Activation of the IGF Axis in Thyroid Cancer: Implications for Tumorigenesis and Treatment
Abstract
:1. Introduction
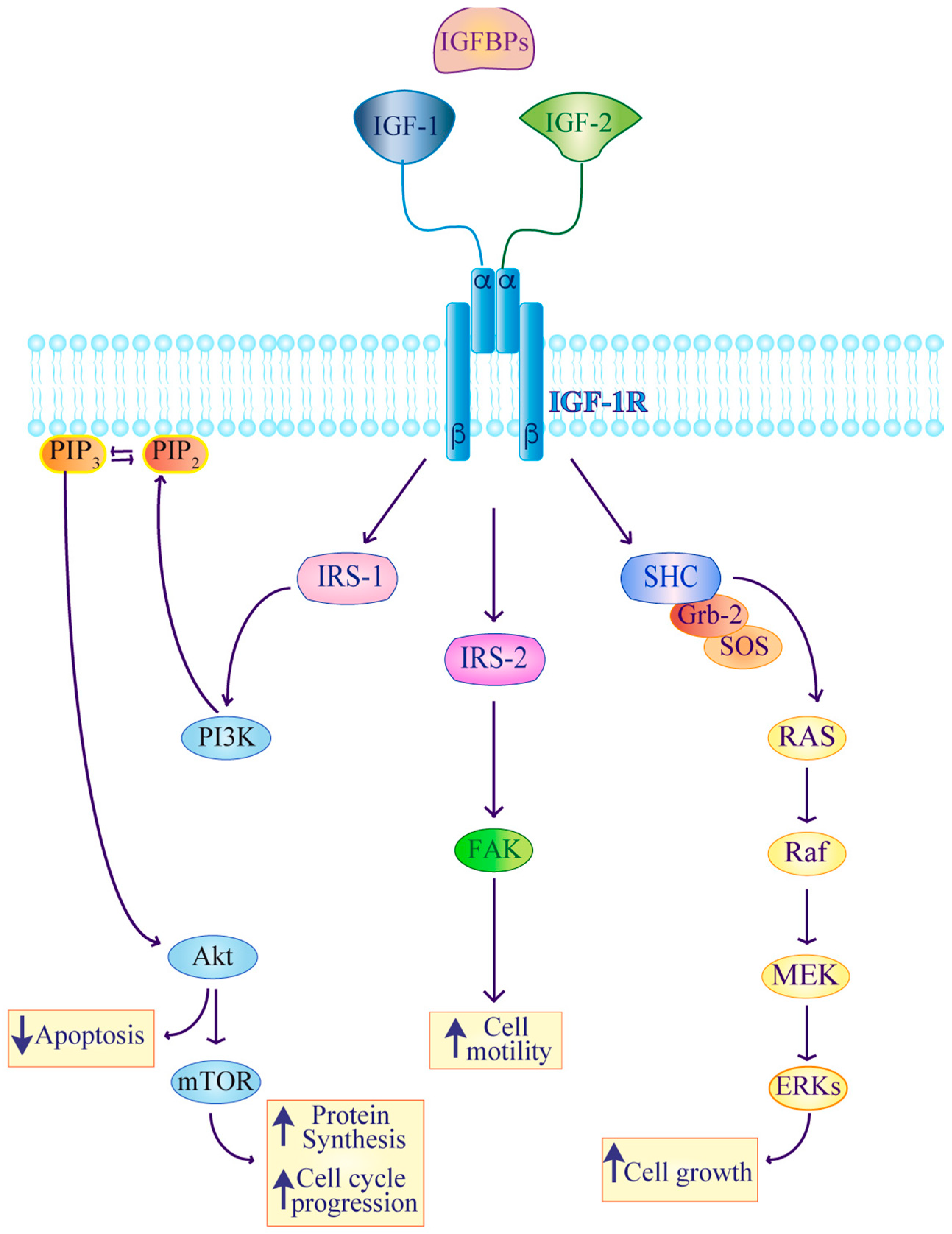
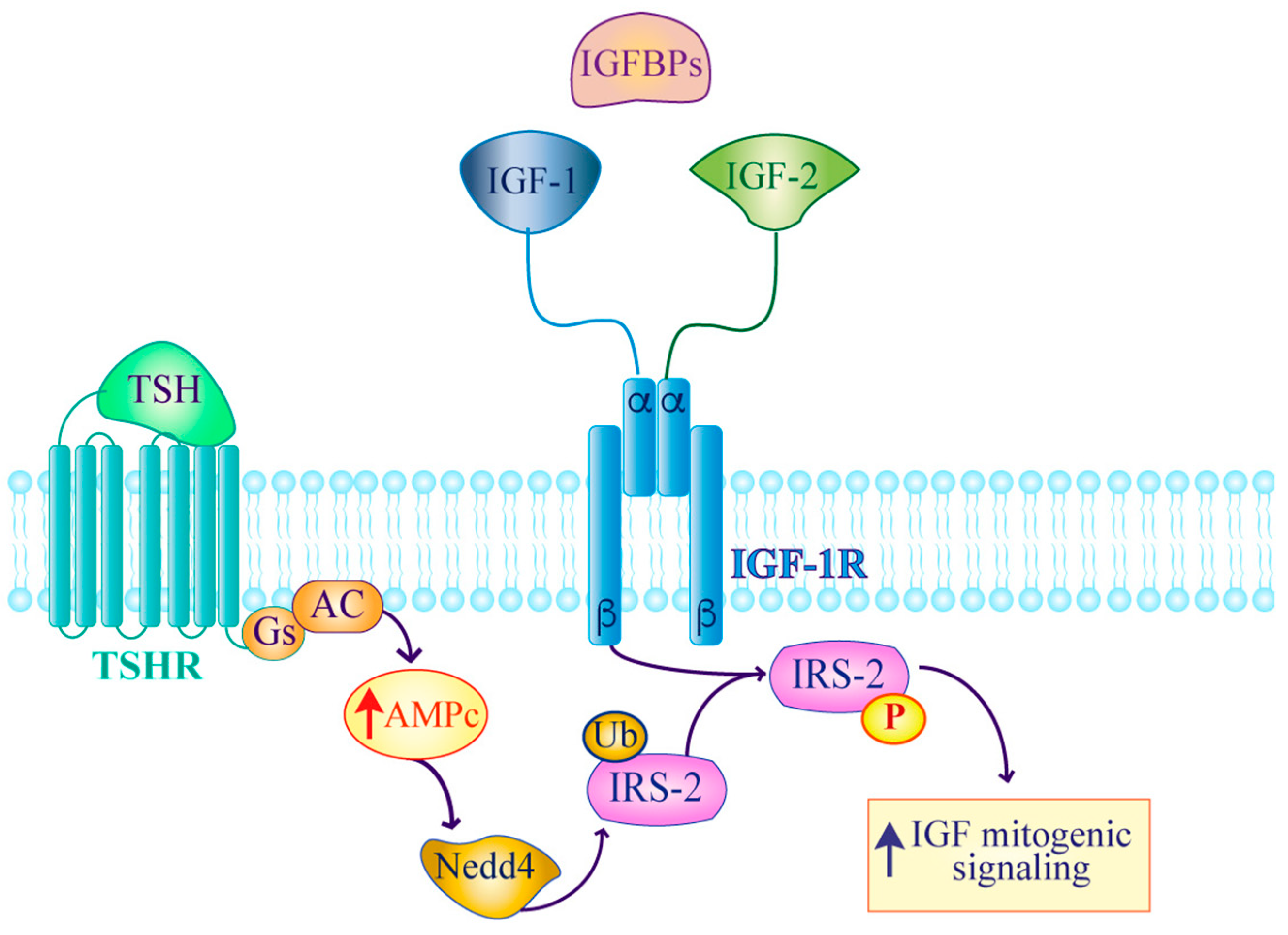
2. IGF Signaling in Thyroid Cancer
3. Targeting IGF Signaling in Thyroid Cancer
3.1. IGF-IR Direct Inhibitors
3.1.1. IGF-IRmAbs
AVE1642
Cixutumumab (IMAC-A12)
Ganitumab (AMG-479)
3.1.2. IGF-IRTKIs
Linsitinib
NVP-ADW742
NVP-AEW541
3.2. Inhibitors Targeting Downstream Signaling Mediators Shared by IGF-IR/RTKs (IGF-IR/RTKs Downstream Inhibitors)
3.2.1. PI3Ki/AKTi/mTORi
Buparlisib (PI3Ki)
Ipatasertib (AKTi)
Everolimus (mTORi)
Sirolimus (mTORi)
Temsirolimus (mTORi)
3.2.2. MEKi
Binimetinib and Pimasertib
Selumetinib
Trametinib
3.2.3. FAKi
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ATC | Anaplastic Thyroid Cancer |
| AKT | V-Akt Murine Thymoma Viral Oncogene Homolog |
| AKTi | V-Akt Murine Thymoma Viral Oncogene Homolog inihibitors |
| ALK | Anaplastic Lymphoma Kinase |
| DDR1 | Discoidin Domain Receptor Tyrosine Kinase 1 |
| DTC | Differentiated Thyroid Cancer |
| FAK | Focal Adhesion Kinase |
| FAKi | Focal Adhesion Kinase inhibitors |
| FTC | Follicular Thyroid Cancer |
| EGFR | Epidermal Growth Factor Receptor |
| ERK | Extracellular Signal-Regulated Kinase |
| IGF-1 | Insulin-like growth ligand 1 |
| IGF-2 | Insulin-like growth ligand 2 |
| IGF-IR | Insulin-like growth factor receptor I |
| IGF-IIR | Insulin-like growth factor receptor II |
| IGFBP | Insulin-like growth factor binding protein |
| IRA | Insulin receptor isoform A |
| IRB | Insulin receptor isoform B |
| IRS | Insulin receptor substrate |
| IGF- | Insulin-like growth factor receptor monoclonal antibodies |
| IRmAbs | Insulin-like growth factor receptor tyrosine kinase inhibitors |
| IGF-IRTKIs | Mitogen-Activated Protein Kinase |
| MEK | Mammalian Target of Rapamycin inhibitors |
| MEKi | Mitogen-Activated Protein Kinase inhibitors |
| MET | MET Proto-Oncogene, Receptor Tyrosine Kinase |
| mTOR | Mammalian Target of Rapamycin |
| MTC | Neural Precursor Cell Expressed, Developmentally Down-Regulated 4 |
| NEDD4 | Sodium Iodide Symporter |
| NIS | Platelet Derived Growth Factor Receptor |
| PDGFR | Poorly Differentiated Thyroid Cancer |
| PDTC | Phosphatidylinositol kinase |
| PI3K | Phosphatidylinositol kinase inhibitors |
| PI3Ki | Papillary Thyroid Cancer |
| PTC | Proto-Oncogene Serine/Threonine-Protein Kinase |
| RECIST | Response Evaluation Criteria in Solid Tumors |
| RTK | Receptor tyrosine kinase |
| SHC | Src Homology 2 Domain Containing) Transforming Protein 1 |
| TSH | Thyroid Stimulating Hormone |
| TKI | Tyrosine Kinase Inhibitors |
| VEGF | Vascular Endothelial Growth Factor |
| VEGFR | Vascular Endothelial Growth Factor Receptor |
References
- Bowers, L.W.; Rossi, E.L.; O’Flanagan, C.H.; deGraffenried, L.A.; Hursting, S.D. The role of the insulin/igf system in cancer: Lessons learned from clinical trials and the energy balance-cancer link. Front. Endocrinol (Lausanne) 2015, 6, 77. [Google Scholar] [CrossRef]
- Delafontaine, P.; Song, Y.H.; Li, Y. Expression, regulation, and function of igf-1, igf-1r, and igf-1 binding proteins in blood vessels. Arterioscl. Throm. Vas. 2004, 24, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Allard, J.B.; Duan, C. Igf-binding proteins: Why do they exist and why are there so many? Front. Endocrinol. (Lausanne) 2018, 9, 117. [Google Scholar] [CrossRef] [PubMed]
- Brahmkhatri, V.P.; Prasanna, C.; Atreya, H.S. Insulin-like growth factor system in cancer: Novel targeted therapies. Biomed. Res. Int. 2015, 2015, 538019. [Google Scholar] [CrossRef] [PubMed]
- Pollak, M. The insulin and insulin-like growth factor receptor family in neoplasia: An update. Nat. Rev. Cancer 2012, 12, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.; Petnga, W.; Macaulay, V.M.; Weyer-Czernilofsky, U.; Bogenrieder, T. Insulin-like growth factor (igf) pathway targeting in cancer: Role of the igf axis and opportunities for future combination studies. Target. Oncol. 2017, 12, 571–597. [Google Scholar] [CrossRef]
- Baxter, R.C. Igf binding proteins in cancer: Mechanistic and clinical insights. Nat. Rev. Cancer 2014, 14, 329–341. [Google Scholar] [CrossRef]
- Li, R.; Pourpak, A.; Morris, S.W. Inhibition of the insulin-like growth factor-1 receptor (igf1r) tyrosine kinase as a novel cancer therapy approach. J. Med. Chem. 2009, 52, 4981–5004. [Google Scholar] [CrossRef]
- Malaguarnera, R.; Belfiore, A. The insulin receptor: A new target for cancer therapy. Front. Endocrinol. (Lausanne) 2011, 2, 93. [Google Scholar] [CrossRef]
- Vella, V.; Malaguarnera, R. The emerging role of insulin receptor isoforms in thyroid cancer: Clinical implications and new perspectives. Int. J. Mol. Sci. 2018, 19, 3814. [Google Scholar] [CrossRef]
