HNF4α and CDX2 Regulate Intestinal YAP1 Promoter Activity
Abstract
:1. Introduction
2. Results
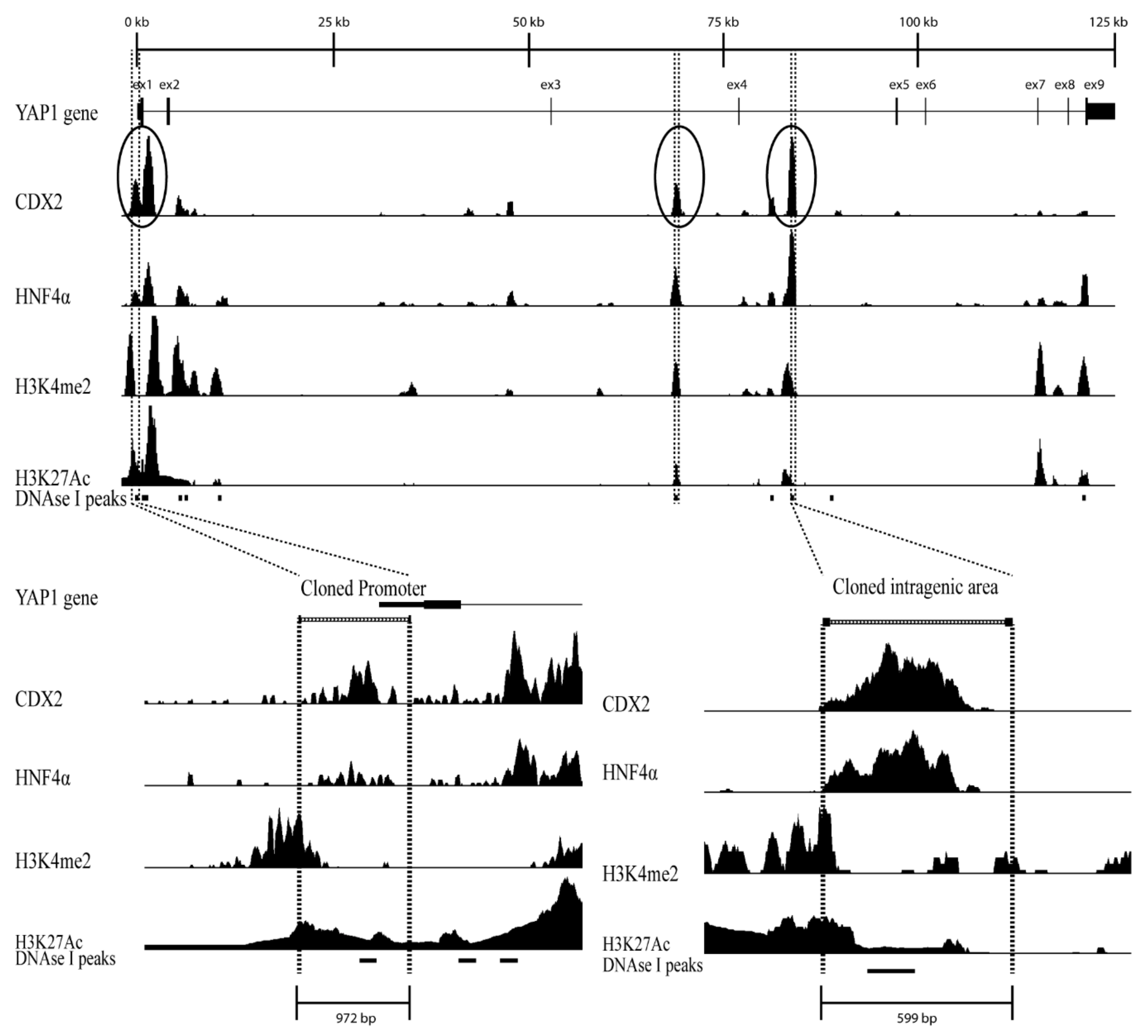
2.1. The Chromatin Landscape of the YAP1 Gene
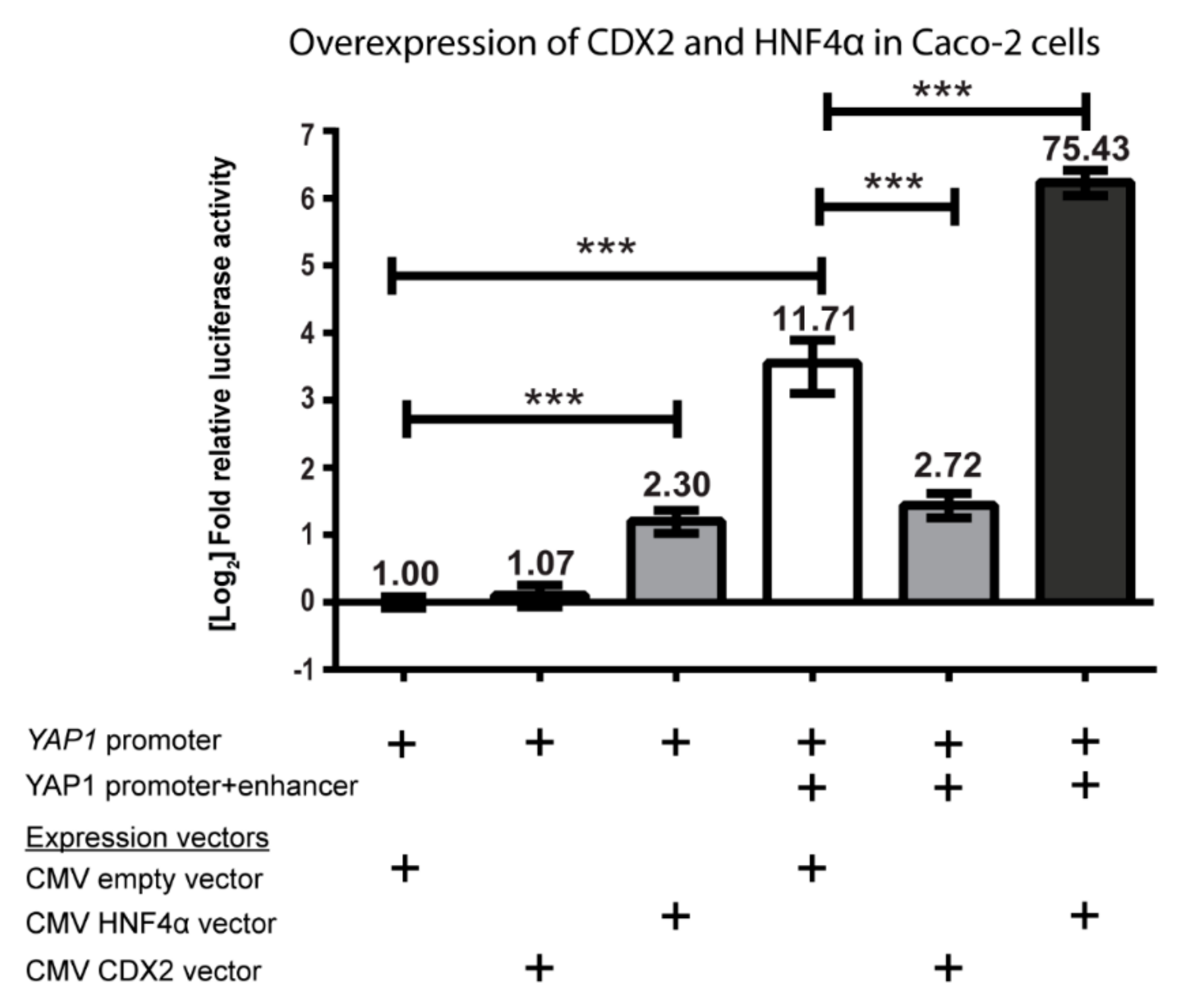
2.1.1. YAP1 Promoter Activity
2.1.2. CDX2 and HNF4α Binding Sites in the YAP1 Intragenic Enhancer
2.1.3. CDX2 Activation of YAP1 Protein Expression
3. Discussion
4. Materials and Methods
4.1. Culture of Human Cell Lines
4.2. Bioinformatic Analysis of the YAP1 Gene
4.3. Construction of Luciferase Reporter Constructs
4.4. Construction of Reporter Constructs with Mutated Binding Sites
4.5. Transfection of Cell Lines
4.6. Measuring Luciferase Activity of Reporter Constructs
4.7. Chromatin Immunoprecipitation Assay and Quantitative PCR
4.8. Electrophoretic Mobility Shift Assay
4.9. Western Blotting
4.10. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| YAP1 | Yes-associated protein 1 |
| WWTR1 | WW domain containing transcription regulator 1 |
| TEAD | TEA domain transcription factor |
| ERG | Transcriptional regulator ERG |
| TEAD4 HA | TEA domain transcription factor 4 Hemagglutinin |
| PCOS GAPDH | Polycystic ovary syndrome Glyceraldehyde-3-phosphate dehydrogenase |
| GATA4/5/6 | GATA-binding proteins 4/5/6 |
| HNF1A/B | HNF1 homeobox proteins |
| TCF7L2 | Transcription factor 7 like 2 |
| HNF4α | Hepatocyte nuclear factor 4 alpha |
| CDX2 | Caudal type homeobox 2 |
| EMSA | Electrophoretic mobility shift assay |
| HA | Hemagglutinin |
| PEI | Polyethyleneimine |
| AMPS | Ammonium persulfate |
References
- Zhao, B.; Li, L.; Lei, Q.; Guan, K.L. The Hippo–YAP pathway in organ size control and tumorigenesis: An updated version. Genes Dev. 2010, 24, 862–874. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Li, L.; Guan, K.L. Hippo signaling at a glance. J. Cell Sci. 2010, 123, 4001–4006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, D. The hippo signaling pathway in development and cancer. Dev. Cell 2010, 19, 491–505. [Google Scholar] [CrossRef]
