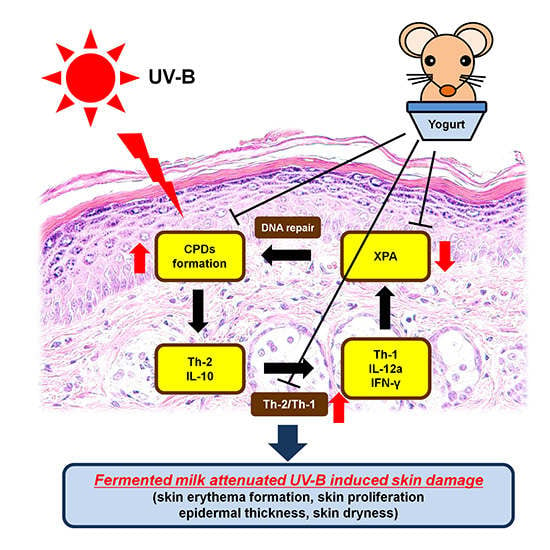
Exopolysaccharides Isolated from Milk Fermented with Lactic Acid Bacteria Prevent Ultraviolet-Induced Skin Damage in Hairless Mice
Abstract
:1. Introduction
2. Results
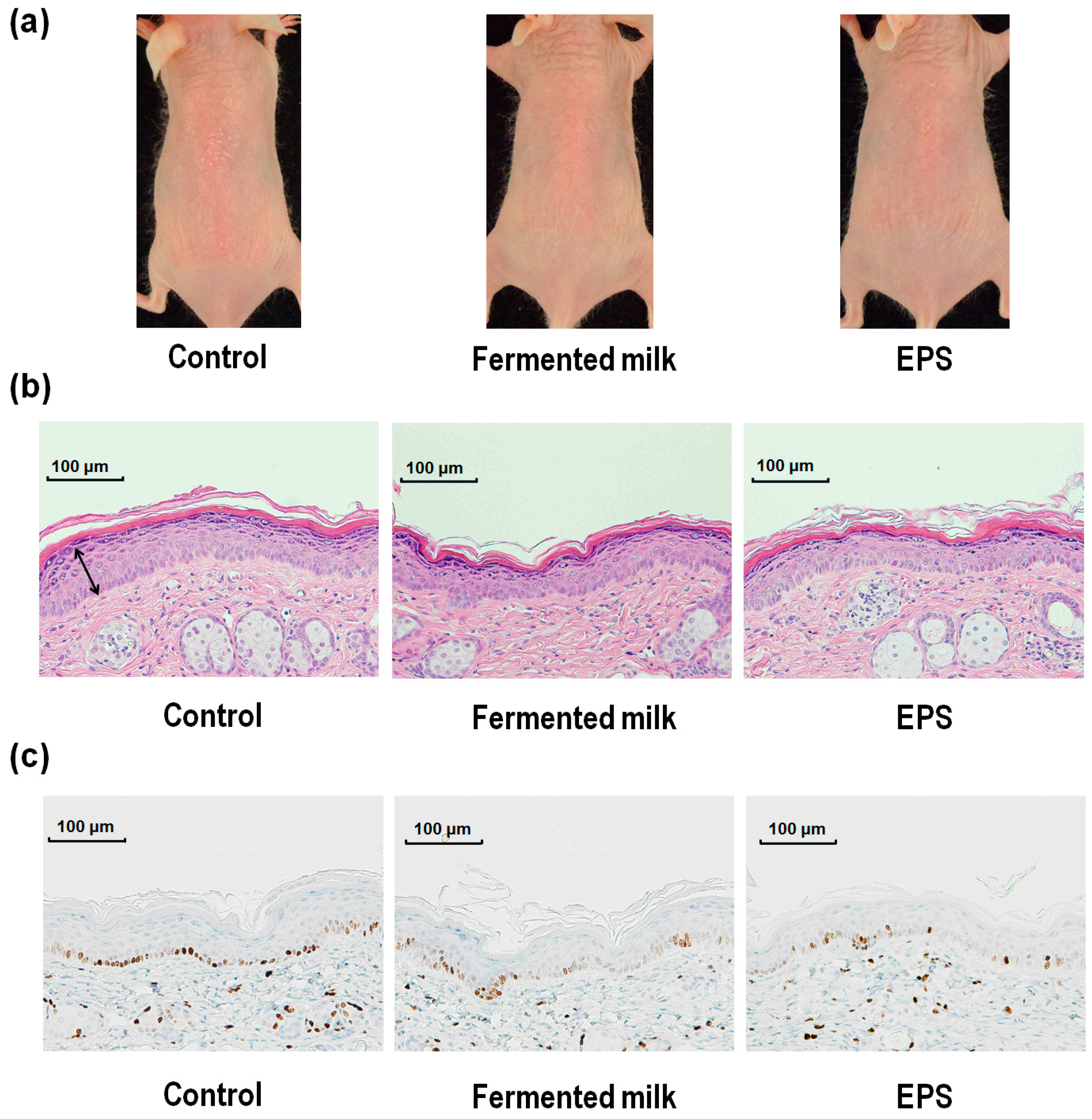
2.1. Effect of Fermented Milk and Exopolysaccharide (EPS) on Skin Erythema Formation and Proliferation in Hairless Mice Following UV-B Exposure
2.2. Effect of Fermented Milk and EPS on Skin Barrier Function in Hairless Mice Following UV-B Exposure
2.3. Effect of Fermented Milk and EPS on DNA Damage, XPA mRNA Levels and the Ratio of Th-2/Th-1 Cytokines in Hairless Mice Exposed to UV-B
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Preparation of Samples
4.3. Animal Study
4.4. Measurement of Transepidermal Water Loss (TEWL) and Stratum Corneum (SC) Water Content
4.5. Analysis of Skin Erythema Formation
4.6. Histological Analysis
4.7. Immunohistochemistry
4.8. Total RNA Isolation, cDNA Synthesis and Quantitative Real-Time Reverse Transcription Polymerase Chain Reaction Analysis
4.9. Analysis of Cyclobutane Pyrimidine Dimers
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Ichihashi, M.; Ueda, M.; Budiyanto, A.; Bito, T.; Oka, M.; Fukunaga, M.; Tsuru, K.; Horikawa, T. UV-induced skin damage. Toxicology 2003, 189, 21–39. [Google Scholar] [CrossRef]
- Fernandez-Garcia, E. Skin protection against UV light by dietary antioxidants. Food Funct. 2014, 5, 1994–2003. [Google Scholar] [CrossRef] [PubMed]
- Jurkiewicz, B.A.; Bissett, D.L.; Buettner, G.R. Effect of topically applied tocopherol on ultraviolet radiation-mediated free radical damage in skin. J. Investig. Dermatol. 1995, 104, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.A.; Katiyar, S.K. Skin photoprotection by natural polyphenols: Anti-inflammatory, antioxidant and DNA repair mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Sawada, J.; Morita, H.; Tanaka, A.; Salminen, S.; He, F.; Matsuda, H. Ingestion of heat-treated Lactobacillus rhamnosus GG prevents development of atopic dermatitis in NC/Nga mice. Clin. Exp. Allergy 2007, 37, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Peguet-Navarro, J.; Dezutter-Dambuyant, C.; Buetler, T.; Leclaire, J.; Smola, H.; Blum, S.; Bastien, P.; Breton, L.; Gueniche, A. Supplementation with oral probiotic bacteria protects human cutaneous immune homeostasis after UV exposure-double blind, randomized, placebo controlled clinical trial. Eur. J. Dermatol. 2008, 18, 504–511. [Google Scholar] [PubMed]
- Gueniche, A.; Benyacoub, J.; Buetler, T.M.; Smola, H.; Blum, S. Supplementation with oral probiotic bacteria maintains cutaneous immune homeostasis after UV exposure. Eur. J. Dermatol. 2006, 16, 511–517. [Google Scholar] [PubMed]
