FSH/LH-Dependent Upregulation of Ahr in Murine Granulosa Cells Is Controlled by PKA Signaling and Involves Epigenetic Regulation
Abstract
:1. Introduction
2. Results
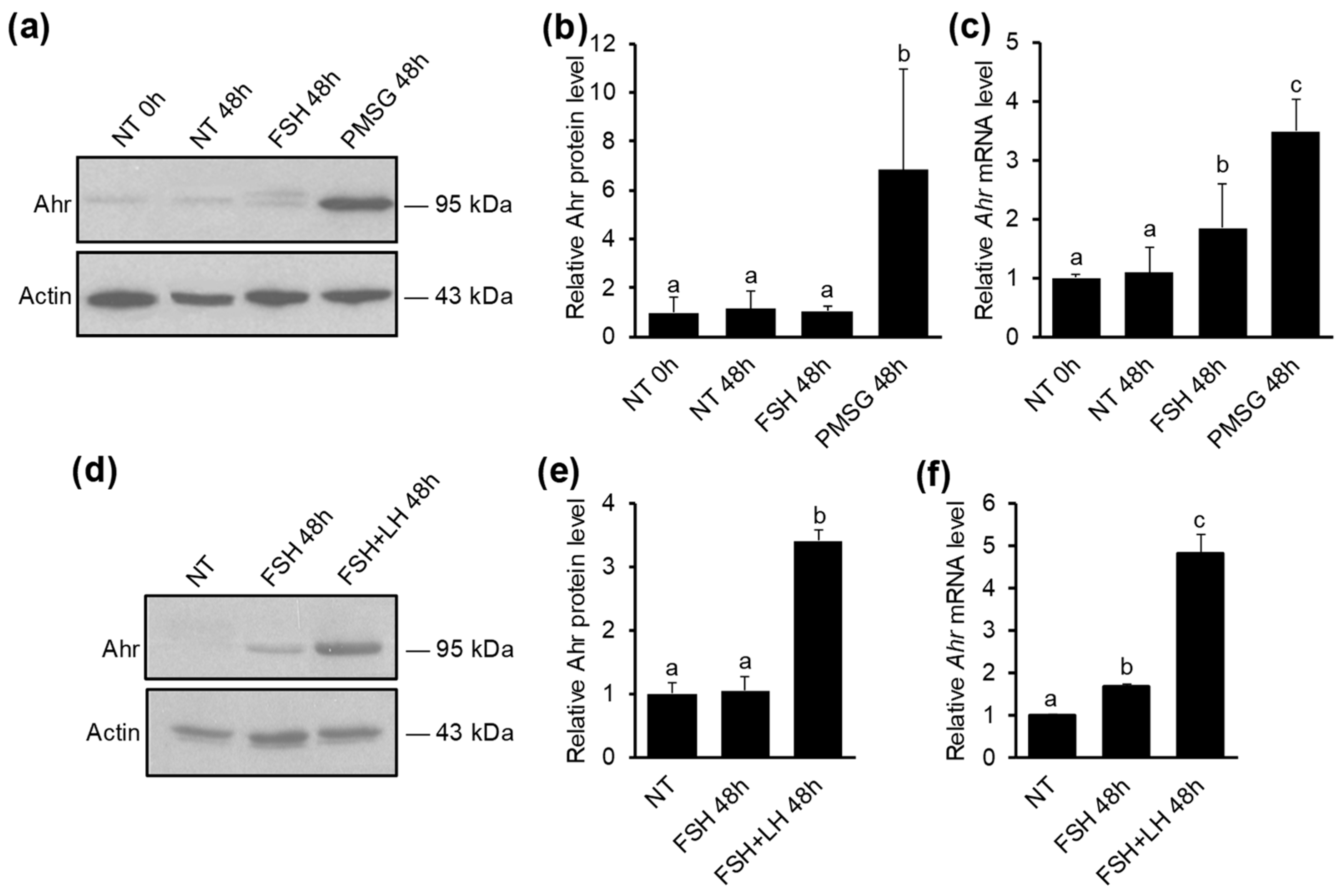
2.1. The Upregulation of Ahr in GCs during Follicular Maturation Requires both FSH and LH Activity
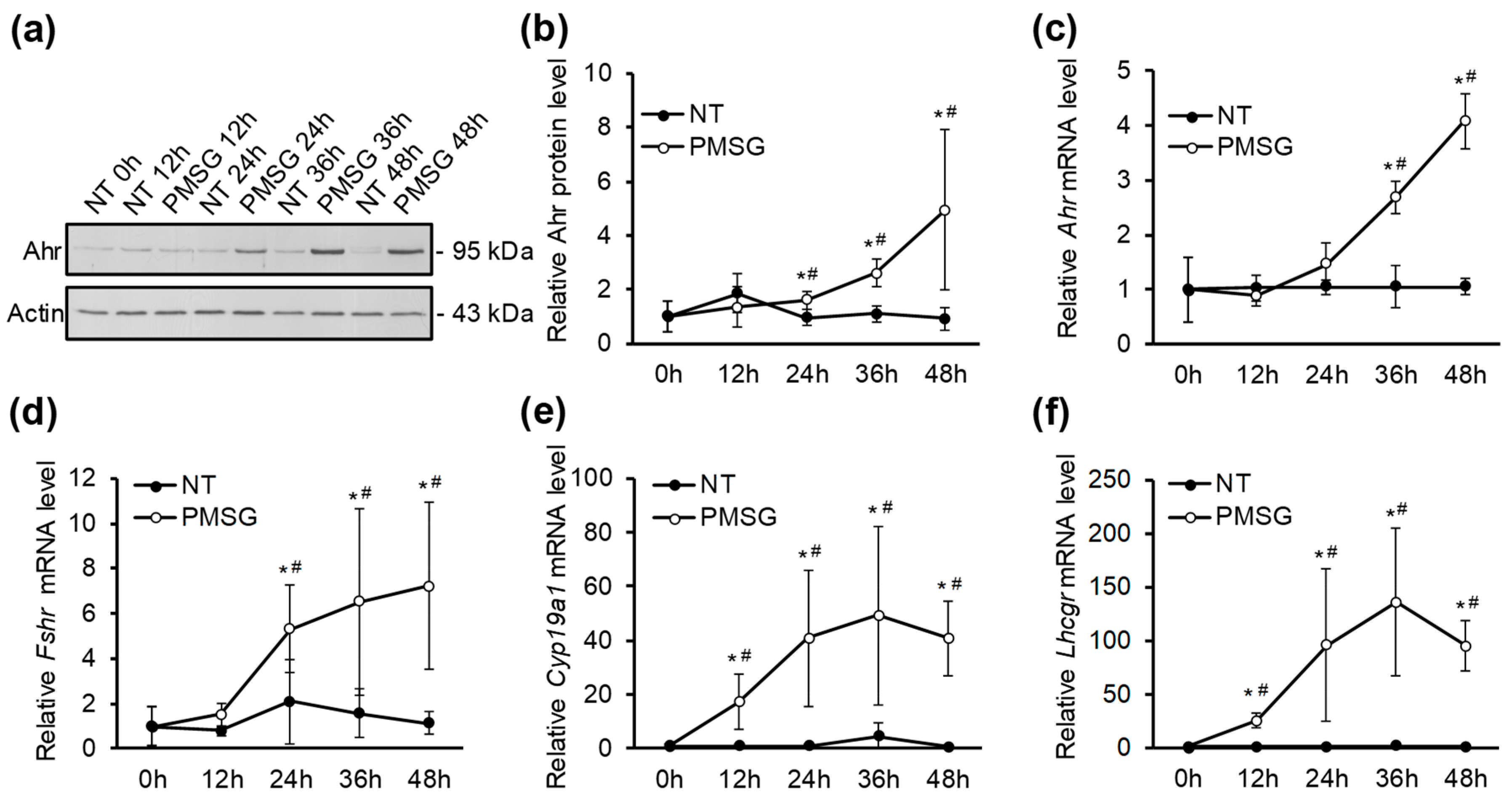
2.2. The Effect of PMSG on The Expression Dynamics of Ahr and Follicle Maturation Marker Genes
2.3. Ahr Is Upregulated in Large Antral Follicles in Response to PMSG
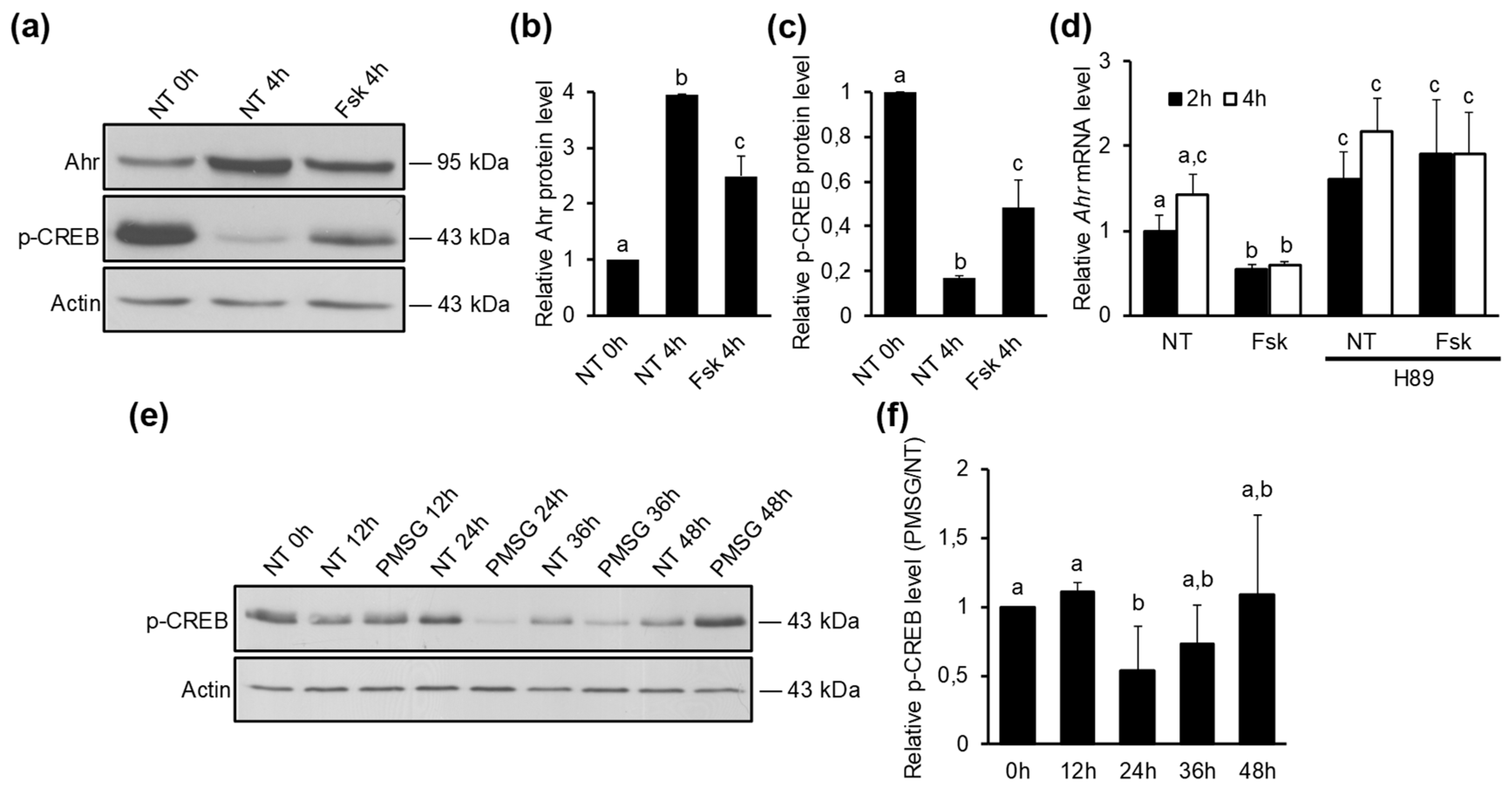
2.4. Ahr Is Downregulated by Protein Kinase A Signaling Pathway
2.5. The Increased Transcription of Ahr in Response to PMSG is Regulated by Chromatin Accessibility
3. Discussion
4. Materials and Methods
4.1. Medium, Chemicals, and Antibodies
4.2. Ethics
4.3. Animals and Granulosa Cell Isolation
4.4. Western Blotting
4.5. Isolation of RNA and mRNA/hnRNA Measurement by RT-qPCR
4.6. Immunofluorescence
4.7. Plasmids, Transient Transfection and Reporter Gene Assay
4.8. Chromatin Accessibility by Real-Time PCR (CHART-PCR)
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AHR | aryl hydrocarbon receptor |
| TCDD | 2,3,7,8-tetrachlorodibenzo-p-dioxin |
| FSH | follicle stimulating hormone |
| LH | luteinizing hormone |
| FSK | forskolin |
| PKA | Protein kinase A |
| hnRNA | heteronuclear RNA |
| ActD | actinomycin D |
| CHART-PCR | chromatin accessibility by real-time PCR |
References
- Denison, M.S.; Nagy, S.R. Activation of the aryl hydrocarbon receptor by structurally diverse exogenous and endogenous chemicals. Annu. Rev. Pharmacol. Toxicol. 2003, 43, 309–334. [Google Scholar] [CrossRef] [PubMed]
- Abbott, B.D.; Schmid, J.E.; Pitt, J.A.; Buckalew, A.R.; Wood, C.R.; Held, G.A.; Diliberto, J.J. Adverse reproductive outcomes in the transgenic Ah receptor-deficient mouse. Toxicol. Appl. Pharmacol. 1999, 15, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Mimura, J.; Fujii-Kuriyama, Y. Functional role of AhR in the expression of toxic effects by TCDD. Biochim. Biophys. Acta 2003, 1619, 263–268. [Google Scholar] [CrossRef]
- Meyer, B.K.; Perdew, G.H. Characterization of the AhR−hsp90−XAP2 Core Complex and the Role of the Immunophilin-Related Protein XAP2 in AhR Stabilization. Biochemistry 1999, 38, 8907–8917. [Google Scholar] [CrossRef] [PubMed]
- Kudo, I.; Hosaka, M.; Haga, A.; Tsuji, N.; Nagata, Y.; Okada, H.; Fukuda, K.; Kakizaki, Y.; Okamoto, T.; Grave, E.; et al. The regulation mechanisms of AhR by molecular chaperone complex. J. Biochem. 2018, 163, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Savas, U.; Alexander, D.L.; Jefcoate, C.R. Characterization of the mouse Cyp1B1 gene. Identification of an enhancer region that directs aryl hydrocarbon receptor-mediated constitutive and induced expression. J. Biol. Chem. 1998, 27, 5174–5183. [Google Scholar] [CrossRef] [PubMed]