- Crudden, C.; Girnita, A.; Girnita, L. Targeting the igf-1r: The tale of the tortoise and the hare. Front. Endocrinol. (Lausanne) 2015, 6, 64. [Google Scholar] [CrossRef] [PubMed]
- Ochnik, A.M.; Baxter, R.C. Combination therapy approaches to target insulin-like growth factor receptor signaling in breast cancer. Endocr. Relat. Cancer 2016, 23, R513–R536. [Google Scholar] [CrossRef] [PubMed]
- Sciacca, L.; Vella, V.; Frittitta, L.; Tumminia, A.; Manzella, L.; Squatrito, S.; Belfiore, A.; Vigneri, R. Long-acting insulin analogs and cancer. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Ciampolillo, A.; De Tullio, C.; Giorgino, F. The igf-i/igf-i receptor pathway: Implications in the pathophysiology of thyroid cancer. Curr. Med. Chem. 2005, 12, 2881–2891. [Google Scholar] [CrossRef]
- Vigneri, P.G.; Tirro, E.; Pennisi, M.S.; Massimino, M.; Stella, S.; Romano, C.; Manzella, L. The insulin/igf system in colorectal cancer development and resistance to therapy. Front. Oncol. 2015, 5, 230. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Borkhuu, O.; Bao, W.; Yang, Y.T. Signaling pathways in thyroid cancer and their therapeutic implications. J. Clin. Med. Res. 2016, 8, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Manzella, L.; Stella, S.; Pennisi, M.S.; Tirro, E.; Massimino, M.; Romano, C.; Puma, A.; Tavarelli, M.; Vigneri, P. New insights in thyroid cancer and p53 family proteins. Int. J. Mol. Sci. 2017, 18, 1325. [Google Scholar] [CrossRef]
- Massimino, M.; Vigneri, P.; Fallica, M.; Fidilio, A.; Aloisi, A.; Frasca, F.; Manzella, L. Irf5 promotes the proliferation of human thyroid cancer cells. Mol. Cancer 2012, 11, 21. [Google Scholar] [CrossRef]
- Vella, V.; Puppin, C.; Damante, G.; Vigneri, R.; Sanfilippo, M.; Vigneri, P.; Tell, G.; Frasca, F. Deltanp73alpha inhibits pten expression in thyroid cancer cells. Int. J. Cancer 2009, 124, 2539–2548. [Google Scholar] [CrossRef]
- Vigneri, R.; Malandrino, P.; Giani, F.; Russo, M.; Vigneri, P. Heavy metals in the volcanic environment and thyroid cancer. Mol. Cell. Endocrinol. 2017, 457, 73–80. [Google Scholar] [CrossRef]
- Massimino, M.; Tirro, E.; Stella, S.; Frasca, F.; Vella, V.; Sciacca, L.; Pennisi, M.S.; Vitale, S.R.; Puma, A.; Romano, C.; et al. Effect of combined epigenetic treatments and ectopic nis expression on undifferentiated thyroid cancer cells. Anticancer Res. 2018, 38, 6653–6662. [Google Scholar] [CrossRef] [PubMed]
- Grimm, D. Current knowledge in thyroid cancer-from bench to bedside. Int. J. Mol. Sci. 2017, 18, 1529. [Google Scholar] [CrossRef] [PubMed]
- Vella, V.; Sciacca, L.; Pandini, G.; Mineo, R.; Squatrito, S.; Vigneri, R.; Belfiore, A. The igf system in thyroid cancer: New concepts. Mol. Pathol. 2001, 54, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Van Keymeulen, A.; Golstein, J.; Fusco, A.; Dumont, J.E.; Roger, P.P. Regulation of thyroid cell proliferation by tsh and other factors: A critical evaluation of in vitro models. Endocr. Rev. 2001, 22, 631–656. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, R.; Morcavallo, A.; Belfiore, A. The insulin and igf-i pathway in endocrine glands carcinogenesis. J. Oncol. 2012, 2012, 635614. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Qiang, W.; Shi, J.; Lv, S.Q.; Ji, M.J.; Shi, B.Y. Expression and significance of igf-1 and igf-1r in thyroid nodules. Endocrine 2013, 44, 158–164. [Google Scholar] [CrossRef]
- Belfiore, A.; Pandini, G.; Vella, V.; Squatrito, S.; Vigneri, R. Insulin/igf-i hybrid receptors play a major role in igf-i signaling in thyroid cancer. Biochimie 1999, 81, 403–407. [Google Scholar] [CrossRef]
- Schmidt, J.A.; Allen, N.E.; Almquist, M.; Franceschi, S.; Rinaldi, S.; Tipper, S.J.; Tsilidis, K.K.; Weiderpass, E.; Overvad, K.; Tjonneland, A.; et al. Insulin-like growth factor-i and risk of differentiated thyroid carcinoma in the european prospective investigation into cancer and nutrition. Cancer Epidemiol. Biomarkers Prev. 2014, 23, 976–985. [Google Scholar] [CrossRef]
- Firth, S.M.; Baxter, R.C. Cellular actions of the insulin-like growth factor binding proteins. Endocr. Rev. 2002, 23, 824–854. [Google Scholar] [CrossRef]
- LeRoith, D.; Roberts, C.T., Jr. The insulin-like growth factor system and cancer. Cancer Lett. 2003, 195, 127–137. [Google Scholar] [CrossRef] [Green Version]