- Kripke, M.L.; Cox, P.A.; Alas, L.G.; Yarosh, D.B. Pyrimidine dimers in DNA initiate systemic immunosuppression in UV-irradiated mice. Proc. Natl. Acad. Sci. USA 1992, 89, 7516–7520. [Google Scholar] [CrossRef] [PubMed]
- Wakasugi, M.; Shimizu, M.; Morioka, H.; Linn, S.; Nikaido, O.; Matsunaga, T. Damaged DNA-binding protein DDB stimulates the excision of cyclobutane pyrimidine dimers in vitro in concert with XPA and replication protein A. J. Biol. Chem. 2001, 276, 15434–15440. [Google Scholar] [CrossRef] [PubMed]
- Hanneman, K.K.; Cooper, K.D.; Baron, E.D. Ultraviolet immunosuppression: Mechanisms and consequences. Dermatol. Clin. 2006, 24, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Bao, S.; Reeve, V.E. Modulation of IL-10, IL-12, and IFN-γ in the epidermis of hairless mice by UVA (320–400 nm) and UVB (280–320 nm) radiation. J. Investig. Dermatol. 1999, 113, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Asadullah, K.; Sterry, W.; Stephanek, K.; Jasulaitis, D.; Leupold, M.; Audring, H.; Volk, H.D.; Docke, W.D. IL-10 is a key cytokine in psoriasis. Proof of principle by IL-10 therapy: A new therapeutic approach. J. Clin. Investig. 1998, 101, 783–794. [Google Scholar] [CrossRef] [PubMed]
- Nishigori, C.; Yarosh, D.B.; Ullrich, S.E.; Vink, A.A.; Bucana, C.D.; Roza, L.; Kripke, M.L. Evidence that DNA damage triggers interleukin 10 cytokine production in UV-irradiated murine keratinocytes. Proc. Natl. Acad. Sci. USA 1996, 93, 10354–10359. [Google Scholar] [CrossRef] [PubMed]
- Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat. Rev. Immunol. 2003, 3, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, D.A.; Walterscheid, J.P.; Ullrich, S.E. Reversal of ultraviolet radiation-induced immune suppression by recombinant interleukin-12: Suppression of cytokine production. Immunology 2000, 101, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Cantabrana, C.; Lopez, P.; Gueimonde, M.; de Los Reyes-Gavilan, C.G.; Suarez, A.; Margolles, A.; Ruas-Madiedo, P. Immune Modulation Capability of Exopolysaccharides Synthesised by Lactic Acid Bacteria and Bifidobacteria. Probiotics Antimicrob. Proteins 2012, 4, 227–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, D.L.; Jen, J.; Cleaver, J.E. Relative induction of cyclobutane dimers and cytosine photohydrates in DNA irradiated in vitro and in vivo with ultraviolet-C and ultraviolet-B light. Photochem. Photobiol. 1991, 54, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.; Matsunaga, T.; Bito, T.; Nikaido, O.; Ichihashi, M. Higher cyclobutane pyrimidine dimer and (6-4) photoproduct yields in epidermis of normal humans with increased sensitivity to ultraviolet B radiation. Photodermatol. Photoimmunol. Photomed. 1996, 12, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.P.; Lou, Y.R.; Yen, P.; Mitchell, D.; Huang, M.T.; Conney, A.H. Time course for early adaptive responses to ultraviolet B light in the epidermis of SKH-1 mice. Cancer Res. 1999, 59, 4591–4602. [Google Scholar] [PubMed]
