Review of Prospects of Biological Fluid Biomarkers in Osteoarthritis
Abstract
:1. Introduction
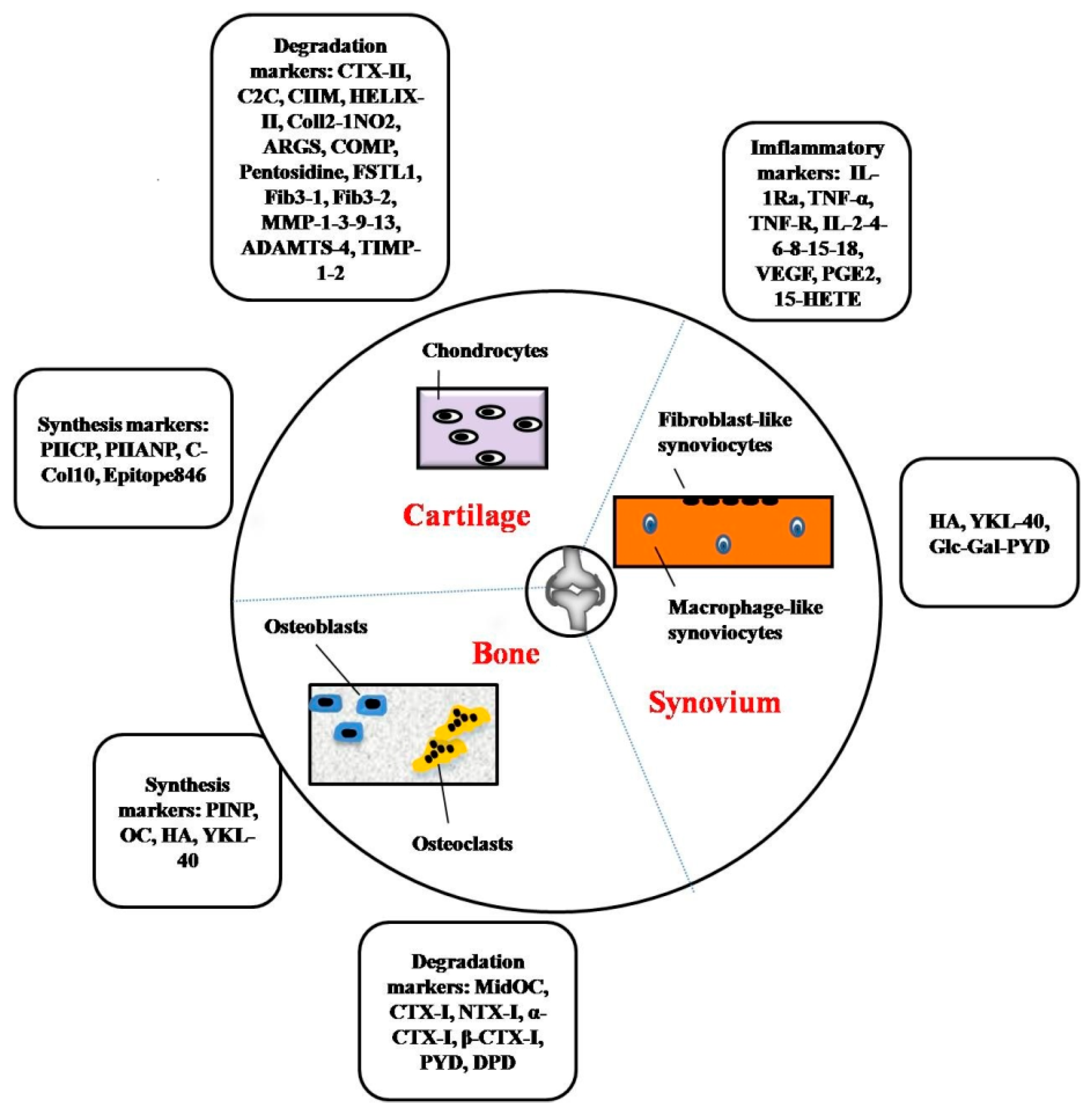
2. Biomarkers for Cartilage, Bone and Synovium Metabolism
2.1. Markers of Cartilage Metabolism
2.2. Markers of Bone Metabolism
2.3. Markers of Synovium Metabolism
3. Inflammatory Markers
3.1. Bone-, Cartilage- and Synovium-Derived Markers
3.1.1. Cytokines
3.1.2. Chemokines and Growth Factors
3.1.3. Lipid Mediators
3.2. Markers Related to Other Tissues
3.2.1. Acute Phase Protein
3.2.2. Obesity-Associated Factors
3.2.3. Other Factors
4. Genetic Markers
5. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
References
- Brandt, K.D.; Radin, E.L.; Dieppe, P.A.; van de Putte, L. Yet more evidence that osteoarthritis is not a cartilage disease. Ann. Rheum. Dis. 2006, 65, 1261–1264. [Google Scholar] [CrossRef] [PubMed]
- Pap, T.; Korb-Pap, A. Cartilage damage in osteoarthritis and rheumatoid arthritis—Two unequal siblings. Nat. Rev. Rheumatol. 2015, 11, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, R.C.; Felson, D.T.; Helmick, C.G.; Arnold, L.M.; Choi, H.; Deyo, R.A.; Gabriel, S.; Hirsch, R.; Hochberg, M.C.; Hunder, G.G.; et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheumatol. 2008, 58, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Massart, F.; Reginster, J.Y.; Brandi, M.L. Genetics of menopause-associated diseases. Maturitas 2001, 40, 103–116. [Google Scholar] [CrossRef]
- Teichtahl, A.J.; Wang, Y.; Wluka, A.E.; Cicuttini, F.M. Obesity and knee osteoarthritis: New insights provided by body composition studies. Obesity (Silver Spring) 2008, 16, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Blom, A.B.; van Lent, P.L.; Libregts, S.; Holthuysen, A.E.; van der Kraan, P.M.; van Rooijen, N.; van den Berg, W.B. Crucial role of macrophages in matrix metalloproteinase-mediated cartilage destruction during experimental osteoarthritis: Involvement of matrix metalloproteinase 3. Arthritis Rheum. 2007, 56, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Zhen, G.; Wen, C.; Jia, X.; Li, Y.; Crane, J.L.; Mears, S.C.; Askin, F.B.; Frassica, F.J.; Chang, W.; Yao, J.; et al. Inhibition of TGF-beta signaling in mesenchymal stem cells of subchondral bone attenuates osteoarthritis. Nat. Med. 2013, 19, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Favero, M.; Ramonda, R.; Goldring, M.B.; Goldring, S.R.; Punzi, L. Early knee osteoarthritis. RMD Open 2015, 1, e000062. [Google Scholar] [CrossRef] [PubMed]
- Ramonda, R.; Favero, M.; Vio, S.; Lacognata, C.; Frallonardo, P.; Belluzzi, E.; Campana, C.; Lorenzin, M.; Ortolan, A.; Angelini, F.; et al. A recently developed MRI scoring system for hand osteoarthritis: Its application in a clinical setting. Clin. Rheumatol. 2016, 35, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Saarakkala, S.; Waris, P.; Waris, V.; Tarkiainen, I.; Karvanen, E.; Aarnio, J.; Koski, J.M. Diagnostic performance of knee ultrasonography for detecting degenerative changes of articular cartilage. Osteoarthr. Cartil. 2012, 20, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Yoshihara, Y.; Yamada, H.; Fujikawa, K. Procollagen IIc-peptide as a marker for assessing mechanical risk factors of knee osteoarthritis: Effect of obesity and varus alignment. Ann. Rheum. Dis. 2000, 59, 982–984. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, J.C.; Sandell, L.J.; Delmas, P.D.; Garnero, P. Development and clinical application in arthritis of a new immunoassay for serum type IIa procollagen NH2 propeptide. Methods Mol. Med. 2004, 101, 25–37. [Google Scholar] [PubMed]
- Rousseau, J.C.; Zhu, Y.; Miossec, P.; Vignon, E.; Sandell, L.J.; Garnero, P.; Delmas, P.D. Serum levels of type IIa procollagen amino terminal propeptide (PIIANP) are decreased in patients with knee osteoarthritis and rheumatoid arthritis. Osteoarthr. Cartil. 2004, 12, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Sharif, M.; Kirwan, J.; Charni, N.; Sandell, L.J.; Whittles, C.; Garnero, P. A 5-yr longitudinal study of type IIa collagen synthesis and total type ii collagen degradation in patients with knee osteoarthritis—Association with disease progression. Rheumatology 2007, 46, 938–943. [Google Scholar] [CrossRef] [PubMed]
- Garnero, P.; Ayral, X.; Rousseau, J.C.; Christgau, S.; Sandell, L.J.; Dougados, M.; Delmas, P.D. Uncoupling of type II collagen synthesis and degradation predicts progression of joint damage in patients with knee osteoarthritis. Arthritis Rheum. 2002, 46, 2613–2624. [Google Scholar] [CrossRef] [PubMed]
