Osteoclast-Released Wnt-10b Underlies Cinacalcet Related Bone Improvement in Chronic Kidney Disease
Abstract
:1. Introduction
2. Results
2.1. Cinacalcet Significantly Improved Bone Quantity and Bone Quality in 5/6 Nephrectomy CKD Mice Through Increased Femoral Bone Wnt 10b Expression
2.2. Cinacalcet Inhibits Osteoclastic Resorption
2.3. Cinacalcet Increased Osteoclastic Wnt 10b Secretion and Improves Osteoblastic Mineralization
3. Discussion
4. Materials and Methods
4.1. 5/6 Nephrectomy CKD Model
4.2. Biochemical Analyses
4.3. Micro-Computed Tomography (μCT)
4.4. Biomechanical Three-Point Bending Test
4.5. Bone Marrow-Derived Monocyte Collection and Osteoclast Differentiation
4.6. Confocal Microscopic Analysis of Osteoclasts
4.7. Tartrate-Resistant Acid Phosphatase (TRAP) Staining
4.8. Alizarin Red Staining
4.9. Western Blotting
4.10. Statistics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Di Benedetto, A.; Marcelli, D.; D’Andrea, A.; Cice, G.; D’Isa, S.; Cappabianca, F.; Pacchiano, G.; D’Amato, R.; Oggero, A.R.; Bonanno, D.; et al. Risk factors and underlying cardiovascular diseases in incident esrd patients. J. Nephrol. 2005, 18, 592–598. [Google Scholar] [PubMed]
- Wu-Wong, J.R.; Nakane, M.; Ma, J.; Ruan, X.; Kroeger, P.E. Elevated phosphorus modulates vitamin d receptor-mediated gene expression in human vascular smooth muscle cells. Am. J. Physiol. Ren. Physiol. 2007, 293, F1592–F1604. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.M.; Lu, K.C.; Wu, C.C.; Hsu, Y.H.; Lin, Y.F. Association of serum phosphate and related factors in esrd-related vascular calcification. Int. J. Nephrol. 2011, 2011, 939613. [Google Scholar] [CrossRef] [PubMed]
- Yajima, A.; Tsuchiya, K.; Yokota, H.; Nitta, K. [bone disease in the field of ckd-mbd]. Clin. Calcium 2016, 26, 875–880. [Google Scholar] [PubMed]
- Zheng, C.M.; Zheng, J.Q.; Wu, C.C.; Lu, C.L.; Shyu, J.F.; Yung-Ho, H.; Wu, M.Y.; Chiu, I.J.; Wang, Y.H.; Lin, Y.F.; et al. Bone loss in chronic kidney disease: Quantity or quality? Bone 2016, 87, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Spaulding, C.M.; Young, G. Osteitis fibrosa cystica and chronic renal failure. J. Am. Podiatr. Med. Assoc. 1997, 87, 238–240. [Google Scholar] [CrossRef]
- Torres, P.U.; Bover, J.; Mazzaferro, S.; De Vernejoul, M.C.; Cohen-Solal, M. When, how, and why a bone biopsy should be performed in patients with chronic kidney disease. Sem. Nephrol. 2014, 34, 612–625. [Google Scholar] [CrossRef]
- Jevtic, V. Imaging of renal osteodystrophy. Eur. J. Radiol. 2003, 46, 85–95. [Google Scholar] [CrossRef]
- Meunier, P.J.; Boivin, G. Bone mineral density reflects bone mass but also the degree of mineralization of bone: Therapeutic implications. Bone 1997, 21, 373–377. [Google Scholar] [CrossRef]
- Malluche, H.H.; Porter, D.S.; Monier-Faugere, M.C.; Mawad, H.; Pienkowski, D. Differences in bone quality in low- and high-turnover renal osteodystrophy. J. Am. Soc. Nephrol. 2012, 23, 525–532. [Google Scholar] [CrossRef]
