1. Introduction
Pancreatic cancer is one of the predominant causes of cancer-associated death in humans worldwide [
1,
2]. On an average, below 8% of patient with pancreatic cancer survives for 5 years after diagnosis [
2]. Little progress has been made in improving the survival rate of pancreatic cancer, mainly due to the fact that pancreatic cancer is often undetected until late stages [
1,
2]. For the pancreatic cancer that is able to be diagnosed at an early stage (stage IA and IB), survival rate is improved with surgery and chemotherapy. The average survival rate five years after surgery is still only 27–31%; 67–71% of patients survive one year and 45–50% of patients survive two years [
3].
Epithelial surfaces form a mucous layer, which functions to shield and protect organ surfaces. An important component in the protection and lubrication of organs is the high molecular weight glycoprotein mucin, which is expressed specifically by epithelial cells [
4]. Mucin1 (MUC1) is a heterodimeric glycoprotein containing an extracellular N-terminal domain (MUC1-N) and a C-terminal domain (MUC1-C). MUC1-C has an extracellular domain, transmembrane domain and cytoplasmic tail and thus spans the cell membrane [
5,
6]. Mucin protein contains tandem repeat structures known as PTS domains, which contain numerous proline, threonine and serine amino acids [
7]. The diversity of mucin genes is derived from variations in the PTS domains. The serine and threonine residues are heavily glycosylated with
O-linked oligosaccharides [
5,
7]. Mucins are divided into either membrane-bound or secreted classes based on their physiological and structural characteristics [
8]. MUC1, MUC3A, MUC3B, MUC4, MUC11, MUC12, MUC13, MUC15, MUC16, MUC17, MUC20 and MUC21 are members of the membrane-bound transmembrane family of mucins, while MUC2, MUC5AC, MUC5B, MUC6 and MUC19 are secreted mucins [
5]. In normal tissue, mucins are heavily glycosylated and are localized to the apical surface of epithelial cells. However, in cancerous tissues, mucins are often hypoglycosylated and dispersed evenly throughout the cell surface [
5,
7], indicating a potential cancer-promoting role for mucins.
Mucins are known to be present in pancreatic cancer, with elevated expression of multiple MUC family including MUC1 [
5]. MUC1 overexpression specifically has been linked to tumor progression, invasion and metastasis in breast and pancreatic cancer cells [
9,
10,
11]. In addition, MUC1 expression is associated with resistance to anti-cancer drugs, presumably leading poor patient prognosis [
12]. Studies have shown increased expression of MUC1 in pancreatic cancer in contrast to the low expression levels observed of MUC1 in the luminal surfaces of a normal control pancreases [
5,
13,
14]. We are thus interested in blocking MUC1′s role in pancreatic cancer.
MUC1 may interact with other known growth factor receptors including EGF receptor (EGFR) to promote pancreatic cancer. Previous studies have demonstrated aberrant expression of both MUC1 and EGFR in pancreatic cancer [
15,
16]. EGFR is a receptor tyrosine kinase known to be involved in multiple cancers. EGFR-mediated signaling leads to activation of multiple pathways, including the mitogen-activated protein kinase (MAPK) and Akt pathways, to enhance cell survival and proliferation. Blocking EGFR signaling by monoclonal antibodies or tyrosine kinase inhibitors induces apoptosis and reduces proliferation in vitro and in cancer xenograft models [
17,
18]. Targeting of MUC1 inhibits EGFR signaling and reduces EGFR-mediated cancer growth [
19].
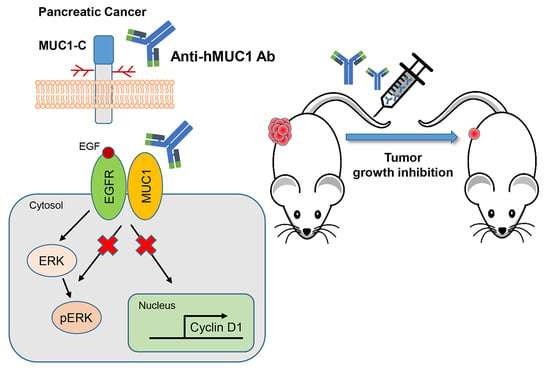
To our knowledge, minimal progress has been made in the development of potential antibodies to use in the treatment of pancreatic cancer. However, we developed a MUC1-C specific monoclonal antibody (anti-hMUC1 monoclonal antibody) that decreases proliferation of breast cancer cells in vitro and efficiently targets MUC1 in breast tumor in a xenograft mouse model [
20]. In this study, we have tested the anti-hMUC1 antibody in pancreatic cancer cells. Here, we observe that the anti-MUC1 monoclonal antibody specifically recognizes MUC1 in both the cell surface and the cytoplasm of pancreatic cells. The anti-hMUC1 monoclonal antibody effectively targeted tumor-specific MUC1 without affecting normal tissue. Importantly, our antibody significantly attenuated the growth of tumors in a pancreatic cancer cell xenograft mouse model. Therefore, this study suggests anti-hMUC1 monoclonal antibody may be a promising novel therapeutic for the treatment of pancreatic cancer.