- De Vries, A.; van Oostrom, C.T.; Hofhuis, F.M.; Dortant, P.M.; Berg, R.J.; de Gruijl, F.R.; Wester, P.W.; van Kreijl, C.F.; Capel, P.J.; van Steeg, H.; et al. Increased susceptibility to ultraviolet-B and carcinogens of mice lacking the DNA excision repair gene XPA. Nature 1995, 377, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Rivas, J.M.; Ullrich, S.E. Systemic suppression of delayed-type hypersensitivity by supernatants from UV-irradiated keratinocytes. An essential role for keratinocyte-derived IL-10. J. Immunol. 1992, 149, 3865–3871. [Google Scholar] [PubMed]
- Shreedhar, V.; Giese, T.; Sung, V.W.; Ullrich, S.E. A cytokine cascade including prostaglandin E2, IL-4, and IL-10 is responsible for UV-induced systemic immune suppression. J. Immunol. 1998, 160, 3783–3789. [Google Scholar] [PubMed]
- Schwarz, A.; Grabbe, S.; Aragane, Y.; Sandkuhl, K.; Riemann, H.; Luger, T.A.; Kubin, M.; Trinchieri, G.; Schwarz, T. Interleukin-12 prevents ultraviolet B-induced local immunosuppression and overcomes UVB-induced tolerance. J. Investig. Dermatol. 1996, 106, 1187–1191. [Google Scholar] [CrossRef] [PubMed]
- Kalliomaki, M.; Salminen, S.; Arvilommi, H.; Kero, P.; Koskinen, P.; Isolauri, E. Probiotics in primary prevention of atopic disease: A randomised placebo-controlled trial. Lancet 2001, 357, 1076–1079. [Google Scholar] [CrossRef]
- Uemura, J.; Itoh, T.; Kaneko, T.; Noda, K. Chemical characterization of exocellular polysaccharide from Lactobacillus delbrueckii subsp. bulgaricus OLL1073R-1. Milchwissenschaft 1998, 53, 443–446. [Google Scholar]
- Nishimura-Uemura, J.; Kitazawa, H.; Kawai, Y.; Itoh, T.; Oda, M.; Saito, T. Functional alteration of murine macrophages stimulated with extracellular polysaccharides from Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. Food Microbiol. 2003, 20, 267–273. [Google Scholar] [CrossRef]
- Kitazawa, H.; Itoh, T.; Tomioka, Y.; Mizugaki, M.; Yamaguchi, T. Induction of IFN-γ and IL-1α production in macrophages stimulated with phosphopolysaccharide produced by Lactococcus lactis ssp. cremoris. Int. J. Food Microbiol. 1996, 31, 99–106. [Google Scholar] [CrossRef]