- Denison, M.S.; Fisher, J.M.; Whitlock, J.P., Jr. Protein-DNA interactions at recognition sites for the dioxin-Ah receptor complex. J. Biol. Chem. 1989, 264, 16478–16482. [Google Scholar]
- Baba, T.; Mimura, J.; Gradin, K.; Kuroiwa, A.; Watanabe, T.; Matsuda, Y.; Inazawa, J.; Sogawa, K.; Fujii-Kuriyama, Y. Structure and expression of the Ah receptor repressor gene. J. Biol. Chem. 2001, 276, 33101–33110. [Google Scholar] [CrossRef]
- Hahn, M.E. Aryl hydrocarbon receptors: Diversity and evolution. Chemico-biol. Interact. 2002, 141, 131–160. [Google Scholar] [CrossRef]
- Harrill, J.A.; Hukkanen, R.R.; Lawson, M.; Martin, G.; Gilger, B.; Soldatow, V.; Lecluyse, E.L.; Budinsky, R.A.; Rowlands, J.C.; Thomas, R.S. Knockout of the aryl hydrocarbon receptor results in distinct hepatic and renal phenotypes in rats and mice. Toxicol. Appl. Pharmacol. 2013, 15, 503–518. [Google Scholar] [CrossRef]
- Gutiérrez-Vázquez, C.; Quintana, F.J. Regulation of the Immune Response by the Aryl Hydrocarbon Receptor. Immunity 2018, 48, 19–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnett, K.R.; Tomic, D.; Gupta, R.K.; Miller, K.P.; Meachum, S.; Paulose, T.; Flaws, J.A. The aryl hydrocarbon receptor affects mouse ovarian follicle growth via mechanisms involving estradiol regulation and responsiveness. Biol. Reprod. 2007, 76, 1062–1070. [Google Scholar] [CrossRef] [PubMed]
- Kolluri, S.K.; Jin, U.H.; Safe, S. Role of the aryl hydrocarbon receptor in carcinogenesis and potential as an anti-cancer drug target. Arch. Toxicol. 2017, 91, 2497–2513. [Google Scholar] [CrossRef] [PubMed]
- Kharat, I.; Saatcioglu, F. Antiestrogenic effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin are mediated by direct transcriptional interference with the liganded estrogen receptor. Cross-talk between aryl hydrocarbon- and estrogen-mediated signaling. J. Biol. Chem. 1996, 271, 10533–10537. [Google Scholar] [CrossRef] [PubMed]
- Son, D.S.; Ushinohama, K.; Gao, X.; Taylor, C.C.; Roby, K.F.; Rozman, K.K.; Terranova, P.F. 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) blocks ovulation by a direct action on the ovary without alteration of ovarian steroidogenesis: Lack of a direct effect on ovarian granulosa and thecal-interstitial cell steroidogenesis in vitro. Reprod. Toxicol. 1999, 13, 521–530. [Google Scholar] [CrossRef]
- Benedict, J.C.; Lin, T.M.; Loeffler, I.K.; Peterson, R.E.; Flaws, J.A. Physiological role of the aryl hydrocarbon receptor in mouse ovary development. Toxicol. Sci. 2000, 56, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Barnett, K.R.; Tomic, D.; Gupta, R.K.; Babus, J.K.; Roby, K.F.; Terranova, P.F.; Flaws, J.A. The aryl hydrocarbon receptor is required for normal gonadotropin responsiveness in the mouse ovary. Toxicol. Appl. Pharmacol. 2007, 223, 66–72. [Google Scholar] [CrossRef] [Green Version]