3. Discussion
Pancreatic cancer is a difficult disease to treat because many patients remain undiagnosed until their cancer is advanced [
2,
22]. Despite extensive efforts to develop pancreatic cancer therapies, there has been no improvement for decades. The most difficult hurdle to treating pancreatic cancer is the dearth of early symptoms and therefore the late diagnosis of advanced disease [
22]. Another reason pancreatic cancer therapies remain often ineffective is that most therapeutics has focused on chemotherapy with cytotoxic drugs, which have limited effect on overall survival [
23,
24]. Therefore, there is an immediate need for effective targeted therapy to better treat pancreatic cancer.
We previously developed the anti-hMUC1 monoclonal antibody targeting MUC1-C. The anti-hMUC1 monoclonal antibody specifically targets breast cancer cells but not normal cells [
20]. To address the clinical need for improved pancreatic cancer therapy, we here evaluated the anti-hMUC1 monoclonal antibody’s potential utility in treating pancreatic cancer. We determined that the anti-hMUC1 monoclonal antibody can specifically target MUC1-C of pancreatic cancer cells in vitro and in vivo and suppresses the growth of pancreatic tumor xenografts.
The anti-hMUC1 monoclonal antibody immunoprecipitated MUC1 in the pancreatic cancer cell lines Capan-1, Capan-2 and CFPAC-1; however, it was unable to detect MUC1-C by western blot analysis. This indicates the anti-hMUC1 monoclonal antibody’s ability to detect endogenous MUC1-C only when the protein maintains its native structure. In addition, confocal microscopy analysis of Capan-1, Capan-2 and CFPAC-1 cells clearly revealed that the anti-hMUC1 monoclonal antibody recognized MUC1-C at the membrane and in the cytoplasm of cells.
The anti-hMUC1 monoclonal antibody targets the MUC1-C subunit, unlike most current MUC1 antibodies that target MUC1-N [
4]. MUC1-N domains are shed from the cell surface and are found freely in the extracellular matrix and blood circulation [
7,
25], which could result in off-target binding of antibodies designed towards MUC1-N. However, MUC1-C remains on the cell surface and is therefore an ideal antibody target. The efficacy of the anti-hMUC1 monoclonal antibody is improved by its ability to pass through the membrane and enter the cytoplasm of the cell. We have further investigated the internalization of the anti-hMUC1 monoclonal antibody by staining the cells with anti-hMUC1 monoclonal antibody conjugated with DyLight 488 fluorescent dye. Confocal analysis showed that the anti-hMUC1 monoclonal antibody induces the movement of MUC1 protein from the membrane into the cytoplasm effectively in both pancreatic cancer cells and xenograft tumors.
For an antibody to be effective as a targeted therapy, it must be specific to tumor and spare normal tissue. Antibodies such as HMFG-1 and C595 are used for the treatment of cancer in preclinical models but they are not shown to have specific affinity to tumor-associated MUC1-C [
26,
27]. Treatment with the commercially available anti-MUC1 antibody GP1.4 resulted in the inhibition of proliferation and migration of pancreatic cancer cells by activating the internalization of EGFR which leads to sequestration of surface receptors and repression of ERK phosphorylation [
19]. However, its tumor specificity or in vivo efficacy has not been examined.