- Kraus, V.B.; Collins, J.E.; Hargrove, D.; Losina, E.; Nevitt, M.; Katz, J.N.; Wang, S.X.; Sandell, L.J.; Hoffmann, S.C.; Hunter, D.J.; et al. Predictive validity of biochemical biomarkers in knee osteoarthritis: Data from the fnih oa biomarkers consortium. Ann. Rheum. Dis. 2017, 76, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.; Christgau, S.; Lukoschek, M.; Henriksen, D.; Richter, W. Increased urinary concentration of collagen type II c-telopeptide fragments in patients with osteoarthritis. Pathobiology 2004, 71, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Meulenbelt, I.; Kloppenburg, M.; Kroon, H.M.; Houwing-Duistermaat, J.J.; Garnero, P.; Hellio Le Graverand, M.P.; Degroot, J.; Slagboom, P.E. Urinary CTX-II levels are associated with radiographic subtypes of osteoarthritis in hip, knee, hand, and facet joints in subject with familial osteoarthritis at multiple sites: The garp study. Ann. Rheum. Dis. 2006, 65, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Rotterud, J.H.; Reinholt, F.P.; Beckstrom, K.J.; Risberg, M.A.; Aroen, A. Relationship between CTX-II and patient characteristics, patient-reported outcome, muscle strength, and rehabilitation in patients with a focal cartilage lesion of the knee: A prospective exploratory cohort study of 48 patients. BMC Musculoskelet. Disord. 2014, 15, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanishi, N.; Yamagiwa, H.; Hayami, T.; Mera, H.; Koga, Y.; Omori, G.; Endo, N. Usefulness of urinary CTX-II and NTX-I in evaluating radiological knee osteoarthritis : The matsudai knee osteoarthritis survey. J. Orthop. Sci. 2014, 19, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Gungen, G.O.; Ardic, F.; Findikoglu, G.; Rota, S. Effect of mud compress therapy on cartilage destruction detected by ctx-ii in patients with knee osteoarthritis. J. Back Musculoskelet. Rehabil. 2016, 29, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Garnero, P.; Conrozier, T.; Christgau, S.; Mathieu, P.; Delmas, P.D.; Vignon, E. Urinary type II collagen c-telopeptide levels are increased in patients with rapidly destructive hip osteoarthritis. Ann. Rheum. Dis. 2003, 62, 939–943. [Google Scholar] [CrossRef] [PubMed]
- Lohmander, L.S.; Atley, L.M.; Pietka, T.A.; Eyre, D.R. The release of crosslinked peptides from type II collagen into human synovial fluid is increased soon after joint injury and in osteoarthritis. Arthritis Rheum. 2003, 48, 3130–3139. [Google Scholar] [CrossRef] [PubMed]
- Conrozier, T.; Poole, A.R.; Ferrand, F.; Mathieu, P.; Vincent, F.; Piperno, M.; Verret, C.; Ionescu, M.; Vignon, E. Serum concentrations of type II collagen biomarkers (C2C, C1, 2C and CPII) suggest different pathophysiologies in patients with hip osteoarthritis. Clin. Exp. Rheumatol. 2008, 26, 430–435. [Google Scholar] [PubMed]
- Kumahashi, N.; Sward, P.; Larsson, S.; Lohmander, L.S.; Frobell, R.; Struglics, A. Type II collagen C2C epitope in human synovial fluid and serum after knee injury—Associations with molecular and structural markers of injury. Osteoarthr. Cartil. 2015, 23, 1506–1512. [Google Scholar] [CrossRef] [PubMed]
- He, G.; Chen, X.; Zhang, G.; Lin, H.; Li, R.; Wu, X. Detection of urine C2C and trace element level in patients with knee osteoarthritis. Cell Biochem. Biophys. 2014, 70, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Bay-Jensen, A.C.; Liu, Q.; Byrjalsen, I.; Li, Y.; Wang, J.; Pedersen, C.; Leeming, D.J.; Dam, E.B.; Zheng, Q.; Qvist, P.; et al. Enzyme-linked immunosorbent assay (elisas) for metalloproteinase derived type II collagen neoepitope, CIIM—Increased serum ciim in subjects with severe radiographic osteoarthritis. Clin. Biochem. 2011, 44, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Charni, N.; Juillet, F.; Garnero, P. Urinary type II collagen helical peptide (helix-II) as a new biochemical marker of cartilage degradation in patients with osteoarthritis and rheumatoid arthritis. Arthritis Rheum. 2005, 52, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Punzi, L.; Ramonda, R.; Deberg, M.; Frallonardo, P.; Campana, C.; Musacchio, E.; Henrotin, Y. Coll2-1, Coll2-1NO2 and myeloperoxidase serum levels in erosive and non-erosive osteoarthritis of the hands. Osteoarthr. Cartil. 2012, 20, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Hosseininia, S.; Weis, M.A.; Rai, J.; Kim, L.; Funk, S.; Dahlberg, L.E.; Eyre, D.R. Evidence for enhanced collagen type III deposition focally in the territorial matrix of osteoarthritic hip articular cartilage. Osteoarthr. Cartil. 2016. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Siebuhr, A.S.; Brandt-Hansen, N.U.; Wang, J.; Su, D.; Zheng, Q.; Simonsen, O.; Petersen, K.K.; Arendt-Nielsen, L.; Eskehave, T.; et al. Type X collagen levels are elevated in serum from human osteoarthritis patients and associated with biomarkers of cartilage degradation and inflammation. BMC Musculoskelet. Disord. 2014, 15, 309. [Google Scholar] [CrossRef] [PubMed]
- Lohmander, L.S.; Ionescu, M.; Jugessur, H.; Poole, A.R. Changes in joint cartilage aggrecan after knee injury and in osteoarthritis. Arthritis Rheum. 1999, 42, 534–544. [Google Scholar] [CrossRef]
- Larsson, S.; Lohmander, L.S.; Struglics, A. Synovial fluid level of aggrecan args fragments is a more sensitive marker of joint disease than glycosaminoglycan or aggrecan levels: A cross-sectional study. Arthritis Res. Ther. 2009, 11, R92. [Google Scholar] [CrossRef] [PubMed]
