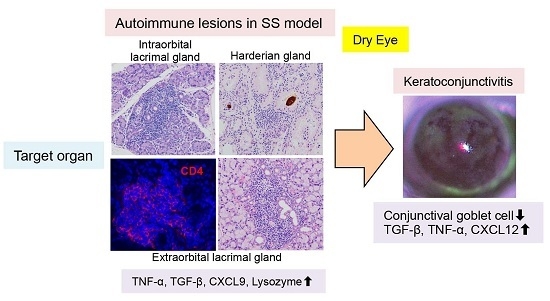
Pathological Analysis of Ocular Lesions in a Murine Model of Sjögren’s Syndrome
Abstract
:1. Introduction
2. Results
2.1. Dry Eye in Sjögren’s Syndrome (SS) Model Mice
2.1.1. Tear Secretion
2.1.2. Cytokines, Chemokines, and Growth Factors in Tears
2.1.3. Corneal Epithelium
2.1.4. Histology of Corneal Epithelium
2.1.5. Conjunctival Goblet Cells
2.1.6. Gene Expression in Corneal Epithelium
2.2. Inflammatory Lesions of Lacrimal Glands in SS Model Mice
2.2.1. Pathology of Lacrimal Glands
2.2.2. Immunological Analysis of Extraorbital Lacrimal Glands in SS Model
2.2.3. Change in Gene Expression in Extraorbital Lacrimal Glands in the SS Model Mice
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Tear Secretion Measurements
4.3. Cytokines, Chemokines, and Growth Factors in Tear
4.4. Evaluation of Ocular Surfaces
4.5. Histological Evaluations
4.6. Immunofluorescence Staining
4.7. Flow Cytometry Analysis
4.8. Quantitative Real-Time Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SS | Sjögren’s syndrome |
| VEGF | Vascular endothelial growth factor |
| MIP-1α | Macrophage inflammatory protein-1α |
| TNF-α | Tumor necrosis factor-α |
| TGF-β | Transforming growth factor-β |
| CXCL9 | C-X-C motif ligand 9 |
| CXCL12 | C-X-C motif ligand 12 |
References
- Fox, R.I. Sjogren’s syndrome. Lancet 2005, 366, 321–331. [Google Scholar] [CrossRef]
- Nocturne, G.; Mariette, X. Advances in understanding the pathogenesis of primary Sjogren’s syndrome. Nat. Rev. Rheumatol. 2013, 9, 544–556. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.S.; Rocha, E.M. Sjogren syndrome: What and where are we looking for? Curr. Opin. Ophthalmol. 2015, 26, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Brito-Zeron, P.; Baldini, C.; Bootsma, H.; Bowman, S.J.; Jonsson, R.; Mariette, X.; Sivils, K.; Theander, E.; Tzioufas, A.; Ramos-Casals, M. Sjögren syndrome. Nat. Rev. Dis. Primers 2016, 2, 16047. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.S.; Gauna, A.E.; Cha, S. Mouse Models of Primary Sjögren’s Syndrome. Curr. Pharm. Des. 2015, 21, 2350–2364. [Google Scholar] [CrossRef] [PubMed]
- Haneji, N.; Hamano, H.; Yanagi, K.; Hayashi, Y. A new animal model for primary Sjögren’s syndrome in NFS/sld mutant mice. J. Immunol. 1994, 153, 2769–2777. [Google Scholar] [PubMed]
- Haneji, N.; Nakamura, T.; Takio, K.; Yanagi, K.; Higashiyama, H.; Saito, I.; Noji, S.; Sugino, H.; Hayashi, Y. Identification of α-fodrin as a candidate autoantigen in primary Sjögren’s syndrome. Science 1997, 276, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, N.; Yoneda, T.; Saegusa, K.; Yanagi, K.; Haneji, N.; Moriyama, K.; Saito, I.; Hayashi, Y. Severe destructive autoimmune lesions with aging in murine Sjögren’s syndrome through Fas-mediated apoptosis. Am. J. Pathol. 2000, 156, 1557–1564. [Google Scholar] [CrossRef]
