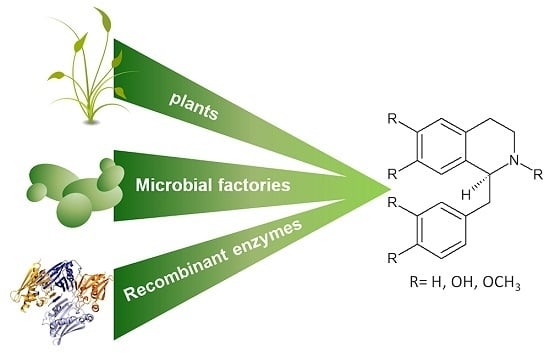
Green Routes for the Production of Enantiopure Benzylisoquinoline Alkaloids
Abstract
:1. Introduction
2. Working with Living Organisms: Biotechnological Approaches in Benzylisoquinoline Alkaloids (BIA) Production
2.1. BIA Production in Plants
2.2. BIA Production in Microbial Systems: Bacteria and Yeast
2.2.1. Bacterial Factories
2.2.2. Yeast Factories
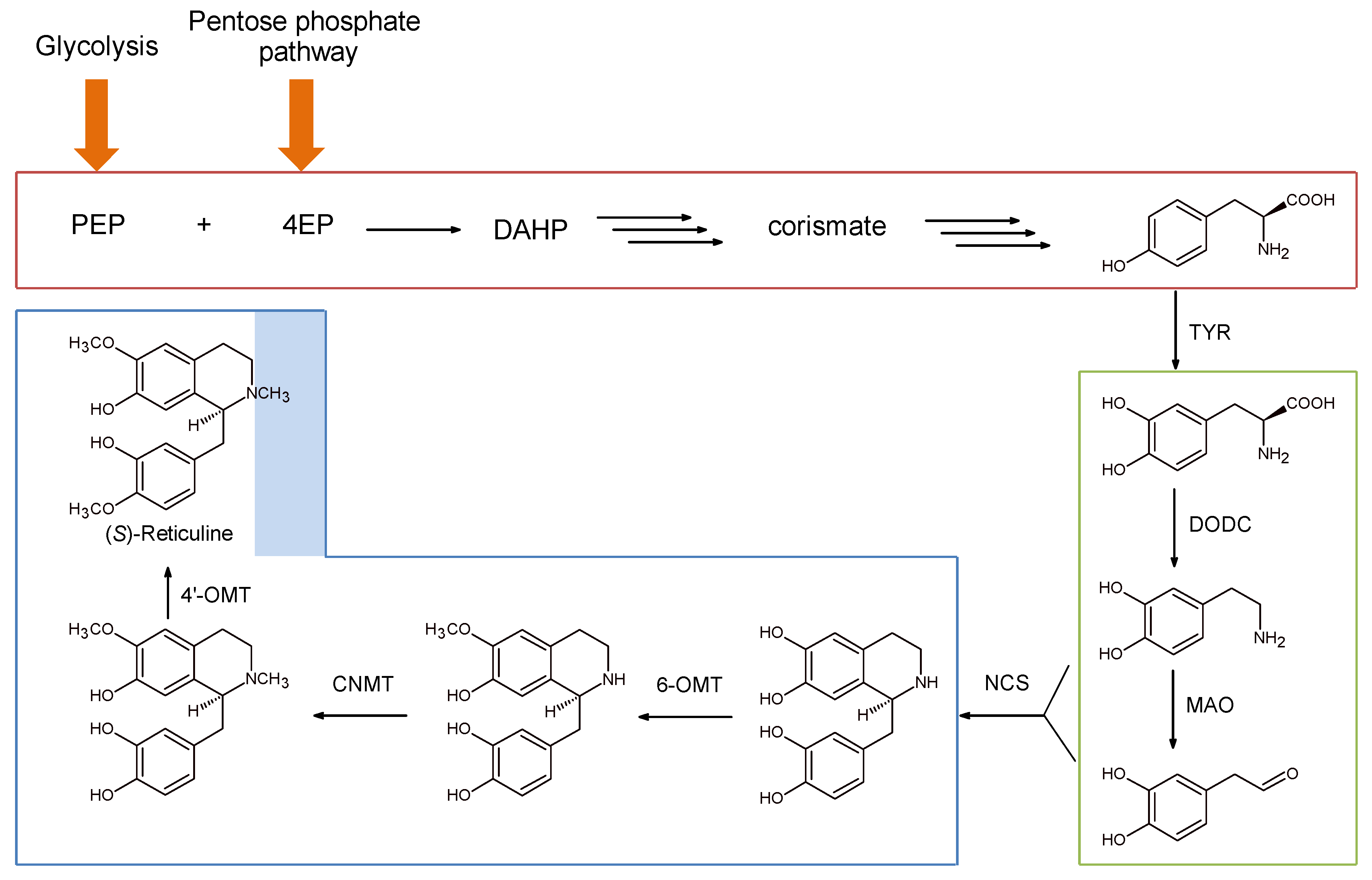
3. Norcolaurine Synthase: The Gate of BIA Chirality
3.1. Chemo-Enzymatic Synthesis of BIAs
3.2. Fully Enzymatic Synthesis of Substituted Tetrahydroisoquinolines
Conflicts of Interest
Abbreviations
| BIAs | Benzylisoquinoline alkaloids |
| THIQs | Tetrahydroisoquinolines |
| 4-HPAA | 4-hydroxyphenylacethaldehyde |
| NCS | Norcoclaurine synthase |
| VIGS | Virus-induced gene silencing |
| CRISPR | Clustered regularly interspaced short palindromic repeats |
| OMT | O-Methyltransferase |
| CMNT | Coclurine-N-methyltransferase |
| TNMT | Tetrahydroprotoberberine-cis-N-methyltransferase |
| BBE | Berberine bridge enzyme |
| DODC | l-DOPA decarboxylase |
| MAO | Monoamine oxidase |
| LCAO | Lathyrus cicera amine oxidase |
| CYP | Cytochrome P450 monooxygenases |
| TAm | Transaminase |
References
- Hagel, J.M.; Facchini, P.J. Benzylisoquinoline alkaloid metabolism: A century of discovery and a brave new world. Plant Cell Physiol. 2013, 54, 647–672. [Google Scholar] [CrossRef] [PubMed]
- Shamma, M. The Isoquinoline Alkaloids: Chemistry and Pharmacology; Academic Press: New York, NY, USA, 1972; Volume 25. [Google Scholar]
- Berényi, S.; Csutorás, C.; Sipos, A. Recent developments in the chemistry of thebaine and its transformation products as pharmacological targets. Curr. Med. Chem. 2009, 16, 3215–3242. [Google Scholar] [CrossRef] [PubMed]
- Beaudoin, G.A.W.; Facchini, P.J. Benzylisoquinoline alkaloid biosynthesis in opium poppy. Planta 2014, 240, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Abbasoglu, U.; Sener, B.; Gunay, Y.; Temizer, H. Antimicrobial activity of some isoquinoline alkaloids. Arch. Pharm. 1991, 324, 379–380. [Google Scholar] [CrossRef]
- Iwasa, K.; Nishiyama, Y.; Ichimaru, M.; Moriyasau, M.; Kim, H.S.; Wataya, Y.; Yamori, T.; Takashi, T.; Lee, D.U. Structure-activity relationships of quaternary protoberberine alkaloids having an antimalarial activity. Eur. J. Med. Chem. 1999, 34, 1077–1083. [Google Scholar] [CrossRef]
- Iwasa, K.; Moriyasau, M.; Tachibana, Y.; Kim, H.-S.; Wataya, Y.; Wiegrebe, W.; Bastow, K.F.; Cosentino, L.M.; Kozuka, M.; Lee, K.-H. Simple isoquinoline and benzylisoquinoline alkaloids as potential antimicrobial, antimalarial, cytotoxic, and anti-HIV agents. Bioorg. Med. Chem. 2001, 9, 2871–2884. [Google Scholar] [CrossRef]