- Lian, I.; Kim, J.; Okazawa, H.; Zhao, J.; Zhao, B.; Yu, J.; Chinnaiyan, A.; Israel, M.A.; Goldstein, L.S.; Abujarour, R.; et al. The role of YAP transcription coactivator in regulating stem cell self-renewal and differentiation. Genes Dev. 2010, 24, 1106–1118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stein, C.; Bardet, A.F.; Roma, G.; Bergling, S.; Clay, I.; Ruchti, A.; Agarinis, C.; Schmelzle, T.; Bouwmeester, T.; Schubeler, D.; et al. YAP1 Exerts Its Transcriptional Control via TEAD-Mediated Activation of Enhancers. PLoS Genet. 2015, 11, e1005465. [Google Scholar] [CrossRef] [PubMed]
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, A.; Kampf, C.; Sjostedt, E.; Asplund, A.; et al. Proteomics. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Seo, E.; Basu-Roy, U.; Gunaratne, P.H.; Coarfa, C.; Lim, D.-S.S.; Basilico, C.; Mansukhani, A. SOX2 regulates YAP1 to maintain stemness and determine cell fate in the osteo-adipo lineage. Cell Rep. 2013, 3, 2075–2087. [Google Scholar] [CrossRef] [PubMed]
- Miao, H.-L.; Pan, Z.-J.; Lei, C.-J.; Wen, J.-Y.; Li, M.-Y.; Liu, Z.-K.; Qiu, Z.-D.; Lin, M.-Z.; Chen, N.-P.; Chen, M. Knockdown of GPC3 inhibits the proliferation of Huh7 hepatocellular carcinoma cells through down-regulation of YAP. J. Cell. Biochem. 2013, 114, 625–631. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Tretiakova, M.S.; Silvis, M.R.; Lucas, J.; Klezovitch, O.; Coleman, I.; Bolouri, H.; Kutyavin, V.I.; Morrissey, C.; True, L.D.; et al. ERG Activates the YAP1 Transcriptional Program and Induces the Development of Age-Related Prostate Tumors. Cancer Cell 2015, 27, 797–808. [Google Scholar] [CrossRef] [Green Version]
- Jiang, L.-L.; Xie, J.-K.; Cui, J.-Q.; Wei, D.; Yin, B.-L.; Zhang, Y.-N.; Chen, Y.-H.; Han, X.; Wang, Q.; Zhang, C.-L. Promoter methylation of yes-associated protein (YAP1) gene in polycystic ovary syndrome. Medicine 2017, 96, e5768. [Google Scholar] [CrossRef]
- Tschaharganeh, D.F.; Chen, X.; Latzko, P.; Malz, M.; Gaida, M.M.; Felix, K.; Ladu, S.; Singer, S.; Pinna, F.; Gretz, N.; et al. Yes-associated protein up-regulates Jagged-1 and activates the Notch pathway in human hepatocellular carcinoma. Gastroenterology 2013, 144, 1530–1542.e12. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.; Zhou, G.; Zhang, Q.; Li, J.; Zhang, C. Expression of hippo pathway in colorectal cancer. Saudi J. Gastroenterol. Off. J. Saudi Gastroenterol. Assoc. 2014, 20, 188–194. [Google Scholar] [CrossRef]