- Benedict, J.C.; Miller, K.P.; Lin, T.M.; Greenfeld, C.; Babus, J.K.; Peterson, R.E.; Flaws, J.A. Aryl hydrocarbon receptor regulates growth, but not atresia, of mouse preantral and antral follicles. Biol. Reprod. 2003, 68, 1511–1517. [Google Scholar] [CrossRef]
- Baba, T.; Mimura, J.; Nakamura, N.; Harada, N.; Yamamoto, M.; Morohashi, K.; Fujii-Kuriyama, Y. Intrinsic function of the aryl hydrocarbon (Dioxin) receptor as a key factor in female reproduction. Mol. Cell. Biol. 2005, 25, 10040–10051. [Google Scholar] [CrossRef]
- Edson, M.A.; Nagaraja, A.K.; Matzuk, M.M. The mammalian ovary from genesis to revelation. Endocr. Rev. 2009, 30, 624–712. [Google Scholar] [CrossRef]
- Bao, B.; Garverick, H.A. Expression of steroidogenic enzyme and gonadotropin receptor genes in bovine follicles during ovarian follicular waves: A review. J. Anim. Sci. 1998, 76, 1903–1921. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, A.J.; Adashi, E.Y.; Jones, P.B.; Welsh, T.H., Jr. Hormonal regulation of the differentiation of cultured ovarian granulosa cells. Endocr. Rev. 1984, 5, 76–127. [Google Scholar] [CrossRef] [PubMed]
- Menon, K.M.J.; Menon, B.; Gulappa, T. Regulation of Luteinizing Hormone Receptor mRNA Expression in the Ovary: The Role of miR-122. Vitam. Horm. 2018, 107, 67–87. [Google Scholar] [PubMed]
- Gates, A.H.; Bozarth, J.L. Ovulation in the PMSG-treated immature mouse: Effect of dose, age, weight, puberty, season and strain (BALB/c, 129 and C129F1 hybrid). Biol. Reprod. 1978, 18, 497–505. [Google Scholar] [CrossRef]
- Richards, J.S. Maturation of ovarian follicles: Actions and interactions of pituitary and ovarian hormones on follicular cell differentiation. Physiol. Rev. 1980, 60, 51–89. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Park-Sarge, O.K.; Mayo, K.E. Gonadotropins induce rapid phosphorylation of the 3′,5′-cyclic adenosine monophosphate response element binding protein in ovarian granulosa cells. Endocrinology 1996, 137, 3234–4325. [Google Scholar] [CrossRef] [PubMed]
- Carlone, D.L.; Richards, J.S. Functional interactions, phosphorylation, and levels of 3′,5′-cyclic adenosine monophosphate-regulatory element binding protein and steroidogenic factor-1 mediate hormone-regulated and constitutive expression of aromatase in gonadal cells. Mol. Endocrinol. 1997, 11, 292–304. [Google Scholar]
- Puri, P.; Little-Ihrig, L.; Chandran, U.; Law, N.C.; Hunzicker-Dunn, M.; Zeleznik, A.J. Protein Kinase A: A Master Kinase of Granulosa Cell Differentiation. Sci. Rep. 2016, 6, 28132. [Google Scholar] [CrossRef] [Green Version]