- Vigneri, R.; Malandrino, P.; Vigneri, P. The changing epidemiology of thyroid cancer: Why is incidence increasing? Curr. Opin. Oncol. 2015, 27, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Modric, T.; Rajkumar, K.; Murphy, L.J. Thyroid gland function and growth in igf binding protein-1 transgenic mice. Eur. J. Endocrinol. 1999, 141, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Fakhruddin, N.; Jabbour, M.; Novy, M.; Tamim, H.; Bahmad, H.; Farhat, F.; Zaatari, G.; Aridi, T.; Kriegshauser, G.; Oberkanins, C.; et al. Braf and nras mutations in papillary thyroid carcinoma and concordance in braf mutations between primary and corresponding lymph node metastases. Sci. Rep. 2017, 7, 4666. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P. The twentieth century struggle to decipher insulin signalling. Nat. Rev. Mol. Cell. Biol. 2006, 7, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, C.M.; Emanuelli, B.; Kahn, C.R. Critical nodes in signalling pathways: Insights into insulin action. Nat. Rev. Mol. Cell. Biol. 2006, 7, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Averous, J.; Fonseca, B.D.; Proud, C.G. Regulation of cyclin d1 expression by mtorc1 signaling requires eukaryotic initiation factor 4e-binding protein 1. Oncogene 2008, 27, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, P.T.; Hay, N. The two torcs and akt. Dev. Cell. 2007, 12, 487–502. [Google Scholar] [CrossRef]
- Chitnis, M.M.; Yuen, J.S.; Protheroe, A.S.; Pollak, M.; Macaulay, V.M. The type 1 insulin-like growth factor receptor pathway. Clin. Cancer Res. 2008, 14, 6364–6370. [Google Scholar] [CrossRef]
- Brewer, C.; Yeager, N.; Di Cristofano, A. Thyroid-stimulating hormone initiated proliferative signals converge in vivo on the mtor kinase without activating akt. Cancer Res. 2007, 67, 8002–8006. [Google Scholar] [CrossRef]
- Fiore, E.; Rago, T.; Provenzale, M.A.; Scutari, M.; Ugolini, C.; Basolo, F.; Di Coscio, G.; Berti, P.; Grasso, L.; Elisei, R.; et al. Lower levels of tsh are associated with a lower risk of papillary thyroid cancer in patients with thyroid nodular disease: Thyroid autonomy may play a protective role. Endocr. Relat. Cancer 2009, 16, 1251–1260. [Google Scholar] [CrossRef]
- Morgan, S.J.; Neumann, S.; Marcus-Samuels, B.; Gershengorn, M.C. Thyrotropin and insulin-like growth factor 1 receptor crosstalk upregulates sodium-iodide symporter expression in primary cultures of human thyrocytes. Thyroid 2016, 26, 1794–1803. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, T.; Yoshihara, H.; Furuta, H.; Kamei, H.; Hakuno, F.; Luan, J.; Duan, C.; Saeki, Y.; Tanaka, K.; Iemura, S.; et al. Nedd4-induced monoubiquitination of irs-2 enhances igf signalling and mitogenic activity. Nat. Commun. 2015, 6, 6780. [Google Scholar] [CrossRef] [PubMed]
- Herkert, B.; Kauffmann, A.; Molle, S.; Schnell, C.; Ferrat, T.; Voshol, H.; Juengert, J.; Erasimus, H.; Marszalek, G.; Kazic-Legueux, M.; et al. Maximizing the efficacy of mapk-targeted treatment in ptenlof/brafmut melanoma through pi3k and igf1r inhibition. Cancer Res. 2016, 76, 390–402. [Google Scholar] [CrossRef] [PubMed]
- Chakravarti, A.; Loeffler, J.S.; Dyson, N.J. Insulin-like growth factor receptor i mediates resistance to anti-epidermal growth factor receptor therapy in primary human glioblastoma cells through continued activation of phosphoinositide 3-kinase signaling. Cancer Res. 2002, 62, 200–207. [Google Scholar] [PubMed]
- Liu, C.; Zhang, Z.; Tang, H.; Jiang, Z.; You, L.; Liao, Y. Crosstalk between igf-1r and other tumor promoting pathways. Curr. Pharm. Des. 2014, 20, 2912–2921. [Google Scholar] [CrossRef] [PubMed]
- Vella, V.; Malaguarnera, R.; Nicolosi, M.L.; Palladino, C.; Spoleti, C.; Massimino, M.; Vigneri, P.; Purrello, M.; Ragusa, M.; Morrione, A.; et al. Discoidin domain receptor 1 modulates insulin receptor signaling and biological responses in breast cancer cells. Oncotarget 2017, 8, 43248–43270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vella, V.; Nicolosi, M.L.; Cantafio, P.; Massimino, M.; Lappano, R.; Vigneri, P.; Ciuni, R.; Gangemi, P.; Morrione, A.; Malaguarnera, R.; et al. Ddr1 regulates thyroid cancer cell differentiation via igf-2/ir-a autocrine signaling loop. Endocr. Relat. Cancer 2019, 26, 197–214. [Google Scholar] [CrossRef]
- Guigon, C.J.; Fozzatti, L.; Lu, C.; Willingham, M.C.; Cheng, S.Y. Inhibition of mtorc1 signaling reduces tumor growth but does not prevent cancer progression in a mouse model of thyroid cancer. Carcinogenesis 2010, 31, 1284–1291. [Google Scholar] [CrossRef]
- Laursen, R.; Wehland, M.; Kopp, S.; Pietsch, J.; Infanger, M.; Grosse, J.; Grimm, D. Effects and role of multikinase inhibitors in thyroid cancer. Curr. Pharm. Des. 2016, 22, 5915–5926. [Google Scholar] [CrossRef]
- Montero-Conde, C.; Ruiz-Llorente, S.; Dominguez, J.M.; Knauf, J.A.; Viale, A.; Sherman, E.J.; Ryder, M.; Ghossein, R.A.; Rosen, N.; Fagin, J.A. Relief of feedback inhibition of her3 transcription by raf and mek inhibitors attenuates their antitumor effects in braf-mutant thyroid carcinomas. Cancer Dis. 2013, 3, 520–533. [Google Scholar] [CrossRef]
- Naoum, G.E.; Morkos, M.; Kim, B.; Arafat, W. Novel targeted therapies and immunotherapy for advanced thyroid cancers. Mol. Cancer 2018, 17, 51. [Google Scholar] [CrossRef] [PubMed]