- Steinhardt, A.A.; Gayyed, M.F.; Klein, A.P.; Dong, J.; Maitra, A.; Pan, D.; Montgomery, E.A.; Anders, R.A. Expression of Yes-associated protein in common solid tumors. Hum. Pathol. 2008, 39, 1582–1589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, J.; Zeng, M.; Zhu, H.; Chen, X.; Weng, Z.; Li, S. Emerging role of Hippo signalling pathway in bladder cancer. J. Cell. Mol. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zygulska, A.L.; Krzemieniecki, K.; Pierzchalski, P. Hippo pathway - brief overview of its relevance in cancer. J. Physiol. Pharmacol. 2017, 68, 311–335. [Google Scholar] [PubMed]
- Yui, S.; Azzolin, L.; Maimets, M.; Pedersen, M.T.; Fordham, R.P.; Hansen, S.L.; Larsen, H.L.; Guiu, J.; Alves, M.R.P.; Rundsten, C.F.; et al. YAP/TAZ-Dependent Reprogramming of Colonic Epithelium Links ECM Remodeling to Tissue Regeneration. Cell Stem Cell 2018, 22, 35–49e.7. [Google Scholar] [CrossRef]
- Olsen, A.K.; Boyd, M.; Danielsen, E.T.; Troelsen, J.T. Current and emerging approaches to define intestinal epithelium-specific transcriptional networks. Am. J. Physiol.-Gastrointest. Liver Physiol. 2012, 302, G277–G286. [Google Scholar] [CrossRef] [Green Version]
- Gregorieff, A.; Wrana, J.L. Hippo signalling in intestinal regeneration and cancer. Curr. Opin. Cell Biol. 2017, 48, 17–25. [Google Scholar] [CrossRef]
- Kriz, V.; Korinek, V. Wnt, RSPO and Hippo Signalling in the Intestine and Intestinal Stem Cells. Genes (Basel) 2018, 9, 20. [Google Scholar] [CrossRef]
- Boyd, M.; Hansen, M.; Jensen, T.G.K.; Perearnau, A.; Olsen, A.K.; Bram, L.L.; Bak, M.; Tommerup, N.; Olsen, J.; Troelsen, J.T. Genome-wide analysis of CDX2 binding in intestinal epithelial cells (Caco-2). J. Biol. Chem. 2010, 285, 25115–25125. [Google Scholar] [CrossRef]
- San Roman, A.K.; Aronson, B.E.; Krasinski, S.D.; Shivdasani, R.A.; Verzi, M.P. Transcription Factors GATA4 and HNF4A Control Distinct Aspects of Intestinal Homeostasis in Conjunction with Transcription Factor CDX2. J. Biol. Chem. 2015, 290, 1850–1860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suh, E.; Traber, P.G. An intestine-specific homeobox gene regulates proliferation and differentiation. Mol. Cell. Biol. 1996, 16, 619–625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silberg, D.G.; Swain, G.P.; Suh, E.R.; Traber, P.G. Cdx1 and cdx2 expression during intestinal development. Gastroenterology 2000, 119, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Simmini, S.; Bialecka, M.; Huch, M.; Kester, L.; van de Wetering, M.; Sato, T.; Beck, F.; van Oudenaarden, A.; Clevers, H.; Deschamps, J. Transformation of intestinal stem cells into gastric stem cells on loss of transcription factor Cdx2. Nat. Commun. 