Effects of Commonly Used Pesticides in China on the Mitochondria and Ubiquitin-Proteasome System in Parkinson’s Disease
Abstract
:1. Introduction
2. Results
2.1. Pesticides Caused Dose-Dependent Apoptotic Cell Death in SH-SY5Y Cells
2.2. Pesticides Induced Morphological Changes of Mitochondria
2.3. Paraquat, Rotenone, Chlorpyrifos, Fenpyroximate and Tebufenpyrad Induced a Significant Dose-Dependent Decrease of Intracellular ATP
2.4. Paraquat, Rotenone and Chlorpyrifos Inhibited NADH Dehydrogenase Activity
2.5. All Present-Study Pesticides Except Trichlorphon and Carbaryl Inhibited 26S Proteasome Activity
2.6. All Present-Study Pesticides Except Trichlorphon and Carbaryl Inhibited 20S Proteasome Activity
2.7. Trichlorphon and Carbaryl Did Not Affect Mitochondrial and UPS Functions
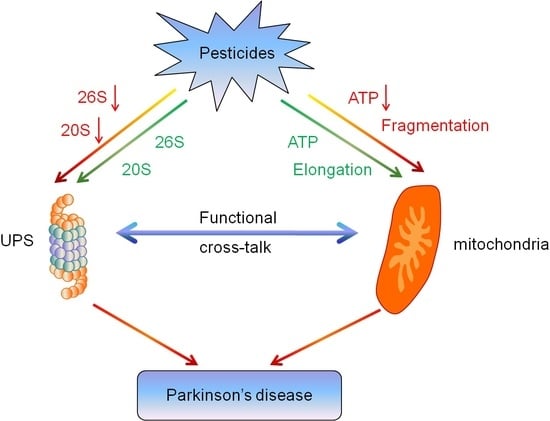
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Apoptosis Assay
4.3. Immunofluorescence and Analysis of Mitochondrial Morphology
4.4. Immunoblotting
4.5. ATP Measurements
4.6. Mitochondrial Complex I Activity Assay
4.7. Determination of Cellular 26S Proteasome Activity
4.8. Determination of 20S Proteasome Activity
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PD | Parkinson’s Disease |
| PI | Propidine Iodide |
| UPS | Ubiquitin-proteasome System |
| WHO | World Health Organization |
| DHR | [3H]dihydrorotenone |
References
- Dawson, T.M.; Dawson, V.L. Molecular pathways of neurodegeneration in Parkinson’s disease. Science 2003, 302, 819–822. [Google Scholar] [CrossRef] [PubMed]
- Valente, E.M.; Abou-Sleiman, P.M.; Caputo, V.; Muqit, M.M.; Harvey, K.; Gispert, S.; Ali, Z.; Del Turco, D.; Bentivoglio, A.R.; Healy, D.G.; et al. Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science 2004, 304, 1158–1160. [Google Scholar] [CrossRef] [PubMed]
- Wirdefeldt, K.; Adami, H.O.; Cole, P.; Trichopoulos, D.; Mandel, J. Epidemiology and etiology of Parkinson’s disease: A review of the evidence. Eur. J. Epidemiol. 2011, 26 (Suppl. 1), S1–S58. [Google Scholar] [CrossRef] [PubMed]
- Engel, L.S.; Checkoway, H.; Keifer, M.C.; Seixas, N.S.; Longstreth, W.T., Jr.; Scott, K.C.; Hudnell, K.; Anger, W.K.; Camicioli, R. Parkinsonism and occupational exposure to pesticides. Occup. Environ. Med. 2001, 58, 582–589. [Google Scholar] [CrossRef] [PubMed]
- McCormack, A.L.; Thiruchelvam, M.; Manning-Bog, A.B.; Thiffault, C.; Langston, J.W.; Cory-Slechta, D.A.; Di Monte, D.A. Environmental risk factors and Parkinson’s disease: Selective degeneration of nigral dopaminergic neurons caused by the herbicide paraquat. Neurobiol. Dis. 2002, 10, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Meco, G.; Bonifati, V.; Vanacore, N.; Fabrizio, E. Parkinsonism after chronic exposure to the fungicide maneb (manganese ethylene-bis-dithiocarbamate). Scand. J. Work Environ. Health 1994, 20, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Ramos, J.; Facca, A.; Basit, A.; Song, S. Toxicity of dieldrin for dopaminergic neurons in mesencephalic cultures. Exp. Neurol. 1998, 150, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Uversky, V.N. Neurotoxicant-induced animal models of Parkinson’s disease: Understanding the role of rotenone, maneb and paraquat in neurodegeneration. Cell Tissue Res. 2004, 318, 225–241. [Google Scholar] [CrossRef] [PubMed]