Our results clearly demonstrate that anti-hMUC1 monoclonal antibody conjugated with DyLight 755 exclusively targets MUC1-C in tumor cells but not in normal tissues. This specificity of our anti-hMUC1 monoclonal antibody could be because it recognizes different posttranslational modifications of MUC1 in normal versus tumor tissue. In normal cells, MUC1 is heavily glycosylated and displays polarized expression on the apical surface of the cells. This modification potentially masks the anti-hMUC1 monoclonal antibody recognition site with bulky carbohydrate chains. However, in tumor cells, MUC1 is hypoglycosylated and dispersed throughout the cell membrane, presenting a better recognition site for the anti-hMUC1 monoclonal antibody to recognize and bind to MUC1-C [
25].
In addition to effectively recognizing and binding cellular MUC1-C, the anti-hMUC1 monoclonal antibody significantly attenuated tumor signaling. Importantly, the anti-hMUC1 monoclonal antibody significantly reduced the growth of tumors in a Capan-2 xenograft mouse model. Therefore, the need for clinical trials to evaluate the use of the anti-hMUC1 monoclonal antibody in patients with pancreatic cancer is clear. Our data showing expression of MUC1-C in 60.6% of human pancreatic tumors also highlight the plausibility of using the anti-hMUC1 monoclonal antibody to target pancreatic cancer. Because of the exclusive specificity to tumor cells, our anti-hMUC1 antibody could be developed into a potent pancreatic cancer therapy.
The potency of our antibody could be further enhanced by conjugation with toxic components. Previous studies have demonstrated enhanced toxicity of antibodies to tumor cells when the murine antibody HMFG1 is conjugated with an Yttrium moiety and when the C595 antibody is conjugated with docetaxel [
26,
27]. Another study shows the effective toxicity of the PankoMab antibody conjugated with the β-amanitin toxin on T47D tumor cells [
28].
We have demonstrated that the anti-hMUC1 monoclonal antibody specifically binds tumor MUC1-C in vitro, in xenograft models and in human pancreatic cancer tissue. The anti-hMUC1 monoclonal antibody passes through the membrane and inactivates MUC1 oncogenic signaling. When used in pancreatic cancer xenografts, the anti-hMUC1 monoclonal antibody attenuated tumor growth. Although application of the anti-hMUC1 monoclonal antibody in other cancer therapies has yet to be explored, we propose production of the humanized anti-hMUC1 monoclonal antibody and conjugating the anti-hMUC1 monoclonal antibody with a toxic group to effectively treat the pancreatic cancer patients who present with currently untreatable advanced disease. Surely, we will further confirm the specificity of the humanized anti-hMUC1 monoclonal antibody to MUC1-C on the membrane of pancreatic cancer cells and not to other normal tissues in the future work.
4. Materials and Methods
4.1. Antibodies
We harvested anti-hMUC1 monoclonal antibodies against the extracellular region of MUC1-C from hybridoma cells, established after immunization of BALB/c mice with rhMUC1-EC192 protein and CpG-DNA co-encapsulated in a phosphatidyl-β-oleoyl-γ-palmitoyl ethanolamine: cholesterol hemisuccinate (DOPE:CHEMS) complex as previously described [
20]. The amino acid sequence homology analysis showed that rhMUC1-EC192 protein has 26–39% identity with the corresponding sequences of other mucin family members. The anti-hMUC1 monoclonal antibody was not reactive to mouse MUC1 C-terminal protein which has 58% identity [
20]. To detect the cytoplasmic tail region of MUC1 protein in cells by western blotting and immunoprecipitation analyses, commercially available anti-MUC1-CT antibody (anti-MUC1-CT Ab, Catalog No. ab109185, rabbit monoclonal antibody to MUC1) was purchased from Abcam (Cambridge, UK). Anti-β-actin antibody was obtained from Sigma-Aldrich (Saint Louis, MO, USA). Anti-phospho-ERK, anti-ERK and anti-cyclin D1 antibodies were obtained from Cell Signaling Technology (Danvers, MA, USA).