- Germaschewski, F.M.; Matheny, C.J.; Larkin, J.; Liu, F.; Thomas, L.R.; Saunders, J.S.; Sully, K.; Whittall, C.; Boyle, Y.; Peters, G.; et al. Quantitation of ARGS aggrecan fragments in synovial fluid, serum and urine from osteoarthritis patients. Osteoarthr. Cartil. 2014, 22, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, F.A.; Pucinelli, M.L.; da Silva, N.P.; Feldman, D. Serum cartilage oligomeric matrix protein (COMP) levels in knee osteoarthritis in a Brazilian population: Clinical and radiological correlation. Scand. J. Rheumatol. 2007, 36, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Dalal, K. Serum cartilage oligomeric matrix protein (COMP) in knee osteoarthritis: A novel diagnostic and prognostic biomarker. J. Orthop. Res. 2013, 31, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Senolt, L.; Braun, M.; Olejarova, M.; Forejtova, S.; Gatterova, J.; Pavelka, K. Increased pentosidine, an advanced glycation end product, in serum and synovial fluid from patients with knee osteoarthritis and its relation with cartilage oligomeric matrix protein. Ann. Rheum. Dis. 2005, 64, 886–890. [Google Scholar] [CrossRef] [PubMed]
- Pavelka, K.; Forejtova, S.; Olejarova, M.; Gatterova, J.; Senolt, L.; Spacek, P.; Braun, M.; Hulejova, M.; Stovickova, J.; Pavelkova, A. Hyaluronic acid levels may have predictive value for the progression of knee osteoarthritis. Osteoarthr. Cartil. 2004, 12, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, D.; Xu, N.; Tao, W.; Zhu, R.; Sun, R.; Fan, W.; Zhang, P.; Dong, T.; Yu, L. Follistatin-like protein 1: A serum biochemical marker reflecting the severity of joint damage in patients with osteoarthritis. Arthritis Res. Ther. 2011, 13, R193. [Google Scholar] [CrossRef] [PubMed]
- Henrotin, Y.; Gharbi, M.; Mazzucchelli, G.; Dubuc, J.E.; De Pauw, E.; Deberg, M. Fibulin 3 peptides Fib3-1 and Fib3-2 are potential biomarkers of osteoarthritis. Arthritis Rheum. 2012, 64, 2260–2267. [Google Scholar] [CrossRef] [PubMed]
- Runhaar, J.; Sanchez, C.; Taralla, S.; Henrotin, Y.; Bierma-Zeinstra, S.M. Fibulin-3 fragments are prognostic biomarkers of osteoarthritis incidence in overweight and obese women. Osteoarthr. Cartil. 2016, 24, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, L.; Min, J.; Yang, H.; Xu, X.; Yuan, Y.; Wang, D. Levels of metalloproteinase (MMP-3, MMP-9), NF-kappab ligand (RANKL), and nitric oxide (NO) in peripheral blood of osteoarthritis (OA) patients. Clin. Lab. 2012, 58, 755–762. [Google Scholar] [PubMed]
- Rubenhagen, R.; Schuttrumpf, J.P.; Sturmer, K.M.; Frosch, K.H. Interleukin-7 levels in synovial fluid increase with age and MMP-1 levels decrease with progression of osteoarthritis. Acta Orthop. 2012, 83, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Ozler, K.; Aktas, E.; Atay, C.; Yilmaz, B.; Arikan, M.; Gungor, S. Serum and knee synovial fluid matrix metalloproteinase-13 and tumor necrosis factor-alpha levels in patients with late-stage osteoarthritis. Acta Orthop. Traumatol. Turc. 2016, 50, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Du, C.; Wang, H.; Zhang, C. Increased serum ADAMTS-4 in knee osteoarthritis: A potential indicator for the diagnosis of osteoarthritis in early stages. Genet. Mol. Res. 2014, 13, 9642–9649. [Google Scholar] [CrossRef] [PubMed]
- Ishiguro, N.; Ito, T.; Ito, H.; Iwata, H.; Jugessur, H.; Ionescu, M.; Poole, A.R. Relationship of matrix metalloproteinases and their inhibitors to cartilage proteoglycan and collagen turnover: Analyses of synovial fluid from patients with osteoarthritis. Arthritis Rheum. 1999, 42, 129–136. [Google Scholar] [CrossRef]
- Kumm, J.; Tamm, A.; Lintrop, M. Diagnostic and prognostic value of bone biomarkers in progressive knee osteoarthritis: A 6-year follow-up study in middle-aged subjects. Osteoarthr. Cartil. 2013, 21, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Bettica, P.; Cline, G.; Hart, D.J.; Meyer, J.; Spector, T.D. Evidence for increased bone resorption in patients with progressive knee osteoarthritis: Longitudinal results from the chingford study. Arthritis Rheum. 2002, 46, 3178–3184. [Google Scholar] [CrossRef] [PubMed]
- Huebner, J.L.; Bay-Jensen, A.C.; Huffman, K.M.; He, Y.; Leeming, D.J.; McDaniel, G.E.; Karsdal, M.A.; Kraus, V.B. Alpha c-telopeptide of type I collagen is associated with subchondral bone turnover and predicts progression of joint space narrowing and osteophytes in osteoarthritis. Arthritis Rheumatol. 2014, 66, 2440–2449. [Google Scholar] [CrossRef] [PubMed]
