Fish Oil Ameliorates High-Fat Diet Induced Male Mouse Reproductive Dysfunction via Modifying the Rhythmic Expression of Testosterone Synthesis Related Genes
Abstract
:1. Introduction
2. Results
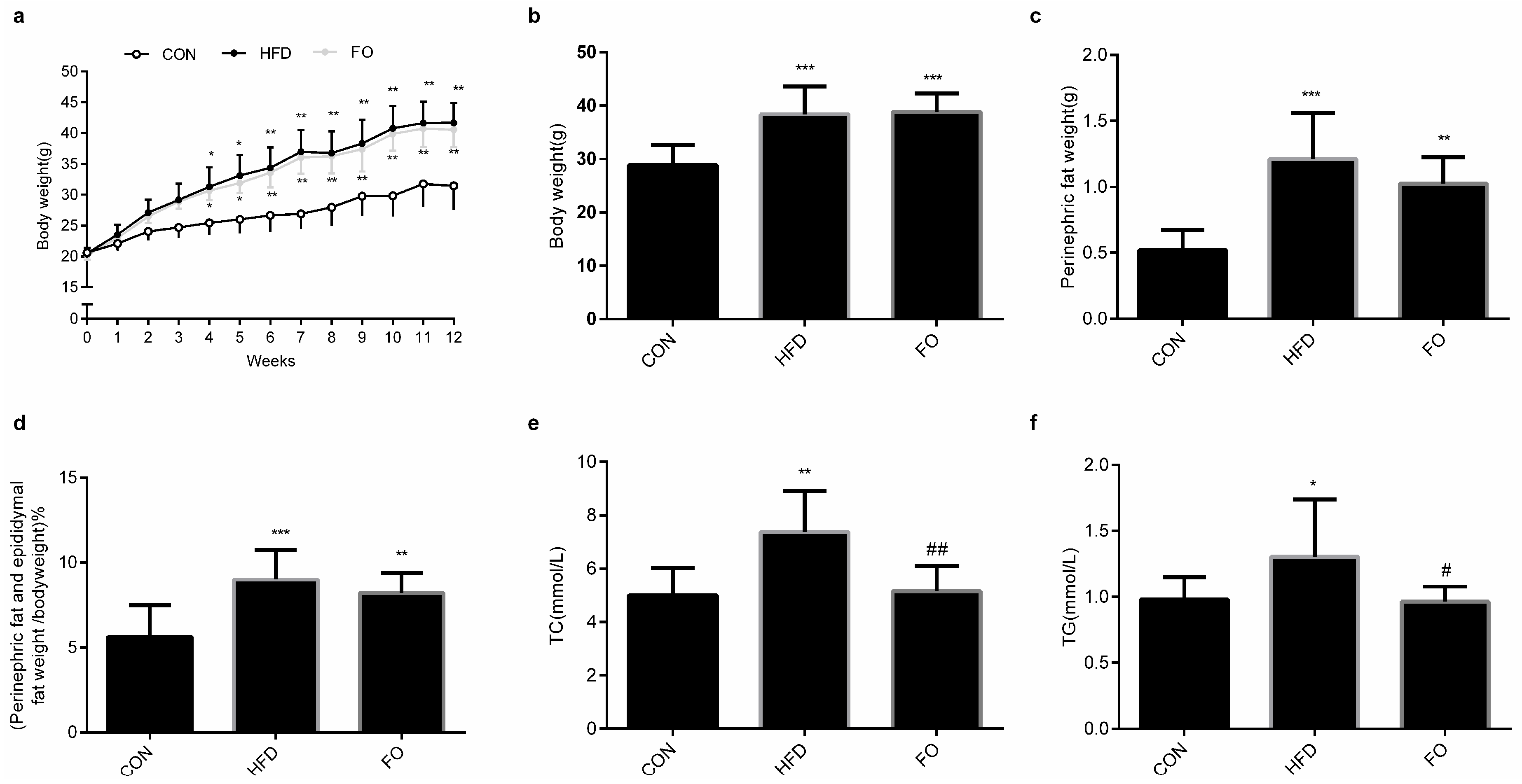
2.1. Effect of High-Fat Diet (HFD) and Fish Oil Diet (FO) on the Body Weight, Perinephric Fat and Epididymal Fat Weight and the Plasma Total Cholesterol (TC), Triglyceride (TG) Levels
2.2. Effect of HFD and FO on Male Reproductive Organ
2.3. Effect of HFD and FO on Histological Study and Testicular Cell Apoptosis
2.4. FO Alleviated Testes Testosterone Synthesis Capacity in HFD Mice
2.5. Different Diets Affect the Circadian Rhythm of Testes Testosterone Synthesis
2.6. Effect of HFD and FO on Rhythmic Expression of Core Clock Genes
3. Discussion
4. Materials and Methods
4.1. Animals and Diets
4.2. Measurement of Plasma Parameters
4.3. Histological Studies and TUNEL Assay
4.4. Real-Time Polymerase Chain Reaction (PCR) Analysis
4.5. Western Blot
4.6. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| AC | Adenylyl cyclase |
| β-Actin | Actin, beta |
| Bmal1 | Aryl hydrocarbon receptor nuclear translocator-like |
| BSA | Bovine serum albumin |
| cAMP | Cyclic adenosine monophosphate |
| Clock | Circadian locomotor output cycles kaput |
| CON | Control diet |
| Cry1/2 | Cryptochrome 1/2 |
| 3/17β-Hsd | 3β-Hydroxysteroid dehydrogenase |
| HE | Hematoxylin-eosin |
| HFD | High fat diet |
| FO | High fat and fish oil diet |
| KO | Knockout |
| Lh | Luteinizing hormone |
| Lhr | Luteinizing hormone receptors |
| Per1/2 | Period circadian clock 1/2 |
| PMSF | Phenylmethylsulfonyl fluoride |
| Pparα | Peroxisome proliferator activated receptor alpha |
| P450scc | Cholesterol side-chain cleavage enzyme |
| PUFAs | Polyunsaturated fatty acids |
| PVDF | Polyvinylidene difluoride |
| Rorα | RAR-related orphan receptor alpha |
| Sf-1 | Steroidogenic factor 1 |
| Sirt1 | Sirtuin 1 |
| Star | Steroidogenic acute regulatory protein |
| TUNEL | Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling |
| RT-qPCR | Quantitative real-time PCR |
| TC | Total cholesterol |
| TG | Triacylglycerol |
| ZT | Zeitgeber time |
References
- Jensen, T.K.; Heitmann, B.L.; Blomberg Jensen, M.; Halldorsson, T.I.; Andersson, A.M.; Skakkebaek, N.E.; Joensen, U.N.; Lauritsen, M.P.; Christiansen, P.; Dalgard, C.; et al. High dietary intake of saturated fat is associated with reduced semen quality among 701 young danish men from the general population. Am. J. Clin. Nutr. 2013, 97, 411–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, W.J.; Mu, Y.; Yu, N.; Yi, T.L.; Zhang, Y.; Pang, X.L.; Cheng, D.; Yang, J. Protective effects of metformin on reproductive function in obese Male rats induced by high-fat diet. J. Assist. Reprod. Genet. 2015, 32, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.K.; Andersson, A.M.; Jorgensen, N.; Andersen, A.G.; Carlsen, E.; Petersen, J.H.; Skakkebaek, N.E. Body mass index in relation to semen quality and reproductive hormones among 1558 danish men. Fertil. Steril. 2004, 82, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Gao, H.; Chen, D.; Tang, D.; Huang, W.; Li, T.; Ma, T.; Chang, B. Effects of obesity and exercise on testicular leptin signal transduction and testosterone biosynthesis in male mice. Am. J. Physiol. Regul. Integr. Compar. Physiol. 2017, 312, R501–R510. [Google Scholar] [CrossRef] [PubMed]