- Ng, A.H.; Hercz, G.; Kandel, R.; Grynpas, M.D. Association between fluoride, magnesium, aluminum and bone quality in renal osteodystrophy. Bone 2004, 34, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Krempien, B.; Geiger, G.; Ritz, E. Structural changes of cortical bone in secondary hyperparathyroidism: Replacement of lamellar bone by woven bone. Virchows Arch. A Pathol. Anat. Histol. 1975, 366, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Yajima, A.; Inaba, M.; Tominaga, Y.; Ito, A. Minimodeling reduces the rate of cortical bone loss in patients with secondary hyperparathyroidism. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2007, 49, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Foldes, A.J.; Arnon, E.; Popovtzer, M.M. Reduced speed of sound in tibial bone of haemodialysed patients: Association with serum pth level. Nephrol. Dial. Transp. Off. Publ. Eur. Dial. Transp. Assoc. Eur. Ren. Assoc. 1996, 11, 1318–1321. [Google Scholar] [CrossRef]
- Isaksson, H.; Turunen, M.J.; Rieppo, L.; Saarakkala, S.; Tamminen, I.S.; Rieppo, J.; Kroger, H.; Jurvelin, J.S. Infrared spectroscopy indicates altered bone turnover and remodeling activity in renal osteodystrophy. J. Bone Miner. Res. 2010, 25, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Mitome, J.; Yamamoto, H.; Saito, M.; Yokoyama, K.; Marumo, K.; Hosoya, T. Nonenzymatic cross-linking pentosidine increase in bone collagen and are associated with disorders of bone mineralization in dialysis patients. Calcif. Tissue Int. 2011, 88, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, E.F.; Steffey, M.E.; Hammerland, L.G.; Hung, B.C.; Van Wagenen, B.C.; DelMar, E.G.; Balandrin, M.F. Calcimimetics with potent and selective activity on the parathyroid calcium receptor. Proc. Natl. Acad. Sci. USA 1998, 95, 4040–4045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colloton, M.; Shatzen, E.; Miller, G.; Stehman-Breen, C.; Wada, M.; Lacey, D.; Martin, D. Cinacalcet hcl attenuates parathyroid hyperplasia in a rat model of secondary hyperparathyroidism. Kidney Int. 2005, 67, 467–476. [Google Scholar] [CrossRef]
- Mizobuchi, M.; Ogata, H.; Hatamura, I.; Saji, F.; Koiwa, F.; Kinugasa, E.; Koshikawa, S.; Akizawa, T. Activation of calcium-sensing receptor accelerates apoptosis in hyperplastic parathyroid cells. Biochem. Biophys. Res. Commun. 2007, 362, 11–16. [Google Scholar] [CrossRef]
- Cunningham, J.; Danese, M.; Olson, K.; Klassen, P.; Chertow, G.M. Effects of the calcimimetic cinacalcet hcl on cardiovascular disease, fracture, and health-related quality of life in secondary hyperparathyroidism. Kidney Int. 2005, 68, 1793–1800. [Google Scholar] [CrossRef]
- Urena Torres, P. Clinical experience with cinacalcet hcl. Nephrol. Dial. Transp. Off. Publ. Eur. Dial. Transp. Assoc. Eur. Ren. Assoc. 2004, 19, V27–V33. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.M.; Wu, C.C.; Hung, C.F.; Liao, M.T.; Shyu, J.F.; Hsu, Y.H.; Lu, C.L.; Wang, Y.H.; Zheng, J.Q.; Chang, T.J.; et al. Cholecalciferol additively reduces serum parathyroid hormone levels in severe secondary hyperparathyroidism treated with calcitriol and cinacalcet among hemodialysis patients. Nutrients 2018, 10, 196. [Google Scholar] [CrossRef] [PubMed]