- Graverand, M.P.; Tron, A.M.; Ichou, M.; Dallard, M.C.; Richard, M.; Uebelhart, D.; Vignon, E. Assessment of urinary hydroxypyridinium cross-links measurement in osteoarthritis. Br. J. Rheumatol. 1996, 35, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, E.; Tsuda, E.; Yamamoto, Y.; Iwasaki, K.; Inoue, R.; Takahashi, I.; Sawada, K.; Fujita, H.; Umeda, T.; Nakaji, S.; et al. Serum hyaluronan levels increase with the total number of osteoarthritic joints and are strongly associated with the presence of knee and finger osteoarthritis. Int. Orthop. 2013, 37, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Filkova, M.; Senolt, L.; Braun, M.; Hulejova, H.; Pavelkova, A.; Sleglova, O.; Kupka, K.; Gatterova, J.; Pavelka, K. Serum hyaluronic acid as a potential marker with a predictive value for further radiographic progression of hand osteoarthritis. Osteoarthr. Cartil. 2009, 17, 1615–1619. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, H.; Ishijima, M.; Doi, T.; Futami, I.; Liu, L.; Sadatsuki, R.; Yusup, A.; Hada, S.; Kubota, M.; Kawasaki, T.; et al. Reference intervals of serum hyaluronic acid corresponding to the radiographic severity of knee osteoarthritis in women. BMC Musculoskelet. Disord. 2013, 14, 34. [Google Scholar] [CrossRef] [PubMed]
- Conrozier, T.; Carlier, M.C.; Mathieu, P.; Colson, F.; Debard, A.L.; Richard, S.; Favret, H.; Bienvenu, J.; Vignon, E. Serum levels of YKL-40 and C reactive protein in patients with hip osteoarthritis and healthy subjects: A cross sectional study. Ann. Rheum. Dis. 2000, 59, 828–831. [Google Scholar] [CrossRef] [PubMed]
- Vaananen, T.; Koskinen, A.; Paukkeri, E.L.; Hamalainen, M.; Moilanen, T.; Moilanen, E.; Vuolteenaho, K. YKL-40 as a novel factor associated with inflammation and catabolic mechanisms in osteoarthritic joints. Mediat. Inflamm. 2014, 2014, 215140. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Liu, Z.; Li, F.; Feng, J.S.; Wang, H.J.; Chu, J.G.; Song, Y.Z.; Xie, L.; Ding, L.B. Increased synovial fluid YKL-40 levels are linked with symptomatic severity in knee osteoarthritis patients. Clin. Lab. 2015, 61, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Jordan, K.M.; Syddall, H.E.; Garnero, P.; Gineyts, E.; Dennison, E.M.; Sayer, A.A.; Delmas, P.D.; Cooper, C.; Arden, N.K. Urinary CTX-II and glucosyl-galactosyl-pyridinoline are associated with the presence and severity of radiographic knee osteoarthritis in men. Ann. Rheum. Dis. 2006, 65, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Garnero, P.; Piperno, M.; Gineyts, E.; Christgau, S.; Delmas, P.D.; Vignon, E. Cross sectional evaluation of biochemical markers of bone, cartilage, and synovial tissue metabolism in patients with knee osteoarthritis: Relations with disease activity and joint damage. Ann. Rheum. Dis. 2001, 60, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Aigner, T.; Zhu, Y.; Chansky, H.H.; Matsen, F.A., 3rd; Maloney, W.J.; Sandell, L.J. Reexpression of type IIa procollagen by adult articular chondrocytes in osteoarthritic cartilage. Arthritis Rheum. 1999, 42, 1443–1450. [Google Scholar] [CrossRef]
- Clutter, S.D.; Wilson, D.C.; Marinov, A.D.; Hirsch, R. Follistatin-like protein 1 promotes arthritis by up-regulating IFN-gamma. J. Immunol. 2009, 182, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Malfait, A.M.; Liu, R.Q.; Ijiri, K.; Komiya, S.; Tortorella, M.D. Inhibition of ADAM-TS4 and ADAM-TS5 prevents aggrecan degradation in osteoarthritic cartilage. J. Biol. Chem. 2002, 277, 22201–22208. [Google Scholar] [CrossRef] [PubMed]
- Neuhold, L.A.; Killar, L.; Zhao, W.; Sung, M.L.; Warner, L.; Kulik, J.; Turner, J.; Wu, W.; Billinghurst, C.; Meijers, T.; et al. Postnatal expression in hyaline cartilage of constitutively active human collagenase-3 (MMP-13) induces osteoarthritis in mice. J. Clin. Investig. 2001, 107, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.; Nagase, H. Reappraising metalloproteinases in rheumatoid arthritis and osteoarthritis: Destruction or repair? Nat. Clin. Pract. Rheumatol. 2008, 4, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.J.; Temenoff, J.S. Engineering orthopedic tissue interfaces. Tissue Eng. Part B Rev. 2009, 15, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Blair-Levy, J.M.; Watts, C.E.; Fiorentino, N.M.; Dimitriadis, E.K.; Marini, J.C.; Lipsky, P.E. A type I collagen defect leads to rapidly progressive osteoarthritis in a mouse model. Arthritis Rheum. 2008, 58, 1096–1106. [Google Scholar] [CrossRef] [PubMed]
- Ivaska, K.K.; Hentunen, T.A.; Vaaraniemi, J.; Ylipahkala, H.; Pettersson, K.; Vaananen, H.K. Release of intact and fragmented osteocalcin molecules from bone matrix during bone resorption in vitro. J. Biol. Chem. 2004, 279, 18361–18369. [Google Scholar] [CrossRef] [PubMed]
- Hui, A.Y.; McCarty, W.J.; Masuda, K.; Firestein, G.S.; Sah, R.L. A systems biology approach to synovial joint lubrication in health, injury, and disease. Wiley Interdiscip. Rev. Syst. Biol. Med. 2012, 4, 15–37. [Google Scholar] [CrossRef] [PubMed]
- Johansen, J.S.; Olee, T.; Price, P.A.; Hashimoto, S.; Ochs, R.L.; Lotz, M. Regulation of YKL-40 production by human articular chondrocytes. Arthritis Rheum. 2001, 44, 826–837. [Google Scholar] [CrossRef]
- Hakala, B.E.; White, C.; Recklies, A.D. Human cartilage GP-39, a major secretory product of articular chondrocytes and synovial cells, is a mammalian member of a chitinase protein family. J. Biol. Chem. 1993, 268, 25803–25810. [Google Scholar] [PubMed]