- Betarbet, R.; Sherer, T.B.; MacKenzie, G.; Garcia-Osuna, M.; Panov, A.V.; Greenamyre, J.T. Chronic systemic pesticide exposure reproduces features of Parkinson’s disease. Nat. Neurosci. 2000, 3, 1301–1306. [Google Scholar] [PubMed]
- Henchcliffe, C.; Beal, M.F. Mitochondrial biology and oxidative stress in Parkinson disease pathogenesis. Nat. Clin. Pract. Neurol. 2008, 4, 600–609. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.L. Ubiquitin-proteasome system dysfunction in Parkinson’s disease: Current evidence and controversies. Expert Rev. Proteom. 2007, 4, 769–781. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.J.; West, A.B.; Dawson, V.L.; Dawson, T.M. Molecular pathophysiology of Parkinson’s disease. Annu. Rev. Neurosci. 2005, 28, 57–87. [Google Scholar] [CrossRef] [PubMed]
- Shimura, H.; Hattori, N.; Kubo, S.; Mizuno, Y.; Asakawa, S.; Minoshima, S.; Shimizu, N.; Iwai, K.; Chiba, T.; Tanaka, K.; et al. Familial Parkinson disease gene product, parkin, is a ubiquitin-protein ligase. Nat. Genet. 2000, 25, 302–305. [Google Scholar] [PubMed]
- Leroy, E.; Boyer, R.; Auburger, G.; Leube, B.; Ulm, G.; Mezey, E.; Harta, G.; Brownstein, M.J.; Jonnalagada, S.; Chernova, T.; et al. The ubiquitin pathway in Parkinson’s disease. Nature 1998, 395, 451–452. [Google Scholar] [CrossRef] [PubMed]
- Kitada, T.; Asakawa, S.; Hattori, N.; Matsumine, H.; Yamamura, Y.; Minoshima, S.; Yokochi, M.; Mizuno, Y.; Shimizu, N. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 1998, 392, 605–608. [Google Scholar] [PubMed]
- McNaught, K.S.; Belizaire, R.; Isacson, O.; Jenner, P.; Olanow, C.W. Altered proteasomal function in sporadic Parkinson’s disease. Exp. Neurol. 2003, 179, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Hoglinger, G.U.; Feger, J.; Prigent, A.; Michel, P.P.; Parain, K.; Champy, P.; Ruberg, M.; Oertel, W.H.; Hirsch, E.C. Chronic systemic complex I inhibition induces a hypokinetic multisystem degeneration in rats. J. Neurochem. 2003, 84, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Sherer, T.B.; Kim, J.H.; Betarbet, R.; Greenamyre, J.T. Subcutaneous rotenone exposure causes highly selective dopaminergic degeneration and alpha-synuclein aggregation. Exp. Neurol. 2003, 179, 9–16. [Google Scholar] [CrossRef] [PubMed]
- McNaught, K.S.; Perl, D.P.; Brownell, A.L.; Olanow, C.W. Systemic exposure to proteasome inhibitors causes a progressive model of Parkinson’s disease. Ann. Neurol. 2004, 56, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, T.; Yamada, K.; Hojo, N.; Isobe, A.; Shiwaku, K.; Yamane, Y. Mechanism of cytotoxicity of paraquat. III. The effects of acute paraquat exposure on the electron transport system in rat mitochondria. Exp. Toxicol. Pathol. 1994, 46, 437–441. [Google Scholar] [CrossRef]
- Tawara, T.; Fukushima, T.; Hojo, N.; Isobe, A.; Shiwaku, K.; Setogawa, T.; Yamane, Y. Effects of paraquat on mitochondrial electron transport system and catecholamine contents in rat brain. Arch. Toxicol. 1996, 70, 585–589. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Ragheb, K.; Lawler, G.; Sturgis, J.; Rajwa, B.; Melendez, J.A.; Robinson, J.P. Mitochondrial complex I inhibitor rotenone induces apoptosis through enhancing mitochondrial reactive oxygen species production. J. Biol. Chem. 2003, 278, 8516–8525. [Google Scholar] [CrossRef] [PubMed]
