GhMAP3K65, a Cotton Raf-Like MAP3K Gene, Enhances Susceptibility to Pathogen Infection and Heat Stress by Negatively Modulating Growth and Development in Transgenic Nicotiana benthamiana
Abstract
:1. Introduction
2. Results
2.1. Isolation and Sequence Analysis of GhMAP3K65
2.2. GhMAP3K65 is Localized to the Cytoplasm and Nucleus
2.3. Relative Expression Profiles of GhMAP3K65
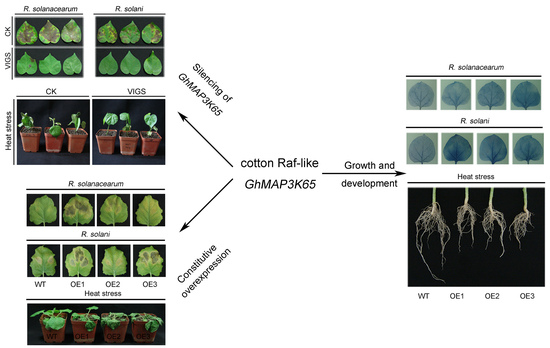
2.4. Effects of GhMAP3K65 Silencing on Pathogen Infection and Heat Stress in Cotton
2.5. Overexpression of GhMAP3K65 Enhanced Susceptibility to Pathogen Infection and Heat Stress
2.6. Overexpression of GhMAP3K65 Activated Transcription of Defence-Related Genes
2.7. Overexpression of GhMAP3K65 Impaired Ligin Biosynthesis and Stomatal Immunity in the Leaves of N. benthamiana
2.8. Overexpression of GhMAP3K65 Repressed Growth and Development of the Root System
3. Discussion
4. Materials and Methods
4.1. Plant Growth and Stress Treatments
4.2. Isolation of GhMAP3K65 Gene, Vector Construction, and Genetic Transformation
4.3. Subcellular Localization of GhMAP3K65
4.4. RNA Extraction and Quantitative PCR
4.5. Virus-Induced Gene Silencing (VIGS) of GhMAP3K65 in Cotton
4.6. Pathogen Infection
4.7. Heat Stress Treatment
4.8. Histochemical Staining
4.9. Structural Defects in Leaves
4.10. Lignin Content
4.11. Determination of Root Vitality by Triphenyl Tetrazolium Chloride (TTC) Method
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dangl, J.L.; Jones, J.D. Plant pathogens and integrated defense responses to infection. Nature 2001, 411, 826–833. [Google Scholar] [CrossRef] [PubMed]
- Virk, N.; Li, D.; Tian, L.; Huang, L.; Hong, Y.; Li, X.; Zhang, Y.; Liu, B.; Zhang, H.; Song, F. Arabidopsis Raf-like mitogen-activated protein kinase kinase kinase gene Raf43 is required for tolerance to multiple abiotic stresses. PLoS ONE 2015, 10, eo133975. [Google Scholar] [CrossRef] [PubMed]
- Nishihama, R.; Banno, H.; Shibata, W.; Hirano, K.; Nakashima, M.; Usami, S.; Machida, Y. Plant homologues of components of MAPK (mitogen-activated protein kinase) signal pathways in yeast and animal cells. Plant Cell Physiol. 1995, 36, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.S.; Tuteja, R.; Tuteja, N. Signalling through MAP kinase networks in plants. Arch. Biochem. Biophys. 2006, 452, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Opdenakker, K.; Remans, T.; Vangronsveld, J.; Cuypers, A. Mitogen-activated protein (MAP) kinases in plant metal stress: Regulation and responses in comparison to other biotic and abiotic stresses. Int. J. Mol. Sci. 2012, 13, 7828–7853. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Wang, J.; Wang, D.; Fan, W.; Wang, S.; Ye, W. The MAPKKK gene family in Gossypium raimondii: Genome-wide identification, classification and expression analysis. Int. J. Mol. Sci. 2013, 14, 18740–18757. [Google Scholar] [CrossRef] [PubMed]
- Taj, G.; Agarwal, P.; Grant, M.; Kumar, A. MAPK machinery in plants: Recognition and response to different stresses through multiple signal transduction pathways. Plant Signal. Behav. 2010, 5, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.A.; Agrawal, G.K.; Rakwal, R.; Han, K.S.; Kim, K.N.; Yun, C.H.; Heu, S.; Park, S.Y.; Lee, Y.H.; Jwa, N.S. Molecular cloning and mRNA expression analysis of novel rice (Oryzasativa L.) MAPK kinase kinase, OsEDR1, an ortholog of Arabidopsis AtEDR1, reveal its role in defense/stress signalling pathways and development. Biochem. Biophys. Res. Commun. 2003, 300, 868–876. [Google Scholar] [CrossRef]
- Lin, Z.; Alexander, L.; Hackett, R.; Grierson, D. LeCTR2, a CTR1-like protein kinase from tomato, plays a role in ethylene signalling, development and defense. Plant J. 2008, 54, 1083–1093. [Google Scholar] [CrossRef] [PubMed]
- Sasayama, D.; Matsuoka, D.; Oka, M.; Shitamichi, N.; Furuya, T.; Azuma, T.; Itoh, K.; Nanmori, T. MAP3Kδ4, an Arabidopsis Raf-like MAP3K, regulates plant growth and shoot branching. Plant Biotechnol. 2011, 28, 463–470. [Google Scholar] [CrossRef]