4.2. Cell Culture
Human pancreatic cancer cell lines Capan-1 (adenocarcinoma, liver metastasis) and PANC-1 (epithelioid carcinoma) were purchased from Korean Cell Line Bank (KCLB, Seoul, Korea). KCLB characterized the cell lines using DNA fingerprinting analysis, species verification test, mycoplasma contamination test and viral contamination test. Human pancreatic cancer cell lines, Capan-2 (adenocarcinoma) and CFPAC-1 (adenocarcinoma, liver metastasis), were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). The cell lines were characterized by the tests for morphology, post-freeze viability, interspecies determination (isoenzyme analysis), cytogenetic analysis, mycoplasma contamination and bacterial and fungal contamination. Capan-1 cells were cultured with RPMI-1640 medium (Thermo Fisher Scientific, Waltham, MA, USA), Capan-2 cells in McCoy’s 5A medium (Thermo Fisher Scientific), PANC-1 cells in Dulbecco’s modified Eagle’s medium (DMEM, Thermo Fisher Scientific) and CFPAC-1 cells in Iscove’s modified Dulbecco’s medium (IMDM, Thermo Fisher Scientific). All cell lines were cultured with 10% fetal bovine serum (FBS, Thermo Fisher Scientific), 100 U/mL penicillin and 100 µg/mL streptomycin and were incubated at 37 °C in 5% CO2.
4.3. Western Blot and Immunoprecipitation Analysis
Cell lysates from pancreatic cancer cell lines were prepared with cell lysis buffer (20 mM Tris·HCl pH = 8.0, 5 mM EDTA, 150 mM NaCl, 100 mM NaF, 2 mM Na
3VO
4, 1% NP-40) and centrifuged at 16,000×
g at 4 °C for 20 min. Proteins from cell lysates were separated in 4–12% Bis-Tris gradient gel (Thermo Fisher Scientific). The separated proteins were transferred onto nitrocellulose membranes and blocked with 3% BSA in PBST for 1 h at room temperature. The nitrocellulose membranes were incubated with anti-hMUC1-CT antibody, anti-hMUC1 monoclonal antibody, or anti-β-actin antibody overnight at 4 °C. Anti-phospho-ERK, anti-ERK and anti-cyclin D1 antibodies were used for analysis of EGF-mediated signaling. The membranes were treated with horseradish peroxidase-conjugated secondary antibody (Jackson ImmunoResearch, West Grove, PA, USA) and the immune-reactive bands were detected by an enhanced chemiluminescence reagent (Thermo Fisher Scientific) as previously described [
20,
29]. To investigate whether the anti-hMUC1 monoclonal antibody recognizes MUC1-C in pancreatic cancer cells, immunoprecipitation analysis was performed. Briefly, cell lysates were treated with mouse anti-hMUC1 monoclonal antibody or mouse normal IgG overnight at 4 °C and then incubated with Protein A beads at 4 °C for 1 h. The immunocomplexes were identified by western blotting using the anti-hMUC1-CT antibody.
4.4. Confocal Microscopy
To obtain confocal images, pancreatic cancer cell lines were cultured on poly-
l-lysine-coated glass cover slips in 12-well culture plates as previously described [
30,
31]. After cells were cultured for 48 h, cells were fixed with 4% paraformaldehyde for 10 min and mouse anti-hMUC1 monoclonal antibody or mouse normal IgG were treated for 4 h on ice for detection of cell surface MUC1-C. For intracellular staining, cells were fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, blocked with 3% BSA and stained with anti-hMUC1 monoclonal antibody for 2 h at room temperature. After washing cells with PBST (0.1% Triton X-100 in PBS) containing 1% BSA, cells were stained with Alexa Flour 488-conjugated secondary antibody (Thermo Fisher Scientific) for 1 h. Nuclei were stained with Hoechst 33258 (Thermo Fisher Scientific). The samples were observed by confocal laser scanning microscope system (CLSM, LSM 710, Carl Zeiss, Jena, Germany) [
30,
31]. To visualize the internalization of the MUC1-anti-hMUC1 monoclonal antibody complex in pancreatic cancer cell lines, the anti-hMUC1 monoclonal antibody was labeled with DyLight 488 according to the manufacturer’s instructions (Thermo Fisher Scientific). The human pancreatic cancer cells were incubated with DyLight 488-labeled anti-hMUC1 monoclonal antibody at 37 °C in 5% CO
2 for the indicated time periods. Fluorescence signals were observed with CLSM (LSM 710, Carl Zeiss).
