Plasma Sphingolipids in Acute Pancreatitis
Abstract
:1. Introduction
2. Results
2.1. Baseline Characteristics of the Patients (Table 1)
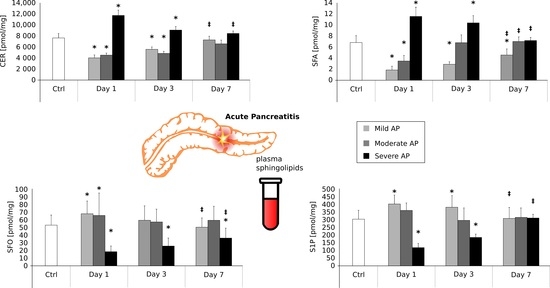
2.2. Plasma Sphinganine Contents (pmol/mg) on the First, Third and Seventh Day after AP Onset (Figure 1)
2.3. Plasma Ceramide Contents (pmol/mg) on the First, Third and Seventh Day after AP Onset (Figure 2)
2.4. Plasma Sphingosine Contents (pmol/mg) on the First, Third and Seventh Day after AP Onset (Figure 3)
2.5. Plasma Sphingosine-1-Phosphate Contents (pmol/mg) on the First, Third and Seventh Day after AP Onset (Figure 4)
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Serum Analyses
4.3. Contents of Plasma Sphinganine (SFA), Sphinganine-1-Phosphate (S1P) and Sphingosine (SFO)
4.4. Plasma Ceramide Content
4.5. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lankisch, P.; Apte, M.; Banks, P. Acute pancreatitis. Lancet 2015, 386, 85–96. [Google Scholar] [CrossRef]
- Singh, V.K.; Wu, B.U.; Bollen, T.L.; Repas, K.; Maurer, R.; Mortele, K.J.; Banks, P.A. Early systemic inflammatory response syndrome is associated with severe acute pancreatitis. Clin. Gastroenterol. Hepatol. 2009, 7, 1247–1251. [Google Scholar] [CrossRef] [PubMed]
- Mofidi, R.; Duff, M.; Wigmore, S.; Madhavan, K.; Garden, O.; Parks, R. Association between early systemic inflammatory response, severity of multiorgan dysfunction and death in acute pancreatitis. Br. J. Surg. 2006, 93, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Windsor, J.A.; Petrov, M.S. Acute pancreatitis reclassified. Gut 2013, 62, 4–5. [Google Scholar] [CrossRef] [PubMed]
- Banks, P.A.; Freeman, M.L. Practice guidelines in acute pancreatitis. Am. J. Gastroenterol. 2006, 101, 2379–2400. [Google Scholar] [CrossRef] [PubMed]
- Arvanitakis, M.; Delhaye, M.; De Maertelaere, V.; Bali, M.; Winant, C.; Coppens, E.; Jeanmart, J.; Zalcman, M.; Van Gansbeke, D.; Devière, J.; et al. Computed tomography and magnetic resonance imaging in the assessment of acute pancreatitis. Gastroenterology 2004, 126, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Banks, P.A.; Bollen, T.L.; Dervenis, C.; Gooszen, H.G.; Johnson, C.D.; Sarr, M.G.; Tsiotos, G.G.; Vege, S.S. Classification of acute pancreatitis—2012: Revision of the atlanta classification and definitions by international consensus. Gut 2013, 62, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Bollen, T.L.; Wu, B.U.; Repas, K.; Maurer, R.; Yu, S.; Mortele, K.J.; Conwell, D.L.; Banks, P.A. An assessment of the severity of interstitial pancreatitis. Clin. Gastroenterol. Hepatol. 2011, 9, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Vege, S.S.; Gardner, T.B.; Chari, S.T.; Munukuti, P.; Pearson, R.K.; Clain, J.E.; Petersen, B.T.; Baron, T.H.; Farnell, M.B.; Sarr, M.G. Low mortality and high morbidity in severe acute pancreatitis without organ failure: A case for revising the atlanta classification to include “moderately severe acute pancreatitis”. Am. J. Gastroenterol. 2009, 104, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Dumnicka, P.; Maduzia, D.; Ceranowicz, P.; Olszanecki, R.; Drożdż, R.; Kuśnierz-Cabala, B. The Interplay between Inflammation, Coagulation and Endothelial Injury in the Early Phase of Acute Pancreatitis: Clinical Implications. Int. J. Mol. Sci. 2017, 18, 354. [Google Scholar] [CrossRef] [PubMed]
