17, 1526. https://doi.org/10.3390/ijms17091526
Wu F, Li Q, Yan H, Zhang D, Jiang G, Jiang Y, Duan X. Characteristics of Three Thioredoxin Genes and Their Role in Chilling Tolerance of Harvested Banana Fruit. International Journal of Molecular Sciences. 2016; 17(9):1526. https://doi.org/10.3390/ijms17091526
Chicago/Turabian StyleWu, Fuwang, Qing Li, Huiling Yan, Dandan Zhang, Guoxiang Jiang, Yueming Jiang, and Xuewu Duan. 2016. "Characteristics of Three Thioredoxin Genes and Their Role in Chilling Tolerance of Harvested Banana Fruit" International Journal of Molecular Sciences 17, no. 9: 1526. https://doi.org/10.3390/ijms17091526