- Richards, J.S.; Kersey, K.A. Changes in Theca and Granulosa Cell Function in Antral Follicles Developing during Pregnancy in the Rat: Gonadotropin Receptors, Cyclic AMP and Estradiol-17β. Biol. Reprod. 1979, 21, 1185–1201. [Google Scholar] [CrossRef]
- Chaffin, C.L.; Stouffer, R.L.; Duffy, D.M. Gonadotropin and steroid regulation of steroid receptor and aryl hydrocarbon receptor messenger ribonucleic acid in macaque granulosa cells during the periovulatory interval. Endocrinology 1999, 140, 4753–4760. [Google Scholar] [CrossRef]
- Chaffin, C.L.; Trewin, A.L.; Hutz, R.J. Estrous cycle-dependent changes in the expression of aromatic hydrocarbon receptor (AHR) and AHR-nuclear translocator (ARNT) MRNAs in the rat ovary and liver. Chem. Biol. Interact. 2000, 124, 205–216. [Google Scholar] [CrossRef]
- Teino, I.; Matvere, A.; Kuuse, S.; Ingerpuu, S.; Maimets, T.; Kristjuhan, A.; Tiido, T. Transcriptional repression of the Ahr gene by LHCGR signaling in preovulatory granulosa cells is controlled by chromatin accessibility. Mol. Cell. Endocrinol. 2014, 382, 292–301. [Google Scholar] [CrossRef] [PubMed]
- LaVoie, H.A. Epigenetic control of ovarian function: The emerging role of histone modifications. Mol. Cell Endocrinol. 2005, 243, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Garrison, P.M.; Rogers, J.M.; Brackney, W.R.; Denison, M.S. Effects of histone deacetylase inhibitors on the Ah receptor gene promoter. Arch. Biochem. Biophys. 2000, 374, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Ko, C.I.; Wang, Q.; Fan, Y.; Xia, Y.; Puga, A. Pluripotency factors and Polycomb Group proteins repress aryl hydrocarbon receptor expression in murine embryonic stem cells. Stem Cell Res. 2014, 12, 296–308. [Google Scholar] [CrossRef]
- Peng, X.R.; Hsueh, A.J.; LaPolt, P.S.; Bjersing, L.; Ny, T. Localization of luteinizing hormone receptor messenger ribonucleic acid expression in ovarian cell types during follicle development and ovulation. Endocrinology 1991, 129, 3200–3207. [Google Scholar] [CrossRef] [PubMed]
- LaPolt, P.S.; Tilly, J.L.; Aihara, T.; Nishimori, K.; Hsueh, A.J. Gonadotropin-induced up- and down-regulation of ovarian follicle-stimulating hormone (FSH) receptor gene expression in immature rats: Effects of pregnant mare’s serum gonadotropin, human chorionic gonadotropin, and recombinant FSH. Endocrinology 1992, 130, 1289–1295. [Google Scholar]
- Sites, C.K.; Patterson, K.; Jamison, C.S.; Degen, S.J.; LaBarbera, A.R. Follicle-stimulating hormone (FSH) increases FSH receptor messenger ribonucleic acid while decreasing FSH binding in cultured porcine granulosa cells. Endocrinology 1994, 134, 411–417. [Google Scholar] [CrossRef]