- Rashid, M.A.; Gustafson, K.R.; Kashman, Y.; Cardellina, J.H., II; McMahon, J.B.; Boyd, M.R. Anti-HIV alkaloids from Toddalia asiatica. Nat. Prod. Res. 1995, 6, 153–156. [Google Scholar]
- Kashiwada, Y.; Aoshima, A.; Ikeshiro, Y.; Chen, Y.-P.; Furukawa, H.; Itoigawa, M.; Fujioka, T.; Mihashi, K.; Cosentino, L.M.; Morris-Natschke, S.L.; et al. Anti-HIV benzylisoquinoline alkaloids and flavonoids from the leaves of Nelumbo nucifera, and structure-activity correlations with related alkaloids. Bioorg. Med. Chem. 2005, 13, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.Z.; Hu, S.-F.; Zaw, K.; Angerhofer, C.K.; Chai, H.; Pezzuto, J.M.; Cordell, G.A.; Lin, J.; Zheng, D.-M. Thalifaberidine, a cytotoxic aporphine-benzylisoquinoline alkaloid from Thalictrum faberi. J. Nat. Prod. 1994, 57, 1430–1436. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Iwasa, K.; Tokuda, H.; Kashihara, A.; Mitani, Y.; Hasegawa, T.; Nishiyama, Y.; Moriyasu, M.; Nishino, H.; Hanaoka, M.; et al. Potential cancer chemopreventive activity of simple isoquinolines, 1-benzylisoquinolines, and protoberberines. Phytochemistry 2006, 67, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudian, M.; Rahimi-Moghaddam, P. The anti-cancer activity of noscapine: A review. Recent Pat. Anticancer Drug Discov. 2009, 4, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.; Wei, J.; Abidi, P.; Lin, M.; Inaba, S.; Li, C.; Wang, Y.; Wang, Z.; Si, S.; Pan, H.; et al. Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nat. Med. 2004, 10, 1344–1351. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.M.; Lee, J.P.; Min, B.S.; Choi, J.S.; Na, M.; Zhang, X.; Ngoc, T.M.; Lee, I.; Bae, K. Magnoflorine from Coptidis rhizoma protects high density lipoprotein during oxidant stress. Biol. Pharm. Bull. 2007, 30, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.M.; Na, M.; Min, B.S.; Zhang, X.; Lee, I.; Ngoc, T.M.; Thuong, P.T.; Sok, D.E.; Bae, K. Protective effect of magnoflorine isolated from Coptidis rhizoma on Cu2+-induced oxidation of human low density lipoprotein. Planta Med. 2007, 73, 1281–1284. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Yao, W.; Zheng, X.; Liao, K. Berberine promotes the development of atherosclerosis and foam cell formation by inducing scavenger receptor A expression in macrophage. Cell Res. 2009, 19, 1006–1017. [Google Scholar] [CrossRef] [PubMed]
- Ruan, H.; Zhan, Y.Y.; Hou, J.; Xu, B.; Chen, B.; Tian, Y.; Wu, D.; Zhao, Y.; Zhang, Y.; Chen, X.; et al. Berberine binds RXRα to suppress β-catenin signaling in colon cancer cells. Oncogene 2017. [Google Scholar] [CrossRef] [PubMed]
- Singla, D.; Sharma, A.; Kaur, J.; Panwar, B.; Raghava, P.S. BIAdb: A curated database of benzylisoquinoline alkaloids. BMC Pharmacol. 2010, 10, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, M.; van Linn, M.L.; Cook, J.M. The asymmetric Pictet-Spengler reaction. Curr. Org. Synth. 2010, 7, 189–223. [Google Scholar] [CrossRef]