- Shamoto-Nagai, M.; Maruyama, W.; Kato, Y.; Isobe, K.; Tanaka, M.; Naoi, M.; Osawa, T. An inhibitor of mitochondrial complex I, rotenone, inactivates proteasome by oxidative modification and induces aggregation of oxidized proteins in SH-SY5Y cells. J. Neurosci. Res. 2003, 74, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Sherer, T.B.; Richardson, J.R.; Testa, C.M.; Seo, B.B.; Panov, A.V.; Yagi, T.; Matsuno-Yagi, A.; Miller, G.W.; Greenamyre, J.T. Mechanism of toxicity of pesticides acting at complex I: Relevance to environmental etiologies of Parkinson’s disease. J. Neurochem. 2007, 100, 1469–1479. [Google Scholar] [CrossRef] [PubMed]
- Chou, A.P.; Li, S.; Fitzmaurice, A.G.; Bronstein, J.M. Mechanisms of rotenone-induced proteasome inhibition. Neurotoxicology 2010, 31, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Chou, A.P.; Maidment, N.; Klintenberg, R.; Casida, J.E.; Li, S.; Fitzmaurice, A.G.; Fernagut, P.O.; Mortazavi, F.; Chesselet, M.F.; Bronstein, J.M. Ziram causes dopaminergic cell damage by inhibiting E1 ligase of the proteasome. J. Biol. Chem. 2008, 283, 34696–34703. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.F.; Li, S.; Chou, A.P.; Bronstein, J.M. Inhibitory effects of pesticides on proteasome activity: Implication in Parkinson’s disease. Neurobiol. Dis. 2006, 23, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Wills, J.; Credle, J.; Oaks, A.W.; Duka, V.; Lee, J.H.; Jones, J.; Sidhu, A. Paraquat, but not maneb, induces synucleinopathy and tauopathy in striata of mice through inhibition of proteasomal and autophagic pathways. PLoS ONE 2012, 7, e30745. [Google Scholar] [CrossRef]
- Rhodes, S.L.; Fitzmaurice, A.G.; Cockburn, M.; Bronstein, J.M.; Sinsheimer, J.S.; Ritz, B. Pesticides that inhibit the ubiquitin-proteasome system: Effect measure modification by genetic variation in SKP1 in Parkinsons disease. Environ. Res. 2013, 126, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gomez, C.; Bandez, M.J.; Navarro, A. Pesticides and impairment of mitochondrial function in relation with the parkinsonian syndrome. Front. Biosci. 2007, 12, 1079–1093. [Google Scholar] [CrossRef] [PubMed]
- Bellou, V.; Belbasis, L.; Tzoulaki, I.; Evangelou, E.; Ioannidis, J.P.A. Environmental risk factors and Parkinson’s disease: An umbrella review of meta-analyses. Parkinsonism Relat. Disord. 2016, 23, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Alavanja, M.C.; Hoppin, J.A.; Kamel, F. Health effects of chronic pesticide exposure: Cancer and neurotoxicity. Annu. Rev. Public Health 2004, 25, 155–197. [Google Scholar] [CrossRef] [PubMed]
- Damalas, C.A.; Eleftherohorinos, I.G. Pesticide exposure, safety issues, and risk assessment indicators. Int. J. Environ. Res. Public Health 2011, 8, 1402–1419. [Google Scholar] [CrossRef] [PubMed]
- Tanner, C.M.; Kamel, F.; Ross, G.W.; Hoppin, J.A.; Goldman, S.M.; Korell, M.; Marras, C.; Bhudhikanok, G.S.; Kasten, M.; Chade, A.R.; et al. Rotenone, paraquat, and Parkinson’s disease. Environ. Health Perspect. 2011, 119, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Pieczenik, S.R.; Neustadt, J. Mitochondrial dysfunction and molecular pathways of disease. Exp. Mol. Pathol. 2007, 83, 84–92. [Google Scholar] [CrossRef] [PubMed]