2014, 5, 5728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olsen, J.; Espersen, M.L.; Jess, P.; Kirkeby, L.T.; Troelsen, J.T. The clinical perspectives of CDX2 expression in colorectal cancer: A qualitative systematic review. Surg. Oncol. 2014, 23, 167–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalerba, P.; Sahoo, D.; Paik, S.; Guo, X.; Yothers, G.; Song, N.; Wilcox-Fogel, N.; Forgo, E.; Rajendran, P.S.; Miranda, S.P.; et al. CDX2 as a Prognostic Biomarker in Stage II and Stage III Colon Cancer. N. Engl. J. Med. 2016, 374, 211–222. [Google Scholar] [CrossRef]
- Bonhomme, C.; Duluc, I.; Martin, E.; Chawengsaksophak, K.; Chenard, M.P.; Kedinger, M.; Beck, F.; Freund, J.N.; Domon-Dell, C. The Cdx2 homeobox gene has a tumour suppressor function in the distal colon in addition to a homeotic role during gut development. Gut 2003, 52, 1465–1471. [Google Scholar] [CrossRef]
- Choi, B.J.; Kim, C.J.; Cho, Y.G.; Song, J.H.; Kim, S.Y.; Nam, S.W.; Lee, S.H.; Yoo, N.J.; Lee, J.Y.; Park, W.S. Altered expression of CDX2 in colorectal cancers. APMIS 2006, 114, 50–54. [Google Scholar] [CrossRef]
- Bakaris, S.; Cetinkaya, A.; Ezberci, F.; Ekerbicer, H. Expression of homeodomain protein CDX2 in colorectal adenoma and adenocarcinoma. Histol. Histopathol. 2008, 23, 1043–1047. [Google Scholar] [CrossRef]
- Drewes, T.; Senkel, S.; Holewa, B.; Ryffel, G.U. Human hepatocyte nuclear factor 4 isoforms are encoded by distinct and differentially expressed genes. Mol. Cell. Biol. 1996, 16, 925–931. [Google Scholar] [CrossRef] [Green Version]
- Garrison, W.D.; Battle, M.A.; Yang, C.; Kaestner, K.H.; Sladek, F.M.; Duncan, S.A. Hepatocyte nuclear factor 4alpha is essential for embryonic development of the mouse colon. Gastroenterology 2006, 130, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Gautier-Stein, A.; Zitoun, C.; Lalli, E.; Mithieux, G.; Rajas, F. Transcriptional regulation of the glucose-6-phosphatase gene by cAMP/vasoactive intestinal peptide in the intestine. Role of HNF4alpha, CREM, HNF1alpha, and C/EBPalpha. J. Biol. Chem. 2006, 281, 31268–31278. [Google Scholar] [CrossRef] [PubMed]