- Shitamichi, N.; Matsuoka, D.; Furuya, T.; Nanmori, T. Overexpression of MAP3Kδ4, an ABA-inducible Raf-like MAP3K that confers salt tolerance in Arabidopsis. Plant Biotechnol. 2013, 30, 111–118. [Google Scholar] [CrossRef]
- Chen, X.; Wang, J.; Zhu, M.; Jia, H.; Liu, D.; Hao, L.; Guo, X. A cotton Raf-like MAP3K gene, GhMAP3K40, mediates reduced tolerance to biotic and abiotic stress in Nicotiana Benthamiana by negatively regulating growth and development. Plant Sci. 2015, 240, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Rizhsky, L.; Liang, H.; Mittler, R. The combined effect of drought stress and heat shock on gene expression in tobacco. Plant Physiol. 2002, 130, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Noutoshi, Y.; Ito, T.; Seki, M.; Nakashita, H.; Yoshida, S.; Marco, Y.; Shirasu, K.; Shinozaki, K. A signal amino acid insertion in the WRKY domain of the Arabidopsis TIR-NBS-LRR-WRKY-type disease resistance protein SLH1 (sensitive to low humidity 1) causes activation of defense responses and hypersensitive cell death. Plant J. 2005, 43, 873–888. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Menke, F.L.; Yoshioka, K.; Moder, W.; Shirano, Y.; Klessig, D.F. High humidity suppresses ssi4-mediated cell death and disease resistance upstream of MAP kinase activation, H2O2 production and defense gene expression. Plant J. 2004, 39, 920–932. [Google Scholar] [CrossRef] [PubMed]
- Loake, G.; Grant, M. Salicylic acid in plant defense—The players and protagonists. Curr. Opin. Plant Biol. 2007, 10, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, O.; Solano, R. Molecular players regulating the jasmonate signalling network. Curr. Opin. Plant Biol. 2005, 8, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Broekaert, W.F.; Delaure, S.L.; De Bolle, M.F.; Cammue, B.P. The role of ethylene in host-pathogen interactions. Annu. Rev. Phytopathol. 2016, 44, 393–416. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.M.; Cristescu, S.M.; Miersch, O.; Harren, F.J.; Wasternack, C.; Mur, L.A. Jasmonates act with salicylic acid to confer basal thermotolerance in Arabidopsis thaliana. New Phytol. 2009, 182, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Dang, F.F.; Wang, Y.N.; Yu, L.; Eulgem, T.; Lai, Y.; Liu, Z.Q.; Wang, X.; Qiu, A.L.; Zhang, T.X.; Lin, J.; et al. CaWRKY40, a WRKY protein of pepper, plays an important role in the regulation of tolerance to heat stress and resistance to Ralstonia solanacearum infection. Plant Cell Environ. 2013, 36, 757–774. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.E.; Pennell, R.I.; Meijer, P.J.; Ishikawa, A.; Dixon, R.A.; Lamb, C. Reactive oxygen intermediates mediate a systemic signal network in the establishment of plant immunity. Cell 1998, 92, 773–784. [Google Scholar] [CrossRef]
- Yoshioka, H.; Numata, N.; Nakajima, K.; Katou, S.; Kawakita, K.; Rowland, O.; Jones, J.D.; Doke, N. Nicotiana benthamiana gp91phox homologs NbrbohA and NbrbohB participate in H2O2 accumulation and resistance to Phytophthora infestans. Plant Cell 2003, 15, 706–718. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Goodwin, P.H.; Hsiang, T. The role of ethylene during the infection of Nicotiana tabacum by Colletotrichum destructivum. J. Exp. Bot. 2003, 54, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Sohn, S.I.; Kim, Y.H.; Kim, B.R.; Lee, S.Y.; Lim, C.K.; Hur, J.H.; Lee, J.Y. Transgenic tobacco expressing the hrpNEP gene from Erwinia pyrifoliae triggers defense responses against Botrytis cinerea. Mol. Cells 2007, 24, 232–239. [Google Scholar] [PubMed]
- Yan, Y.; Jia, H.; Wang, F.; Wang, C.; Liu, S.; Guo, X. Overexpression of GhWRKY27a reduces tolerance to drought stress and resistance to Rhizoctonia solani infection in transgenic Nicotiana benthamiana. Front. Physiol. 2015, 6, 265. [Google Scholar] [CrossRef] [PubMed]