4.5. Animals
Six-week-old female BALB/c nu/nu mice were obtained from Nara Biotech, Inc. (Seoul, Korea). The mice were maintained under specific-pathogen-free conditions in a controlled environment (20–25 °C, 40–45% humidity, 12-h light/dark cycle; ad libitum access to food and water). All animal experimental procedures are in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Veterinary Research & Quarantine Service of Korea. The experimental procedures were approved by the Institutional Animal Care and Use Committee of Hallym University (Permit Number: Hallym2015-81, 17, 02, 2016). The mice were anesthetized under isoflurane (2–3%) inhalation with RC2-Rodent Circuit Controller (Lab Etc Inc. Store, Clayton, MO, USA) and all efforts were made to minimize suffering.
4.6. In Vivo Imaging
To analyze the distribution profiles of anti-hMUC1 monoclonal antibody, 5 × 10
6 Capan-2 cells in 50% Matrigel (PBS/Matrigel, 1:1
v/
v, BD Biosciences, Bedford, MA, USA) were subcutaneously injected into the right flank of six-week-old female BALB/c nu/nu mice. The anti-hMUC1 monoclonal antibody and normal mouse IgG were conjugated with DyLight 755 in accordance with manufacturer’s recommendation (Thermo Fisher Scientific). The mouse normal IgG-DyLight 755 (5 mg/kg) or anti-hMUC1 monoclonal antibody-DyLight 755 (5 mg/kg) was intravenously injected into the mice after tumor volumes averaged 300 mm
3. The antibody distribution profiles were examined with an in vivo imaging system (IVIS 200; Xenogen Corporation, Hopkinton, MA, USA) at 0, 24 and 48 h. Intracellular localization of the anti-hMUC1 monoclonal antibody in tumor tissues in vivo 2 days after injection with DyLight 488-labeled antibody was monitored with CLSM (LSM 710, Carl Zeiss) as described previously [
30,
31]. The sensitivity of the assay was adjusted to the extent where autoflorescence may not come up.
4.7. Xenograft Mouse Studies
Six-week-old female BALB/c nu/nu mice (n = 8) were injected subcutaneously in the dorsal right flank with 5 × 106 of Capan-2 cells in a 50% Matrigel solution (PBS/Matrigel, 1:1 v/v). After tumor volumes averaged 80 mm3, mice were injected intravenously with PBS or anti-hMUC1 monoclonal antibody twice weekly. Tumor volumes were measured for 11 weeks at 7 day intervals. The tumor volumes were calculated as width2 × length/2 with calipers in three dimensions. The mice were sacrificed 11 weeks after antibody injection and the tumors were surgically excised and weighed. To observe the histopathology, tumors were isolated, fixed in 4% buffered formalin solution and embedded in paraffin by conventional methods. The tissues were cut into 4 µm thick sections. The specimens were stained with hematoxylin and eosin. To identify the expression of hMUC1, the specimens were stained with anti-hMUC1 monoclonal antibody using standard procedures. Mice were euthanized by CO2 inhalation (CO2 inhalation was performed with 100% CO2 at a fill rate of 10–30% of the chamber volume per min) when the tumor size reached 600 mm3, the mice lost >20% of initial body weight exhibited evidence of debilitation, pain or distress, such as a hunched posture, rough hair coat, decreased food consumption, emaciation, inactivity, difficulty ambulating and respiratory problems in accordance with experimental procedures approved by the Institutional Animal Care and Use Committee of Hallym University.
4.8. Tissue Array and Immunohistochemistry
To investigate the expression of MUC1-C, we obtained human pancreatic cancer tissue sections (AccuMax Array A207(IV)) from ISU ABXIS (Seoul, Korea) with the approval of the Institutional Review Board in Hallym University (approval number: HIRB-2014-114; approval data: 32 December 2014). This study was performed in accordance with the ethical standards of the Declaration of Helsinki. The tissue sections were stained with the anti-hMUC1 monoclonal antibody (1 μg/slide) or anti-MUC1-CT2 antibody (1 μg/slide) in accordance with standard procedures using the Histostain Plus kit (Invitrogen, Carlsbad, CA, USA). Sections were developed with 3,3-diaminobenzidine (DAB, Thermo Fisher Scientific) and stained with hematoxylin to counterstain (Muto Pure Chemicals, Tokyo, Japan). All images were scanned with a light microscope (Eclipse E-200, Nikon, Tokyo, Japan).
4.9. Statistical Analysis
Results are shown as a mean ± standard deviation. Statistical significance of differences between two samples was evaluated using Student’s t-test; p < 0.05 was considered statistically significant.