- De Ceuninck, F.; Gaufillier, S.; Bonnaud, A.; Sabatini, M.; Lesur, C.; Pastoureau, P. YKL-40 (cartilage GP-39) induces proliferative events in cultured chondrocytes and synoviocytes and increases glycosaminoglycan synthesis in chondrocytes. Biochem. Biophys. Res. Commun. 2001, 285, 926–931. [Google Scholar] [CrossRef] [PubMed]
- Gineyts, E.; Garnero, P.; Delmas, P.D. Urinary excretion of glucosyl-galactosyl pyridinoline: A specific biochemical marker of synovium degradation. Rheumatology 2001, 40, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.P.; Martel-Pelletier, J.; Abramson, S.B. Osteoarthritis, an inflammatory disease: Potential implication for the selection of new therapeutic targets. Arthritis Rheum. 2001, 44, 1237–1247. [Google Scholar] [CrossRef]
- Benito, M.J.; Veale, D.J.; FitzGerald, O.; van den Berg, W.B.; Bresnihan, B. Synovial tissue inflammation in early and late osteoarthritis. Ann. Rheum. Dis. 2005, 64, 1263–1267. [Google Scholar] [CrossRef] [PubMed]
- Myers, S.L.; Brandt, K.D.; Ehlich, J.W.; Braunstein, E.M.; Shelbourne, K.D.; Heck, D.A.; Kalasinski, L.A. Synovial inflammation in patients with early osteoarthritis of the knee. J. Rheumatol. 1990, 17, 1662–1669. [Google Scholar] [PubMed]
- Hennerbichler, A.; Moutos, F.T.; Hennerbichler, D.; Weinberg, J.B.; Guilak, F. Interleukin-1 and tumor necrosis factor alpha inhibit repair of the porcine meniscus in vitro. Osteoarthr. Cartil. 2007, 15, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Hiraoka, K.; Hoshino, T.; Okamoto, M.; Iwanaga, T.; Zenmyo, M.; Shoda, T.; Aizawa, H.; Nagata, K. High levels of serum IL-18 promote cartilage loss through suppression of aggrecan synthesis. Bone 2008, 42, 1102–1110. [Google Scholar] [CrossRef] [PubMed]
- Attur, M.; Statnikov, A.; Samuels, J.; Li, Z.; Alekseyenko, A.V.; Greenberg, J.D.; Krasnokutsky, S.; Rybak, L.; Lu, Q.A.; Todd, J.; et al. Plasma levels of interleukin-1 receptor antagonist (IL-1Ra) predict radiographic progression of symptomatic knee osteoarthritis. Osteoarthr. Cartil. 2015, 23, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Stannus, O.; Jones, G.; Cicuttini, F.; Parameswaran, V.; Quinn, S.; Burgess, J.; Ding, C. Circulating levels of IL-6 and TNF-alpha are associated with knee radiographic osteoarthritis and knee cartilage loss in older adults. Osteoarthr. Cartil. 2010, 18, 1441–1447. [Google Scholar] [CrossRef] [PubMed]
- Penninx, B.W.; Abbas, H.; Ambrosius, W.; Nicklas, B.J.; Davis, C.; Messier, S.P.; Pahor, M. Inflammatory markers and physical function among older adults with knee osteoarthritis. J. Rheumatol. 2004, 31, 2027–2031. [Google Scholar] [PubMed]
- Stannus, O.P.; Jones, G.; Quinn, S.J.; Cicuttini, F.M.; Dore, D.; Ding, C. The association between leptin, interleukin-6, and hip radiographic osteoarthritis in older people: A cross-sectional study. Arthritis Res. Ther. 2010, 12, R95. [Google Scholar] [CrossRef] [PubMed]
- Shimura, Y.; Kurosawa, H.; Sugawara, Y.; Tsuchiya, M.; Sawa, M.; Kaneko, H.; Futami, I.; Liu, L.; Sadatsuki, R.; Hada, S.; et al. The factors associated with pain severity in patients with knee osteoarthritis vary according to the radiographic disease severity: A cross-sectional study. Osteoarthr. Cartil. 2013, 21, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- Livshits, G.; Zhai, G.; Hart, D.J.; Kato, B.S.; Wang, H.; Williams, F.M.; Spector, T.D. Interleukin-6 is a significant predictor of radiographic knee osteoarthritis: The Chingford study. Arthritis Rheum. 2009, 60, 2037–2045. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.M.; Sun, L.Z.; Liu, J.; Su, B.H.; Shi, L. Serum interleukin-15 levels are associated with severity of pain in patients with knee osteoarthritis. Dis. Markers 2013, 35, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, D.; Long, L.; Deng, X.; Tao, R.; Huang, G. Correlation between plasma, synovial fluid and articular cartilage interleukin-18 with radiographic severity in 33 patients with osteoarthritis of the knee. Clin. Exp. Med. 2014, 14, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Mabey, T.; Honsawek, S.; Tanavalee, A.; Yuktanandana, P.; Wilairatana, V.; Poovorawan, Y. Plasma and synovial fluid inflammatory cytokine profiles in primary knee osteoarthritis. Biomarkers 2016, 21, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.Y.; Yang, Z.B.; Zhang, Z.J.; Zhang, Z.Q.; Kang, Y.; Huang, G.X.; Wang, S.W.; Huang, H.; Liao, W.M. CCL3 serves as a potential plasma biomarker in knee degeneration (osteoarthritis). Osteoarthr. Cartil. 2015, 23, 1405–1411. [Google Scholar] [CrossRef] [PubMed]