- Saegusa, K.; Ishimaru, N.; Yanagi, K.; Arakaki, R.; Ogawa, K.; Saito, I.; Katunuma, N.; Hayashi, Y. Cathepsin S inhibitor prevents autoantigen presentation and autoimmunity. J. Clin. Investig. 2002, 110, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Kohashi, M.; Ishimaru, N.; Arakaki, R.; Hayashi, Y. Effective treatment with oral administration of rebamipide in a mouse model of Sjögren’s syndrome. Arthritis Rheum. 2008, 58, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Arakaki, R.; Eguchi, H.; Yamada, A.; Kudo, Y.; Iwasa, A.; Enkhmaa, T.; Hotta, F.; Mitamura-Aizawa, S.; Mitamura, Y.; Hayashi, Y.; et al. Anti-inflammatory effects of rebamipide eyedrop administration on ocular lesions in a murine model of primary Sjögren’s syndrome. PLoS ONE 2014, 9, e98390. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, G.; Baccari, G.C.; di Matteo, L.; d’Istria, M.; Minucci, S.; Varriale, B. Cell biology of the harderian gland. Int. Rev. Cytol. 1996, 168, 1–80. [Google Scholar] [PubMed]
- Kittridge, A.; Routhouska, S.B.; Korman, N.J. Dermatologic manifestations of Sjogren syndrome. J. Cutan. Med. Surg. 2011, 15, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Hatron, P.Y.; Tillie-Leblond, I.; Launay, D.; Hachulla, E.; Fauchais, A.L.; Wallaert, B. Pulmonary manifestations of Sjögren’s syndrome. Press. Med. 2011, 40, e49–e64. [Google Scholar] [CrossRef] [PubMed]
- Segal, B.M.; Pogatchnik, B.; Holker, E.; Liu, H.; Sloan, J.; Rhodus, N.; Moser, K.L. Primary Sjögren’s syndrome: Cognitive symptoms, mood, and cognitive performance. Acta Neurol. Scand. 2012, 125, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Francois, H.; Mariette, X. Renal involvement in primary Sjögren syndrome. Nat. Rev. Nephrol. 2016, 12, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Cornec, D.; Jamin, C.; Pers, J.O. Sjögren’s syndrome: Where do we stand, and where shall we go? J. Autoimmun. 2014, 51, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Carlsoo, B.; Ostberg, Y. The autoimmune submandibular sialoadenitis of the NZB/NZW hybrid mice. A light and electron microscopical investigation. Arch. Otorhinolaryngol. 1979, 225, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Talal, N.; Fischbach, M. Interleukins in experimental autoimmune disease. Adv. Exp. Med. Biol. 1983, 166, 105–115. [Google Scholar] [PubMed]
- Wicker, L.S.; Appel, M.C.; Dotta, F.; Pressey, A.; Miller, B.J.; DeLarato, N.H.; Fischer, P.A.; Boltz, R.C., Jr.; Peterson, L.B. Autoimmune syndromes in major histocompatibility complex (MHC) congenic strains of nonobese diabetic (NOD) mice. The NOD MHC is dominant for insulitis and cyclophosphamide-induced diabetes. J. Exp. Med. 1992, 176, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.Q.; Cornelius, J.G.; Cooper, L.; Neff, J.; Tao, J.; Lee, B.H.; Peck, A.B. Identification of possible candidate genes regulating Sjogren’s syndrome-associated autoimmunity: A potential role for TNFSF4 in autoimmune exocrinopathy. Arthritis Res. Ther. 2008, 10, R137. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.Q.; Sharma, A.; She, J.X.; McIndoe, R.A.; Peck, A.B. Differential gene expressions in the lacrimal gland during development and onset of keratoconjunctivitis sicca in Sjögren’s syndrome (SJS)-like disease of the C57BL/6.NOD-Aec1Aec2 mouse. Exp. Eye Res. 2009, 88, 398–409. [Google Scholar] [CrossRef] [PubMed]
- Tsubata, R.; Tsubata, T.; Hiai, H.; Shinkura, R.; Matsumura, R.; Sumida, T.; Miyawaki, S.; Ishida, H.; Kumagai, S.; Nakao, K.; et al. Autoimmune disease of exocrine organs in immunodeficient alymphoplasia mice: A spontaneous model for Sjögren’s syndrome. Eur. J. Immunol. 1996, 26, 2742–2748. [Google Scholar] [CrossRef] [PubMed]