- Richards, J.S. Hormonal control of gene expression in the ovary. Endocr. Rev. 1994, 15, 725–751. [Google Scholar] [CrossRef]
- Richards, J.S.; Fitzpatrick, S.L.; Clemens, J.W.; Morris, J.K.; Alliston, T.; Sirois, J. Ovarian cell differentiation: A cascade of multiple hormones, cellular signals, and regulated genes. Rec. Prog. Horm. Res. 1995, 50, 223–254. [Google Scholar]
- Findlay, J.K.; Drummond, A.E. Regulation of the FSH Receptor in the Ovary. Trend. Endocrinol. Metab. 1999, 10, 183–188. [Google Scholar] [CrossRef]
- Chakraborty, P.; Roy, S.K. Expression of FSH receptor in the hamster ovary during perinatal development. Mol. Cell. Endocrinol. 2015, 400, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Teino, I.; Kuuse, S.; Ingerpuu, S.; Maimets, T.; Tiido, T. The aryl hydrocarbon receptor regulates mouse Fshr promoter activity through an E-box binding site. Biol. Reprod. 2012, 86, 77. [Google Scholar] [CrossRef]
- McGee, E.A.; Hsueh, A.J. Initial and cyclic recruitment of ovarian follicles. Endocr. Rev. 2000, 21, 200–214. [Google Scholar] [CrossRef] [PubMed]
- Salvador, L.M.; Park, Y.; Cottom, J.; Maizels, E.T.; Jones, J.C.; Schillace, R.V.; Carr, D.W.; Cheung, P.; Allis, C.D.; Jameson, J.L.; et al. Follicle stimulating hormone stimulates protein kinase A-mediated histone H3 phosphorylation and acetylation leading to select gene activation in ovarian granulosa cells. J. Biol. Chem. 2001, 276, 40146–40155. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Robayna, I.J.; Alliston, T.N.; Buse, P.; Firestone, G.L.; Richards, J.S. Functional and subcellular changes in the A-kinase-signaling pathway: Relation to aromatase and Sgk expression during the transition of granulosa cells to luteal cells. Mol. Endocrinol. 1999, 13, 1318–1337. [Google Scholar] [CrossRef]
- Ranta, T.; Knecht, M.; Darbon, J.M.; Baukal, A.J.; Catt, K.J. Induction of granulosa cell differentiation by forskolin: Stimulation of adenosine 3′,5′-monophosphate production, progesterone synthesis, and luteinizing hormone receptor expression. Endocrinology 1984, 114, 845–850. [Google Scholar] [CrossRef]
- Hunzicker-Dunn, M.; Maizels, E.T. FSH signaling pathways in immature granulosa cells that regulate target gene expression: Branching out from protein kinase A. Cell. Signal. 2006, 18, 1351–1359. [Google Scholar] [CrossRef] [Green Version]
- Minegishi, T.; Tano, M.; Kishi, H.; Kameda, T.; Miyamoto, K. Follicle-stimulating hormone regulation on its receptor messenger ribonucleic acid levels in cultured rat granulosa cells. Biochim. Biophys. Acta. 1997, 1359, 165–173. [Google Scholar] [CrossRef] [Green Version]