- Malluche, H.H.; Monier-Faugere, M.C.; Wang, G.; Fraza, O.J.; Charytan, C.; Coburn, J.W.; Coyne, D.W.; Kaplan, M.R.; Baker, N.; McCary, L.C.; et al. An assessment of cinacalcet hcl effects on bone histology in dialysis patients with secondary hyperparathyroidism. Clin. Nephrol. 2008, 69, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Yajima, A.; Akizawa, T.; Tsukamoto, Y.; Kurihara, S.; Ito, A.; Group, K.S. Impact of cinacalcet hydrochloride on bone histology in patients with secondary hyperparathyroidism. Ther. Apher. Dial. 2008, 12, S38–S43. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, S.; Yamaguchi, T.; Yano, S.; Yamauchi, M.; Sugimoto, T. The calcium-sensing receptor (car) is involved in strontium ranelate-induced osteoblast differentiation and mineralization. Horm. Metab. Res. 2010, 42, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, N.; Yano, S.; Tfelt-Hansen, J.; Rooney, P.; Kanuparthi, D.; Bandyopadhyay, S.; Ren, X.; Terwilliger, E.; Brown, E.M. Mitogenic action of calcium-sensing receptor on rat calvarial osteoblasts. Endocrinology 2004, 145, 3451–3462. [Google Scholar] [CrossRef] [PubMed]
- Ward, B.K.; Magno, A.L.; Davis, E.A.; Hanyaloglu, A.C.; Stuckey, B.G.; Burrows, M.; Eidne, K.A.; Charles, A.K.; Ratajczak, T. Functional deletion of the calcium-sensing receptor in a case of neonatal severe hyperparathyroidism. J. Clin. Endocrinol. Metab. 2004, 89, 3721–3730. [Google Scholar] [CrossRef]
- Datta, H.K.; MacIntyre, I.; Zaidi, M. The effect of extracellular calcium elevation on morphology and function of isolated rat osteoclasts. Biosci. Rep. 1989, 9, 747–751. [Google Scholar] [CrossRef]
- Kanatani, M.; Sugimoto, T.; Kanzawa, M.; Yano, S.; Chihara, K. High extracellular calcium inhibits osteoclast-like cell formation by directly acting on the calcium-sensing receptor existing in osteoclast precursor cells. Biochem. Biophys. Res. Commun. 1999, 261, 144–148. [Google Scholar] [CrossRef]
- Lerner, U.H.; Ohlsson, C. The wnt system: Background and its role in bone. J. Intern. Med. 2015, 277, 630–649. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.P.; Paulson, C.; Shao, J.Z.; Zhang, X.; Wu, M.; Chen, W. Wnt and the wnt signaling pathway in bone development and disease. Front. Biosci. (Landmark Ed.) 2014, 19, 379–407. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, V.; Bryant, H.U.; Macdougald, O.A. Regulation of bone mass by wnt signaling. J. Clin. Investig. 2006, 116, 1202–1209. [Google Scholar] [CrossRef] [PubMed]
- Cadigan, K.M.; Peifer, M. Wnt signaling from development to disease: Insights from model systems. Cold Spring Harb. Perspect. Biol. 2009, 1, a002881. [Google Scholar] [CrossRef] [PubMed]
- Jenny, A. Planar cell polarity signaling in the drosophila eye. Curr. Top. Dev. Biol. 2010, 93, 189–227. [Google Scholar] [PubMed]
- Kohn, A.D.; Moon, R.T. Wnt and calcium signaling: Beta-catenin-independent pathways. Cell Calcium 2005, 38, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Westendorf, J.J.; Kahler, R.A.; Schroeder, T.M. Wnt signaling in osteoblasts and bone diseases. Gene 2004, 341, 19–39. [Google Scholar] [CrossRef] [PubMed]
- Monroe, D.G.; McGee-Lawrence, M.E.; Oursler, M.J.; Westendorf, J.J. Update on wnt signaling in bone cell biology and bone disease. Gene 2012, 492, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Bodine, P.V.; Billiard, J.; Moran, R.A.; Ponce-de-Leon, H.; McLarney, S.; Mangine, A.; Scrimo, M.J.; Bhat, R.A.; Stauffer, B.; Green, J.; et al. The wnt antagonist secreted frizzled-related protein-1 controls osteoblast and osteocyte apoptosis. J. Cell Biochem. 2005, 96, 1212–1230. [Google Scholar] [CrossRef] [PubMed]
- Sabbagh, Y.; Graciolli, F.G.; O’Brien, S.; Tang, W.; Dos Reis, L.M.; Ryan, S.; Phillips, L.; Boulanger, J.; Song, W.; Bracken, C.; et al. Repression of osteocyte wnt/beta-catenin signaling is an early event in the progression of renal osteodystrophy. J. Bone Miner. Res. 2012, 27, 1757–1772. [Google Scholar] [CrossRef]
