Enhanced Tolerance against a Fungal Pathogen and Insect Resistance in Transgenic Tobacco Plants Overexpressing an Endochitinase Gene from Serratia marcescens
Abstract
:1. Introduction
2. Results
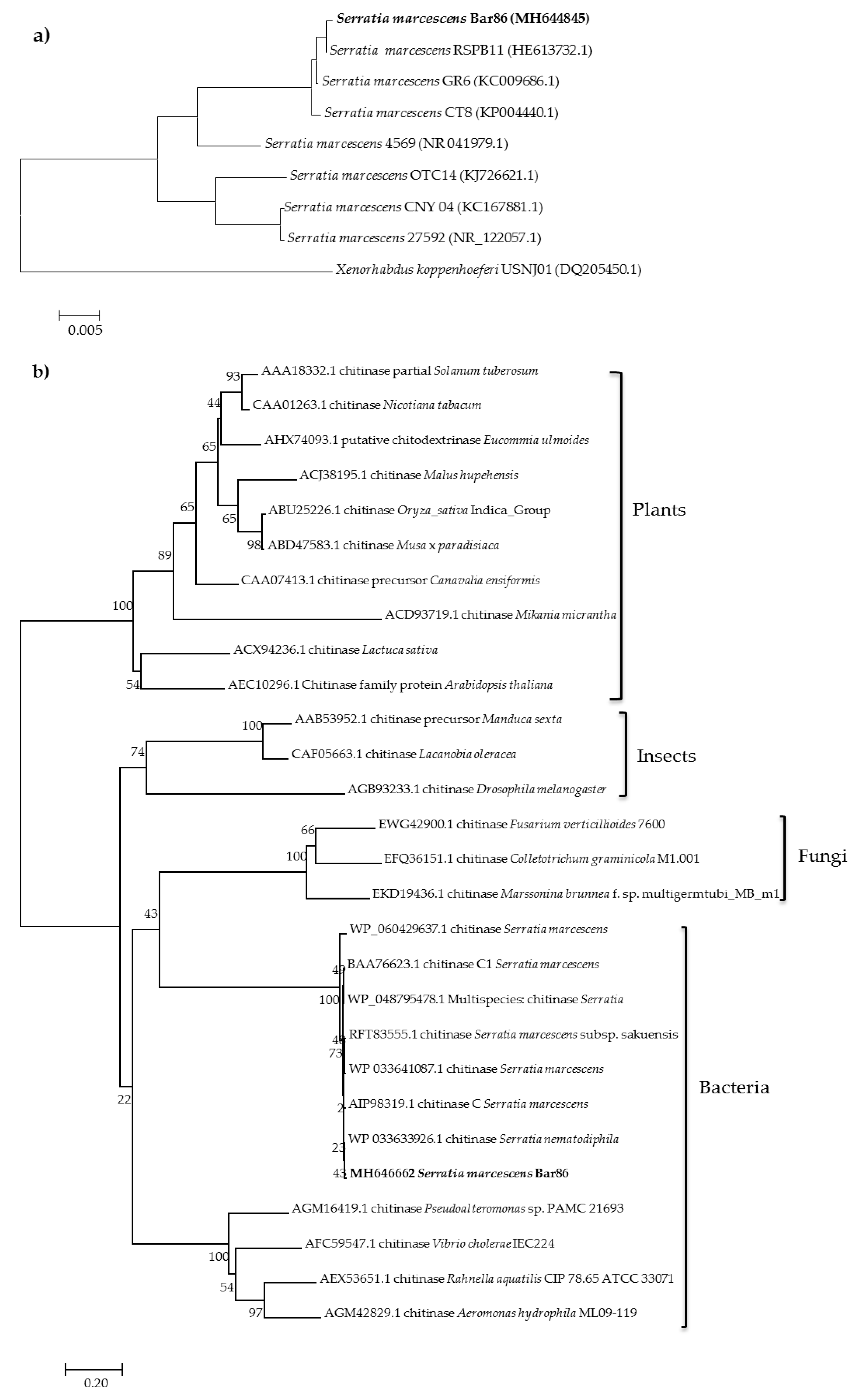
2.1. Bacteria Strain Identification and Phylogenetic Analysis
2.2. Cloning of Chitinase Genes
2.3. Tobacco Transformation and Molecular Analysis
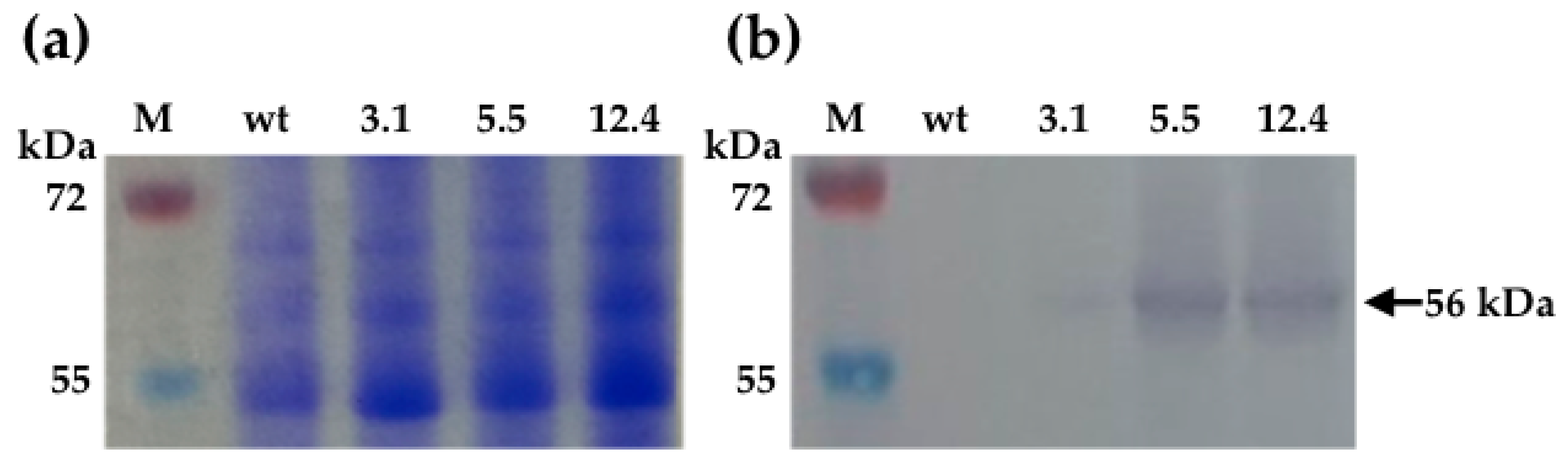
2.4. Western Blot Analysis
2.5. Endochitinase Activity
2.6. Antifungal Inhibition Activity Assays on Crude Protein Extracts
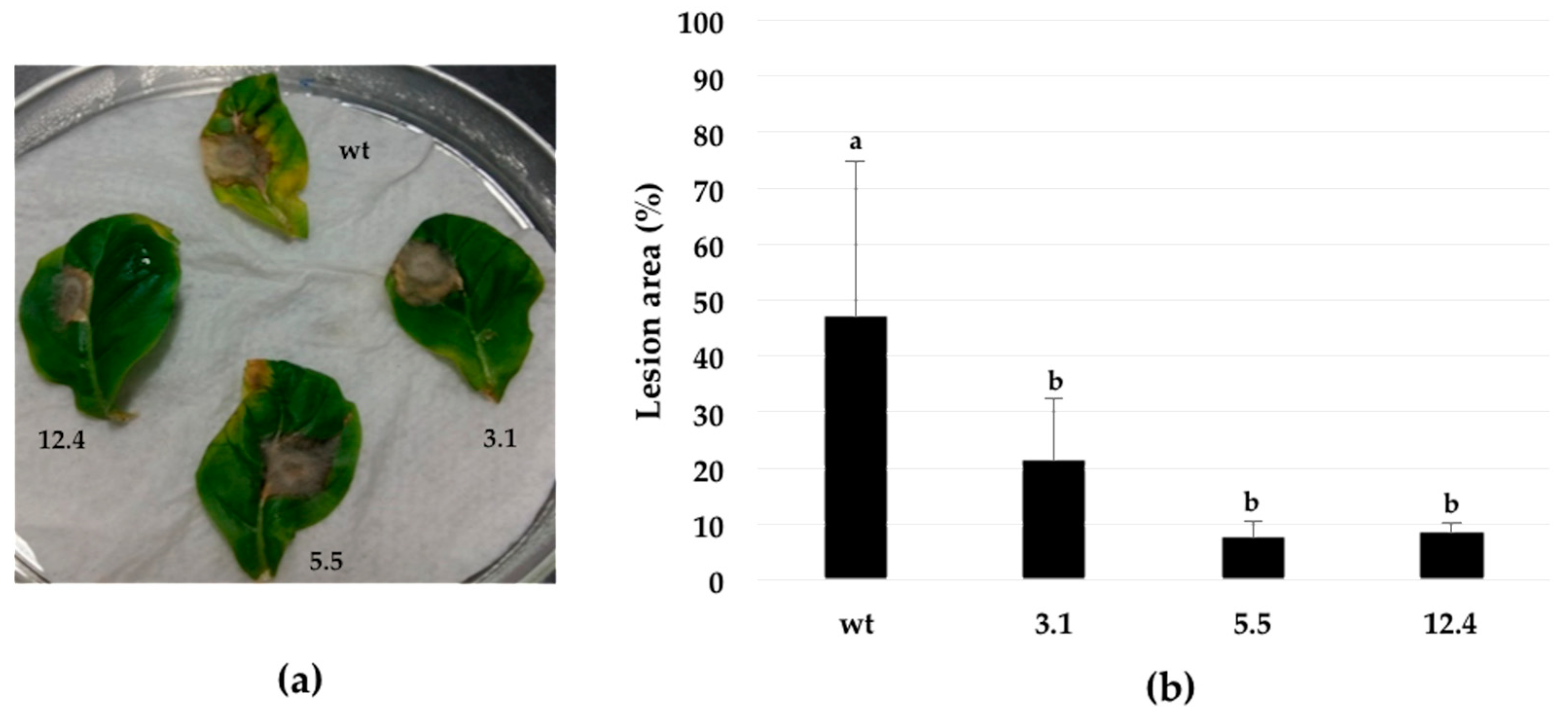
2.7. Botrytis Cinerea Resistance Assays on Detached Leaves
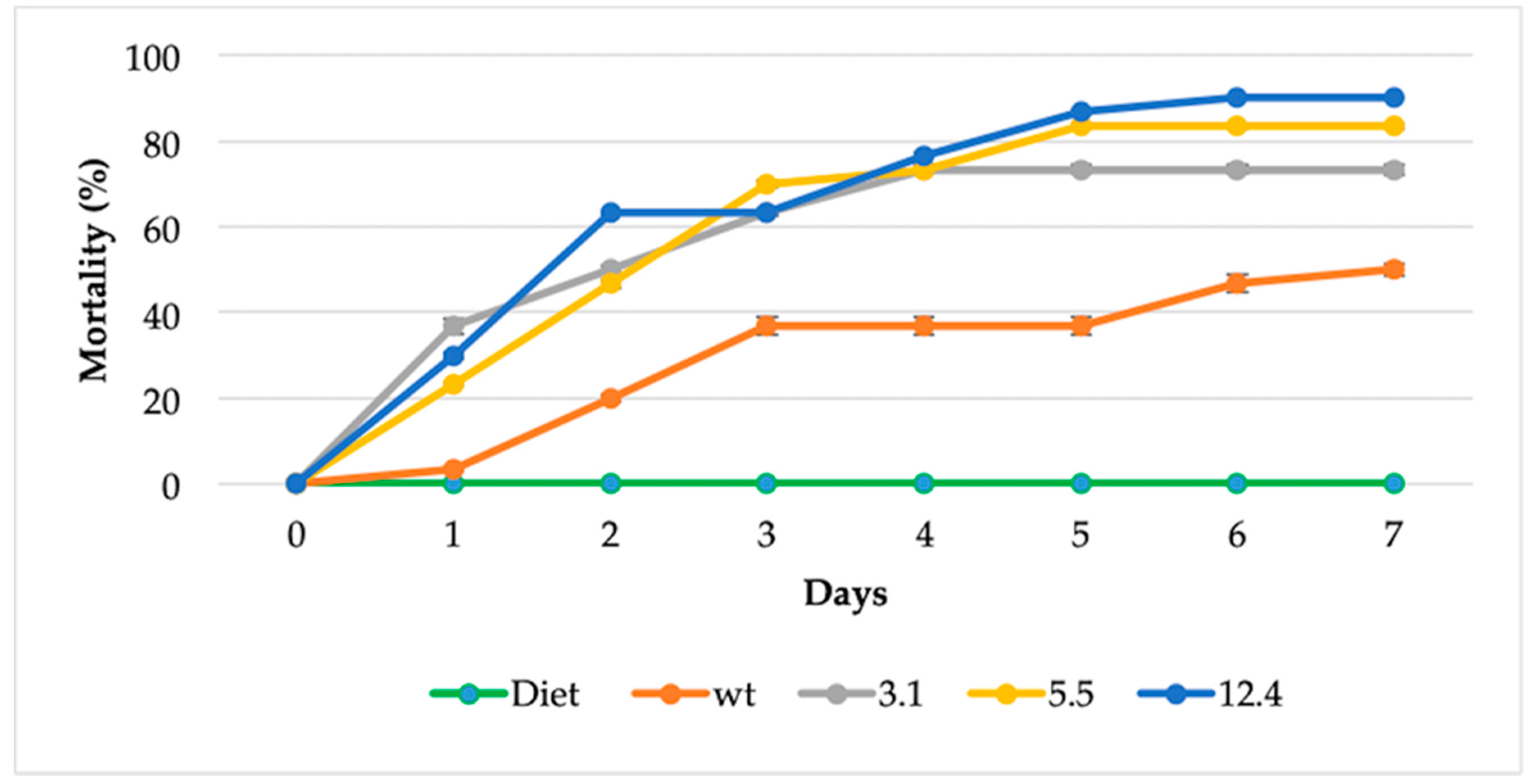
2.8. Insect Toxicity Assay
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Screening of Bacterial Isolate for Chitinolytic Aactivity
4.3. Bacterial Identification
4.4. Chitinase Gene Isolation
4.5. Tobacco Transformations
4.6. Molecular Analyses of Tobacco Transgenic Lines
4.7. Progeny Segregation
4.8. Preparation of Crude Protein Extracts from Transgenic Tobacco Lines
4.9. Western Blotting