- Campos-Silva, P.; Furriel, A.; Costa, W.S.; Sampaio, F.J.; Gregorio, B.M. Metabolic and testicular effects of the long-term administration of different high-fat diets in adult rats. Int. Braz. J. Urol. 2015, 41, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Chambers, T.J.; Richard, R.A. The impact of obesity on male fertility. Hormones 2015, 14, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Fui, M.N.; Dupuis, P.; Grossmann, M. Lowered testosterone in male obesity: Mechanisms, morbidity and management. Asian J. Androl. 2014, 16, 223–231. [Google Scholar] [PubMed]
- Miller, W.L. Star search—What we know about how the steroidogenic acute regulatory protein mediates mitochondrial cholesterol import. Mol. Endocrinol. 2007, 21, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Christenson, L.K.; Strauss, J.F., III. Steroidogenic acute regulatory protein: An update on its regulation and mechanism of action. Arch. Med. Res. 2001, 32, 576–586. [Google Scholar] [CrossRef]
- Miller, W.L. Molecular biology of steroid hormone synthesis. Endocr. Rev. 1988, 9, 295–318. [Google Scholar] [CrossRef] [PubMed]
- Payne, A.H.; Youngblood, G.L. Regulation of expression of steroidogenic enzymes in leydig cells. Biol. Reprod. 1995, 52, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, L.; Wang, B.; Xiong, J.; Li, Q.; Wang, J.; Chen, D. Impairment of reproductive function in a male rat model of non-alcoholic fatty liver disease and beneficial effect of n-3 fatty acid supplementation. Toxicol. Lett. 2013, 222, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Wang, H.; Tian, Y.; Li, Q.; He, L.; Li, N.; Liu, Z. Fish oil alleviated high-fat diet-induced non-alcoholic fatty liver disease via regulating hepatic lipids metabolism and metaflammation: A transcriptomic study. Lipids Health Dis. 2016, 15, 20. [Google Scholar] [CrossRef] [PubMed]
- Mohawk, J.A.; Green, C.B.; Takahashi, J.S. Central and peripheral circadian clocks in mammals. Annu. Rev. Neurosci. 2012, 35, 445–462. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.X.; Chen, S.Y.; Liu, C. Regulation of reproduction by the circadian rhythms. Sheng Li Xue Bao 2016, 68, 799–808. [Google Scholar] [PubMed]
- Dibner, C.; Schibler, U.; Albrecht, U. The mammalian circadian timing system: Organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 2010, 72, 517–549. [Google Scholar] [CrossRef] [PubMed]
- Partch, C.L.; Green, C.B.; Takahashi, J.S. Molecular architecture of the mammalian circadian clock. Trends Cell Biol. 2014, 24, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Panda, S. Circadian physiology of metabolism. Science 2016, 354, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Salas-Huetos, A.; Bullo, M.; Salas-Salvado, J. Dietary patterns, foods and nutrients in male fertility parameters and fecundability: A systematic review of observational studies. Hum. Reprod. Update 2017, 23, 371–389. [Google Scholar] [CrossRef] [PubMed]