- Pulka, K. Pictet-Spengler reactions for the synthesis of pharmaceutically relevant heterocycles. Curr. Opin. Drug Discov. Dev. 2010, 13, 669–684. [Google Scholar]
- Stöckigt, J.; Antonchick, A.P.; Wu, F.; Waldmann, H. The Pictet-Spengler reaction in nature and in organic synthesis. Angew. Chem. Int. Ed. 2011, 50, 8538–8564. [Google Scholar] [CrossRef] [PubMed]
- Ruijter, E.; Scheffelaar, R.; Orru, R.V.A. Multicomponent reaction design in the quest for molecular complexity and diversity. Angew. Chem. Int. Ed. 2011, 50, 6234–6246. [Google Scholar] [CrossRef] [PubMed]
- Menendez, P.; D’Acquarica, I.; Delle Monache, G.; Ghirga, F.; Calcaterra, A.; Barba, M.; Macone, A.; Boffi, A.; Bonamore, A.; Botta, B. Plant Bioactives and Drug Discovery: Principles, Practice, Perspectives; Cechinel Filho, V., Ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2012; pp. 453–487. [Google Scholar]
- Ingallina, C.; D’Acquarica, I.; Delle Monache, G.; Ghirga, F.; Quaglio, D.; Ghirga, P.; Berardozzi, S.; Markovic, V.; Botta, B. The Pictet-Spengler reaction still on stage. Curr. Pharm. Des. 2016, 22, 1808–1850. [Google Scholar] [CrossRef] [PubMed]
- Pictet, A.; Spengler, T. Formation of isoquinoline derivatives by the action of methylal on phenylethylamine, phenylalanine and tyrosine. Ber. Dtsch. Chem. Ges. 1911, 44, 2030–2036. [Google Scholar] [CrossRef]
- Lee, E.J.; Facchini, P.J. Norcoclaurine Synthase Is a Member of the Pathogenesis-Related 10/Bet v1 Protein Family. Plant Cell 2010, 22, 3489–3503. [Google Scholar] [CrossRef] [PubMed]
- Ghirga, I.; Quaglio, D.; Ghirga, P.; Berardozzi, S.; Zappia, G.; Botta, B.; Mori, M.; D’Acquarica, I. Occurrence of enantioselectivity in nature: The case of (S)-norcoclaurine. Chirality 2016, 28, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Finefield, J.M.; Sherman, D.H.; Kreitman, M.; Williams, R.M. Enantiomeric natural products: Occurrence and biogenesis. Angew. Chem. Int. Ed. 2012, 51, 4802–4836. [Google Scholar] [CrossRef] [PubMed]
- Chrzanowska, M.; Grajewska, A.; Rozwadowska, M.D. Asymmetric synthesis of isoquinoline alkaloids: 2004–2015. Chem. Rev. 2016, 116, 12369–12465. [Google Scholar] [CrossRef] [PubMed]
- Uematsu, N.; Fujii, A.; Hashiguchi, S.; Ikariya, T.; Noyori, R. Asymmetric transfer hydrogenation of imines. J. Am. Chem. Soc. 1996, 118, 4916–4917. [Google Scholar] [CrossRef]