- McBride, H.M.; Neuspiel, M.; Wasiak, S. Mitochondria: More than just a powerhouse. Curr. Biol. 2006, 16, R551–R560. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; Shirihai, O.S.; Gentil, B.J.; Burelle, Y. Mitochondrial morphology transitions and functions: Implications for retrograde signaling? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R393–R406. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.; Verma, R.S.; Srivastava, N. Chlorpyrifos induced alterations in the levels of hydrogen peroxide, nitrate and nitrite in rat brain and liver. Pestic. Biochem. Physiol. 2009, 94, 55–59. [Google Scholar] [CrossRef]
- Verma, R.S.; Mehta, A.; Srivastava, N. In vivo chlorpyrifos induced oxidative stress: Attenuation by antioxidant vitamins. Pestic. Biochem. Physiol. 2007, 88, 191–196. [Google Scholar] [CrossRef]
- Binukumar, B.K.; Bal, A.; Kandimalla, R.J.; Gill, K.D. Nigrostriatal neuronal death following chronic dichlorvos exposure: Crosstalk between mitochondrial impairments, alpha synuclein aggregation, oxidative damage and behavioral changes. Mol. Brain 2010, 3, 35. [Google Scholar] [PubMed]
- Cocheme, H.M.; Murphy, M.P. Complex I is the major site of mitochondrial superoxide production by paraquat. J. Biol. Chem. 2008, 283, 1786–1798. [Google Scholar] [CrossRef] [PubMed]
- Popovic, D.; Vucic, D.; Dikic, I. Ubiquitination in disease pathogenesis and treatment. Nat. Med. 2014, 20, 1242–1253. [Google Scholar] [CrossRef] [PubMed]
- Gilon, T.; Chomsky, O.; Kulka, R.G. Degradation signals for ubiquitin system proteolysis in Saccharomyces cerevisiae. EMBO J. 1998, 17, 2759–2766. [Google Scholar] [CrossRef] [PubMed]
- Preux, P.M.; Condet, A.; Anglade, C.; Druet-Cabanac, M.; Debrock, C.; Macharia, W.; Couratier, P.; Boutros-Toni, F.; Dumas, M. Parkinson’s disease and environmental factors. Matched case-control study in the Limousin region, France. Neuroepidemiology 2000, 19, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Jonasson, S.B.; Ullen, S.; Iwarsson, S.; Lexell, J.; Nilsson, M.H. Concerns About Falling in Parkinson’s Disease: Associations with Disabilities and Personal and Environmental Factors. J. Parkinson’s Dis. 2015, 5, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Keifer, M.C.; Mahurin, R.K. Chronic neurologic effects of pesticide overexposure. Occup. Med. 1997, 12, 291–304. [Google Scholar] [PubMed]
- He, F. Neurotoxic effects of insecticides—Current and future research: A review. Neurotoxicology 2000, 21, 829–835. [Google Scholar] [PubMed]
- Gomes, J.; Lloyd, O.L.; Revitt, D.M. The influence of personal protection, environmental hygiene and exposure to pesticides on the health of immigrant farm workers in a desert country. Int. Arch. Occup. Environ. Health 1999, 72, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Ohayo-Mitoko, G.J.A. Self reported symptoms and inhibition of acetylcholinesterase activity among Kenyan agricultural workers. Occup. Environ. Med. 2000, 57, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Poole, A.C.; Thomas, R.E.; Andrews, L.A.; McBride, H.M.; Whitworth, A.J.; Pallanck, L.J. The PINK1/Parkin pathway regulates mitochondrial morphology. Proc. Natl. Acad. Sci. USA 2008, 105, 1638–1643. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, G.; Chung, J. The PINK1-Parkin pathway is involved in the regulation of mitochondrial remodeling process. Biochem. Biophys. Res. Commun. 2009, 378, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Janetzky, B.; Hauck, S.; Youdim, M.B.; Riederer, P.; Jellinger, K.; Pantucek, F.; Zochling, R.; Boissl, K.W.; Reichmann, H. Unaltered aconitase activity, but decreased complex I activity in substantia nigra pars compacta of patients with Parkinson’s disease. Neurosci. Lett. 1994, 169, 126–128. [Google Scholar] [CrossRef]