- Arcaro, A. Targeting the insulin-like growth factor-1 receptor in human cancer. Front. Pharm. 2013, 4, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Denduluri, S.K.; Idowu, O.; Wang, Z.; Liao, Z.; Yan, Z.; Mohammed, M.K.; Ye, J.; Wei, Q.; Wang, J.; Zhao, L.; et al. Insulin-like growth factor (igf) signaling in tumorigenesis and the development of cancer drug resistance. Genes Dis. 2015, 2, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Janssen, J.A.; Varewijck, A.J. Igf-ir targeted therapy: Past, present and future. Front. Endocrinol. (Lausanne) 2014, 5, 224. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.R.; Fridlyand, J.; Yan, Y.; Penuel, E.; Burton, L.; Chan, E.; Peng, J.; Lin, E.; Wang, Y.; Sosman, J.; et al. Widespread potential for growth-factor-driven resistance to anticancer kinase inhibitors. Nature 2012, 487, 505–509. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Awasthi, N.; Zhang, C.; Ruan, W.; Schwarz, M.A.; Schwarz, R.E. Bms-754807, a small-molecule inhibitor of insulin-like growth factor-1 receptor/insulin receptor, enhances gemcitabine response in pancreatic cancer. Mol. Cancer. Ther. 2012, 11, 2644–2653. [Google Scholar] [CrossRef] [PubMed]
- Chitnis, M.M.; Lodhia, K.A.; Aleksic, T.; Gao, S.; Protheroe, A.S.; Macaulay, V.M. Igf-1r inhibition enhances radiosensitivity and delays double-strand break repair by both non-homologous end-joining and homologous recombination. Oncogene 2014, 33, 5262–5273. [Google Scholar] [CrossRef]
- Ferte, C.; Loriot, Y.; Clemenson, C.; Commo, F.; Gombos, A.; Bibault, J.E.; Fumagalli, I.; Hamama, S.; Auger, N.; Lahon, B.; et al. Igf-1r targeting increases the antitumor effects of DNA-damaging agents in sclc model: An opportunity to increase the efficacy of standard therapy. Mol. Cancer Ther. 2013, 12, 1213–1222. [Google Scholar] [CrossRef]
- Ireland, L.; Santos, A.; Ahmed, M.S.; Rainer, C.; Nielsen, S.R.; Quaranta, V.; Weyer-Czernilofsky, U.; Engle, D.D.; Perez-Mancera, P.A.; Coupland, S.E.; et al. Chemoresistance in pancreatic cancer is driven by stroma-derived insulin-like growth factors. Cancer Res. 2016, 76, 6851–6863. [Google Scholar] [CrossRef]
- Davis, S.L.; Eckhardt, S.G.; Diamond, J.R.; Messersmith, W.A.; Dasari, A.; Weekes, C.D.; Lieu, C.H.; Kane, M.; Choon Tan, A.; Pitts, T.M.; et al. A phase i dose-escalation study of linsitinib (osi-906), a small-molecule dual insulin-like growth factor-1 receptor/insulin receptor kinase inhibitor, in combination with irinotecan in patients with advanced cancer. Oncologist 2018, 23, 1409–e1140. [Google Scholar] [CrossRef]
- Macaulay, V.M.; Middleton, M.R.; Protheroe, A.S.; Tolcher, A.; Dieras, V.; Sessa, C.; Bahleda, R.; Blay, J.Y.; LoRusso, P.; Mery-Mignard, D.; et al. Phase i study of humanized monoclonal antibody ave1642 directed against the type 1 insulin-like growth factor receptor (igf-1r), administered in combination with anticancer therapies to patients with advanced solid tumors. Ann. Oncol. 2013, 24, 784–791. [Google Scholar] [CrossRef]
- Wilky, B.A.; Rudek, M.A.; Ahmed, S.; Laheru, D.A.; Cosgrove, D.; Donehower, R.C.; Nelkin, B.; Ball, D.; Doyle, L.A.; Chen, H.; et al. A phase i trial of vertical inhibition of igf signalling using cixutumumab, an anti-igf-1r antibody, and selumetinib, an mek 1/2 inhibitor, in advanced solid tumours. Br. J. Cancer 2015, 112, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.S.; Puzanov, I.; Friberg, G.; Chan, E.; Hwang, Y.C.; Deng, H.; Gilbert, J.; Mahalingam, D.; McCaffery, I.; Michael, S.A.; et al. Safety and pharmacokinetics of ganitumab (amg 479) combined with sorafenib, panitumumab, erlotinib, or gemcitabine in patients with advanced solid tumors. Clin. Cancer Res. 2012, 18, 3414–3427. [Google Scholar] [CrossRef] [PubMed]
- Rodon, J.; Brana, I.; Siu, L.L.; De Jonge, M.J.; Homji, N.; Mills, D.; Di Tomaso, E.; Sarr, C.; Trandafir, L.; Massacesi, C.; et al. Phase I dose-escalation and –expansion study of buparlisib (BKM120), an oral pan-Class I PI3K inhibitor, in patients with advanced solid tumors. Invest. New Drugs. 2014, 32, 670–681. [Google Scholar] [CrossRef] [PubMed]
- Borson-Chazot, F.; Dantony, E.; Illouz, F.; Lopez, J.; Niccoli, P.; Wasserman, J.; Do Cao, C.; Leboulleux, S.; Klein, M.; Tabarin, A.; et al. The TUTHYREF Network. Thyroic Cancer Nodules 2018, 28, 1174–1179. [Google Scholar]
- Saura, C.; Roda, D.; Rosello, S.; Oliveira, M.; Macarulla, T.; Perez-Fidalgo, J.A.; Morales-Barrera, R.; Sanchis-Garcia, J.M.; Musib, L.; Budha, N.; et al. A first-in-human phase i study of the atp-competitive akt inhibitor ipatasertib demonstrates robust and safe targeting of akt in patients with solid tumors. Cancer Discov. 2017, 7, 102–113. [Google Scholar] [CrossRef]
- Jones, S.F.; Siu, L.L.; Bendell, J.C.; Cleary, J.M.; Razak, A.R.; Infante, J.R.; Pandya, S.S.; Bedard, P.L.; Pierce, K.J.; Houk, B.; et al. A phase i study of vs-6063, a second-generation focal adhesion kinase inhibitor, in patients with advanced solid tumors. Invest. New Drugs 2015, 33, 1100–1107. [Google Scholar] [CrossRef]