- Wolters, H.; Jürgens, G. Survival of the flexible: Hormonal growth control and adaptation in plant development. Nat. Rev. Genet. 2009, 10, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Wu, W.; Abrams, S.R.; Cutler, A.J. The relationship of drought-related gene expression in Arabidopsis thaliana to hormonal and environmental factors. J. Exp. Bot. 2008, 59, 2991–3007. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Fujita, Y.; Noutoshi, Y.; Takahashi, F.; Narusaka, Y.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Crosstalk between abiotic and biotic stress responses: A current view from the points of convergence in the stress signalling networks. Curr. Opin. Plant Biol. 2006, 9, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.P.; Jermakow, A.M.; Swain, S.M. Gibberellins are required for seed development and pollen tube growth in Arabidopsis. Plant Cell 2002, 14, 3133–3147. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, D.; Hwang, I. A sophisticated network of signalling pathways regulates stomatal defenses to bacterial pathogens. Mol. Plant 2014, 129. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, L.; Lu, W.; Wang, X.; Wu, C.A.; Guo, X. Overexpression of cotton GhMKK4 enhances disease susceptibility and affects abscisic acid, gibberellin and hydrogen peroxide signalling in transgenic Nicotiana benthmiana. Mol. Plant Pathol. 2014, 15, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, Y.; Lu, W.; Meng, F.; Wu, C.A.; Guo, X. Cotton GhMKK5 affects disease resistance, induces HR-like cell death, and reduces the tolerance to salt and drought stress in transgenic Nicotiana benthamiana. J. Exp. Bot. 2012, 63, 3935–3951. [Google Scholar] [CrossRef] [PubMed]
- Furuya, T.; Matsuoka, D.; Nanmori, T. Membrane rigidification functions upstream of the MEKK-MEKK2-MEKK4 cascade during cold acclimation in Arabidopsis thaliana. FEBS Lett. 2014, 588, 2025–2030. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zu, X.; Zhang, H.; Wu, L.; Xi, Z.; Chen, Y. Overexpression of ZmMAPK1 enhances drought and heat stress in transgenic Arabidopsis thaliana. Plant Mol. Biol. 2015, 88, 429–443. [Google Scholar] [CrossRef] [PubMed]
- Ning, J.; Li, X.; Hicks, L.M.; Xiong, L. A Raf-like MAPKKK gene DSM1 mediates drought resistance through reactive oxygen species scavenging in rice. Plant Physiol. 2010, 152, 876–890. [Google Scholar] [CrossRef] [PubMed]
- Czernic, P.; Huang, H.C.; Marco, Y. Characterization of hsr201 and hsr515, two tobacco genes preferentially expressed during the hypersensitive reaction provoked by phytopathogenic bacteria. Plant Mol. Biol. 1996, 31, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Draper, J. Salicylate, superoxide synthesis and cell suicide in plant defence. Trends Plant Sci. 1997, 2, 162–165. [Google Scholar] [CrossRef]
- About-Attia, M.A.; Wang, X.; Nashaat Al-Attala, M.; Xu, Q.; Zhan, G.; Kang, Z. TaMDAR6 acts as a negative regulator of plant cell death and participates indirectly in stomatal regulation during the wheat stripe rust-fungus interaction. Physiol. Plant 2016, 156, 262–277. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 2–16. [Google Scholar] [CrossRef]
- Govrin, E.M.; Levine, A. The hypersensitive response facilitates plant infection by the necrotrophic pathogen Botrytis cinerea. Curr. Biol. 2000, 10, 751–757. [Google Scholar] [CrossRef]
- Lujan, R.; Lledias, F.; Martinez, L.M.; Barreto, R.; Cassab, G.I.; Nieto-Sotelo, J. Small heat-shock proteins and leaf cooling capacity account for the unusual heat tolerance of the central spike leaves in Agave tequilana var. Weber. Plant Cell Environ. 2009, 32, 1791–1803. [Google Scholar] [CrossRef] [PubMed]
- Locato, V.; Gadaleta, C.; De Gara, L.; De Pinto, M.C. Production of reactive species and modulation of antioxidant network in response to heat shock: A critical balance for cell fate. Plant Cell Environ. 2008, 31, 1606–1619. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.Y.; Liu, W.; Zhang, S.Y. Different responses of plant growth and antioxidant system to the combination of cadmium and heat stress in transgenic and non-transgenic rice. J. Integr. Plant Biol. 2009, 51, 942–950. [Google Scholar] [CrossRef] [PubMed]