- Fitzgerald, C.T.; Nebert, D.W.; Puga, A. Regulation of mouse Ah receptor (Ahr) gene basal expression by members of the Sp family of transcription factors. DNA Cell Biol. 1998, 17, 811–822. [Google Scholar] [CrossRef]
- Garrison, P.M.; Denison, M.S. Analysis of the murine AhR gene promoter. J. Biochem. Mol. Toxicol. 2000, 14, 1–10. [Google Scholar] [CrossRef]
- Zhang, J.; Watson, A.J.; Probst, M.R.; Minehart, E.; Hankinson, O. Basis for the loss of aryl hydrocarbon receptor gene expression in clones of a mouse hepatoma cell line. Mol. Pharmacol. 1996, 50, 1454–1462. [Google Scholar] [PubMed]
- Englert, N.A.; Turesky, R.J.; Han, W.; Bessette, E.E.; Spivack, S.D.; Caggana, M.; Spink, D.C.; Spink, B.C. Genetic and epigenetic regulation of AHR gene expression in MCF-7 breast cancer cells: Role of the proximal promoter GC-rich region. Biochem. Pharmacol. 2012, 84, 722–735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, L.; Asada, H.; Kizuka, F.; Tamura, I.; Maekawa, R.; Taketani, T.; Sato, S.; Yamagata, Y.; Tamura, H.; Sugino, N. Changes in histone modification and DNA methylation of the StAR and Cyp19a1 promoter regions in granulosa cells undergoing luteinization during ovulation in rats. Endocrinology 2013, 154, 458–470. [Google Scholar] [CrossRef]
- Bussmann, U.A.; Baranao, J.L. Regulation of aryl hydrocarbon receptor expression in rat granulosa cells. Biol. Reprod. 2006, 75, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Combarnous, Y.; Guillou, F.; Martinat, N.; Cahoreau, C. Origin of the FSH + LH double activity of equine chorionic gonadotropin (eCG/PMSG). Ann. Endocrinol. 1984, 45, 261–268. [Google Scholar]
- Palermo, R. Differential actions of FSH and LH during folliculogenesis. Reprod. Biomed. Online. 2007, 15, 326–337. [Google Scholar] [CrossRef]
- Ruman, J.I.; Pollak, S.; Trousdale, R.K.; Klein, J.; Lustbader, J.W. Effects of long-acting recombinant human follicle-stimulating hormone analogs containing N-linked glycosylation on murine folliculogenesis. Fertil. Steril. 2005, 83, 1303–1309. [Google Scholar] [CrossRef]
- Khan, H.A.; Zhao, Y.; Wang, L.; Li, Q.; Du, Y.A.; Dan, Y.; Huo, L.J. Identification of miRNAs during mouse postnatal ovarian development and superovulation. J. Ovar. Res. 2015, 8, 44. [Google Scholar] [CrossRef]
- Li, Y.; Fang, Y.; Liu, Y.; Yang, X. MicroRNAs in ovarian function and disorders. J. Ovar. Res. 2015, 8, 51. [Google Scholar] [CrossRef]