- Soria, J.C.; Gan, H.K.; Blagden, S.P.; Plummer, R.; Arkenau, H.T.; Ranson, M.; Evans, T.R.; Zalcman, G.; Bahleda, R.; Hollebecque, A.; et al. A phase i, pharmacokinetic and pharmacodynamic study of gsk2256098, a focal adhesion kinase inhibitor, in patients with advanced solid tumors. Ann. Oncol. 2016, 27, 2268–2274. [Google Scholar] [CrossRef]
- Watanabe, K.; Otsu, S.; Hirashima, Y.; Morinaga, R.; Nishikawa, K.; Hisamatsu, Y.; Shimokata, T.; Inada-Inoue, M.; Shibata, T.; Takeuchi, H.; et al. A phase i study of binimetinib (mek162) in japanese patients with advanced solid tumors. Cancer Chemother. Pharmacol. 2016, 77, 1157–1164. [Google Scholar] [CrossRef]
- Mita, M.; Fu, S.; Piha-Paul, S.A.; Janku, F.; Mita, A.; Natale, R.; Guo, W.; Zhao, C.; Kurzrock, R.; Naing, A. Phase i trial of mek 1/2 inhibitor pimasertib combined with mtor inhibitor temsirolimus in patients with advanced solid tumors. Invest. New Drugs 2017, 35, 616–626. [Google Scholar] [CrossRef]
- Infante, J.R.; Cohen, R.B.; Kim, K.B.; Burris, H.A., 3rd; Curt, G.; Emeribe, U.; Clemett, D.; Tomkinson, H.K.; LoRusso, P.M. A phase i dose-escalation study of selumetinib in combination with erlotinib or temsirolimus in patients with advanced solid tumors. Invest. New Drugs 2017, 35, 576–588. [Google Scholar] [CrossRef]
- Hayes, D.N.; Lucas, A.S.; Tanvetyanon, T.; Krzyzanowska, M.K.; Chung, C.H.; Murphy, B.A.; Gilbert, J.; Mehra, R.; Moore, D.T.; Sheikh, A.; et al. Phase ii efficacy and pharmacogenomic study of selumetinib (azd6244; arry-142886) in iodine-131 refractory papillary thyroid carcinoma with or without follicular elements. Clin. Cancer Res. 2012, 18, 2056–2065. [Google Scholar] [CrossRef]
- Infante, J.R.; Fecher, L.A.; Falchook, G.S.; Nallapareddy, S.; Gordon, M.S.; Becerra, C.; DeMarini, D.J.; Cox, D.S.; Xu, Y.; Morris, S.R.; et al. Safety, pharmacokinetic, pharmacodynamic, and efficacy data for the oral mek inhibitor trametinib: A phase 1 dose-escalation trial. Lancet Oncol. 2012, 13, 773–781. [Google Scholar] [CrossRef]
- Tolcher, A.W.; Bendell, J.C.; Papadopoulos, K.P.; Burris, H.A., 3rd; Patnaik, A.; Jones, S.F.; Rasco, D.; Cox, D.S.; Durante, M.; Bellew, K.M.; et al. A phase ib trial of the oral mek inhibitor trametinib (gsk1120212) in combination with everolimus in patients with advanced solid tumors. Ann. Oncol. 2015, 26, 58–64. [Google Scholar] [CrossRef]
- Harris, E.J.; Hanna, G.J.; Chau, N.; Rabinowits, G.; Haddad, R.; Margalit, D.N.; Schoenfeld, J.; Tishler, R.B.; Barletta, J.A.; Nehs, M.; et al. Everolimus in anaplastic thyroid cancer: A case series. Front. Oncol 2019, 9, 106. [Google Scholar] [CrossRef] [PubMed]
- Fury, M.G.; Sherman, E.; Haque, S.; Korte, S.; Lisa, D.; Shen, R.; Wu, N.; Pfister, D. A phase i study of daily everolimus plus low-dose weekly cisplatin for patients with advanced solid tumors. Cancer Chemother. Pharmacol. 2012, 69, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.M.; Chang, H.; Yoon, M.J.; Hong, Y.K.; Kim, H.; Chung, W.Y.; Park, C.S.; Nam, K.H.; Kang, S.W.; Kim, M.K.; et al. A multicenter, phase ii trial of everolimus in locally advanced or metastatic thyroid cancer of all histologic subtypes. Ann. Oncol. 2013, 24, 3089–3094. [Google Scholar] [CrossRef] [PubMed]
- Schneider, T.C.; de Wit, D.; Links, T.P.; van Erp, N.P.; van der Hoeven, J.J.; Gelderblom, H.; van Wezel, T.; van Eijk, R.; Morreau, H.; Guchelaar, H.J.; et al. Beneficial effects of the mtor inhibitor everolimus in patients with advanced medullary thyroid carcinoma: Subgroup results of a phase ii trial. Int. J. Endocrinol. 2015, 2015, 348124. [Google Scholar] [CrossRef] [PubMed]
- Sherman, E.J.; Dunn, L.A.; Ho, A.L.; Baxi, S.S.; Ghossein, R.A.; Fury, M.G.; Haque, S.; Sima, C.S.; Cullen, G.; Fagin, J.A.; et al. Phase 2 study evaluating the combination of sorafenib and temsirolimus in the treatment of radioactive iodine-refractory thyroid cancer. Cancer 2017, 123, 4114–4121. [Google Scholar] [CrossRef] [Green Version]
- Schneider, T.C.; de Wit, D.; Links, T.P.; van Erp, N.P.; van der Hoeven, J.J.; Gelderblom, H.; Roozen, I.C.; Bos, M.; Corver, W.E.; van Wezel, T.; et al. Everolimus in patients with advanced follicular-derived thyroid cancer: Results of a phase ii clinical trial. J. Clin. Endocrinol. Metab. 2017, 102, 698–707. [Google Scholar] [CrossRef] [PubMed]
- Chiu, J.W.; Hotte, S.J.; Kollmannsberger, C.K.; Renouf, D.J.; Cescon, D.W.; Hedley, D.; Chow, S.; Moscow, J.; Chen, Z.; Perry, M.; et al. A phase i trial of ang1/2-tie2 inhibitor trebaninib (amg386) and temsirolimus in advanced solid tumors (pjc008/ncimusical sharp9041). Invest. New Drugs 2016, 34, 104–111. [Google Scholar] [CrossRef]
- Wang, Z.; Chakravarty, G.; Kim, S.; Yazici, Y.D.; Younes, M.N.; Jasser, S.A.; Santillan, A.A.; Bucana, C.D.; El-Naggar, A.K.; Myers, J.N. Growth-inhibitory effects of human anti-insulin-like growth factor-i receptor antibody (a12) in an orthotopic nude mouse model of anaplastic thyroid carcinoma. Clin. Cancer Res. 2006, 12, 4755–4765. [Google Scholar] [CrossRef]