- Pierzchala, A.W.; Kusz, D.J.; Hajduk, G. CXCL8 and CCL5 expression in synovial fluid and blood serum in patients with osteoarthritis of the knee. Arch. Immunol. Ther. Exp. 2011, 59, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Saetan, N.; Honsawek, S.; Tanavalee, A.; Yuktanandana, P.; Meknavin, S.; Ngarmukos, S.; Tanpowpong, T.; Parkpian, V. Relationship of plasma and synovial fluid vascular endothelial growth factor with radiographic severity in primary knee osteoarthritis. Int. Orthop. 2014, 38, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Attur, M.; Krasnokutsky, S.; Statnikov, A.; Samuels, J.; Li, Z.; Friese, O.; Hellio Le Graverand-Gastineau, M.P.; Rybak, L.; Kraus, V.B.; Jordan, J.M.; et al. Low-grade inflammation in symptomatic knee osteoarthritis: Prognostic value of inflammatory plasma lipids and peripheral blood leukocyte biomarkers. Arthritis Rheumatol. 2015, 67, 2905–2915. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Ramirez, D.C.; van der Leeden, M.; van der Esch, M.; Gerritsen, M.; Roorda, L.D.; Verschueren, S.; van Dieen, J.; Dekker, J.; Lems, W.F. Association of serum C-reactive protein and erythrocyte sedimentation rate with muscle strength in patients with knee osteoarthritis. Rheumatology 2013, 52, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Stannus, O.P.; Jones, G.; Blizzard, L.; Cicuttini, F.M.; Ding, C. Associations between serum levels of inflammatory markers and change in knee pain over 5 years in older adults: A prospective cohort study. Ann. Rheum. Dis. 2013, 72, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Arendt-Nielsen, L.; Eskehave, T.N.; Egsgaard, L.L.; Petersen, K.K.; Graven-Nielsen, T.; Hoeck, H.C.; Simonsen, O.; Siebuhr, A.S.; Karsdal, M.; Bay-Jensen, A.C. Association between experimental pain biomarkers and serologic markers in patients with different degrees of painful knee osteoarthritis. Arthritis Rheumatol. 2014, 66, 3317–3326. [Google Scholar] [CrossRef] [PubMed]
- Punzi, L.; Ramonda, R.; Oliviero, F.; Sfriso, P.; Mussap, M.; Plebani, M.; Podswiadek, M.; Todesco, S. Value of C reactive protein in the assessment of erosive osteoarthritis of the hand. Ann. Rheum. Dis. 2005, 64, 955–957. [Google Scholar] [CrossRef] [PubMed]
- Saberi Hosnijeh, F.; Siebuhr, A.S.; Uitterlinden, A.G.; Oei, E.H.; Hofman, A.; Karsdal, M.A.; Bierma-Zeinstra, S.M.; Bay-Jensen, A.C.; van Meurs, J.B. Association between biomarkers of tissue inflammation and progression of osteoarthritis: Evidence from the Rotterdam study cohort. Arthritis Res. Ther. 2016, 18, 81. [Google Scholar] [CrossRef] [PubMed]
- Koskinen, A.; Juslin, S.; Nieminen, R.; Moilanen, T.; Vuolteenaho, K.; Moilanen, E. Adiponectin associates with markers of cartilage degradation in osteoarthritis and induces production of proinflammatory and catabolic factors through mitogen-activated protein kinase pathways. Arthritis Res. Ther. 2011, 13, R184. [Google Scholar] [CrossRef] [PubMed]
- Oliviero, F.; Sfriso, P.; Baldo, G.; Dayer, J.M.; Giunco, S.; Scanu, A.; Bernardi, D.; Ramonda, R.; Plebani, M.; Punzi, L. Apolipoprotein A-I and cholesterol in synovial fluid of patients with rheumatoid arthritis, psoriatic arthritis and osteoarthritis. Clin. Exp. Rheumatol. 2009, 27, 79–83. [Google Scholar] [PubMed]
- Li, L.; Jiang, B.E. Serum and synovial fluid chemokine ligand 2/monocyte chemoattractant protein 1 concentrations correlates with symptomatic severity in patients with knee osteoarthritis. Ann. Clin. Biochem. 2015, 52, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Guerne, P.A.; Carson, D.A.; Lotz, M. IL-6 production by human articular chondrocytes. Modulation of its synthesis by cytokines, growth factors, and hormones in vitro. J. Immunol. 1990, 144, 499–505. [Google Scholar] [PubMed]
- Lotz, M.; Terkeltaub, R.; Villiger, P.M. Cartilage and joint inflammation. Regulation of IL-8 expression by human articular chondrocytes. J. Immunol. 1992, 148, 466–473. [Google Scholar] [PubMed]
- Daheshia, M.; Yao, J.Q. The interleukin 1beta pathway in the pathogenesis of osteoarthritis. J. Rheumatol. 2008, 35, 2306–2312. [Google Scholar] [CrossRef] [PubMed]
- Kunisch, E.; Kinne, R.W.; Alsalameh, R.J.; Alsalameh, S. Pro-inflammatory IL-1beta and/or TNF-alpha up-regulate matrix metalloproteases-1 and -3 mRNA in chondrocyte subpopulations potentially pathogenic in osteoarthritis: In situ hybridization studies on a single cell level. Int. J. Rheum. Dis. 2016, 19, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Poree, B.; Kypriotou, M.; Chadjichristos, C.; Beauchef, G.; Renard, E.; Legendre, F.; Melin, M.; Gueret, S.; Hartmann, D.J.; Mallein-Gerin, F.; et al. Interleukin-6 (IL-6) and/or soluble IL-6 receptor down-regulation of human type II collagen gene expression in articular chondrocytes requires a decrease of Sp1.Sp3 ratio and of the binding activity of both factors to the COL2A1 promoter. J. Biol. Chem. 2008, 283, 4850–4865. [Google Scholar] [CrossRef] [PubMed]
- Murata, M.; Yudoh, K.; Masuko, K. The potential role of vascular endothelial growth factor (VEGF) in cartilage: How the angiogenic factor could be involved in the pathogenesis of osteoarthritis? Osteoarthr. Cartil. 2008, 16, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, P.C.; Castell, J.V.; Andus, T. Interleukin-6 and the acute phase response. Biochem. J. 1990, 265, 621–636. [Google Scholar] [CrossRef] [PubMed]