- Bahrami, A.; Miraie-Ashtiani, S.R.; Sadeghi, M.; Najafi, A. miRNA-mRNA network involved in folliculogenesis interactome: Systems biology approach. Reproduction 2017, 154, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Carletti, M.Z.; Christenson, L.K. Rapid effects of LH on gene expression in the mural granulosa cells of mouse periovulatory follicles. Reproduction 2009, 137, 843–855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiedler, S.D.; Carletti, M.Z.; Hong, X.; Christenson, L.K. Hormonal regulation of MicroRNA expression in periovulatory mouse mural granulosa cells. Biol. Reprod. 2008, 79, 1030–1037. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.C.; Chang, H.Y.; Chen, C.Y.; Wu, P.Y.; Lee, H.; Liao, Y.F.; Hsu, W.M.; Huang, H.C.; Juan, H.F. Silencing of MiR-124 induces neuroblastoma SK-N-SH cell differentiation, cell cycle arrest and apoptosis through promoting AHR. FEBS Lett. 2011, 585, 3582–3586. [Google Scholar] [CrossRef] [PubMed]
- Chowdhary, V.; Teng, K.Y.; Thakral, S.; Zhang, B.; Lin, C.H.; Wani, N.; Bruschweiler-Li, L.; Zhang, X.; James, L.; Yang, D.; et al. miRNA-122 Protects Mice and Human Hepatocytes from Acetaminophen Toxicity by Regulating Cytochrome P450 Family 1 Subfamily A Member 2 and Family 2 Subfamily E Member 1 Expression. Am. J. Pathol. 2017, 187, 2758–2774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.C.; Xia, M.; Zhang, Y.J.; Jin, P.; Zhao, L.; Zhang, J.; Li, T.; Zhou, X.M.; Tu, Y.Y.; Kong, F.; et al. Micro124-mediated AHR expression regulates the inflammatory response of chronic rhinosinusitis (CRS) with nasal polyps. Biochem. Biophys. Res. Commun. 2018, 500, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Harper, P.A.; Riddick, D.S.; Okey, A.B. Regulating the regulator: Factors that control levels and activity of the aryl hydrocarbon receptor. Biochem. Pharmacol. 2006, 72, 267–279. [Google Scholar] [CrossRef]
- Shi, Z.; Valdez, K.E.; Ting, A.Y.; Franczak, A.; Gum, S.L.; Petroff, B.K. Ovarian endocrine disruption underlies premature reproductive senescence following environmentally relevant chronic exposure to the aryl hydrocarbon receptor agonist 2,3,7,8-tetrachlorodibenzo-p-dioxin. Biol. Reprod. 2007, 76, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Wigglesworth, K.; Lee, K.B.; Emori, C.; Sugiura, K.; Eppig, J.J. Transcriptomic diversification of developing cumulus and mural granulosa cells in mouse ovarian follicles. Biol. Reprod. 2015, 92, 23. [Google Scholar] [CrossRef]
- Maizels, E.T.; Mukherjee, A.; Sithanandam, G.; Peters, C.A.; Cottom, J.; Mayo, K.E.; Hunzicker-Dunn, M. Developmental regulation of mitogen-activated protein kinase-activated kinases-2 and -3 (MAPKAPK-2/-3) in vivo during corpus luteum formation in the rat. Mol. Endocrinol. 2001, 15, 716–733. [Google Scholar] [CrossRef]
- Elferink, C.J.; Reiners, J.J., Jr. Quantitative RT-PCR on CYP1A1 heterogeneous nuclear RNA: A surrogate for the in vitro transcription run-on assay. Biotechniques 1996, 20, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Wu, C. Chromatin remodeling and the control of gene expression. J. Biol. Chem. 1997, 272, 28171–28174. [Google Scholar] [CrossRef] [PubMed]