- Langer, C.J.; Novello, S.; Park, K.; Krzakowski, M.; Karp, D.D.; Mok, T.; Benner, R.J.; Scranton, J.R.; Olszanski, A.J.; Jassem, J. Randomized, phase iii trial of first-line figitumumab in combination with paclitaxel and carboplatin versus paclitaxel and carboplatin alone in patients with advanced non-small-cell lung cancer. J. Clin. Oncol. 2014, 32, 2059–2066. [Google Scholar] [CrossRef]
- Ma, H.; Zhang, T.; Shen, H.; Cao, H.; Du, J. The adverse events profile of anti-igf-1r monoclonal antibodies in cancer therapy. Br. J. Clin. Pharmacol. 2014, 77, 917–928. [Google Scholar] [CrossRef] [PubMed]
- Mitsiades, C.S.; Mitsiades, N.S.; McMullan, C.J.; Poulaki, V.; Shringarpure, R.; Akiyama, M.; Hideshima, T.; Chauhan, D.; Joseph, M.; Libermann, T.A.; et al. Inhibition of the insulin-like growth factor receptor-1 tyrosine kinase activity as a therapeutic strategy for multiple myeloma, other hematologic malignancies, and solid tumors. Cancer Cell 2004, 5, 221–230. [Google Scholar] [CrossRef] [Green Version]
- Chakravarty, G.; Mondal, D. Insulin-Like Growth Factor Receptor Signaling in Thyroid Cancers: Clinical Implications and Therapeutic Potential; IntechOpen: London, UK, 2012. [Google Scholar]
- Licitra, L.; Locati, L.D.; Greco, A.; Granata, R.; Bossi, P. Multikinase inhibitors in thyroid cancer. Eur. J. Cancer 2010, 46, 1012–1018. [Google Scholar] [CrossRef]
- Liu, Z.; Hou, P.; Ji, M.; Guan, H.; Studeman, K.; Jensen, K.; Vasko, V.; El-Naggar, A.K.; Xing, M. Highly prevalent genetic alterations in receptor tyrosine kinases and phosphatidylinositol 3-kinase/akt and mitogen-activated protein kinase pathways in anaplastic and follicular thyroid cancers. J. Clin. Endocrinol. Metab. 2008, 93, 3106–3116. [Google Scholar] [CrossRef] [PubMed]
- Bozorg-Ghalati, F.; Hedayati, M.; Dianatpour, M.; Azizi, F.; Mosaffa, N.; Mehrabani, D. Effects of a phosphoinositide-3-kinase inhibitor on anaplastic thyroid cancer stem cells. Asian. Pac. J. Cancer Prev. 2017, 18, 2287–2291. [Google Scholar] [PubMed]
- Nozhat, Z.; Hedayati, M. Pi3k/akt pathway and its mediators in thyroid carcinomas. Mol. Diagn. Ther. 2016, 20, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, M.; Chung, Y.J.; Saji, M.; Ringel, M.D. Akt in thyroid tumorigenesis and progression. Endocrinology 2007, 148, 942–947. [Google Scholar] [CrossRef] [PubMed]
- Petrulea, M.S.; Plantinga, T.S.; Smit, J.W.; Georgescu, C.E.; Netea-Maier, R.T. Pi3k/akt/mtor: A promising therapeutic target for non-medullary thyroid carcinoma. Cancer Treat. Rev. 2015, 41, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Manfredi, G.I.; Dicitore, A.; Gaudenzi, G.; Caraglia, M.; Persani, L.; Vitale, G. Pi3k/akt/mtor signaling in medullary thyroid cancer: A promising molecular target for cancer therapy. Endocrine 2015, 48, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Manohar, P.M.; Beesley, L.J.; Taylor, J.M.; Hesseltine, E.; Haymart, M.R.; Esfandiari, N.H.; Hanauer, D.A.; Worden, F.P. Retrospective study of sirolimus and cyclophosphamide in patients with advanced differentiated thyroid cancers. J. Thyroid. Disord. Ther. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.M.; Ahn, B.C. Redifferentiation of radioiodine refractory differentiated thyroid cancer for reapplication of i-131 therapy. Front. Endocrinol. (Lausanne) 2017, 8, 260. [Google Scholar] [CrossRef] [PubMed]
- Wachter, S.; Wunderlich, A.; Greene, B.H.; Roth, S.; Elxnat, M.; Fellinger, S.A.; Verburg, F.A.; Luster, M.; Bartsch, D.K.; Di Fazio, P. Selumetinib activity in thyroid cancer cells: Modulation of sodium iodide symporter and associated mirnas. Int. J. Mol. Sci. 2018, 19, 2077. [Google Scholar] [CrossRef] [PubMed]
- Ball, D.W.; Jin, N.; Xue, P.; Bhan, S.; Ahmed, S.R.; Rosen, D.M.; Schayowitz, A.; Clark, D.P.; Nelkin, B.D. Trametinib with and without pazopanib has potent preclinical activity in thyroid cancer. Oncol. Rep. 2015, 34, 2319–2324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, D.; Xing, M. Potent inhibition of thyroid cancer cells by the mek inhibitor pd0325901 and its potentiation by suppression of the pi3k and nf-kappab pathways. Thyroid 2008, 18, 853–864. [Google Scholar] [CrossRef]
- Zhao, Y.; Adjei, A.A. The clinical development of mek inhibitors. Nat. Rev. Clin. Oncol. 2014, 11, 385–400. [Google Scholar] [CrossRef]
- Ho, A.L.; Grewal, R.K.; Leboeuf, R.; Sherman, E.J.; Pfister, D.G.; Deandreis, D.; Pentlow, K.S.; Zanzonico, P.B.; Haque, S.; Gavane, S.; et al. Selumetinib-enhanced radioiodine uptake in advanced thyroid cancer. N. Engl. J. Med. 2013, 368, 623–632. [Google Scholar] [CrossRef]
- Kessler, B.E.; Sharma, V.; Zhou, Q.; Jing, X.; Pike, L.A.; Kerege, A.A.; Sams, S.B.; Schweppe, R.E. Fak expression, not kinase activity, is a key mediator of thyroid tumorigenesis and protumorigenic processes. Mol. Cancer Res. 2016, 14, 869–882. [Google Scholar] [CrossRef]