- Guimond, N.; Fagnou, K. Isoquinoline synthesis via rhodium-catalyzed oxidative cross-coupling/cyclization of aryl aldimines and alkynes. J. Am. Chem. Soc. 2009, 131, 12050–12051. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.-H.; Yan, P.-C.; Zhang, Q.-Q.; Yuan, K.-X.; Zhou, Q.-L. Asymmetric hydrogenation of cyclic imines catalyzed by chiral spiro iridium phosphoramidite complexes for enantioselective synthesis of tetrahydroisoquinolines. ACS Catal. 2012, 2, 561–564. [Google Scholar] [CrossRef]
- Ruiz-Olalla, A.; Würdemann, M.A.; Wanner, M.J.; Ingemann, S.; van Maarseveen, J.H.; Hiemstra, H. Organocatalytic enantioselective Pictet-Spengler approach to biologically relevant 1-benzyl-1,2,3,4-tetrahydroisoquinoline alkaloids. J. Org. Chem. 2015, 80, 5125–5132. [Google Scholar] [CrossRef] [PubMed]
- Kato, E.; Iwata, R.; Kawabata, J. Synthesis and detailed examination of spectral properties of (S)- and (R)-higenamine 4′-O-β-d-glucoside and HPLC analytical conditions to distinguish the diastereomers. Molecules 2017, 22, 1450. [Google Scholar] [CrossRef] [PubMed]
- Kato, E.; Inagaki, Y.; Kawabata, J. Higenamine 4’-O-β-d-glucoside in the lotus plumule induces glucose uptake of L6 cells through β2-adrenergic receptor. Bioorg. Med. Chem. 2015, 23, 3317–3321. [Google Scholar] [CrossRef] [PubMed]
- Gober, C.M.; Joullié, M.M. Joining forces: Fermentation and organic synthesis for the production of complex heterocycles. J. Org. Chem. 2016, 81, 10136–10144. [Google Scholar] [CrossRef] [PubMed]
- Larkin, P.J.; Miller, J.A.; Allen, R.S.; Chitty, J.A.; Gerlach, W.L.; Frick, S.; Kutchan, T.M.; Fist, A.J. Increasing morphinan alkaloid production by over-expressing codeinone reductase in transgenic Papaver somniferum. Plant Biotechnol. J. 2007, 5, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Sharafi, A.; Sohi, H.H.; Mousavi, A.; Azadi, P.; Khalifani, B.H.; Razavi, K. Metabolic engineering of morphinan alkaloids by over-expression of codeinone reductase in transgenic hairy roots of Papaver bracteatum, the Iranian poppy. Biotechnol. Lett. 2013, 35, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Sato, F.; Takeshihta, N.; Fujiwara, H.; Katagiri, Y.; Huan, L.; Yamada, Y. Characterization of Coptis japonica cells with different alkaloid productivities. Plant Cell Tissue Organ Cult. 1994, 38, 249–256. [Google Scholar] [CrossRef]
- Inui, T.; Kawano, N.; Shitan, N.; Yazaki, K.; Kiuchi, F.; Kawahara, N.; Sato, F.; Yoshimatsu, K. Improvement of benzylisoquinoline alkaloid productivity by overexpression of 3′-hydroxy-N-methylcoclaurine 4′-O-methyltransferase in transgenic Coptis japonica plants. Biol. Pharm. Bull. 2012, 35, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Park, S.U.; Yu, M.; Facchini, P.J. Modulation of berberine bridge enzyme levels in transgenic root cultures of California poppy alters the accumulation of benzophenanthridine alkaloids. Plant Mol. Biol. 2003, 51, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.S.; Miller, J.A.; Chitty, J.A.; Fist, A.J.; Gerlach, W.L.; Larkin, P.J. Metabolic