- Mann, V.M.; Cooper, J.M.; Daniel, S.E.; Srai, K.; Jenner, P.; Marsden, C.D.; Schapira, A.H. Complex I, iron, and ferritin in Parkinson’s disease substantia nigra. Ann. Neurol. 1994, 36, 876–881. [Google Scholar] [CrossRef] [PubMed]
- Reeve, A.K.; Krishnan, K.J.; Turnbull, D. Mitochondrial DNA mutations in disease, aging, and neurodegeneration. Ann. N. Y. Acad. Sci. 2008, 1147, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Khrapko, K.; Vijg, J. Mitochondrial DNA mutations and aging: Devils in the details? Trends Genet. TIG 2009, 25, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Dagda, R.K.; Cherra, S.J., 3rd; Kulich, S.M.; Tandon, A.; Park, D.; Chu, C.T. Loss of PINK1 function promotes mitophagy through effects on oxidative stress and mitochondrial fission. J. Biol. Chem. 2009, 284, 13843–13855. [Google Scholar] [CrossRef] [PubMed]
- Narendra, D.; Tanaka, A.; Suen, D.F.; Youle, R.J. Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J. Cell Biol. 2008, 183, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Ross, C.A.; Poirier, M.A. Protein aggregation and neurodegenerative disease. Nat. Med. 2004, 10, S10–S17. [Google Scholar] [CrossRef] [PubMed]
- Breydo, L.; Wu, J.W.; Uversky, V.N. Alpha-synuclein misfolding and Parkinson’s disease. Biochim. Biophys. Acta 2012, 1822, 261–285. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, E.R. Protein oxidation and aging. Free Radic. Res. 2006, 40, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Bochem, M.-C.; Herbert, M.; Kuiperij, H.B.; Verbeek, M.M. Oxidized Pin1 and alpha-synuclein in the cerebrospinal fluid as putative biomarkers for Alzheimer’s and Parkinson’s diseases. Alzheimer’s Dement. 2012, 8, P279. [Google Scholar] [CrossRef]
- Ramachandiran, S.; Hansen, J.M.; Jones, D.P.; Richardson, J.R.; Miller, G.W. Divergent mechanisms of paraquat, MPP+, and rotenone toxicity: Oxidation of thioredoxin and caspase-3 activation. Toxicol. Sci. 2007, 95, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Berry, C.; La Vecchia, C.; Nicotera, P. Paraquat and Parkinson’s disease. Cell Death Differ. 2010, 17, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- Livnat-Levanon, N.; Glickman, M.H. Ubiquitin-proteasome system and mitochondria—Reciprocity. Biochim. Biophys. Acta 2011, 1809, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Nunomura, A.; Perry, G.; Aliev, G.; Hirai, K.; Takeda, A.; Balraj, E.K.; Jones, P.K.; Ghanbari, H.; Wataya, T.; Shimohama, S.; et al. Oxidative Damage Is the Earliest Event in Alzheimer Disease. J. Neuropathol. Exp. Neurol. 2001, 60, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Montine, K.S.; Sidell, K.R.; Zhang, J.; Montine, T.J. Dopamine thioethers: Formation in brain and neurotoxicity. Neurotox. Res. 2002, 4, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Sakurai, T.; Usami, H.; Uchida, K. Oxidative modification of proteasome: Identification of an oxidation-sensitive subunit in 26 S proteasome. Biochemistry 2005, 44, 13893–13901. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.M.; Rutter, J. Ubiquitin-dependent mitochondrial protein degradation. Int. J. Biochem. Cell Biol. 2011, 43, 1422–1426. [Google Scholar] [CrossRef] [PubMed]