- Vigushin, D.M.; Pepys, M.B.; Hawkins, P.N. Metabolic and scintigraphic studies of radioiodinated human C-reactive protein in health and disease. J. Clin. Investig. 1993, 91, 1351–1357. [Google Scholar] [CrossRef] [PubMed]
- Oliviero, F.; Ramonda, R.; Punzi, L. New horizons in osteoarthritis. Swiss Med. Wkly. 2010, 140, w13098. [Google Scholar] [CrossRef] [PubMed]
- Belluzzi, E.; El Hadi, H.; Granzotto, M.; Rossato, M.; Ramonda, R.; Macchi, V.; de Caro, R.; Vettor, R.; Favero, M. Systemic and local adipose tissue in knee osteoarthritis. J. Cell. Physiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Tong, K.M.; Chen, C.P.; Huang, K.C.; Shieh, D.C.; Cheng, H.C.; Tzeng, C.Y.; Chen, K.H.; Chiu, Y.C.; Tang, C.H. Adiponectin increases MMP-3 expression in human chondrocytes through Adipor1 signaling pathway. J. Cell. Biochem. 2011, 112, 1431–1440. [Google Scholar] [CrossRef] [PubMed]
- Koskinen, A.; Vuolteenaho, K.; Nieminen, R.; Moilanen, T.; Moilanen, E. Leptin enhances MMP-1, MMP-3 and MMP-13 production in human osteoarthritic cartilage and correlates with MMP-1 and MMP-3 in synovial fluid from oa patients. Clin. Exp. Rheumatol. 2011, 29, 57–64. [Google Scholar] [PubMed]
- De Boer, T.N.; van Spil, W.E.; Huisman, A.M.; Polak, A.A.; Bijlsma, J.W.; Lafeber, F.P.; Mastbergen, S.C. Serum adipokines in osteoarthritis; comparison with controls and relationship with local parameters of synovial inflammation and cartilage damage. Osteoarthr. Cartil. 2012, 20, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.K.; Ke, Y.; Wang, B.; Lin, J.H. The role of MCP-1-CCR2 ligand-receptor axis in chondrocyte degradation and disease progress in knee osteoarthritis. Biol. Res. 2015, 48, 64. [Google Scholar] [CrossRef] [PubMed]
- Al-Mughales, J.; Blyth, T.H.; Hunter, J.A.; Wilkinson, P.C. The chemoattractant activity of rheumatoid synovial fluid for human lymphocytes is due to multiple cytokines. Clin. Exp. Immunol. 1996, 106, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Yuan, G.H.; Masuko-Hongo, K.; Sakata, M.; Tsuruha, J.; Onuma, H.; Nakamura, H.; Aoki, H.; Kato, T.; Nishioka, K. The role of C-C chemokines and their receptors in osteoarthritis. Arthritis Rheum. 2001, 44, 1056–1070. [Google Scholar] [CrossRef]
- Ramonda, R.; Lorenzin, M.; Modesti, V.; Campana, C.; Ortolan, A.; Frallonardo, P.; Punzi, L. Serological markers of erosive hand osteoarthritis. Eur. J. Intern. Med. 2013, 24, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, N.; Rasheed, Z.; Ramamurthy, S.; Anbazhagan, A.N.; Voss, F.R.; Haqqi, T.M. Microrna-27b regulates the expression of matrix metalloproteinase 13 in human osteoarthritis chondrocytes. Arthritis Rheum. 2010, 62, 1361–1371. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Tian, B.; Qu, X.; Liu, F.; Tang, T.; Qin, A.; Zhu, Z.; Dai, K. MicroRNAs play a role in chondrogenesis and osteoarthritis (review). Int. J. Mol. Med. 2014, 34, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Nugent, M. Micrornas: Exploring new horizons in osteoarthritis. Osteoarthr. Cartil. 2016, 24, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.W.; Watkins, G.; Le Good, N.; Roberts, S.; Murphy, C.L.; Brockbank, S.M.; Needham, M.R.; Read, S.J.; Newham, P. The identification of differentially expressed microRNA in osteoarthritic tissue that modulate the production of TNF-alpha and MMP13. Osteoarthr. Cartil. 2009, 17, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Miyaki, S.; Sato, T.; Inoue, A.; Otsuki, S.; Ito, Y.; Yokoyama, S.; Kato, Y.; Takemoto, F.; Nakasa, T.; Yamashita, S.; et al. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev. 2010, 24, 1173–1185. [Google Scholar] [CrossRef] [PubMed]
- Matsukawa, T.; Sakai, T.; Yonezawa, T.; Hiraiwa, H.; Hamada, T.; Nakashima, M.; Ono, Y.; Ishizuka, S.; Nakahara, H.; Lotz, M.K.; et al. MicroRNA-125b regulates the expression of aggrecanase-1 (ADAMTS-4) in human osteoarthritic chondrocytes. Arthritis Res. Ther. 2013, 15, R28. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kroin, J.S.; Kc, R.; Gibson, G.; Chen, D.; Corbett, G.T.; Pahan, K.; Fayyaz, S.; Kim, J.S.; van Wijnen, A.J.; et al. Altered spinal microRNA-146a and the microRNA-183 cluster contribute to osteoarthritic pain in knee joints. J. Bone Miner. Res. 2013, 28, 2512–2522. [Google Scholar] [CrossRef] [PubMed]
- Murata, K.; Yoshitomi, H.; Tanida, S.; Ishikawa, M.; Nishitani, K.; Ito, H.; Nakamura, T. Plasma and synovial fluid micrornas as potential biomarkers of rheumatoid arthritis and osteoarthritis. Arthritis Res. Ther. 2010, 12, R86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borgonio Cuadra, V.M.; Gonzalez-Huerta, N.C.; Romero-Cordoba, S.; Hidalgo-Miranda, A.; Miranda-Duarte, A. Altered expression of circulating microrna in plasma of patients with primary osteoarthritis and in silico analysis of their pathways. PLoS ONE 2014, 9, e97690. [Google Scholar] [CrossRef] [PubMed]