- Stegmann, A.; Hansen, M.; Wang, Y.; Larsen, J.B.; Lund, L.R.; Ritie, L.; Nicholson, J.K.; Quistorff, B.; Simon-Assmann, P.; Troelsen, J.T.; et al. Metabolome, transcriptome, and bioinformatic cis-element analyses point to HNF-4 as a central regulator of gene expression during enterocyte differentiation. Physiol. Genom. 2006, 27, 141–155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oshima, T.; Kawasaki, T.; Ohashi, R.; Hasegawa, G.; Jiang, S.; Umezu, H.; Aoyagi, Y.; Iwanari, H.; Tanaka, T.; Hamakubo, T.; et al. Downregulated P1 promoter-driven hepatocyte nuclear factor-4alpha expression in human colorectal carcinoma is a new prognostic factor against liver metastasis. Pathol. Int. 2007, 57, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Darsigny, M.; Babeu, J.P.; Seidman, E.G.; Gendron, F.P.; Levy, E.; Carrier, J.; Perreault, N.; Boudreau, F. Hepatocyte nuclear factor-4alpha promotes gut neoplasia in mice and protects against the production of reactive oxygen species. Cancer Res. 2010, 70, 9423–9433. [Google Scholar] [CrossRef] [PubMed]
- Boyd, M.; Bressendorff, S.; Møller, J.; Olsen, J.; Troelsen, J.T. Mapping of HNF4alpha target genes in intestinal epithelial cells. BMC Gastroenterol. 2009, 9, 68. [Google Scholar] [CrossRef]
- Xu, F. Cell Type-specific Autoregulation of the Caudal-related Homeobox Gene Cdx-2/3. J. Biol. Chem. 1999, 274, 34310–34316. [Google Scholar] [CrossRef]
- Saandi, T.; Baraille, F.; Derbal-Wolfrom, L.; Cattin, A.-L.; Benahmed, F.; Martin, E.; Cardot, P.; Duclos, B.; Ribeiro, A.; Freund, J.N.; et al. Regulation of the tumor suppressor homeogene Cdx2 by HNF4α in intestinal cancer. Oncogene 2013, 32, 3782–3788. [Google Scholar] [CrossRef]
- Verzi, M.P.; Hatzis, P.; Sulahian, R.; Philips, J.; Schuijers, J.; Shin, H.; Freed, E.; Lynch, J.P.; Dang, D.T.; Brown, M.; et al. TCF4 and CDX2, major transcription factors for intestinal function, converge on the same cis-regulatory regions. Proc. Natl. Acad. Sci. USA 2010, 107, 15157–15162. [Google Scholar] [CrossRef]
- Guo, R.-J.; Funakoshi, S.; Lee, H.H.; Kong, J.; Lynch, J.P. The intestine-specific transcription factor Cdx2 inhibits β-catenin/TCF transcriptional activity by disrupting the β-catenin–TCF protein complex. Carcinogenesis 2010, 31, 159–166. [Google Scholar] [CrossRef]