- Ceranowicz, P.; Dembinski, A.; Warzecha, Z.; Dembinski, M.; Cieszkowski, J.; Rembisz, K.; Konturek, S.; Kusnierz-Cabala, B.; Tomaszewska, R.; Pawlik, W. Protective and therapeutic effect of heparin in acute pancreatitis. J. Physiol. Pharmacol. 2008, 59, 103–125. [Google Scholar] [PubMed]
- Dumnicka, P.; Kuśnierz-Cabala, B.; Sporek, M.; Mazur-Laskowska, M.; Gil, K.; Kuźniewski, M.; Ceranowicz, P.; Warzecha, Z.; Dembiński, A.; Bonior, J.; et al. Serum concentrations of angiopoietin-2 and soluble fms-like tyrosine kinase 1 (sFlt-1) are associated with coagulopathy among patients with acute pancreatitis. Int. J. Mol. Sci. 2017, 18, 753. [Google Scholar] [CrossRef] [PubMed]
- Minkov, G.A.; Halacheva, K.S.; Yovtchev, Y.P.; Gulubova, M.V. Pathophysiological mechanisms of acute pancreatitis define inflammatory markers of clinical prognosis. Pancreas 2015, 44, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Tenner, S.; Baillie, J.; DeWitt, J.; Vege, S.S. American college of gastroenterology guideline: Management of acute pancreatitis. Am. J. Gastroenterol. 2013, 108, 1400–1415. [Google Scholar] [CrossRef] [PubMed]
- Coant, N.; Sakamoto, W.; Mao, C.; Hannun, Y.A. Ceramidases, roles in sphingolipid metabolism and in health and disease. Adv. Biol. Regul. 2017, 63, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Ogretmen, B.; Hannun, Y.A. Biologically active sphingolipids in cancer pathogenesis and treatment. Nat. Rev. Cancer 2004, 4, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Hannun, Y.A.; Obeid, L.M. Principles of bioactive lipid signalling: Lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 2008, 9, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Hanada, K. Serine palmitoyltransferase, a key enzyme of sphingolipid metabolism. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2003, 1632, 16–30. [Google Scholar] [CrossRef]
- Spiegel, S.; Milstien, S. Sphingosine-1-phosphate: An enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol. 2003, 4, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Gukovskaya, A.S.; Gukovsky, I.; Algül, H.; Habtezion, A. Autophagy, inflammation, and immune dysfunction in the pathogenesis of pancreatitis. Gastroenterology 2017, 153, 1212–1226. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, C.; Zhang, Q.; Tang, C.; Li, N.; Li, J. The role of sphingosine kinase 1 in patients with severe acute pancreatitis. Ann. Surg. 2012, 255, 954–962. [Google Scholar] [CrossRef] [PubMed]
- Fabre, A.; Boulogne, O.; Gaudart, J.; Mas, E.; Olives, J.-P.; Sarles, J. Evaluation of serum lipase as predictor of severity of acute pancreatitis in children. J. Pediatr. Gastroenterol. Nutr. 2014, 58, e41–e42. [Google Scholar] [CrossRef] [PubMed]
- Pezzilli, R.; Brighi, N.; Calculli, L. Quality of life in patients with long-standing chronic non-pathological pancreatic hyperenzymemia. Pancreatology 2015, 15, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Choung, B.S.; Kim, S.H.; Seo, S.Y.; Kim, I.H.; Kim, S.W.; Lee, S.-O.; Lee, S.T. Pancreatic hyperenzymemia is associated with bacterial culture positivity, more severe and right-sided colitis. Dig. Dis. Sci. 2014, 59, 2272–2279. [Google Scholar] [CrossRef] [PubMed]
