Exploring the Mechanism of Inhibition of Au Nanoparticles on the Aggregation of Amyloid-β(16-22) Peptides at the Atom Level by All-Atom Molecular Dynamics
Abstract
:1. Introduction
2. Results and Discussion
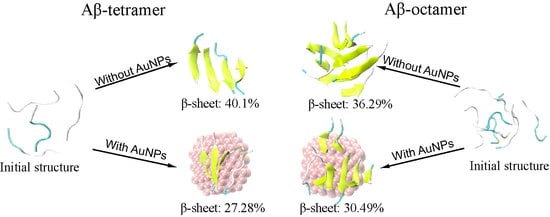
2.1. AuNPs Prevent the β-Sheet Formation of Aβ Peptides and Prolong the Progress of the Aggregation of Aβ Peptides
2.2. Interactions of AuNPs with Aβ(16-22) Peptides Competes with the Aβ-Aβ Interaction
3. Materials and Methods
3.1. Aβ Tetramer and Aβ Octamer Systems
3.2. Aβ Tetramer + AuNPs Complex and Aβ Octamer + AuNPs Complex Systems
3.3. MD Simulation Details
3.4. Analysis Methods
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chiti, F.; Dobson, C.M. Protein misfolding, functional amyloid, and human disease. Annu. Rev. Biochem. 2006, 75, 333–366. [Google Scholar] [CrossRef] [PubMed]
- Pitschke, M.; Prior, R.; Haupt, M.; Riesner, D. Detection of single amyloid β-protein aggregates in the cerebrospinal fluid of Alzheimer’s patients by fluorescence correlation spectroscopy. Nat. Med. 1998, 4, 832–834. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Ma, B.; McElheny, D.; Parthasarathy, S.; Long, F.; Hoshi, M.; Nussinov, R.; Ishii, Y. Aβ(1–42) fibril structure illuminates self-recognition and replication of amyloid in Alzheimer’s disease. Nat. Struct. Mol. Biol. 2015, 22, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Stefani, M.; Dobson, C.M. Protein aggregation and aggregate toxicity: New insights into protein folding, misfolding diseases and biological evolution. J. Mol. Med. 2003, 81, 678–699. [Google Scholar] [CrossRef] [PubMed]
- Lomakin, A.; Teplow, D.B.; Kirschner, D.A.; Benedek, G.B. Kinetic theory of fibrillogenesis of amyloid β-protein. Proc. Natl. Acad. Sci. USA 1997, 94, 7942–7947. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.M.; Hartley, D.M.; Kusumoto, Y.; Fezoui, Y.; Condron, M.M.; Lomakin, A.; Benedek, G.B.; Selkoe, D.J.; Teplow, D.B. Amyloid β-protein fibrillogenesis. Structure and biological activity of protofibrillar intermediates. J. Biol. Chem. 1999, 274, 25945–25952. [Google Scholar] [CrossRef] [PubMed]
- Hilbich, C.; Kisters-Woike, B.; Reed, J.; Masters, C.L.; Beyreuther, K. Substitutions of hydrophobic amino acids reduce the amyloidogenicity of Alzheimer’s disease β a4 peptides. J. Mol. Biol. 1992, 228, 460–473. [Google Scholar] [CrossRef]
- Balbach, J.J.; Ishii, Y.; Antzutkin, O.N.; Leapman, R.D.; Rizzo, N.W.; Dyda, F.; Reed, J.; Tycko, R. Amyloid fibril formation by a β 16–22, a seven-residue fragment of the Alzheimer’s β-amyloid peptide, and structural characterization by solid state nmr. Biochemistry 2000, 39, 13748–13759. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Luo, Y.; Lin, D.; Xi, W.; Yang, X.; Wei, G. The molecular mechanism of fullerene-inhibited aggregation of Alzheimer’s β-amyloid peptide fragment. Nanoscale 2014, 6, 9752–9762. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Qian, Z.; Wei, G. The inhibitory mechanism of a fullerene derivative against amyloid-β peptide aggregation: An atomistic simulation study. Phys. Chem. Chem. Phys. PCCP 2016, 18, 12582–12591. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Luo, Y.; Derreumaux, P.; Wei, G. Carbon nanotube inhibits the formation of β-sheet-rich oligomers of the Alzheimer’s amyloid-β(16–22) peptide. Biophys. J. 2011, 101, 2267–2276. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Zhu, Y.; Wei, G.; Li, H. Carbon nanotube prevents the secondary structure formation of amyloid-β trimers: An all-atom molecular dynamics study. Mol. Simul. 2017, 43, 1189–1195. [Google Scholar] [CrossRef]