4.10. Endochitinase Assay
4.11. Botrytis cinerea Spore Harvest for Antifungal Inhibition Assays
4.12. Assays of Antifungal Inhibition Activity on Crude Protein Extracts
4.13. Resistance Assays of Botrytis cinerea on Detached Leaves
4.14. Bioassays for Toxicity to Insects
4.15. Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PR- | Pathogenesis-related |
| GlcNAc | β-1,4-N-acetylglucosamine |
| S. marcescens | Serratia marcescens |
| B. cinerea | Botrytis cinerea |
| S. frugiperda | Spodoptera frugiperda |
| B. thuringiensis | Bacillus thuringiensis |
| PM | Peritrophic matrix |
References
- Oerke, E.C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31. [Google Scholar] [CrossRef]
- Abulreesh, H.H.; Osman, G.E.H.; Assaeedi, A.S.A. Characterization of Insecticidal Genes of Bacillus thuringiensis Strains Isolated from Arid Environments. Indian J. Microbiol. 2012, 52, 500–503. [Google Scholar] [CrossRef]
- El-Menofy, W.H.; Osman, G.H.; Assaeedi, A.; Salama, M.S. Construction of A Novel recombinant baculovirus containing Cry1Abb insecticidal protein from Bacillus thuringiensis. Biol. Proced. Online 2014, 4, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Kupiec, R.; Chet, I. The molecular biology of chitin digestion. Curr. Opin. Biotechnol. 1998, 9, 270–277. [Google Scholar] [CrossRef]
- Nagpure, A.; Choudhary, B.; Gupta, R.K. Chitinases: In agriculture and human healthcare. Crit. Rev. Biotechnol. 2014, 34, 215–232. [Google Scholar] [CrossRef] [PubMed]
- Takenaka, Y.; Nakano, S.; Tamoi, M.; Sakuda, S.; Fukamizo, T. Chitinase Gene Expression in Response to Environmental Stresses in Arabidopsis thaliana: Chitinase Inhibitor Allosamidin Enhances Stress Tolerance. Biosci. Biotechnol. Biochem. 2009, 73, 1066–1071. [Google Scholar] [CrossRef]
- De las Mercedes, D.M.; Pintor-Toro, J.A.; Cubero, B. Transgenic Tobacco Plants Overexpressing Chitinases of Fungal Origin Show Enhanced Resistance to Biotic and Abiotic Stress Agents. Plant Physiol. 2006, 142, 722–730. [Google Scholar] [Green Version]
- Melchers, L.S.; Stuiver, M.H. Novel genes for disease-resistance breeding. Curr. Opin. Plant Biol. 2000, 3, 147–152. [Google Scholar] [CrossRef]