- Carrillo-Lopez, N.; Panizo, S.; Alonso-Montes, C.; Roman-Garcia, P.; Rodriguez, I.; Martinez-Salgado, C.; Dusso, A.S.; Naves, M.; Cannata-Andia, J.B. Direct inhibition of osteoblastic wnt pathway by fibroblast growth factor 23 contributes to bone loss in chronic kidney disease. Kidney Int. 2016, 90, 77–89. [Google Scholar] [CrossRef]
- Fang, Y.; Ginsberg, C.; Seifert, M.; Agapova, O.; Sugatani, T.; Register, T.C.; Freedman, B.I.; Monier-Faugere, M.C.; Malluche, H.; Hruska, K.A. Ckd-induced wingless/integration1 inhibitors and phosphorus cause the ckd-mineral and bone disorder. J. Am. Soc. Nephrol. JASN 2014, 25, 1760–1773. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.Q.; Cao, G.; Chen, H.; Liu, D.; Su, W.; Yu, X.Y.; Vaziri, N.D.; Liu, X.H.; Bai, X.; Zhang, L.; et al. Gene and protein expressions and metabolomics exhibit activated redox signaling and wnt/beta-catenin pathway are associated with metabolite dysfunction in patients with chronic kidney disease. Redox Biol. 2017, 12, 505–521. [Google Scholar] [CrossRef] [PubMed]
- Evenepoel, P.; D’Haese, P.; Brandenburg, V. Sclerostin and dkk1: New players in renal bone and vascular disease. Kidney Int. 2015, 88, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Bennett, C.N.; Ouyang, H.; Ma, Y.L.; Zeng, Q.; Gerin, I.; Sousa, K.M.; Lane, T.F.; Krishnan, V.; Hankenson, K.D.; MacDougald, O.A. Wnt10b increases postnatal bone formation by enhancing osteoblast differentiation. J. Bone Miner. Res. 2007, 22, 1924–1932. [Google Scholar] [CrossRef] [PubMed]
- Bennett, C.N.; Longo, K.A.; Wright, W.S.; Suva, L.J.; Lane, T.F.; Hankenson, K.D.; MacDougald, O.A. Regulation of osteoblastogenesis and bone mass by wnt10b. Proc. Natl. Acad. Sci. USA 2005, 102, 3324–3329. [Google Scholar] [CrossRef] [PubMed]
- Ota, K.; Quint, P.; Ruan, M.; Pederson, L.; Westendorf, J.J.; Khosla, S.; Oursler, M.J. Tgf-beta induces wnt10b in osteoclasts from female mice to enhance coupling to osteoblasts. Endocrinology 2013, 154, 3745–3752. [Google Scholar] [CrossRef] [PubMed]
- Pederson, L.; Ruan, M.; Westendorf, J.J.; Khosla, S.; Oursler, M.J. Regulation of bone formation by osteoclasts involves wnt/bmp signaling and the chemokine sphingosine-1-phosphate. Proc. Natl. Acad. Sci. USA 2008, 105, 20764–20769. [Google Scholar] [CrossRef] [PubMed]
- Chin, J.; Miller, S.C.; Wada, M.; Nagano, N.; Nemeth, E.F.; Fox, J. Activation of the calcium receptor by a calcimimetic compound halts the progression of secondary hyperparathyroidism in uremic rats. J. Am. Soc. Nephrol. JASN 2000, 11, 903–911. [Google Scholar]
- Komaba, H.; Nakanishi, S.; Fujimori, A.; Tanaka, M.; Shin, J.; Shibuya, K.; Nishioka, M.; Hasegawa, H.; Kurosawa, T.; Fukagawa, M. Cinacalcet effectively reduces parathyroid hormone secretion and gland volume regardless of pretreatment gland size in patients with secondary hyperparathyroidism. Clin. J. Am. Soc. Nephrol. CJASN 2010, 5, 2305–2314. [Google Scholar] [CrossRef]
- Nickolas, T.L.; Stein, E.M.; Dworakowski, E.; Nishiyama, K.K.; Komandah-Kosseh, M.; Zhang, C.A.; McMahon, D.J.; Liu, X.S.; Boutroy, S.; Cremers, S.; et al. Rapid cortical bone loss in patients with chronic kidney disease. J. Bone Miner. Res. 2013, 28, 1811–1820. [Google Scholar] [CrossRef]