engineering of morphinan alkaloids by over-expression and RNAi suppression of salutaridinol 7-O-acetyltransferase in opium poppy. Plant Biotechnol. J. 2008, 6, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Sharafi, A.; Hashemi, S.H.; Mousavi, A.; Azadi, P.; Dehsara, B.; Hosseini, K.B. Enhanced morphinan alkaloid production in hairy root cultures of Papaver bracteatum by over-expression of salutaridinol 7-O-acetyltransferase gene via Agrobacterium rhizogenes mediated transformation. World J. Microbiol. Biotechnol. 2013, 29, 2125–2131. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.S.; Millgate, A.G.; Chitty, J.A.; Thisleton, J.; Miller, J.A.; Fist, A.J.; Gerlach, W.L.; Larkin, P.J. RNAi-mediated replacement of morphine with the nonnarcotic alkaloid reticuline in opium poppy. Nat. Biotechnol. 2004, 22, 1559–1566. [Google Scholar] [CrossRef] [PubMed]
- Champa, P.W.; Facchini, P.J. Systematic knockdown of morphine pathway enzymes in opium poppy using virus-induced gene silencing. Plant J. 2012, 69, 1052–1063. [Google Scholar]
- Mohan, C. Genome Editing in Sugarcane: Challenges Ahead. Front. Plant Sci. 2016, 7, 1542. [Google Scholar] [CrossRef] [PubMed]
- Alagoz, Y.; Gurkok, T.; Zhang, B.; Unver, T. Manipulating the Biosynthesis of Bioactive Compound Alkaloids for Next-Generation Metabolic Engineering in Opium Poppy Using CRISPR-Cas 9 Genome Editing Technology. Sci. Rep. 2016, 6, 30910. [Google Scholar] [CrossRef] [PubMed]
- Facchini, P.J. Alkaloid biosynthesis in plants: Biochemistry, Cell Biology, Molecular Regulation, and Metabolic Engineering Applications. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001, 52, 29–66. [Google Scholar] [CrossRef] [PubMed]
- Sato, F.; Hashimoto, T.; Hachiya, A.; Tamura, K.; Choi, K.; Morishige, T.; Fujimoto, H.; Yamada, Y. Metabolic engineering of plant alkaloid biosynthesis. Proc. Natl. Acad. Sci. USA 2001, 98, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Staniek, A.; Bouwmeester, H.; Fraser, P.D.; Kayser, O.; Martens, S.; Tissier, A.; van der Krol, S.; Wessjohann, L.; Warzecha, H. Natural products—Modifying metabolite pathways in plants. Biotechnol. J. 2013, 8, 1159–1171. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.S.; Thodey, K.; Trenchard, I.; Smolke, C.D. Advancing secondary metabolite biosynthesis in yeast with synthetic biology tools. FEMS Yeast Res. 2012, 12, 144–170. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, A.; Minami, H.; Kim, J.S.; Koyanagi, T.; Katayama, T.; Sato, F.; Kumagai, H. A bacterial platform for fermentative production of plant alkaloids. Nat. Commun. 2011, 2, 326. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, E.; Nakagawa, A.; Tomabechi, Y.; Koyanagi, T.; Kumagai, H.; Yamamoto, K.; Katayama, T.; Sato, F.; Minami, H. Laboratory-scale production of (S)-reticuline, an important intermediate of benzylisoquinoline alkaloids, using a bacterial-based method. Biosci. Biotechnol. Biochem. 2017, 81, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, K.M.; Smolke, C.D. Production of benzylisoquinoline alkaloids in Saccharomyces cerevisiae. Nat. Chem. Biol. 2008, 4, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Trenchard, I.J.; Siddiqui, M.S.; Thodey, K.; Smolke, C.D. De novo production of the key branch point benzylisoquinoline alkaloid reticuline in yeast. Metab. Eng. 2015, 31, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Galanie, S.; Smolke, C.D. Optimization of yeast-based production of medicinal protoberberine alkaloids. Microb. Cell Fact. 2015, 14, 144. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Smolke, C.D. Engineering biosynthesis of the anticancer alkaloid noscapine in yeast. Nat. Commun. 2016, 7, 12137. [Google Scholar] [CrossRef] [PubMed]
- Hori, K.; Okano, S.; Sato, F. Efficient microbial production of stylopine using a Pichia pastoris expression system. Sci. Rep. 2016, 6, 22201. [Google Scholar] [CrossRef] [PubMed]
- Fossati, E.; Ekins, A.; Narcross, L.; Zhu, Y.; Falgueyret, J.P.; Beaudoin, G.A.; Facchini, P.J.; Martin, V.J. Reconstitution of a 10-gene pathway for synthesis of the plant alkaloid dihydrosanguinarine in Saccharomyces cerevisiae. Nat. Commun. 2014, 5, 3283. [Google Scholar] [CrossRef] [PubMed]
- Trenchard, I.J.; Smolke, C.D. Engineering strategies for the fermentative production of plant alkaloids in yeast. Metab. Eng. 2015, 30, 96–104. [Google Scholar] [CrossRef] [PubMed]
- DeLoache, W.C.; Russ, Z.N.; Narcross, L.; Gonzales, A.M.; Martin, V.J.; Dueber, J.E. An enzyme-coupled biosensor enables (S)-reticuline production in yeast from glucose. Nat. Chem. Biol. 2015, 11, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Fossati, E.; Narcross, L.; Ekins, A.; Falgueyret, J.P.; Martin, V.J. Synthesis of Morphinan Alkaloids in Saccharomyces cerevisiae. PLoS ONE 2015, 10, e0124459. [Google Scholar] [CrossRef] [PubMed]
- Thodey, K.; Galanie, S.; Smolke, C.D. A microbial biomanufacturing platform for natural and semisynthetic opioids. Nat. Chem. Biol. 2014, 10, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Farrow, S.C.; Hagel, J.M.; Beaudoin, G.A.W.; Burns, D.C.; Facchini, P.J. Stereochemical inversion of (S)-reticuline by a cytochrome P450 fusion in opium poppy. Nat. Chem. Biol. 2015, 11, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Kraus, P.F.; Kutchan, T.M. Molecular cloning and heterologous expression of a cDNA encoding berbamunine synthase, a C–O phenol-coupling cytochrome P450 from the higher plant Berberis stolonifera. Proc. Natl. Acad. Sci. USA 1995, 92, 2071–2075. [Google Scholar] [CrossRef] [PubMed]