- Kinson, G.A.; Liu, C.C. Diurnal variation in plasma testosterone of the male laboratory rat. Horm. Metab. Res. 1973, 5, 233–234. [Google Scholar] [CrossRef] [PubMed]
- Faiman, C.; Winter, J.S. Diurnal cycles in plasma fsh, testosterone and cortisol in men. J. Clin. Endocrinol. Metabol. 1971, 33, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Sayegh, J.F.; Kobor, G.; Lajtha, A.; Vadasz, C. Effects of social isolation and the time of day on testosterone levels in plasma of c57bl/6by and balb/cby mice. Steroids 1990, 55, 79–82. [Google Scholar] [CrossRef]
- Cano, P.; Jimenez-Ortega, V.; Larrad, A.; Reyes Toso, C.F.; Cardinali, D.P.; Esquifino, A.I. Effect of a high-fat diet on 24-h pattern of circulating levels of prolactin, luteinizing hormone, testosterone, corticosterone, thyroid-stimulating hormone and glucose, and pineal melatonin content, in rats. Endocrine 2008, 33, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Froy, O. Circadian rhythms and obesity in mammals. ISRN Obes. 2012, 2012, 437198. [Google Scholar] [CrossRef] [PubMed]
- Ando, H.; Yanagihara, H.; Hayashi, Y.; Obi, Y.; Tsuruoka, S.; Takamura, T.; Kaneko, S.; Fujimura, A. Rhythmic messenger ribonucleic acid expression of clock genes and adipocytokines in mouse visceral adipose tissue. Endocrinology 2005, 146, 5631–5636. [Google Scholar] [CrossRef] [PubMed]
- Blancas-Velazquez, A.; Mendoza, J.; Garcia, A.N.; la Fleur, S.E. Diet-induced obesity and circadian disruption of feeding behavior. Front. Neurosci. 2017, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Kohsaka, A.; Laposky, A.D.; Ramsey, K.M.; Estrada, C.; Joshu, C.; Kobayashi, Y.; Turek, F.W.; Bass, J. High-fat diet disrupts behavioral and molecular circadian rhythms in mice. Cell Metab. 2007, 6, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, J.; Pevet, P.; Challet, E. High-fat feeding alters the clock synchronization to light. J. Physiol. 2008, 586, 5901–5910. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi Roushandeh, A.; Salehi, I.; Mortazavi, M. Protective effects of restricted diet and antioxidants on testis tissue in rats fed with high-fat diet. Iran. Biomed. J. 2015, 19, 96–101. [Google Scholar] [PubMed]
- MInguez-Alarcón, L.; Chavarro, J.E.; Mendiola, J.; Roca, M.; Tanrikut, C.; Vioque, J.; Jorgensen, N.; Torres-Cantero, A.M. Fatty acid intake in relation to reproductive hormones and testicular volume among young healthy men. Asian J. Androl. 2017, 19, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Gould, M.L.; Hurst, P.R.; Nicholson, H.D. The effects of oestrogen receptors alpha and beta on testicular cell number and steroidogenesis in mice. Reproduction 2007, 134, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Hatano, M.; Migita, T.; Ohishi, T.; Shima, Y.; Ogawa, Y.; Morohashi, K.I.; Hasegawa, Y.; Shibasaki, F. Sf-1 deficiency causes lipid accumulation in leydig cells via suppression of star and cyp11a1. Endocrine 2016, 54, 484–496. [Google Scholar] [CrossRef] [PubMed]