- Trombetti, A.; Stoermann, C.; Chevalley, T.; Van Rietbergen, B.; Herrmann, F.R.; Martin, P.Y.; Rizzoli, R. Alterations of bone microstructure and strength in end-stage renal failure. Osteoporos. Int. 2013, 24, 1721–1732. [Google Scholar] [CrossRef] [PubMed]
- Augat, P.; Reeb, H.; Claes, L.E. Prediction of fracture load at different skeletal sites by geometric properties of the cortical shell. J. Bone Miner. Res. 1996, 11, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Pistoia, W.; Van Rietbergen, B.; Ruegsegger, P. Mechanical consequences of different scenarios for simulated bone atrophy and recovery in the distal radius. Bone 2003, 33, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Nickolas, T.L.; Stein, E.; Cohen, A.; Thomas, V.; Staron, R.B.; McMahon, D.J.; Leonard, M.B.; Shane, E. Bone mass and microarchitecture in ckd patients with fracture. J. Am. Soc. Nephrol. 2010, 21, 1371–1380. [Google Scholar] [CrossRef] [PubMed]
- Cejka, D.; Patsch, J.M.; Weber, M.; Diarra, D.; Riegersperger, M.; Kikic, Z.; Krestan, C.; Schueller-Weidekamm, C.; Kainberger, F.; Haas, M. Bone microarchitecture in hemodialysis patients assessed by hr-pqct. Clin. J. Am. Soc. Nephrol. 2011, 6, 2264–2271. [Google Scholar] [CrossRef]
- Jamal, S.; Cheung, A.M.; West, S.; Lok, C. Bone mineral density by dxa and hr pqct can discriminate fracture status in men and women with stages 3 to 5 chronic kidney disease. Osteoporos. Int. 2012, 23, 2805–2813. [Google Scholar] [CrossRef] [PubMed]
- Teti, A. Mechanisms of osteoclast-dependent bone formation. BoneKEy Rep. 2013, 2, 449. [Google Scholar] [CrossRef]
- Tang, Y.; Wu, X.; Lei, W.; Pang, L.; Wan, C.; Shi, Z.; Zhao, L.; Nagy, T.R.; Peng, X.; Hu, J.; et al. Tgf-beta1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat. Med. 2009, 15, 757–765. [Google Scholar] [CrossRef]
- Dvorak, M.M.; Riccardi, D. Ca2+ as an extracellular signal in bone. Cell Calcium 2004, 35, 249–255. [Google Scholar] [CrossRef]
- Kameda, T.; Mano, H.; Yamada, Y.; Takai, H.; Amizuka, N.; Kobori, M.; Izumi, N.; Kawashima, H.; Ozawa, H.; Ikeda, K.; et al. Calcium-sensing receptor in mature osteoclasts, which are bone resorbing cells. Biochem. Biophys. Res. Commun. 1998, 245, 419–422. [Google Scholar] [CrossRef]
- Lin, K.I.; Chattopadhyay, N.; Bai, M.; Alvarez, R.; Dang, C.V.; Baraban, J.M.; Brown, E.M.; Ratan, R.R. Elevated extracellular calcium can prevent apoptosis via the calcium-sensing receptor. Biochem. Biophys. Res. Commun. 1998, 249, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Zhu, B.; Rittling, S.R.; Denhardt, D.T.; Goldberg, H.A.; McCulloch, C.A.; Sodek, J. Colocalization of intracellular osteopontin with cd44 is associated with migration, cell fusion, and resorption in osteoclasts. J. Bone Miner. Res. 2002, 17, 1486–1497. [Google Scholar] [CrossRef] [PubMed]
- Stacey, E.; Korkia, P.; Hukkanen, M.V.; Polak, J.M.; Rutherford, O.M. Decreased nitric oxide levels and bone turnover in amenorrheic athletes with spinal osteopenia. J. Clin. Endocrinol. Metab. 1998, 83, 3056–3061. [Google Scholar] [CrossRef] [PubMed]
- Lakkakorpi, P.T.; Vaananen, H.K. Kinetics of the osteoclast cytoskeleton during the resorption cycle in vitro. J. Bone Miner. Res. 1991, 6, 817–826. [Google Scholar] [CrossRef] [PubMed]






 Decreased;
Decreased;  Increased.