- Samanani, N.; Facchini, P.J. Isolation and partial characterization of norcoclaurine synthase, the first committed step in benzylisoquinoline alkaloid biosynthesis, from opium poppy. Planta 2001, 213, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Samanani, N.; Liscombe, D.K.; Facchini, P.J. Molecular cloning and characterization of norcoclaurine synthase, an enzyme catalyzing the first committed step in benzylisoquinoline alkaloid biosynthesis. Plant J. 2004, 40, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Samanani, N.; Facchini, P.J. Purification and characterization of norcoclaurine synthase. The first committed enzyme in benzylisoquinoline alkaloid biosynthesis in plants. J. Biol. Chem. 2002, 277, 33878–33883. [Google Scholar] [CrossRef] [PubMed]
- Pasquo, A.; Bonamore, A.; Franceschini, S.; Macone, A.; Boffi, A.; Ilari, A. Cloning, expression, crystallization and preliminary X-ray data analysis of norcoclaurine synthase from Thalictrum flavum. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2008, 64, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Ilari, A.; Franceschini, S.; Bonamore, A.; Arenghi, F.; Botta, B.; Macone, A.; Pasquo, A.; Bellucci, L.; Boffi, A. Structural basis of enzymatic (S)-norcoclaurine biosynthesis. J. Biol. Chem. 2009, 284, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Bonamore, A.; Barba, M.; Botta, B.; Boffi, A.; Macone, A. Norcoclaurine synthase: Mechanism of an enantioselective pictet-spengler catalyzing enzyme. Molecules 2010, 15, 2070–2078. [Google Scholar] [CrossRef] [PubMed]
- Lichman, B.R.; Gershater, M.C.; Lamming, E.D.; Pesnot, T.; Sula, A.; Keep, N.H.; Hailes, H.C.; Ward, J.M. ‘Dopamine-first’ mechanism enables the rational engineering of the norcoclaurine synthase aldehyde activity profile. FEBS J. 2015, 282, 1137–1151. [Google Scholar] [CrossRef] [PubMed]
- Lichman, B.R.; Sula, A.; Pesnot, T.; Hailes, H.C.; Ward, J.M.; Keep, N.H. Structural Evidence for the Dopamine-First Mechanism of Norcoclaurine Synthase. Biochemistry 2017, 56, 5274–5277. [Google Scholar] [CrossRef] [PubMed]
- Bonamore, A.; Rovardi, I.; Gasparrini, F.; Baiocco, P.; Barba, M.; Molinaro, C.; Botta, B.; Boffi, A.; Macone, A. An enzymatic, stereoselective synthesis of (S)-norcoclaurine. Green Chem. 2010, 12, 1623–1627. [Google Scholar] [CrossRef]
- Pesnot, T.; Gershater, M.C.; Wardb, J.M.; Hailes, H.C. Phosphate mediated biomimetic synthesis of tetrahydroisoquinoline alkaloid. Chem. Commun. 2011, 47, 3242–3244. [Google Scholar] [CrossRef] [PubMed]