- Khanna-Chopra, R.; Sabarinath, S. Heat-stable chloroplastic Cu/Zn superoxide dismutase in Chenopodium murale. Biochem. Biophys. Res. Commun. 2004, 320, 1187–1192. [Google Scholar] [CrossRef] [PubMed]
- Jaskiewicz, M.; Conrath, U.; Peterhansel, C. Chromatin modification acts as a memory for systemic acquired resistance in the plant stress. EMBO Rep. 2011, 12, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Sani, E.; Herzyk, P.; Perrella, G.; Colot, V.; Amtmann, A. Hyperosmotic priming of Arabidopsis seedlings establishes a long-term somatic memory accompanied by specific changes of the epigenome. Genome Biol. 2013, 14, R59. [Google Scholar] [CrossRef] [PubMed]
- Bäurle, I. Plant Heat Adaptation: Priming in response to heat stress. F1000Research 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Narusaka, Y.; Narusaka, M.; Seki, M.; Umezawa, T.; Ishida, J.; Nakajima, M.; Enju, A.; Shinozaki, K. Crosstalk in the responses to abiotic and biotic stresses in Arabidopsis: Analysis of gene expression in cytochrome P450 gene superfamily by cDNA microarray. Plant Mol. Biol. 2004, 55, 327–342. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Cai, X.; Zheng, Z. High humidity represses cf-4/Avr4- and cf-9/AVR9-dependent hypersensitive cell death and defense gene expression. Planta 2005, 222, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Glazebrook, J. Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef] [PubMed]
- Devadas, S.K.; Enyedi, A.; Raina, R. The Arabidopsis hrl1 mutation reveals novel overlapping roles for salicylic acid, jasmonic acid and ethylene signalling in cell death and defence against pathogens. Plant J. 2002, 30, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Mur, L.A.; Kenton, P.; Atzorn, R.; Miersch, O.; Wasternack, C. The outcomes of concentration-specific interactions between salicylate and jasmonate signalling include synergy, antagonism, and oxidative stress leading to cell death. Plant Physiol. 2006, 140, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Schramm, F.; Larkindale, J.; Kiehlmann, E.; Ganguli, A.; Englich, G.; Vierling, E.; von Koskull-Doring, P. A cascade of transcription factor DREB2A and heat stress transcription factor HsfA3 regulates the heat stress response of Arabidopsis. Plant J. 2008, 53, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Collinge, D.B. Cell wall appositions: The first line of defense. J. Exp. Bot. 2009, 60, 351–352. [Google Scholar] [CrossRef] [PubMed]
- Frei, M. Lignin: Characterization of a multifaceted crop component. Sci. World J. 2013, 2013, 436517. [Google Scholar] [CrossRef] [PubMed]
- Vance, C.P.; Kirk, T.K.; Sherwood, R.T. Ligniication as a mechanism of disease resistance. Annu. Rev. Phytopathol. 1980, 18, 259–288. [Google Scholar] [CrossRef]
- Moura, J.C.; Bonine, C.A.; de Oliveira Fernandes Viana, J.; Dornelas, M.C.; Mazzafera, P. Abiotic and biotic stresses and changes in the lignin content and composition in plants. J. Integer. Plant Biol. 2010, 52, 360–376. [Google Scholar] [CrossRef] [PubMed]
- Derikvand, M.M.; Sierra, J.B.; Ruel, K.; Pollet, B.; Do, C.T.; Thévenin, J.; Buffard, D.; Jouanin, L.; Lapierre, C. Redirection of the phenylpropanoid pathway to feruloyl malate in Arabidopsis mutants deficient for cinnamoyl-CoA reductase 1. Planta 2008, 227, 943–956. [Google Scholar] [CrossRef] [PubMed]
- Do, C.T.; Pollet, B.; Thévenin, J.; Sibout, R.; Denoue, D.; Barrière, Y.; Lapierre, C.; Jouanin, L. Both caffeoyl Coenzyme A 3-O-methyltransferase 1 and caffeic acid O-methyltransferase 1 are involved in redundant functions for lignin, flavonoids and sinapoyl malate biosynthesis in Arabidopsis. Planta 2007, 226, 1117–1129. [Google Scholar] [CrossRef] [PubMed]