- Byeon, S.W.; Pelley, R.P.; Ullrich, S.E.; Waller, T.A.; Bucana, C.D.; Strickland, F.M. Aloe barbadensis extracts reduce the production of interleukin-10 after exposure to ultraviolet radiation. J. Investig. Dermatol. 1998, 110, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Strickland, F.M.; Darvill, A.; Albersheim, P.; Eberhard, S.; Pauly, M.; Pelley, R.P. Inhibition of UV-induced immune suppression and interleukin-10 production by plant oligosaccharides and polysaccharides. Photochem. Photobiol. 1999, 69, 141–147. [Google Scholar] [CrossRef]
- Cerning, J.; Renard, C.M.; Thibault, J.F.; Bouillanne, C.; Landon, M.; Desmazeaud, M.; Topisirovic, L. Carbon Source Requirements for Exopolysaccharide Production by Lactobacillus casei CG11 and Partial Structure Analysis of the Polymer. Appl. Environ. Microbiol. 1994, 60, 3914–3919. [Google Scholar] [PubMed]
- Oba, C.; Ohara, H.; Morifuji, M.; Ito, K.; Ichikawa, S.; Kawahata, K.; Koga, J. Collagen hydrolysate intake improves the loss of epidermal barrier function and skin elasticity induced by UVB irradiation in hairless mice. Photodermatol. Photoimmunol. Photomed. 2013, 29, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Oba, C.; Morifuji, M.; Ichikawa, S.; Ito, K.; Kawahata, K.; Yamaji, T.; Asami, Y.; Itou, H.; Sugawara, T. Dietary milk sphingomyelin prevents disruption of skin barrier function in hairless mice after UV-B irradiation. PLoS ONE 2015, 24, e0136377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative Ct method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]




© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morifuji, M.; Kitade, M.; Fukasawa, T.; Yamaji, T.; Ichihashi, M. Exopolysaccharides Isolated from Milk Fermented with Lactic Acid Bacteria Prevent Ultraviolet-Induced Skin Damage in Hairless Mice. Int. J. Mol. Sci. 2017, 18, 146. https://doi.org/10.3390/ijms18010146
Morifuji M, Kitade M, Fukasawa T, Yamaji T, Ichihashi M. Exopolysaccharides Isolated from Milk Fermented with Lactic Acid Bacteria Prevent Ultraviolet-Induced Skin Damage in Hairless Mice. International Journal of Molecular Sciences. 2017; 18(1):146. https://doi.org/10.3390/ijms18010146
Chicago/Turabian StyleMorifuji, Masashi, Masami Kitade, Tomoyuki Fukasawa, Taketo Yamaji, and Masamitsu Ichihashi. 2017. "Exopolysaccharides Isolated from Milk Fermented with Lactic Acid Bacteria Prevent Ultraviolet-Induced Skin Damage in Hairless Mice" International Journal of Molecular Sciences 18, no. 1: 146. https://doi.org/10.3390/ijms18010146
APA StyleMorifuji, M., Kitade, M., Fukasawa, T., Yamaji, T., & Ichihashi, M. (2017). Exopolysaccharides Isolated from Milk Fermented with Lactic Acid Bacteria Prevent Ultraviolet-Induced Skin Damage in Hairless Mice. International Journal of Molecular Sciences, 18(1), 146. https://doi.org/10.3390/ijms18010146