- Hancock, D.B.; Martin, E.R.; Mayhew, G.M.; Stajich, J.M.; Jewett, R.; Stacy, M.A.; Scott, B.L.; Vance, J.M.; Scott, W.K. Pesticide exposure and risk of Parkinson’s disease: A family-based case-control study. BMC Neurol. 2008, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Elbaz, A.; Clavel, J.; Rathouz, P.J.; Moisan, F.; Galanaud, J.P.; Delemotte, B.; Alperovitch, A.; Tzourio, C. Professional exposure to pesticides and Parkinson disease. Ann. Neurol. 2009, 66, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, R.L. Insect Control. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2000. [Google Scholar]
- Lopez-Arrieta, J.M.; Schneider, L. Metrifonate for Alzheimer’s disease. Cochrane Database Syst. Rev. 2006, 2, CD003155. [Google Scholar]
- Popovska-Gorevski, M.; Dubocovich, M.L.; Rajnarayanan, R.V. Carbamate Insecticides Target Human Melatonin Receptors. Chem. Res. Toxicol. 2017, 30, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Zhang, T.; Jiang, L.; Chi, J.; Hu, D.; Pan, Q.; Wang, D.; Zhang, Z. Regulation of intracellular manganese homeostasis by Kufor-Rakeb syndrome-associated ATP13A2 protein. J. Biol. Chem. 2011, 286, 29654–29662. [Google Scholar] [CrossRef] [PubMed]
- De Vos, K.J.; Sheetz, M.P. Visualization and quantification of mitochondrial dynamics in living animal cells. Methods Cell Biol. 2007, 80, 627–682. [Google Scholar] [PubMed]






| Name | Chemical Class | Mitochondria | Ubiquitin-Proteasome System | |||
|---|---|---|---|---|---|---|
| Morphology | ATP | Complex I | 26S | 20S | ||
| Paraquat | Dipyridyl compounds | Fragmentation | Depletion | Decrease | Inhibition | Inhibition |
| Rotenone | Rotenoids | Fragmentation | Depletion | Decrease | Inhibition | Inhibition |
| Chlorpyrifos | Organophosphates | Fragmentation | Depletion | Decrease | Inhibition | Inhibition |
| Pendimethalin | Dinitroanilines | Fragmentation | No influence | No influence | Inhibition | Inhibition |
| Endosulfan | Organochlorides | Fragmentation | No influence | No influence | Inhibition | Inhibition |
| Fenpyroximate | Acaricides | Fragmentation | Depletion | No influence | Inhibition | Inhibition |
| Tebufenpyrad | Acaricides | Fragmentation | Depletion | No influence | Inhibition | Inhibition |
| Trichlorphon | Organophosphates | Elongation | No influence | No influence | No influence | No influence |
| Carbaryl | Carbamates | Elongation | No influence | No influence | No influence | No influence |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, T.; Tan, J.; Wan, Z.; Zou, Y.; Kessete Afewerky, H.; Zhang, Z.; Zhang, T. Effects of Commonly Used Pesticides in China on the Mitochondria and Ubiquitin-Proteasome System in Parkinson’s Disease. Int. J. Mol. Sci. 2017, 18, 2507. https://doi.org/10.3390/ijms18122507
Chen T, Tan J, Wan Z, Zou Y, Kessete Afewerky H, Zhang Z, Zhang T. Effects of Commonly Used Pesticides in China on the Mitochondria and Ubiquitin-Proteasome System in Parkinson’s Disease. International Journal of Molecular Sciences. 2017; 18(12):2507. https://doi.org/10.3390/ijms18122507
Chicago/Turabian StyleChen, Tingting, Jieqiong Tan, Zhengqing Wan, Yongyi Zou, Henok Kessete Afewerky, Zhuohua Zhang, and Tongmei Zhang. 2017. "Effects of Commonly Used Pesticides in China on the Mitochondria and Ubiquitin-Proteasome System in Parkinson’s Disease" International Journal of Molecular Sciences 18, no. 12: 2507. https://doi.org/10.3390/ijms18122507