1. Drug Hypersensitivity Reactions: New Clinical Approach Through Phenotypes, Endotypes, and Biomarkers
Drug hypersensitivity reactions (HSRs) are adverse effects of drugs [
1,
2]. Among the four most common HSRs described by Gell and Coombs, the most studied reactions are IgE (Immunoglobuline E)/mast cell mediated reactions which can cause cardiovascular collapse and anaphylaxis, leading to drug discontinuation which decrease quality of life and/or life expectancy [
3,
4,
5,
6].
The classification of HSRs relies on the clinical presentation of typical symptoms and their timing [
2,
7], and were originally described by Gell and Coombs [
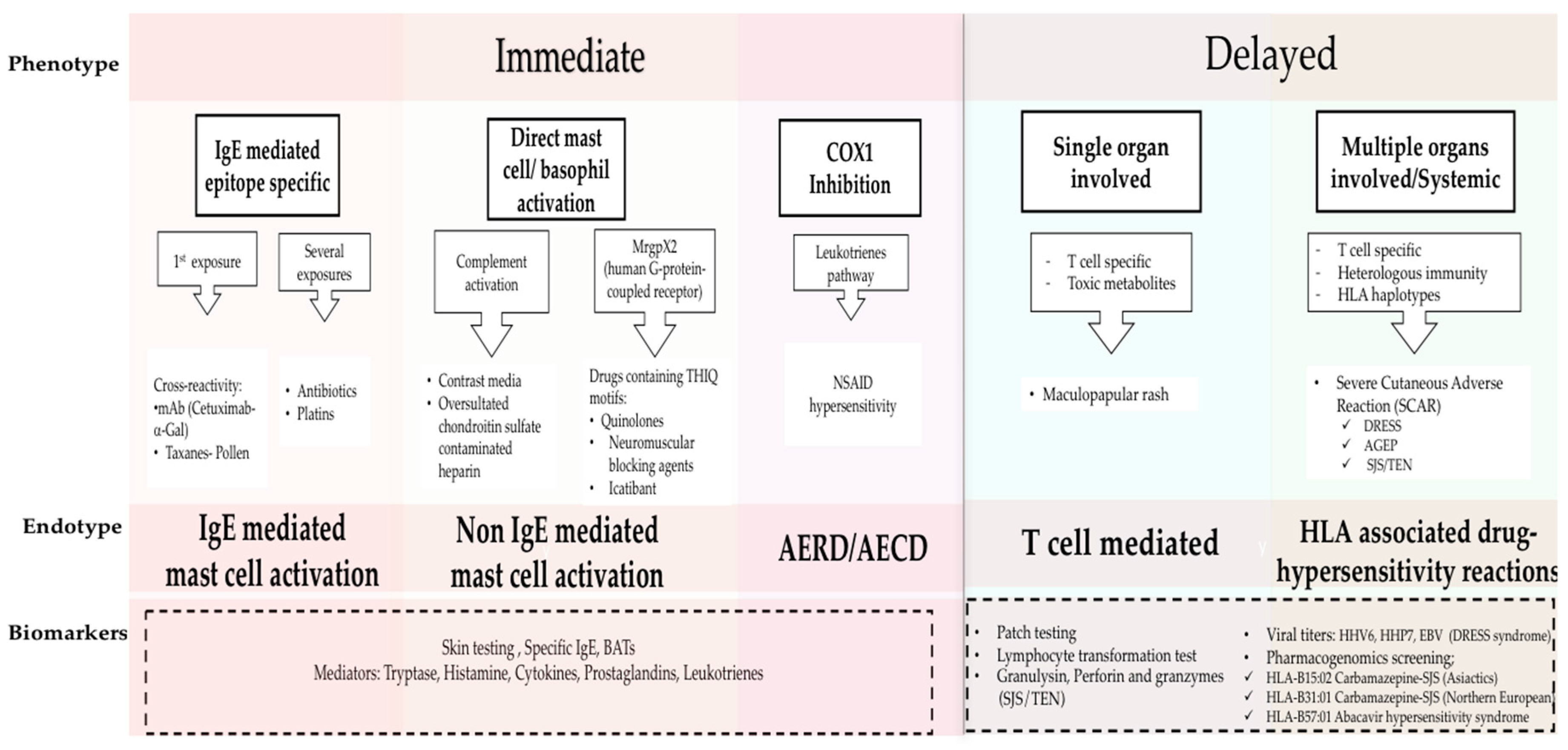
8]: namely Type I (IgE mediated reactions), Type II (antibody mediated cytotoxicity reactions), Type III (immune complex-mediated reactions), and Type IV for delayed type hypersensitivity. Recently phenotypes, endotypes, and genotypes for these HSRs are being elucidated and applied to provide personalized approaches to treating and managing HSRs (
Figure 1) [
1]. Phenotypes in drug allergy focus on symptoms and timing, classifying the reactions as immediate or delayed, depending on the time between treatment administration and the onset of symptoms. Endotypes, based on cellular and biological mediators as well as biomarkers, have become vital to elucidate the molecular pathways as well as to evaluate the risk for reaction during re-exposure to the culprit drug [
1,
9,
10,
11,
12]. Genetic predisposition has been shown to play a role in the development of HSRs to anticonvulsants, sulfonamides, and abacavir among others. Further investigation into pharmacogenetics will lead to prevention and better management of severe reactions such as Steven Johnsons Syndrome (SJS) and Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [
13,
14]. A holistic understanding of drug HSRs can be achieved from combining classic and modern approaches: here we consolidate the new findings on molecular pathways with immediate (Type 1) and delayed (Type IV) phenotypes.
Cutaneous symptoms (like flushing, pruritus, or urticaria/angioedema), as well as respiratory and gastrointestinal symptoms are the most common mast cell activation related clinical presentations. More severe reactions with vital sign changes, throat tightness, or swelling can also appear [
1,
2,
7,