- DeManno, D.A.; Cottom, J.E.; Kline, M.P.; Peters, C.A.; Maizels, E.T.; Hunzicker-Dunn, M. Follicle-stimulating hormone promotes histone H3 phosphorylation on serine-10. Mol. Endocrinol. 1999, 13, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Collas, P.; Le Guellec, K.; Taskén, K. The A-kinase-anchoring protein AKAP95 is a multivalent protein with a key role in chromatin condensation at mitosis. J. Cell Biol. 1999, 147, 1167–1180. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Cruickshank, M.; Fenwick, E.; Abraham, L.J.; Ulgiati, D. Quantitative differences in chromatin accessibility across regulatory regions can be directly compared in distinct cell-types. Biochem. Biophys. Res. Commun. 2008, 367, 349–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Application | Oligonucleotide | Sequence (5′–3′) | |
|---|---|---|---|
| qPCR | Ahr mRNA | Forward | GGCCAAGAGCTTCTTTGATG |
| Reverse | TGCCAGTCTCTGATTTGTGC | ||
| Ahr hnRNA E2-I2 | Forward | TAGGCTCAGCGTCAGCTACC | |
| Reverse | GTCACCAACATTTAAAGGACCAC | ||
| Fshr mRNA | Forward | GCGGCAAACCTCTGAACTTC | |
| Reverse | TGATCCCCAGGCTGAGTCAT | ||
| Cyp19a1 mRNA | Forward | GCCTCCTTCTCCTGATTTGGA | |
| Reverse | CTGCCATGGGAAATGAGGG | ||
| Lhcgr mRNA | Forward | AGTCACAGCTGCACTCTCC | |
| Reverse | GTGAGAGATAGTCGGGCGAG | ||
| Tbp mRNA | Forward | GGCCTCTCAGAAGCATCACT | |
| Reverse | GCCAAGCCCTGAGCATAA | ||
| CHART-PCR | Ahr promoter | Forward | TTTTGAGGCTGGAAAACAGGTACT |
| Reverse | ACGTGATGACGCAGGACGTA | ||
| Cyp19a1 promoter | Forward | CCAATCCAGCACCCTTCCAA | |
| Reverse | GGGAAGAAAGCAGTGGTGGA | ||
| Pax7 promoter | Forward | GTTATCGGTCCCCTCTCCCT | |
| Reverse | CTCAACGGCAGGGAAGGG | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matvere, A.; Teino, I.; Varik, I.; Kuuse, S.; Tiido, T.; Kristjuhan, A.; Maimets, T. FSH/LH-Dependent Upregulation of Ahr in Murine Granulosa Cells Is Controlled by PKA Signaling and Involves Epigenetic Regulation. Int. J. Mol. Sci. 2019, 20, 3068. https://doi.org/10.3390/ijms20123068
Matvere A, Teino I, Varik I, Kuuse S, Tiido T, Kristjuhan A, Maimets T. FSH/LH-Dependent Upregulation of Ahr in Murine Granulosa Cells Is Controlled by PKA Signaling and Involves Epigenetic Regulation. International Journal of Molecular Sciences. 2019; 20(12):3068. https://doi.org/10.3390/ijms20123068
Chicago/Turabian StyleMatvere, Antti, Indrek Teino, Inge Varik, Sulev Kuuse, Tarmo Tiido, Arnold Kristjuhan, and Toivo Maimets. 2019. "FSH/LH-Dependent Upregulation of Ahr in Murine Granulosa Cells Is Controlled by PKA Signaling and Involves Epigenetic Regulation" International Journal of Molecular Sciences 20, no. 12: 3068. https://doi.org/10.3390/ijms20123068