- Verzi, M.P.; Shin, H.; He, H.H.; Sulahian, R.; Meyer, C.A.; Montgomery, R.K.; Fleet, J.C.; Brown, M.; Liu, X.S.; Shivdasani, R.A. Differentiation-specific histone modifications reveal dynamic chromatin interactions and partners for the intestinal transcription factor CDX2. Dev. Cell 2010, 19, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Sabo, P.J.; Kuehn, M.S.; Thurman, R.; Johnson, B.E.; Johnson, E.M.; Cao, H.; Yu, M.; Rosenzweig, E.; Goldy, J.; Haydock, A.; et al. Genome-scale mapping of DNase I sensitivity in vivo using tiling DNA microarrays. Nat. Methods 2006, 3, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Danielsen, E.T.; Olsen, A.K.; Coskun, M.; Nonboe, A.W.; Larsen, S.; Dahlgaard, K.; Bennett, E.P.; Mitchelmore, C.; Vogel, L.K.; Troelsen, J.T. Intestinal regulation of suppression of tumorigenicity 14 (ST14) and serine peptidase inhibitor, Kunitz type -1 (SPINT1) by transcription factor CDX2. Sci. Rep. 2018, 8, 11813. [Google Scholar] [CrossRef] [PubMed]
- Salari, K.; Spulak, M.E.; Cuff, J.; Forster, A.D.; Giacomini, C.P.; Huang, S.; Ko, M.E.; Lin, A.Y.; van de Rijn, M.; Pollack, J.R. CDX2 is an amplified lineage-survival oncogene in colorectal cancer. Proc. Natl. Acad. Sci. USA 2012, 109, E3196–E3205. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.; Hansen, L.; Hintze, J.; Almeida, R.; Larsen, S.; Coskun, M.; Davidsen, J.; Mitchelmore, C.; David, L.; Troelsen, J.T.; et al. Precise integration of inducible transcriptional elements (PrIITE) enables absolute control of gene expression. Nucl. Acids Res. 2017, 40, 439–452. [Google Scholar] [CrossRef]
- Devriese, S.; Van den Bossche, L.; Van Welden, S.; Holvoet, T.; Pinheiro, I.; Hindryckx, P.; De Vos, M.; Laukens, D. T84 monolayers are superior to Caco-2 as a model system of colonocytes. Histochem. Cell Biol. 2017, 148, 85–93. [Google Scholar] [CrossRef]
- Chantret, I.; Barbat, A.; Dussaulx, E.; Brattain, M.G.; Zweibaum, A. Epithelial polarity, villin expression, and enterocytic differentiation of cultured human colon carcinoma cells: A survey of twenty cell lines. Cancer Res. 1988, 48, 1936–1942. [Google Scholar]