- Luk, L.Y.; Bunn, S.; Liscombe, D.K.; Facchini, P.J.; Tanner, M.E. Mechanistic studies on norcoclaurine synthase of benzylisoquinoline alkaloid biosynthesis: An enzymatic Pictet-Spengler reaction. Biochemistry 2007, 46, 10153–10161. [Google Scholar] [CrossRef] [PubMed]
- Ruff, B.M.; Bräse, S.; O’Connor, S.E. Biocatalytic production of tetrahydroisoquinolines. Tetrahedron Lett. 2012, 53, 1071–1074. [Google Scholar] [CrossRef] [PubMed]
- Pesnot, T.; Gershater, M.C.; Ward, J.M.; Hailes, H.C. The Catalytic Potential of Coptis japonica NCS2 Revealed—Development and Utilisation of a Fluorescamine-Based Assay. Adv. Synth. Catal. 2012, 354, 2997–3008. [Google Scholar] [CrossRef]
- Lichman, B.R.; Lamming, E.D.; Pesnot, T.; Smith, J.M.; Hailes, H.C.; Ward, J.M. One-pot triangular chemoenzymatic cascades for the syntheses of chiral alkaloids from dopamine. Green Chem. 2015, 17, 852–855. [Google Scholar] [CrossRef]
- Bonamore, A.; Calisti, L.; Calcaterra, A.; Ismail, O.H.; Gargano, M.; D’Acquarica, I.; Botta, B.; Boffi, A.; Macone, A. A Novel Enzymatic Strategy for the Synthesis of Substituted Tetrahydroisoquinolines. ChemistrySelect 2016, 1, 1525–1528. [Google Scholar] [CrossRef]
- Pietrangeli, P.; Federico, R.; Mondovì, B.; Morpurgo, L. Substrate specificity of copper-containing plant amine oxidases. J. Inorg. Biochem. 2007, 101, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Erdmann, V.; Lichman, B.R.; Zhao, J.; Simon, R.C.; Kroutil, W.; Ward, J.M.; Hailes, H.C.; Rother, D. Enzymatic and Chemoenzymatic Three-Step Cascades for the Synthesis of Stereochemically Complementary Trisubstituted Tetrahydroisoquinolines. Angew. Chem. Int. Ed. Engl. 2017, 56, 12503–12507. [Google Scholar] [CrossRef] [PubMed]
- Lichman, B.R.; Zhao, J.; Hailes, H.C.; Ward, J.M. Enzyme catalysed Pictet-Spengler formation of chiral 1,1′-disubstituted- and spiro-tetrahydroisoquinolines. Nat. Commun. 2017, 8, 14883. [Google Scholar] [CrossRef] [PubMed]















© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghirga, F.; Bonamore, A.; Calisti, L.; D’Acquarica, I.; Mori, M.; Botta, B.; Boffi, A.; Macone, A. Green Routes for the Production of Enantiopure Benzylisoquinoline Alkaloids. Int. J. Mol. Sci. 2017, 18, 2464. https://doi.org/10.3390/ijms18112464
Ghirga F, Bonamore A, Calisti L, D’Acquarica I, Mori M, Botta B, Boffi A, Macone A. Green Routes for the Production of Enantiopure Benzylisoquinoline Alkaloids. International Journal of Molecular Sciences. 2017; 18(11):2464. https://doi.org/10.3390/ijms18112464
Chicago/Turabian StyleGhirga, Francesca, Alessandra Bonamore, Lorenzo Calisti, Ilaria D’Acquarica, Mattia Mori, Bruno Botta, Alberto Boffi, and Alberto Macone. 2017. "Green Routes for the Production of Enantiopure Benzylisoquinoline Alkaloids" International Journal of Molecular Sciences 18, no. 11: 2464. https://doi.org/10.3390/ijms18112464