- Ellis, J.S.; Wan, X.; Braley-Mullen, H. Transient depletion of CD4+ CD25+ regulatory T cells results in multiple autoimmune diseases in wild-type and B-cell-deficient NOD mice. Immunology 2013, 139, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Saegusa, K.; Ishimaru, N.; Yanagi, K.; Haneji, N.; Nishino, M.; Azuma, M.; Saito, I.; Hayashi, Y. Autoantigen-specific CD4+CD28low T cell subset prevents autoimmune exocrinopathy in murine Sjögren’s syndrome. J. Immunol. 2000, 165, 2251–2257. [Google Scholar] [CrossRef] [PubMed]
- Saito, I.; Haruta, K.; Shimuta, M.; Inoue, H.; Sakurai, H.; Yamada, K.; Ishimaru, N.; Higashiyama, H.; Sumida, T.; Ishida, H.; et al. Fas ligand-mediated exocrinopathy resembling Sjögren’s syndrome in mice transgenic for IL-10. J. Immunol. 1999, 162, 2488–2494. [Google Scholar] [PubMed]
- Shen, L.; Zhang, C.; Wang, T.; Brooks, S.; Ford, R.J.; Lin-Lee, Y.C.; Kasianowicz, A.; Kumar, V.; Martin, L.; Liang, P.; et al. Development of autoimmunity in IL-14α-transgenic mice. J. Immunol. 2006, 177, 5676–5686. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.S.; Volpe, E.A.; Gandhi, N.B.; Zhang, X.; Zheng, X.; Pitcher, J.D., 3rd; Farley, W.J.; Stern, M.E.; Niederkorn, J.Y.; Li, D.Q.; et al. Disruption of TGF-β signaling improves ocular surface epithelial disease in experimental autoimmune keratoconjunctivitis sicca. PLoS ONE 2011, 6, e29017. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, N.; Mitani, T.; Takeda, N.; Ishimaru, N.; Arakaki, R.; Hayashi, Y.; Bando, Y.; Izumi, K.; Takahashi, T.; Nomura, T.; et al. Development of autoimmunity against transcriptionally unrepressed target antigen in the thymus of Aire-deficient mice. J. Immunol. 2005, 174, 1862–1870. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Nishikawa, Y.; Nishijima, H.; Morimoto, J.; Matsumoto, M.; Mouri, Y. Which model better fits the role of aire in the establishment of self-tolerance: The transcription model or the maturation model? Front. Immunol. 2013, 4, 210. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, N.; Kishimoto, H.; Hayashi, Y.; Sprent, J. Regulation of naive T cell function by the NF-κB2 pathway. Nat. Immunol. 2006, 7, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, N.; Arakaki, R.; Yoshida, S.; Yamada, A.; Noji, S.; Hayashi, Y. Expression of the retinoblastoma protein RbAp48 in exocrine glands leads to Sjögren’s syndrome-like autoimmune exocrinopathy. J. Exp. Med. 2008, 205, 2915–2927. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, N.; Yamada, A.; Nitta, T.; Arakaki, R.; Lipp, M.; Takahama, Y.; Hayashi, Y. CCR7 with S1P1 signaling through AP-1 for migration of Foxp3+ regulatory T-cells controls autoimmune exocrinopathy. Am. J. Pathol. 2012, 180, 199–208. [Google Scholar] [CrossRef] [PubMed]
- DeVoss, J.J.; LeClair, N.P.; Hou, Y.; Grewal, N.K.; Johannes, K.P.; Lu, W.; Yang, T.; Meagher, C.; Fong, L.; Strauss, E.C.; et al. An autoimmune response to odorant binding protein 1a is associated with dry eye in the Aire-deficient mouse. J. Immunol. 2010, 184, 4236–4246. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.T.; Chen, F.Y.; Vijmasi, T.; Stephens, D.N.; Gallup, M.; McNamara, N.A. Pax6 downregulation mediates abnormal lineage commitment of the ocular surface epithelium in aqueous-deficient dry eye disease. PLoS ONE 2013, 8, e77286. [Google Scholar] [CrossRef] [PubMed]