15]. Previous sensitization to the drug is usually required but cross reactivity has been described between drugs and other allergens (for example in pollen allergic patients who develop taxane hypersensitivity) [
9]. Grading of the reaction severity is done with two classification systems: by Brown et al. [
16] and modified Ring and Messemer [
17] proposed criteria. HSRs are graded as mild/grade I (cutaneous symptoms or with only one symptomatic organ system), moderate/grade II (two or more systems involved without vital sign changes), and severe/grade III when more than two systems are affected with vital sign changes [
16].
Reactions which occur during or within 1–6 h after the drug administration are classified as immediate reactions [
1,
2,
8]. This phenotype typically includes the mast cell activation IgE mediated endotype, driven by epitope-specific IgE with mast cells as the main players. Other endotypes include direct complement activation [
1,
18], HSRs mediated by cyclooxygenase-1 inhibition in Aspirin Exacerbated Respiratory Disease (AERD) and Aspirin Exacerbated Cutaneous Disease (AECD) [
19,
20], or reactions due to some drugs with THIQ (tetrahydroisoquinoline)motifs which signal through the human G-protein-coupled receptor (MrgprX2) may also induce histamine release are included in the mast cell activation endotype [
21,
22]. In the past, symptoms such as fever, chills, and pain were not typically associated with allergic reactions, however, they have been reported during HSRs to monoclonals, oxaliplatin, and taxanes [
3,
6,
9,
23]. These type of reactions known as “cytokine storm-like reactions”, are mediated by the release of proinflammatory cytokines which activate macrophages and other immune cells with FcγR receptors [
21].
Type IV reactions, classically known as delayed reactions, have a more heterogeneous presentation and typically appear several days or weeks after the exposure from the start date of drug administration [
1,
2,
8]. They are related to T cell-mediated symptoms such as maculopapular exanthema or delayed urticaria and can also involve other organs such as liver, lungs, kidneys, or hematological alterations [
1,
8,
13,
24]. Severe Cutaneous Adverse Reactions (SCAR) which include Acute Generalized Exanthematous Pustulosis (AGEP), Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), Stevens-Johnson Syndrome (SJS), and Toxic Epidermal Necrolysis (TEN) have a severe clinical presentation with different treatment and poor clinical outcomes [
13,
14].