- Beyer, C.; Zampetaki, A.; Lin, N.Y.; Kleyer, A.; Perricone, C.; Iagnocco, A.; Distler, A.; Langley, S.R.; Gelse, K.; Sesselmann, S.; et al. Signature of circulating microRNAs in osteoarthritis. Ann. Rheum. Dis. 2015, 74, e18. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yang, M.; Marks, P.; White, L.M.; Hurtig, M.; Mi, Q.S.; Divine, G.; Gibson, G. Serum non-coding RNAs as biomarkers for osteoarthritis progression after Acl injury. Osteoarthr. Cartil. 2012, 20, 1631–1637. [Google Scholar] [CrossRef] [PubMed]
- Pattrick, M.; Manhire, A.; Ward, A.M.; Doherty, M. HLA-a, b antigens and alpha 1-antitrypsin phenotypes in nodal generalised osteoarthritis and erosive osteoarthritis. Ann. Rheum. Dis. 1989, 48, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Moos, V.; Menard, J.; Sieper, J.; Sparmann, M.; Müller, B. Association of HLA-DRB1*02 with osteoarthritis in a cohort of 106 patients. Rheumatology 2002, 41, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Riyazi, N.; Spee, J.; Huizinga, T.W.; Schreuder, G.M.; de Vries, R.R.; Dekker, F.W.; Kloppenburg, M. HLA class II is associated with distal interphalangeal osteoarthritis. Ann. Rheum. Dis. 2003, 62, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Ramonda, R.; Musacchio, E.; Campana, C.; Frigato, M.; Frallonardo, P.; Barbieri, V.; Piccoli, A.; Valvason, C.; Bronte, V.; Zanovello, P.; et al. Immunogenetic aspects of erosive osteoarthritis of the hand in patients from northern Italy. Scand. J. Rheumatol. 2011, 40, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Stern, A.G.; de Carvalho, M.R.; Buck, G.A.; Adler, R.A.; Rao, T.P.; Disler, D.; Moxley, G.; Network, I.-N. Association of erosive hand osteoarthritis with a single nucleotide polymorphism on the gene encoding interleukin-1 beta. Osteoarthr. Cartil. 2003, 11, 394–402. [Google Scholar] [CrossRef]

| Tissue Origination | Molecule Type Origination | Markers of Synthesis | Markers of Degradation | Sample Type | References |
|---|---|---|---|---|---|
| Cartilage | Type II collagen | PIICP 2 | SF | [11] | |
| PIIANP 2 | S | [12,13,14,15,16] | |||
| CTX-II 1,2,3,4 | U | [17,18,19,20,21,22] | |||
| CTX-II 2 | SF | [23] | |||
| C2C 3 | S | [24] | |||
| C2C 2 | U, SF | [25,26] | |||
| CIIM 2 | S | [27] | |||
| HELIX-II 2 | U | [28] | |||
| Coll 2-1 NO2 1 | S | [28,29] | |||
| Type X collagen | C-Col10 2 | S | [30,31] | ||
| Aggrecan | Epitope 846 2 | SF | [32] | ||
| ARGS 2 | SF | [33,34] | |||
| Non-collagenous and non-agrrecan proteins | COMP 2 | S | [35,36] | ||
| Pentosidine 2 | S, SF | [37,38] | |||
| FSTL1 2,3 | S, SF | [39] | |||
| Fib3-1 2 | S | [40,41] | |||
| Fib3-2 2 | S | [40] | |||
| Proteolytic enzymes | MMP-3, -9 2 | S | [42] | ||
| MMP-1, -13 2 | SF | [43,44] | |||
| ADAMTS-4 2 | S | [45] | |||
| Proteolytic enzyme inhibitors | TIMP-1, -2 2 | SF | [46] | ||
| Bone | Type I collagen | PINP 2 | S | [47] | |
| Non-collagenous protein | OC 2 | S | [47] | ||
| MidOC 2 | U | [47] | |||
| CTX-I 2 | U | [48] | |||
| NTX-I 2 | U | ||||
| Alpha-CTX-I 2 | U | [16,49] | |||
| Beta-CTX-I 2 | U | [16] | |||
| PYD 2,3 | U | [50] | |||
| DPD 2,3 | U | [50] | |||
| Synovium | Non-collagenous proteins | HA 1,2 | S | [38,51,52,53] | |
| YKL-40 3 | S | [54] | |||
| YKL-40 2 | SF | [54,55,56] | |||
| Type III collagen | Glc-Gal-PYD 2 | U | [57,58] |
| Tissue Origination | Classification of Inflammatory Markers | Biomarkers | Sample Type | References |
|---|---|---|---|---|
| Cartilage, bone, synovium-deprived markers | Cytokines | IL-1Ra 2 | S | [77] |
| TNF-α 2 | S | [44,78] | ||
| TNF-Rs 2 | S | [79] | ||
| IL-6 2,3 | S | [80,81,82] | ||
| IL-15 2 | S | [83] | ||
| IL-18 2 | S | [84] | ||
| IL-2, -4 2 | S | [85] | ||
| Chemokines and growth factors | IL-8 2 | S, SF | [86,87] | |
| VEGF 2 | S, SF | [43,88] | ||
| Lipid mediators | PGE2 2 | S | [89] | |
| 15-HETE 2 | S | [89] | ||
| Liver | Acute phase protein | CRP 1,2 | S | [90,91,92,93] |
| CRPM | S | [94] | ||
| Adipose tissue | Obesity-related inflammatory factors | Resistin 2 | S | [86] |
| Leptin 3 | S | [80] | ||
| Adioponectin 2 | S | [95] | ||
| ApoA1 | S | [96] | ||
| TC | S | [96] | ||
| Macrophages | Cytokines | CCL3 2 | S | [86] |
| CCL4 2 | S | [86] | ||
| Neutrophils | Enzyme | CCL2 2 | SF | [97] |
| MPO 1 | S | [29] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, L.T.; Sharma, A.R.; Chakraborty, C.; Saibaba, B.; Ahn, M.-E.; Lee, S.-S. Review of Prospects of Biological Fluid Biomarkers in Osteoarthritis. Int. J. Mol. Sci. 2017, 18, 601. https://doi.org/10.3390/ijms18030601
Nguyen LT, Sharma AR, Chakraborty C, Saibaba B, Ahn M-E, Lee S-S. Review of Prospects of Biological Fluid Biomarkers in Osteoarthritis. International Journal of Molecular Sciences. 2017; 18(3):601. https://doi.org/10.3390/ijms18030601
Chicago/Turabian StyleNguyen, Lich Thi, Ashish Ranjan Sharma, Chiranjib Chakraborty, Balaji Saibaba, Moo-Eob Ahn, and Sang-Soo Lee. 2017. "Review of Prospects of Biological Fluid Biomarkers in Osteoarthritis" International Journal of Molecular Sciences 18, no. 3: 601. https://doi.org/10.3390/ijms18030601