- Rosołowski, M.; Lipiński, M.; Dobosz, M.; Durlik, M.; Głuszek, S.; Kuśnierz, K.; Lampe, P.; Małecka-Panas, E.; Nowakowska-Duława, E.; Nowak-Niezgoda, M.; et al. Management of acute pancreatitis (ap)–Polish pancreatic club recommendations. Prz. Gastroenterol. 2016, 11, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, Z.; Sendur, P.; Ceranowicz, P.; Cieszkowski, J.; Dembiński, M.; Sendur, R.; Bonior, J.; Jaworek, J.; Ambroży, T.; Olszanecki, R.; et al. Therapeutic effect of low doses of acenocoumarol in the course of ischemia/reperfusion-induced acute pancreatitis in rats. Int. J. Mol. Sci. 2017, 18, 882. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, Z.; Sendur, P.; Ceranowicz, P.; Dembiński, M.; Cieszkowski, J.; Kuśnierz-Cabala, B.; Olszanecki, R.; Tomaszewska, R.; Ambroży, T.; Dembiński, A. Protective effect of pretreatment with acenocoumarol in cerulein-induced acute pancreatitis. Int. J. Mol. Sci. 2016, 17, 1709. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, Z.; Sendur, P.; Ceranowicz, P.; Dembinski, M.; Cieszkowski, J.; Kusnierz-Cabala, B.; Tomaszewska, R.; Dembinski, A. Pretreatment with low doses of acenocoumarol inhibits the development of acute ischemia/reperfusion-induced pancreatitis. J. Physiol. Pharmacol. 2015, 66, 731–740. [Google Scholar] [PubMed]
- Dumnicka, P.; Sporek, M.; Mazur-Laskowska, M.; Ceranowicz, P.; Kuźniewski, M.; Drożdż, R.; Ambroży, T.; Olszanecki, R.; Kuśnierz-Cabala, B. Serum soluble fms-like tyrosine kinase 1 (sFlt-1) predicts the severity of acute pancreatitis. Int. J. Mol. Sci. 2016, 17, 2038. [Google Scholar] [CrossRef] [PubMed]
- Kuśnierz-Cabala, B.; Gala-Błdzińska, A.; Mazur-Laskowska, M.; Dumnicka, P.; Sporek, M.; Matuszyk, A.; Gil, K.; Ceranowicz, P.; Walocha, J.; Kucharz, J.; et al. Serum uromodulin levels in prediction of acute kidney injury in the early phase of acute pancreatitis. Molecules 2017, 22, 988. [Google Scholar] [CrossRef] [PubMed]
- Sporek, M.; Dumnicka, P.; Gala-Błdzińska, A.; Mazur-Laskowska, M.; Walocha, J.; Ceranowicz, P.; Warzecha, Z.; Dembiński, A.; Kuźniewski, M.; Olszanecki, R.; et al. Determination of serum neutrophil gelatinase-associated lipocalin at the early stage of acute pancreatitis. Folia Med. Cracov. 2016, 56, 5–16. [Google Scholar] [PubMed]
- Kurek, K.; Łukaszuk, B.; Piotrowska, D.M.; Wiesiołek, P.; Chabowska, A.M.; Żendzian-Piotrowska, M. Metabolism, physiological role, and clinical implications of sphingolipids in gastrointestinal tract. BioMed Res. Int. 2013, 2013, 908907. [Google Scholar] [CrossRef] [PubMed]
- Kurek, K.; Łukaszuk, B.; Świdnicka-Siergiejko, A.; Rogalski, P.; Wróblewski, E.; Chabowski, A.; Dbrowski, A.; Żendzian-Piotrowska, M. Sphingolipid metabolism in colorectal adenomas varies depending on histological architecture of polyps and grade of nuclear dysplasia. Lipids 2015, 50, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Kurek, K.; Piotrowska, D.M.; Wiesiołek-Kurek, P.; Łukaszuk, B.; Chabowski, A.; Górski, J.; Żendzian-Piotrowska, M. Inhibition of ceramide de novo synthesis reduces liver lipid accumulation in rats with nonalcoholic fatty liver disease. Liver Int. 2014, 34, 1074–1083. [Google Scholar] [CrossRef] [PubMed]