- Maes, C.; Carmeliet, G.; Schipani, E. Hypoxia-driven pathways in bone development, regeneration and disease. Nat. Rev. Rheumatol. 2012, 8, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Colligris, B.; Alkozi, H.A.; Pintor, J. Recent developments on dry eye disease treatment compounds. Saudi J. Ophthalmol. 2014, 28, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Scofield, R.H.; Hyon, J.Y.; Yun, P.Y.; Lee, H.J.; Lee, E.Y.; Lee, E.B.; Song, Y.W. Salivary chemokine levels in patients with primary Sjögren’s syndrome. Rheumatology 2010, 49, 1747–1752. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-β: The master regulator of fibrosis. Nat. Rev. Nephrol. 2016, 12, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Monroy, D.; Ji, Z.; Yoshino, K.; Huang, A.; Pflugfelder, S.C. Transforming growth factor β-1 and β-2 in human tear fluid. Curr. Eye Res. 1996, 15, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Bourcier, T.; Berbar, T.; Paquet, S.; Rondeau, N.; Thomas, F.; Borderie, V.; Laroche, L.; Rostene, W.; Haour, F.; Lombet, A. Characterization and functionality of CXCR4 chemokine receptor and SDF-1 in human corneal fibroblasts. Mol. Vis. 2003, 9, 96–102. [Google Scholar] [PubMed]
- Ojeda, A.F.; Munjaal, R.P.; Lwigale, P.Y. Expression of CXCL12 and CXCL14 during eye development in chick and mouse. Gene Expr. Patterns 2013, 13, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Lu, P.; Li, L.; Jin, H.; He, X.; Mukaida, N.; Zhang, X. Critical role of SDF-1α-induced progenitor cell recruitment and macrophage VEGF production in the experimental corneal neovascularization. Mol. Vis. 2011, 17, 2129–2138. [Google Scholar] [PubMed]
- Azuma, M.; Motegi, K.; Aota, K.; Hayashi, Y.; Sato, M. Role of cytokines in the destruction of acinar structure in Sjögren’s syndrome salivary glands. Lab. Investig. 1997, 77, 269–280. [Google Scholar] [PubMed]
- Azuma, M.; Ashida, Y.; Tamatani, T.; Motegi, K.; Takamaru, N.; Ishimaru, N.; Hayashi, Y.; Sato, M. Cepharanthin, a biscoclaurine alkaloid, prevents destruction of acinar tissues in murine Sjögren’s syndrome. J. Rheumatol. 2006, 33, 912–920. [Google Scholar] [PubMed]






© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ushio, A.; Arakaki, R.; Eguchi, H.; Hotta, F.; Yamada, A.; Kudo, Y.; Ishimaru, N. Pathological Analysis of Ocular Lesions in a Murine Model of Sjögren’s Syndrome. Int. J. Mol. Sci. 2017, 18, 1209. https://doi.org/10.3390/ijms18061209
Ushio A, Arakaki R, Eguchi H, Hotta F, Yamada A, Kudo Y, Ishimaru N. Pathological Analysis of Ocular Lesions in a Murine Model of Sjögren’s Syndrome. International Journal of Molecular Sciences. 2017; 18(6):1209. https://doi.org/10.3390/ijms18061209
Chicago/Turabian StyleUshio, Aya, Rieko Arakaki, Hiroshi Eguchi, Fumika Hotta, Akiko Yamada, Yasusei Kudo, and Naozumi Ishimaru. 2017. "Pathological Analysis of Ocular Lesions in a Murine Model of Sjögren’s Syndrome" International Journal of Molecular Sciences 18, no. 6: 1209. https://doi.org/10.3390/ijms18061209