- Cabaleiro-Lago, C.; Quinlan-Pluck, F.; Lynch, I.; Lindman, S.; Minogue, A.M.; Thulin, E.; Walsh, D.M.; Dawson, K.A.; Linse, S. Inhibition of amyloid β protein fibrillation by polymeric nanoparticles. J. Am. Chem. Soc. 2008, 130, 15437–15443. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Wei, G.; Yang, X. Influence of au nanoparticles on the aggregation of amyloid-β-(25–35) peptides. Nanoscale 2013, 5, 10397–10403. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, J.J.; Zhang, J.; Wang, X.; Kawazoe, N.; Chen, G. Gold nanoparticle size and shape influence on osteogenesis of mesenchymal stem cells. Nanoscale 2016, 8, 7992–8007. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Zhang, M.; Gong, D.; Chen, R.; Hu, X.; Sun, T. The size-effect of gold nanoparticles and nanoclusters in the inhibition of amyloid-β fibrillation. Nanoscale 2017, 9, 4107–4113. [Google Scholar] [CrossRef] [PubMed]
- Bartczak, D.; Nitti, S.; Millar, T.M.; Kanaras, A.G. Exocytosis of peptide functionalized gold nanoparticles in endothelial cells. Nanoscale 2012, 4, 4470–4472. [Google Scholar] [CrossRef] [PubMed]
- Ko, W.K.; Heo, D.N.; Moon, H.J.; Lee, S.J.; Bae, M.S.; Lee, J.B.; Sun, I.C.; Jeon, H.B.; Park, H.K.; Kwon, I.K. The effect of gold nanoparticle size on osteogenic differentiation of adipose-derived stem cells. J. Colloid Interface Sci. 2015, 438, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Huang, H.; Liu, G.; Zhou, W.; Chen, Y.; Jin, Q.; Ji, J. Multidentate zwitterionic chitosan oligosaccharide modified gold nanoparticles: Stability, biocompatibility and cell interactions. Nanoscale 2013, 5, 3982–3991. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Sun, H.; Dong, K.; Ren, J.; Qu, X. Gold-nanoparticle-based multifunctional amyloid-β inhibitor against Alzheimer’s disease. Chemistry 2015, 21, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.H.; Chang, Y.J.; Yoshiike, Y.; Chang, Y.C.; Chen, Y.R. Negatively charged gold nanoparticles inhibit Alzheimer’s amyloid-β fibrillization, induce fibril dissociation, and mitigate neurotoxicity. Small 2012, 8, 3631–3639. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.T.; Lin, Y.; Todorova, N.; Xu, Y.; Mazo, M.; Rana, S.; Leonardo, V.; Amdursky, N.; Spicer, C.D.; Alexander, B.D.; et al. Facet-dependent interactions of islet amyloid polypeptide with gold nanoparticles: Implications for fibril formation and peptide-induced lipid membrane disruption. Chem. Mater. 2017, 29, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- Rohrig, U.F.; Laio, A.; Tantalo, N.; Parrinello, M.; Petronzio, R. Stability and structure of oligomers of the Alzheimer peptide aβ16–22: From the dimer to the 32-mer. Biophys. J. 2006, 91, 3217–3229. [Google Scholar] [CrossRef] [PubMed]
- Peelle, B.R.; Krauland, E.M.; Wittrup, K.D.; Belcher, A.M. Design criteria for engineering inorganic material-specific peptides. Langmuir ACS J. Surf. Colloids 2005, 21, 6929–6933. [Google Scholar] [CrossRef] [PubMed]
- Kyrychenko, A. Nanogold decorated by phlip peptide: Comparative force field study. Phys. Chem. Chem. Phys. PCCP 2015, 17, 12648–12660. [Google Scholar] [CrossRef] [PubMed]