- Dillehay, D.L.; Webb, S.K.; Schmelz, E.-M.; Merrill, A.H., Jr. Dietary sphingomyelin inhibits 1,2-dimethylhydrazine-induced colon cancer in cf1 mice. J. Nutr. 1994, 124, 615. [Google Scholar] [PubMed]
- Jiang, Y.; DiVittore, N.A.; Young, M.M.; Jia, Z.; Xie, K.; Ritty, T.M.; Kester, M.; Fox, T.E. Altered sphingolipid metabolism in patients with metastatic pancreatic cancer. Biomolecules 2013, 3, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Ruiz, C.; Mato, J.M.; Vance, D.; Kaplowitz, N.; Fernández-Checa, J.C. Acid sphingomyelinase-ceramide system in steatohepatitis: A novel target regulating multiple pathways. J. Hepatol. 2015, 62, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Kitano, M.; Hla, T.; Sekiguchi, M.; Kawahito, Y.; Yoshimura, R.; Miyazawa, K.; Iwasaki, T.; Sano, H. Sphingosine 1-phosphate/sphingosine 1-phosphate receptor 1 signaling in rheumatoid synovium: Regulation of synovial proliferation and inflammatory gene expression. Arthritis Rheumatol. 2006, 54, 742–753. [Google Scholar] [CrossRef] [PubMed]
- Puneet, P.; Yap, C.T.; Wong, L.; Yulin, L.; Koh, D.R.; Moochhala, S.; Pfeilschifter, J.; Huwiler, A.; Melendez, A.J. SphK1 regulates proinflammatory responses associated with endotoxin and polymicrobial sepsis. Science 2010, 328, 1290–1294. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.-Q.; Goh, H.H.; Bao, Z.; Wong, W.F.; Melendez, A.J.; Leung, B.P. The role of sphingosine kinase in a murine model of allergic asthma. J. Immunol. 2008, 180, 4323–4329. [Google Scholar] [CrossRef] [PubMed]
- Deguchi, Y.; Andoh, A.; Yagi, Y.; Bamba, S.; Inatomi, O.; Tsujikawa, T.; Fujiyama, Y. The s1p receptor modulator fty720 prevents the development of experimental colitis in mice. Oncol. Rep. 2006, 16, 699–703. [Google Scholar] [CrossRef] [PubMed]
- Snider, A.J.; Kawamori, T.; Bradshaw, S.G.; Orr, K.A.; Gilkeson, G.S.; Hannun, Y.A.; Obeid, L.M. A role for sphingosine kinase 1 in dextran sulfate sodium-induced colitis. FASEB J. 2009, 23, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Anada, Y.; Tani, M.; Ikeda, M.; Sano, T.; Kihara, A.; Igarashi, Y. Lack of sphingosine 1-phosphate-degrading enzymes in erythrocytes. Biochem. Biophys. Res. Commun. 2007, 357, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Knapp, M.; Baranowski, M.; Czarnowski, D.; Lisowska, A.; Zabielski, P.; Gorski, J.; Musial, W. Plasma sphingosine-1-phosphate concentration is reduced in patients with myocardial infarction. Med. Sci. Monit. 2009, 15, CR490–CR493. [Google Scholar] [PubMed]
- Liu, H.-B.; Cui, N.-Q.; Wang, Q.; Li, D.-H.; Xue, X.-P. Sphingosine-1-phosphate and its analogue fty720 diminish acute pulmonary injury in rats with acute necrotizing pancreatitis. Pancreas 2008, 36, e10–e15. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.A.; Belyaev, O.; Burr, W.; Munding, J.; McArthur, N.; Bergmann, U.; Werner, J.; Tannapfel, A.; Uhl, W. Effects of fty720 and rapamycin on inflammation in taurocholate-induced acute pancreatitis in the rat. Pancreas 2012, 41, 1086–1091. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xu, P.; Zhang, L.; Kayoumu, A.; Wang, Y.; Wang, M.; Gao, M.; Zhang, X.; Wang, Y.; Liu, G. FTY720 attenuates acute pancreatitis in hypertriglyceridemic apolipoprotein CIII transgenic mice. Shock 2015, 44, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Min, J.-K.; Yoo, H.-S.; Lee, E.-Y.; Lee, W.-J.; Lee, Y.-M. Simultaneous quantitative analysis of sphingoid base 1-phosphates in biological samples by o-phthalaldehyde precolumn derivatization after dephosphorylation with alkaline phosphatase. Anal. Biochem. 2002, 303, 167–175. [Google Scholar] [CrossRef] [PubMed]