- Tai, Y.L.; Chen, L.C.; Shen, T.L. Emerging roles of focal adhesion kinase in cancer. Biomed. Res. Int. 2015, 2015, 690690. [Google Scholar] [CrossRef] [PubMed]
- Owens, L.V.; Xu, L.; Dent, G.A.; Yang, X.; Sturge, G.C.; Craven, R.J.; Cance, W.G. Focal adhesion kinase as a marker of invasive potential in differentiated human thyroid cancer. Ann. Surg. Oncol. 1996, 3, 6. [Google Scholar] [CrossRef]
- Kim, S.J.; Park, J.W.; Yoon, J.S.; Mok, J.O.; Kim, Y.J.; Park, H.K.; Kim, C.H.; Byun, D.W.; Lee, Y.J.; Jin, S.Y.; et al. Increased expression of focal adhesion kinase in thyroid cancer: Immunohistochemical study. J. Korean Medi. Sci. 2004, 19, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Vella, V.; Pandini, G.; Sciacca, L.; Mineo, R.; Vigneri, R.; Pezzino, V.; Belfiore, A. A novel autocrine loop involving igf-ii and the insulin receptor isoform-a stimulates growth of thyroid cancer. J. Clin. Endocrinol. Metab. 2002, 87, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.S.; Banerji, U. Combine and conquer: Challenges for targeted therapy combinations in early phase trials. Nat. Rev. Clin. Oncol. 2017, 14, 57–66. [Google Scholar] [CrossRef]
- Pirosa, M.C.; Leotta, S.; Cupri, A.; Stella, S.; Martino, E.A.; Scalise, L.; Sapienza, G.; Calafiore, V.; Mauro, E.; Spadaro, A.; et al. Long-term molecular remission achieved by antibody anti-cd22 and ponatinib in a patient affected by ph’+ acute lymphoblastic leukemia relapsed after second allogeneic hematopoietic stem cell transplantation: A case report. Chemotherapy 2018, 63, 220–224. [Google Scholar] [CrossRef]
- Tirro, E.; Massimino, M.; Romano, C.; Pennisi, M.S.; Stella, S.; Vitale, S.R.; Fidilio, A.; Manzella, L.; Parrinello, N.L.; Stagno, F.; et al. Chk1 inhibition restores inotuzumab ozogamicin citotoxicity in cd22-positive cells expressing mutant p53. Front. Oncol. 2019, 9, 57. [Google Scholar] [CrossRef]
- Ghosh, D.; Nandi, S.; Bhattacharjee, S. Combination therapy to checkmate glioblastoma: Clinical challenges and advances. Clin. Transl. Med. 2018, 7, 33. [Google Scholar] [CrossRef]
- Zanardi, E.; Bregni, G.; de Braud, F.; Di Cosimo, S. Better together: Targeted combination therapies in breast cancer. Semin. Oncol. 2015, 42, 887–895. [Google Scholar] [CrossRef]

| Class of Drugs | Drug Name | Published Data | Phase | Tumor Type | N°* | Regimen |
|---|---|---|---|---|---|---|
| IGF-IRi | AVE1642 | [61] | I | Advanced Solid | 1 | AVE1642+Docetaxel |
| Cixutumumab | [62] | I | Advanced Solid | 4 | Cixutumumab + Selumetinib | |
| Ganitumab | [63] | I | Advanced Solid | 2 | Ganitumab + Sorafenib or Panitumumab | |
| PI3Ki | Buparlisib | [64] | I | Advanced Solid | 1 | Monotherapy |
| [65] | II | FTC + PDTC | 43 | Monotherapy | ||
| AKTi | Ipatasertib | [66] | I | Advanced Solid | 1 | Monotherapy |
| FAKi | VS-6063 | [67] | I | Advanced Solid | 1 | Monotherapy |
| GSK2256098 | [68] | I | Advanced Solid | 2 | Monotherapy | |
| MEKi | Binimetinib | [69] | II | Advanced Solid | 2 | Monotherapy |
| Pimasertib | [70] | I | Advanced Solid | 1 | Pimasertib+Temsirolimus | |
| Selumetinib | [71] | I | Advanced Solid | 2 | Selumetinib + Temsirolimus | |
| [72] | I | PTC | 39 | Monotherapy | ||
| Trametinib | [73] | I | Advanced Solid | 5 | Monotherapy | |
| [74] | I | Advanced Solid | 1 | Trametinib + Everolimus | ||
| mToRi | Everolimus | [75] | II | ATC | 5 | Monotherapy |
| [76] | I | Advanced Solid | 7 | Everolimus + Cisplatin | ||
| [77] | II | DTC+ATC+MTC | 40 | Monotherapy | ||
| [78] | II | MTC | 7 | Monotherapy | ||
| [79] | II | DTC+MTC | 41 | Everolimus + Sorafenib | ||
| [80] | II | DTC+ATC | 28 | Monotherapy | ||
| Temsirolimus | [81] | I | Advanced Solid | 1 | Temsirolimus + Trebananib | |
| [79] | II | DTC+ATC | 36 | Temsirolimus + Sorafenib |
| Intervention | Population | Design | Pts (n) | Primary End Point | Status | Identifier |
|---|---|---|---|---|---|---|
| CixutumumabIGF-IRmAb EverolimusmTORi Octreotide somatostatin analogue | Advanced low- or intermediate-grade neuroendocrine cancers | Nonrandomized, Open label, phase I | 27 actual | DLTs, PD, PK, SP | Completed | NCT01204476 |
| Binimetinib MEKi Capivasertib Akt Copanlisib PI3Ki TaselisibPI3Ki GSK2636771PI3Ki Trametinib MEKi | Genetic testing-directed targeted therapy in patients with advanced refractory solid tumors, lymphomas, or multiple myeloma | Nonrandomized, Open label, phase II (molecular analysis for therapy choice) | 6452 estimated | ORR | Recruiting | NCT02465060 |
| Everolimus mTORi Sorafenib MKi | Metastatic differentiated thyroid cancer progressed on Sorafenib | Nonrandomized, Open label, phase II | 40 estimated | ORR, PFS | Active, not Recruiting | NCT01263951 |