- Cohavi, O.; Reichmann, D.; Abramovich, R.; Tesler, A.B.; Bellapadrona, G.; Kokh, D.B.; Wade, R.C.; Vaskevich, A.; Rubinstein, I.; Schreiber, G. A quantitative, real-time assessment of binding of peptides and proteins to gold surfaces. Chemistry 2011, 17, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Chowdhury, P.; Gai, F. Effect of dehydration on the aggregation kinetics of two amyloid peptides. J. Phys. Chem. B 2009, 113, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Krone, M.G.; Hua, L.; Soto, P.; Zhou, R.; Berne, B.J.; Shea, J.E. Role of water in mediating the assembly of Alzheimer amyloid-b ab16–22 protofilaments. J. Am. Chem. Soc. 2008, 130, 11066–11072. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Luo, Y.; Derreumaux, P.; Wei, G. Induced b-barrel formation of the Alzheimer’s ab25–35 oligomers on carbon nanotube surfaces: Implication for amyloid fibril inhibition. Biophys. J. 2009, 97, 1795–1803. [Google Scholar] [CrossRef] [PubMed]
- Makin, O.S.; Atkins, E.; Sikorski, P.; Johansson, J.; Serpell, L.C. Molecular basis for amyloid fibril formation and stability. Proc. Natl. Acad. Sci. USA 2005, 102, 315–320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, B.; Nussinov, R. Stabilities and conformations of Alzheimer’s β-amyloid peptide oligomers (Aβ16–22, Aβ16–35, and Aβ10–35): sequence effects. Proc. Natl. Acad. Sci. USA 2002, 99, 14126–14131. [Google Scholar] [CrossRef] [PubMed]
- Berendsen, H.J.; Postma, J.P.; van Gunsteren, W.F.; Hermans, J. Intermolecular Forces, Interaction Models for Water in Relation to Protein Hydration; D. Reidel Publishing Company: Dordrecht, The Netherlands, 1981; pp. 331–342. [Google Scholar]
- Lindahl, E.; Hess, B.; Van Der Spoel, D. Gromacs 3.0: A package for molecular simulation and trajectory analysis. J. Mol. Model. 2001, 7, 306–317. [Google Scholar] [CrossRef]
- Soto, P.; Griffin, M.A.; Shea, J.E. New insights into the mechanism of Alzheimer amyloid-b fibrillogenesis inhibition by n-methylated peptides. Biophys. J. 2007, 93, 3015–3025. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.; Li, M.S.; Stock, G.; Straub, J.E.; Thirumalai, D. Monomer adds to preformed structured oligomers of ab-peptides by a two-stage dock-lock mechanism. Proc. Natl. Acad. Sci. USA 2007, 104, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.K.; Lu, K.; Childers, W.S.; Liang, Y.; Dublin, S.N.; Dong, J.; Snyder, J.P.; Pingali, S.V.; Thiyagarajan, P.; Lynn, D.G. Facial symmetry in protein self-assembly. J. Am. Chem. Soc. 2008, 130, 9829–9835. [Google Scholar] [CrossRef] [PubMed]
- Van Gunsteren, W.F.; Billeter, S.R.; Eising, A.A.; Hünenberger, P.H.; Krüger, P.K.; Mark, A.E.; Scott, W.R.; Tironi, I.G. Biomolecular Simulation: The Gromos96 Manual and User Guide; Vdf Hochschulverland, ETH: Zurich, Switzerland, 1996. [Google Scholar]
- Bussi, G.; Donadio, D.; Parrinello, M. Canonical sampling through velocity rescaling. J. Chem. Phys. 2007, 126, 014101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miyamoto, S.; Kollman, P.A. Settle: An analytical version of the shake and rattle algorithm for rigid water models. J. Comput. Chem. 1992, 13, 952–962. [Google Scholar] [CrossRef]
- Hess, B.; Bekker, H.; Berendsen, H.J.C.; Fraaije, J.G.E.M. Lincs: A linear constraint solver for molecular simulations. J. Comput. Chem. 1997, 18, 1463–1472. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. Vmd: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Hummer, G.; Rasaiah, J.C.; Noworyta, J.P. Water conduction through the hydrophobic channel of a carbon nanotube. Nature 2001, 414, 188–190. [Google Scholar] [CrossRef] [PubMed]