These phenotypes and endotypes can be further assessed through biomarkers such as skin testing (ST), and specific IgE and basophil activation tests (BAT), which help identify mast cell involvement in the HSRs as well as cross reactivity between drugs [
7,
9,
10,
11,
25,
26,
27,
28,
29]. Levels of basophil/mast cell activation mediators during HSRs, such as tryptase and histamine, or cytokine and leukotriene production, may be helpful in identifying cells involved in the HSR. These mediators can also been usedin risk stratifying the patient [
4,
7,
9,
20,
27,
30,
31]. In the last few years the relevance of genotypes in drug allergy has increased significantly; for example, specific HLA alleles have been associated with the development of hypersensitivity reactions to antibiotics, retrovirals, and anticonvulsant drugs [
13,
32]. Prescreening before abacavir treatment in HIV positive patients is required to identify potential reactors expressing HLA-B 57:01 [
14]. To identify T-cell mediated hypersensitivity, diagnostic tools such as patch testing and lymphocyte transformation tests (LTT) have been proposed [
1,
24,
33], and more recently, cytotoxic T-cell proteins such as granulysin, perforin, and granzyme B [
34].
2. Drug Desensitization: A Revolutionary Approach to the Management of Type I and Type IV Drug Hypersensitivity Reactions
Drug desensitization (DS) was developed due to the pressing need to reintroduce drugs in a safe fashion in patients who had developed IgE/non IgE type I HSRs to critical antibiotics and/or other drugs. The first reported case was in 1942 in an English soldier in urgent need of penicillin at a time when no alternatives existed; further advances were made in the 1980s when the first oral and intravenous protocols for penicillin were created. The safety and efficacy of penicillin desensitization was widely described without reports of deaths or anaphylaxis to such a degree that it was even used in high risk populations such as pregnant women who had prick positive penicillin allergy and required treatment for syphilis (
Table 1a) [
35,
36]. The first intravenous protocol was also reported with penicillin desensitization in 1987, and the protocol used a 10-fold escalation in solution concentration with 20 min intervals and was the prototype for modern desensitizations [
37].
Desensitization today is indicated when patients have HSRs mediated by mast cell activation to their first line therapy without comparable alternatives [
3,
4,
5,
26,
30,
38,
39]. Desensitization is achieved by incrementally escalating the sub-optimal doses of the culprit drug until the required dose is reached, and DS induces a temporary tolerance which protects the patient from anaphylaxis [
6,
23,
38]. Currently valid DS protocols have been established for other chronic diseases such as Cystic fibrosis, which has poor patient prognosis if antibiotic restrictions are present [
5]. Recently these procedures have been developed for new and innovative drugs for oncologic and chronic inflammatory diseases which were continued first line therapies that were critical for patients’ quality of life and life expectancy [
3,
4]. Previously published data have shown that more than 20% of oncology patients who receive platin chemotherapy developed an allergic reaction. Equally concerning is the increasing number of unique monoclonal antibody (mAbs) therapies which have high rates of immunogenicity due to non-human mAb parts and glycosylation [
3]. Preventing patients from using first line therapy can be taxing both in terms of cost but also in the reduction of quality of life, life expectancy, and disease progression or management. In terms of safety and efficacy, previously published data have shown that DS is the best option when indicated. Successful protocols have been described for different HSRs to culprit drugs, for example antibiotics, biologics, chemotherapy, progesterone, as well as many other treatments [
6,
23,
27,
39,
40,
41]. A cost/efficacy analysis was also shown by Sloane et al. [
6] indicating that DS does not increase health costs compared to standard treatment.
Understanding the molecular mechanisms of the HSR, patient’s comorbidities, skin testing, and genetic markers are critical to determine whether desensitization is indicated as well as the potential risk of reaction during the procedure.
DS has been established to be safe and effective for IgE mediated drug HSRs. Patients with immediate reactions to taxanes and other chemotherapies in which the IgE mechanism cannot be demonstrated have also been successfully desensitized [
4,
6]. HIV positive patients who present with delayed maculopapular exanthem have also been successfully desensitized, however protocols for delayed reactions have not yet been standardized [
40]. DS is contraindicated in immune thrombocytopenia, serum sickness-like reactions, or SCAR (Severe Cutaneous Adverse Reactions) due to high toxicity [
7,
38,
42]. Comorbidities, current prescriptions, and premedications should be evaluated to understand the initial reaction and appropriately risk stratify the patients because some medications may change the risk of reaction, treatment plan, or mask the severity of the initial symptoms. Skin testing and mediator levels are also key factors for evaluating the risk of reaction during desensitization and for understanding the original HSR [
2,
9,
25,
42]. Usually mild symptoms and negative skin tests are associated with lower risk of reaction, while moderate to severe reactions and/or positive skin prick tests indicate higher risk. This has been shown with the published taxane, carboplatin, and penicillin algorithms, however further research is necessary to validate skin testing with chemotherapies, monoclonals, and other drugs [
9,
12,
27,
28]. In vitro test results (such as specific IgE and basophil test activation) and pharmacogenetics have also been proposed to predict outcomes of DS [
10,
11,
29]. Recent data has linked BRCA1/2 mutation in patients being treated with carboplatin with increased rates of IgE mediated HSRs [
11,
43]. These findings illuminate new insights in HSR prediction, management, and desensitization.