- Lopez, D.; Shea-Eaton, W.; McLean, M.P. Characterization of a steroidogenic factor-1-binding site found in promoter of sterol carrier protein-2 gene. Endocrine 2001, 14, 253–261. [Google Scholar] [CrossRef]
- Zhang, F.P.; Pakarainen, T.; Zhu, F.; Poutanen, M.; Huhtaniemi, I. Molecular characterization of postnatal development of testicular steroidogenesis in luteinizing hormone receptor knockout mice. Endocrinology 2004, 145, 1453–1463. [Google Scholar] [CrossRef] [PubMed]
- Mazzoccoli, G.; Francavilla, M.; Giuliani, F.; Aucella, F.; Vinciguerra, M.; Pazienza, V.; Piepoli, A.; Benegiamo, G.; Liu, S.; Cai, Y. Clock gene expression in mouse kidney and testis: Analysis of periodical and dynamical patterns. J. Biol. Regul. Homeost. Agents 2012, 26, 303–311. [Google Scholar] [PubMed]
- Alvarez, J.D.; Chen, D.; Storer, E.; Sehgal, A. Non-cyclic and developmental stage-specific expression of circadian clock proteins during murine spermatogenesis. Biol. Reprod. 2003, 69, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, J.D.; Sehgal, A. The thymus is similar to the testis in its pattern of circadian clock gene expression. J. Biol. Rhythms 2005, 20, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Gao, L.; Xiong, Y.; Yang, D.; Li, C.; Wang, A.; Jin, Y. Circadian clock and steroidogenic-related gene expression profiles in mouse leydig cells following dexamethasone stimulation. Biochem. Biophys. Res. Commun. 2017, 483, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Zylka, M.J.; Shearman, L.P.; Weaver, D.R.; Reppert, S.M. Three period homologs in mammals: Differential light responses in the suprachiasmatic circadian clock and oscillating transcripts outside of brain. Neuron 1998, 20, 1103–1110. [Google Scholar] [CrossRef]
- Yamamoto, T.; Nakahata, Y.; Soma, H.; Akashi, M.; Mamine, T.; Takumi, T. Transcriptional oscillation of canonical clock genes in mouse peripheral tissues. BMC Mol. Biol. 2004, 5, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morse, D.; Cermakian, N.; Brancorsini, S.; Parvinen, M.; Sassone-Corsi, P. No circadian rhythms in testis: Period1 expression is clock independent and developmentally regulated in the mouse. Mol. Endocrinol. 2003, 17, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Bebas, P.; Goodall, C.P.; Majewska, M.; Neumann, A.; Giebultowicz, J.M.; Chappell, P.E. Circadian clock and output genes are rhythmically expressed in extratesticular ducts and accessory organs of mice. FASEB J. 2009, 23, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, J.D.; Hansen, A.; Ord, T.; Bebas, P.; Chappell, P.E.; Giebultowicz, J.M.; Williams, C.; Moss, S.; Sehgal, A. The circadian clock protein bmal1 is necessary for fertility and proper testosterone production in mice. J. Biol. Rhythms 2008, 23, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Schoeller, E.L.; Clark, D.D.; Dey, S.; Cao, N.V.; Semaan, S.J.; Chao, L.W.; Kauffman, A.S.; Stowers, L.; Mellon, P.L. Bmal1 is required for normal reproductive behaviors in male mice. Endocrinology 2016, 157, 4914–4929. [Google Scholar] [CrossRef] [PubMed]