Increased.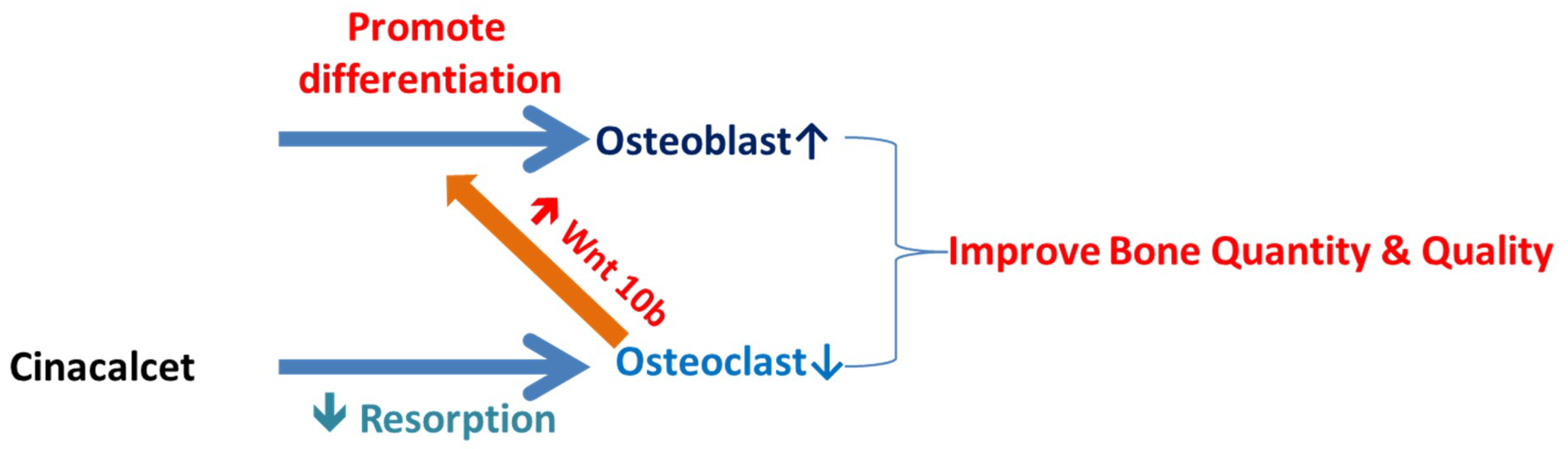
| Cortical Bone Parameters | Total Porosity (%) | Bone Mineral Density (BMD) (g/cm3) | Maximum Load (N) | Post-Yield Displacement (mm) | Stiffness (N/mm) |
|---|---|---|---|---|---|
| Cortical Thickness | −0.883 *** | 0.621 * | 0.406 | −0.586 * | 0.553 |
| Total porosity | −0.500 | −0.178 | 0.380 | −0.216 | |
| Bone Mineral Denity | 0.251 | −0.713 ** | 0.536 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, C.-M.; Hsu, Y.-H.; Wu, C.-C.; Lu, C.-L.; Liu, W.-C.; Zheng, J.-Q.; Lin, Y.-F.; Chiu, H.-W.; Chang, T.-J.; Shyu, J.-F.; et al. Osteoclast-Released Wnt-10b Underlies Cinacalcet Related Bone Improvement in Chronic Kidney Disease. Int. J. Mol. Sci. 2019, 20, 2800. https://doi.org/10.3390/ijms20112800
Zheng C-M, Hsu Y-H, Wu C-C, Lu C-L, Liu W-C, Zheng J-Q, Lin Y-F, Chiu H-W, Chang T-J, Shyu J-F, et al. Osteoclast-Released Wnt-10b Underlies Cinacalcet Related Bone Improvement in Chronic Kidney Disease. International Journal of Molecular Sciences. 2019; 20(11):2800. https://doi.org/10.3390/ijms20112800
Chicago/Turabian StyleZheng, Cai-Mei, Yung-Ho Hsu, Chia-Chao Wu, Chien-Lin Lu, Wen-Chih Liu, Jing-Quan Zheng, Yuh-Feng Lin, Hui-Wen Chiu, Tian-Jong Chang, Jia-Fwu Shyu, and et al. 2019. "Osteoclast-Released Wnt-10b Underlies Cinacalcet Related Bone Improvement in Chronic Kidney Disease" International Journal of Molecular Sciences 20, no. 11: 2800. https://doi.org/10.3390/ijms20112800