| Everolimus mTORi Sorafenib MKi | Advanced thyroid cancer naive to m-TOR inhibitors or Sorafenib | Nonrandomized, Open label, phase II | 41 actual | ORR | Active, not Recruiting | NCT01141309 |
| Everolimus mTORi Pasireotide somatostatin analogue | Radioiodine-refractory differentiated and medullary thyroid cancer | Randomized, Open label, phase II | 42 actual | ORR | Completed | NCT01270321 |
| Everolimus mTORi | Radioiodine-refractory thyroid cancer | Nonrandomized, Open label, phase II | 33 estimated | PFS | Active, not Recruiting | NCT00936858 |
| Everolimus mTORi Sorafenib MKi | Advanced radioiodine-refractory Hurthle cell thyroid cancer | Randomized, Open label, phase II | 34 estimated | PFS | Recruiting | NCT02143726 |
| Everolimus mTORi Lenvatinib MKi | Metastatic differentiated thyroid cancer progressed on Lenvatinib | Nonrandomized, Open label, phase II | 40 estimated | PFS | Recruiting | NCT03139747 |
| Everolimus mTORi Pasireotide somatostatin analogue | Advanced medullary thyroid cancer | Nonrandomized, Open label, phase II | 19 actual | PFS | Completed | NCT01625520 |
| Everolimus mTORi | Locally advanced or metastatic thyroid cancer | Nonrandomized, Open label, phase II | 40 actual | ORR | Completed | NCT01164176 |
| Everolimus mTORi Neratinib EGFRi or Neratinib EGFRi Trametinib MEKi | Advanced cancer with EGFR mutation/amplification, HER2 mutation/amplification, HER3/4 mutation or KRAS mutation | Nonrandomized, Open label, phase I | 120 estimated | DLTs | Recruiting | NCT03065387 |
| Everolimus mTORi Vatalanib VEGFi | Advanced solid tumors | Nonrandomized, Open label, phase I | 96 estimated | DLTs, SP | Completed | NCT00655655 |
| BevacizumabVEGFmAbs Temsirolimus mTORi | Advanced or metastatic malignancies or other benign diseases | Nonrandomized, Open label, phase I | 216 estimated | DLTs | Recruiting | NCT01552434 |
| Temsirolimus mTORi Vinorelbine | Unresectable or metastatic solid tumors | Nonrandomized, Open label, phase I | 19 actual | DLTs, ORR | Completed | NCT01155258 |
| Ciclophosfamide Sirolimus mTORi | Metastatic, RAI-refractory, differentiated thyroid cancer | Nonrandomized, Open label, phase II | 19 estimated | ORR | Recruiting | NCT03099356 |
| Grapefruit juice Sirolimus mTORi | Advanced malignancies | Nonrandomized, Open label, phase Ib | 41 actual | PK | Completed | NCT00375245 |
| Iodine I-131 Selumetinib MEKi | Recurrent or metastatic thyroid cancer | Randomized, Double blind, phase II | 60 estimated | ORR | Recruiting | NCT02393690 |
| Olaparib PARPi Selumetinib MEKi | Endometrial, ovarian and other solid tumors with RAS pathway alterations and ovarian tumors with resistance to PARPis | Nonrandomized, Open label, phase I | 90 estimated | DLTs | Recruiting | NCT03162627 |
| Paclitaxel Trametinib MEKi | Anaplastic thyroid cancer | Nonrandomized, Open label, early phase I | 12 estimated | PFS | Recruiting | NCT03085056 |
| Dabrafenib BRAFi Trametinib MEKi | Recurrent thyroid cancer | Randomized, Open label, phase II | 53 actual | ORR | Active, not Recruiting | NCT01723202 |
| Dabrafenib BRAFi Trametinib MEKi | Refractory metastatic differentiated thyroid cancer with RAS or BRAFV600E mutations | Nonrandomized, Open label, phase II | 87 estimated | ORR | Recruiting | NCT03244956 |
| Pazopanib MKi Trametinib MEKi | Advanced solid tumors enriched for patients with differentiated thyroid cancer, soft tissue sarcoma, and cholangiocarcinoma | Nonrandomized, Open label, phase I | 89 actual | DLTs, SP | Completed | NCT01438554 |
| RAI TrametinibMEKi | Mutant RAS or wild-type RAS/RAF, RAI-refractory recurrent and/or metastatic thyroid cancer | Nonrandomized, Open label, phase II | 35 estimated | PFS, ORR | Recruiting | NCT02152995 |
| CobimetinibMEKi | Differentiated, poorly differentiated and anaplastic thyroid carcinomas | Nonrandomized, Open label, phase II | 50 estimated | OS | Recruiting | NCT03181100 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzella, L.; Massimino, M.; Stella, S.; Tirrò, E.; Pennisi, M.S.; Martorana, F.; Motta, G.; Vitale, S.R.; Puma, A.; Romano, C.; et al. Activation of the IGF Axis in Thyroid Cancer: Implications for Tumorigenesis and Treatment. Int. J. Mol. Sci. 2019, 20, 3258. https://doi.org/10.3390/ijms20133258
Manzella L, Massimino M, Stella S, Tirrò E, Pennisi MS, Martorana F, Motta G, Vitale SR, Puma A, Romano C, et al. Activation of the IGF Axis in Thyroid Cancer: Implications for Tumorigenesis and Treatment. International Journal of Molecular Sciences. 2019; 20(13):3258. https://doi.org/10.3390/ijms20133258
Chicago/Turabian StyleManzella, Livia, Michele Massimino, Stefania Stella, Elena Tirrò, Maria Stella Pennisi, Federica Martorana, Gianmarco Motta, Silvia Rita Vitale, Adriana Puma, Chiara Romano, and et al. 2019. "Activation of the IGF Axis in Thyroid Cancer: Implications for Tumorigenesis and Treatment" International Journal of Molecular Sciences 20, no. 13: 3258. https://doi.org/10.3390/ijms20133258