| Feed Formulation (g) | Control Diet (CON) | High-Fat Diet (HFD) | Fish Oil Diet (FO) |
|---|---|---|---|
| casein | 141.8 | 178 | 178 |
| corn starch | 565.692 | 489 | 489 |
| dextrin | 155 | 0 | 0 |
| cream | 0 | 148 | 110 |
| corn oil | 40 | 60 | 33 |
| fish oil | 0 | 0 | 65 |
| fibrin | 50 | 60 | 60 |
| vitamin premix | 45 | 60 | 60 |
| TBHQ | 0.042 | 0.042 | 0.042 |
| choline chloride | 2.5 | 0 | 0 |
| cholesterin | 0 | 4.63 | 4.725 |
| total | 1000 | 1000 | 1000 |
| Calories (kcal/g) | |||
| protein | 14.2% | 14% | 14% |
| carbohydrate | 75% | 44% | 44% |
| fat | 10% | 42% | 42% |
| total | 3.6 | 4.5 | 4.5 |
| cholesterin | 0 | 0.5% | 0.5% |
| Fatty acid composition | |||
| ∑SFA | 15.5% | 49.70% | 47.76% |
| ∑MFA | 32% | 32.83% | 30.30% |
| ∑n−6 | 53% | 17.14% | 10.98% |
| ∑n−3 | 1% | 0.33% | 10.96% |
| ∑PUFA | 54% | 17.47% | 21.94% |
| n−6/n−3 | 53 | 52 | 1.002 |
| Name | Sequence (5′ → 3′) |
|---|---|
| β-Actin-F | GGAGATTACTGCCCTGGCTCCTA |
| β-Actin-R | GACTCATCGTACTCCTGCTTGCTG |
| Bmal1-F | CGTTCACTCAGGACAGACAGATAAG |
| Bmal1-R | TGTGGCGAAGGTAGGATAGGC |
| Clock-F | CGTTCACTCAGGACAGACAGATAAG |
| Clock-R | TGTGGCGAAGGTAGGATAGGC |
| Per 1-F | CCTCCTCCTACACTGCCTCTTC |
| Per 1-R | CACCACGCTCTCTGCCTTATTG |
| Per 2-F | GCCAACACAGACGACAGCATC |
| Per2-R | TCTCCTGGTCCTCCTTCAACAC |
| Cry 1-F | GCACCAGAAGGCATCCAGAAG |
| Cry 1-R | GGACCGAGGCGAGAAGACC |
| Cry 2-F | TGGCAAGGAGGAGAGACAGAAG |
| Cry2-R | GAAGAGGCGGCAGGAGAGG |
| Pparα-F | ATTTCCCTGTTTGTGGCTGC |
| Pparα-R | CGAAGGTCCACCATTTTTTG |
| Rorα-F | GTGGCTTCAGGAAAAGGTAAA |
| Rorα-R | GTCGCACAATGTCTGGGTAT |
| Sirt1-F | TGGTTCATTTATCAGAGTTGCC |
| Sirt1-R | CATTGTTGTTTGTTGCTTGGTC |
| Lhr-F | TTGTCGTCATCTGTGCTTGCTAC |
| Lhr-R | TTTGAGTTGGTGACAGTGATAAGGG |
| Sf1-F | GGAGCGGCACACCCTTATTA |
| Sf1-R | CCAACTTTCCCTTCTTTCACT |
| Star-F | AACTCACTTGGCTGCTCAGTATTG |
| Star-R | CAGGTGGTTGGCGAACTCTATC |
| P450scc-F | TGCTGCGGGCTGAAGTCC |
| P450scc-R | TGTCTCCTTGATGCTGGCTTTG |
| 3β-Hsd-F | CAAGGTGACAGTGTTGGAAGGAG |
| 3β-Hsd-R | AATGATGGCAGCAGTATGGATGAC |
| 17β-Hsd-F | GTGGTTATGAGCAAGCCCTGAG |
| 17β-Hsd-R | GAAGCGGTTCGTGGAGAAGTAG |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Cai, Y.; Shao, Y.; Zhang, X.; Li, N.; Zhang, H.; Liu, Z. Fish Oil Ameliorates High-Fat Diet Induced Male Mouse Reproductive Dysfunction via Modifying the Rhythmic Expression of Testosterone Synthesis Related Genes. Int. J. Mol. Sci. 2018, 19, 1325. https://doi.org/10.3390/ijms19051325
Wang H, Cai Y, Shao Y, Zhang X, Li N, Zhang H, Liu Z. Fish Oil Ameliorates High-Fat Diet Induced Male Mouse Reproductive Dysfunction via Modifying the Rhythmic Expression of Testosterone Synthesis Related Genes. International Journal of Molecular Sciences. 2018; 19(5):1325. https://doi.org/10.3390/ijms19051325
Chicago/Turabian StyleWang, Hualin, Yazheng Cai, Yang Shao, Xifeng Zhang, Na Li, Hongyu Zhang, and Zhiguo Liu. 2018. "Fish Oil Ameliorates High-Fat Diet Induced Male Mouse Reproductive Dysfunction via Modifying the Rhythmic Expression of Testosterone Synthesis Related Genes" International Journal of Molecular Sciences 19, no. 5: 1325. https://doi.org/10.3390/ijms19051325