After risk stratification, a flexible protocol with 4 to 16 steps (typically 12) and escalating the dose 2 to 2.5 times every 15 min is applied. Usually the starting concentration of the solution in a 4 bags/16 steps protocol is 1/1000 to 1/100 in a 3 bags/12 steps protocol, reaching the target dose at the end of the procedure (
Table 1b) [
3,
5,
6,
23,
36,
42,
44].
Recently, algorithms to manage HSRs through DS have been described for antibiotics and chemotherapies [
9,
12,
25,
27], to allow some patients to tolerate the offending drug via regular infusion without safety concerns [
9]. Other algorithms, such as that for platins, have focused on repeated skin testing to avoid false negative results [
12,
25].
While empiric desensitization protocols were established clinically to treat patients in need, the mechanisms of desensitization were evaluated in vitro to understand the cellular and moelcular players. The first in vitro studies occurred in animals; later, human basophils were used for proof of concept in vitro experiments by performing BAT (basophil activation test). Currently both in vivo and in vitro studies are used to understand the cellular and molecular pathways of mast cell and basophils involved in HSRs and anaphylaxis. A major step in improving clinical desensitization protocols was insight from in vitro studies on how dosing and timing during the desensitization protocol inhibited cell degranulation and cytokine production.
5. Conclusions
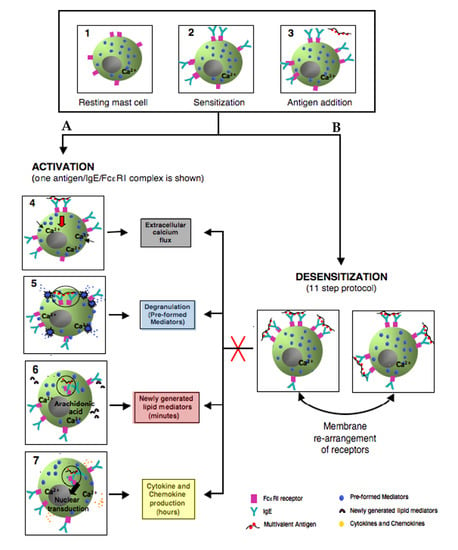
The classical description of drug HSRs from Gell and Coombs is now complemented by the new understanding of phenotypes, endotypes, and corresponding biomarkers. This allows an expanded reaction classification, such as “cytokine storm-like reactions” to be recognized in HSRs to moAbs, oxaliplatin, and taxanes. New biomarkers from mast cells and other immune cell mediators (chymases, Carboxypeptidase A (CPA), platelet activating factor (PAF)) will be added in the future to allow for better categorization (
Figure 5).
Desensitization is a revolutionary approach for the safe reintroduction of immunogenic drugs. Mast cells and basophils have long been known to be the cellular targets involved in desensitization; however the inhibitory mechanisms of desensitization are still being elucidated. DS takes advantage of inhibitory mechanisms which prevent activated mast cell signal transduction and pro-inflammatory mediator release.
Impairment of Ag/IgE/FεcRI complex internalization during desensitization can lead to these complexes remaining mobile on the cell surface. Low antigen doses administered incrementally along with actin remodeling could lead to an aggregation of these mobile complexes forming a secluded compartment of desensitized antigen specific receptors, Ca
2+ channels, and signaling molecules. This compartment is specific to the antigen and excludes non-desensitized receptors (
Figure 5).
Successful human DS protocols are based on in vitro IgE mast cell desensitization models and provide outstanding safety for all patients with severe allergic reactions in need of first line therapies. Future research will uncover the molecular pathway of in vitro desensitization which will permit more effective and safer human protocols.