- Gmenez-lbanez, S.; Boter, M.; Ortigosa, A.; García-Casado, G.; Chini, A.; Lewsey, M.G.; Ecker, J.R.; Ntoukakis, V.; Solano, R. JAZ2 controls stomata dynamics during bacterial invasion. New Phytol. 2017, 213, 1378–1392. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Huang, B. Root physiological factors involved in cool-season grass response to high soil temperature. Environ. Exp. Bot. 2005, 53, 233–245. [Google Scholar] [CrossRef]
- Yang, J.Y.; Sun, Y.; Sun, A.Q.; Yi, S.Y.; Qin, J.; Li, M.H.; Liu, J. The involvement of chloroplast HSP100/ClpB in the acquired thermotolerance in tomato. Plant Mol. Biol. 2006, 62, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Zhang, L.; An, H.; Wu, C.; Guo, X. GhMPK16, a novel stress-responsive group D MAPK gene from cotton, is involved in disease resistance and drought sensitivity. BMC Mol. Biol. 2011, 12, 22. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Chu, X.; Li, Y.; Wang, C.; Guo, X. Cotton GhMKK1 induces the tolerance of salt and drought stress, and mediates defence responses to pathogen infection in transgenic Nicotiana benthamiana. PLoS ONE 2013, 8, e68503. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Huaxia, Y.; Lu, W.; Wu, C.; Cao, X.; Guo, X. GhWRKY15, a member of the WRKY transcription factor family identified from cotton (Gossypium hirsutum L.) is involved in disease resistance and plant development. BMC Plant Biol. 2012, 12, 144. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Hao, L.; Li, J.; Liu, D.; Guo, X.; Li, H. The Gossypium hirsutum WRKY gene GhWRKY39-1 promottes pathogen infection defense responses and mediates salt stress tolerance in transgenic Nicotiana benthamiana. Plant Cell Rep. 2014, 33, 483–498. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Shang, H.; Tamplin, M.; Ross, T.; Bowman, J.P. Culture-dependent and culture-independent assessment of spoilage community growth on VP lamb meat from packaging to past end of shelf-life. Food Microbiol. 2017, 68, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Zhang, H.; Strelkov, S.E.; Hwang, S.F. The LmSNF1 gene is required for pathogenicity in the canola blackleg pathogen Leptosphaeria maculans. PLoS ONE 2014, 9, e92503. [Google Scholar] [CrossRef] [PubMed]
- Fahlgren, N.; Howell, M.D.; Kasschau, K.D.; Chapman, E.J.; Sullivan, C.M.; Cumbie, J.S.; Givan, S.A.; Law, T.F.; Grant, S.R.; Dangl, J.L.; et al. High-throughput sequencing of Arabidopsis microRNAs: Evidence for frequent birth and death of MIRNA genes. PLoS ONE 2007, 2, e219. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, X.; Hu, Z.; Wang, S.; Zhang, J.; Wang, X.; Wang, Q.; Zhang, B. Lack of K-Dependent Oxidative Stress in Cotton Roots Following Coronatine-Induced ROS Accumulation. PLoS ONE 2015, 10, e0126476. [Google Scholar] [CrossRef] [PubMed]











© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhai, N.; Jia, H.; Liu, D.; Liu, S.; Ma, M.; Guo, X.; Li, H. GhMAP3K65, a Cotton Raf-Like MAP3K Gene, Enhances Susceptibility to Pathogen Infection and Heat Stress by Negatively Modulating Growth and Development in Transgenic Nicotiana benthamiana. Int. J. Mol. Sci. 2017, 18, 2462. https://doi.org/10.3390/ijms18112462
Zhai N, Jia H, Liu D, Liu S, Ma M, Guo X, Li H. GhMAP3K65, a Cotton Raf-Like MAP3K Gene, Enhances Susceptibility to Pathogen Infection and Heat Stress by Negatively Modulating Growth and Development in Transgenic Nicotiana benthamiana. International Journal of Molecular Sciences. 2017; 18(11):2462. https://doi.org/10.3390/ijms18112462
Chicago/Turabian StyleZhai, Na, Haihong Jia, Dongdong Liu, Shuchang Liu, Manli Ma, Xingqi Guo, and Han Li. 2017. "GhMAP3K65, a Cotton Raf-Like MAP3K Gene, Enhances Susceptibility to Pathogen Infection and Heat Stress by Negatively Modulating Growth and Development in Transgenic Nicotiana benthamiana" International Journal of Molecular Sciences 18, no. 11: 2462. https://doi.org/10.3390/ijms18112462