- Rousset, M. The human colon carcinoma cell lines HT-29 and Caco-2: Two in vitro models for the study of intestinal differentiation. Biochimie 1986, 68, 1035–1040. [Google Scholar] [CrossRef]
- Bolte, G.; Wolburg, H.; Beuermann, K.; Stocker, S.; Stern, M. Specific interaction of food proteins with apical membranes of the human intestinal cell lines Caco-2 and T84. Clin. Chim. Acta 1998, 270, 151–167. [Google Scholar] [CrossRef]
- James, R.; Kazenwadel, J. Homeobox gene expression in the intestinal epithelium of adult mice. J. Biol. Chem. 1991, 266, 3246–3251. [Google Scholar]
- Suh, E.; Chen, L.; Taylor, J.; Traber, P.G. A homeodomain protein related to caudal regulates intestine-specific gene transcription. Mol. Cell. Biol. 1994, 14, 7340–7351. [Google Scholar] [CrossRef] [PubMed]
- Mathelier, A.; Fornes, O.; Arenillas, D.J.; Chen, C.Y.; Denay, G.; Lee, J.; Shi, W.; Shyr, C.; Tan, G.; Worsley-Hunt, R.; et al. JASPAR 2016: A major expansion and update of the open-access database of transcription factor binding profiles. Nucl. Acids Res. 2016, 44, D110–D115. [Google Scholar] [CrossRef] [PubMed]
- Coskun, M.; Olsen, A.K.; Holm, T.L.; Kvist, P.H.; Nielsen, O.H.; Riis, L.B.; Olsen, J.; Troelsen, J.T. TNF-α-induced down-regulation of CDX2 suppresses MEP1A expression in colitis. Biochimica et Biophysica Acta 2012, 1822, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Troelsen, J.T.; Mitchelmore, C.; Olsen, J. An enhancer activates the pig lactase phlorizin hydrolase promoter in intestinal cells. Gene 2003, 305, 101–111. [Google Scholar] [CrossRef]





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larsen, S.; Davidsen, J.; Dahlgaard, K.; Pedersen, O.B.; Troelsen, J.T. HNF4α and CDX2 Regulate Intestinal YAP1 Promoter Activity. Int. J. Mol. Sci. 2019, 20, 2981. https://doi.org/10.3390/ijms20122981
Larsen S, Davidsen J, Dahlgaard K, Pedersen OB, Troelsen JT. HNF4α and CDX2 Regulate Intestinal YAP1 Promoter Activity. International Journal of Molecular Sciences. 2019; 20(12):2981. https://doi.org/10.3390/ijms20122981
Chicago/Turabian StyleLarsen, Sylvester, Johanne Davidsen, Katja Dahlgaard, Ole B. Pedersen, and Jesper T. Troelsen. 2019. "HNF4α and CDX2 Regulate Intestinal YAP1 Promoter Activity" International Journal of Molecular Sciences 20, no. 12: 2981. https://doi.org/10.3390/ijms20122981