- Hirschfelder, J.O.; Curtiss, C.F.; Byron Bird, R. Molecular Theory of Gases and Liquids; John Wiley and Sons: New York, NY, USA, 1954. [Google Scholar]
- Kabsch, W.; Sander, C. Dictionary of protein secondary structure: Pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 1983, 22, 2577–2637. [Google Scholar] [CrossRef] [PubMed]
- Kumari, R.; Kumar, R.; Open Source Drug Discovery Consortium; Lynn, A. g_Mmpbsa—A GROMACS tool for high-throughput MM-PBSA calculations. J. Chem. Inf. Model. 2014, 54, 1951–1962. [Google Scholar] [CrossRef] [PubMed]
- Azriel, R.; Gazit, E. Analysis of the minimal amyloid-forming fragment of the islet amyloid polypeptide. An experimental support for the key role of the phenylalanine residue in amyloid formation. J. Biol. Chem. 2001, 276, 34156–34161. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhang, J.; Derreumaux, P.; Mu, Y. Molecular mechanism of the inhibition of egcg on the Alzheimer aβ(1–42) dimer. J. Phys. Chem. B 2013, 117, 3993–4002. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Xi, W.; Wei, G. Atomic-level study of the effects of o4 molecules on the structural properties of protofibrillar aβ trimer: β-sheet stabilization, salt bridge protection, and binding mechanism. J. Phys. Chem. B 2015, 119, 2786–2794. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yu, X.; Patal, K.; Hu, R.; Chuang, S.; Zhang, G.; Zheng, J. Tanshinones inhibit amyloid aggregation by amyloid-β peptide, disaggregate amyloid fibrils, and protect cultured cells. ACS Chem. Neurosci. 2013, 4, 1004–1015. [Google Scholar] [CrossRef] [PubMed]
- Gazit, E. A possible role for pi-stacking in the self-assembly of amyloid fibrils. FASEB J. 2002, 16, 77–83. [Google Scholar] [CrossRef] [PubMed]





| Reasearch System | α-Helix | β-Sheet | β-Strand | Turn | Random Coil | Bend |
|---|---|---|---|---|---|---|
| Abeta tetramer | <0.01 ± 0.730 | 40.01 ± 1.837 | 2.61 ± 1.094 | 1.52 ± 0.068 | 53.12 ± 0.078 | 2.35 ± 1.094 |
| Abeta tetramer + AuNPs | <0.01 ± 0.139 | 27.28 ± 3.360 | 5.11 ± 0.087 | 1.48 ± 0.348 | 60.95 ± 2.785 | 5.19 ± 0.087 |
| Abeta octamer | <0.01 ± 0.354 | 36.29 ± 0.142 | 4.18 ± 0.794 | 1.70 ± 1.580 | 53.65 ± 0.303 | 3.33 ± 0.794 |
| Abeta octamer + AuNPs | <0.01 ± 0.001 | 30.49 ± 3.367 | 4.79 ± 0.811 | 0.28 ± 0.098 | 59.66 ± 2.656 | 3.85 ± 0.811 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, M.; Sun, Y.; Luo, Y.; Zhu, Y.; Liu, Y.; Li, H. Exploring the Mechanism of Inhibition of Au Nanoparticles on the Aggregation of Amyloid-β(16-22) Peptides at the Atom Level by All-Atom Molecular Dynamics. Int. J. Mol. Sci. 2018, 19, 1815. https://doi.org/10.3390/ijms19061815
Song M, Sun Y, Luo Y, Zhu Y, Liu Y, Li H. Exploring the Mechanism of Inhibition of Au Nanoparticles on the Aggregation of Amyloid-β(16-22) Peptides at the Atom Level by All-Atom Molecular Dynamics. International Journal of Molecular Sciences. 2018; 19(6):1815. https://doi.org/10.3390/ijms19061815
Chicago/Turabian StyleSong, Menghua, Yunxiang Sun, Yin Luo, Yanyan Zhu, Yongsheng Liu, and Huiyu Li. 2018. "Exploring the Mechanism of Inhibition of Au Nanoparticles on the Aggregation of Amyloid-β(16-22) Peptides at the Atom Level by All-Atom Molecular Dynamics" International Journal of Molecular Sciences 19, no. 6: 1815. https://doi.org/10.3390/ijms19061815