| Variable | Mean Value | ||
|---|---|---|---|
| Number of patients | n = 44 | ||
| Age (y) | |||
| - mean (±SD) | 45 ± 15.6 | ||
| - range, no. | 23–78 | ||
| Sex, no. (%) | |||
| - male | 28 (63.6) | ||
| - female | 16 (36.4) | ||
| Acute pancreatitis aetiology: | |||
| - alcohol abuse | n = 21 (47.7%) | ||
| - biliary | n = 17 (38.6%) | ||
| - hypertriglyceridemia | n = 2 (4.5%) | ||
| - autoimmune pancreatitis | n = 2 (4.5%) | ||
| - groove pancreatitis | n = 1 (2.3%) | ||
| - azathioprine-induced | n = 1 (2.3%) | ||
| Acute pancreatitis severity: | |||
| - mild | n = 18 (40.9%) | ||
| - moderately severe | n = 15 (34.1%) | ||
| - severe | n = 11 (25.0%) | ||
| Day 1 | Day 3 | Day 7 | |
| WBC (103/mm3) (n = 4.0–10.0) | 12.89 ± 4.56 | 8.02 ± 6.36 | 7.38 ± 5.28 |
| HgB (g/dL) (n = 12.5–18.0) | 15.35 ± 3.18 | 13.93 ± 4.37 | 14.21 ± 4.16 |
| RBC (106/mm3) (n = 4.2–5.4) | 5.08 ± 1.09 | 5.26 ± 2.13 | 4.87 ± 2.35 |
| PLT (103/mm3) (n = 130–450) | 237.26 ± 156.97 | 326.83 ± 138.46 | 190.04 ± 167.41 |
| CRP (mg/L) (n ≤ 5) | 151.63 ± 80.24 | 326.35 ± 100.93 * | 40.72 ± 38.31 * |
| PCT (ng/ml) (n ≤ 0.5) | 0.46 ± 2.46 | 0.23 ± 0.15 | 0.11 ± 0.73 |
| Amylase (U/L) (n ≤ 90) | 651.73 ± 156.23 | 206.44 ± 63.94 * | 53.12 ± 46.63 * |
| Lipase (U/L) (n ≤ 70) | 1427.62 ± 648.26 | 430.27 ± 158.73 * | 68.32 ± 45.82 * |
| ALT (U/L) (n ≤ 50) | 117.85 ± 70.82 | 92.34 ± 26.03 | 64.12 ± 30.22 |
| AST (U/L) (n ≤ 50) | 149.96 ± 54.12 | 110.03 ± 41.86 | 93.02 ± 23.74 |
| Bilirubin (mg/dL) (n ≤ 1.1) | 1.26 ± 4.74 | 0.78 ± 2.52 | 1.02 ± 0.83 |
| Na (mmol/L) (n = 135–145) | 136.04 ± 6.92 | 134.82 ± 3.15 | 133.54 ± 4.15 |
| K (mmol/L) (n = 3.7–5.3) | 4.25 ± 1.76 | 4.02 ± 1.82 | 3.98 ± 1.45 |
| Fibrinogen (mg/dL) (n = 200–400) | 581.16 ± 282.34 | 403.26 ± 173.83 | 390.72 ± 210.48 |
| D-dimer (mg/L) (n ≤ 0.5) | 6.12 ± 2.43 | 3.36 ± 0.14 * | 1.92 ± 1.23 * |
| Creatinine (mg/dL) (n = 0.5–1.0) | 1.33 ± 1.04 | 1.00 ± 1.45 | 0.8 ± 1.15 |
| Urea (mg/dL) (n ≤ 50) | 37.57 ± 34.27 | 45.18 ± 23.72 | 29.36 ± 27.03 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konończuk, T.; Łukaszuk, B.; Żendzian-Piotrowska, M.; Dąbrowski, A.; Krzyżak, M.; Ostrowska, L.; Kurek, K. Plasma Sphingolipids in Acute Pancreatitis. Int. J. Mol. Sci. 2017, 18, 2606. https://doi.org/10.3390/ijms18122606
Konończuk T, Łukaszuk B, Żendzian-Piotrowska M, Dąbrowski A, Krzyżak M, Ostrowska L, Kurek K. Plasma Sphingolipids in Acute Pancreatitis. International Journal of Molecular Sciences. 2017; 18(12):2606. https://doi.org/10.3390/ijms18122606
Chicago/Turabian StyleKonończuk, Tomasz, Bartłomiej Łukaszuk, Małgorzata Żendzian-Piotrowska, Andrzej Dąbrowski, Michalina Krzyżak, Lucyna Ostrowska, and Krzysztof Kurek. 2017. "Plasma Sphingolipids in Acute Pancreatitis" International Journal of Molecular Sciences 18, no. 12: 2606. https://doi.org/10.3390/ijms18122606