- Benito, E.P.; ten Have, A.; van’t Klooster, J.W.; Van Kan, J.A.L. Fungal and plant gene expression during synchronized infection of tomato leaves by Botrytis cinerea. Eur. J. Plant Pathol. 1998, 104, 207–220. [Google Scholar] [CrossRef]
- Hong, J.K.; Hwang, B.K. Promoter activation of pepper class II basic chitinase gene, CAChi2, and enhanced bacterial disease resistance and osmotic stress tolerance in the CAChi2-overexpressing Arabidopsis. Planta 2006, 223, 433–448. [Google Scholar] [CrossRef]
- Alexander, D.; Goodman, R.M.; Gut-Rella, M.; Glascock, C.; Weymann, K.; Friedrich, L.; Maddox, D.; Ahl-Goy, P.; Luntz, T.; Ward, E.; et al. Increased tolerance to two oomycete pathogens in transgenic tobacco expressing pathogenesis-related protein 1a. Plant Biol. 1993, 90, 7327–7331. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Sharma, K.P.; Gaur, R.K.; Gupta, V.K. Role of Chitinase in Plant Defense. Asian J. Biochem. 2011, 6, 29–37. [Google Scholar] [CrossRef]
- Lorito, M.; Woo, S.L.; Fernandez, I.G.; Colucci, G.; Harman, G.E.; Pintor-Toro, J.A.; Filippone, E.; Muccifora, S.; Lawrence, C.B.; Zoina, A.; et al. Genes from mycoparasitic fungi as a source for improving plant resistance to fungal pathogens. Proc. Natl. Acad. Sci. USA 1998, 95, 7860–7865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Limón, M.C.; Lora, J.M.; García, I.; de la Cruz, J.; Llobell, A.; Benítez, T.; Pintor-Toro, J.A. Primary structure and expression pattern of the 33-kDa chitinase gene from the mycoparasitic fungus Trichoderma harzianum. Curr. Genet. 1995, 28, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Jach, G.; Görnhardt, B.; Mundy, J.; Logemann, J.; Pinsdorf, E.; Leah, R.; Schell, J.; Mass, C. Enhanced quantitative resistance against fungal disease by combinatorial expression of different barley antifungal proteins in transgenic tobacco. Plant J. 1995, 8, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Eissa, H.F.; Hassanien, S.E.; Ramadan, A.M.; El-Shamy, M.M.; Saleh, O.M.; Shokry, A.M.; Abdelsattar, M.; Morsy, Y.B.; El-Maghraby, M.A.; Alameldin, H.F.; et al. Developing transgenic wheat to encounter rusts and powdery mildew by overexpressing barley chi26 gene for fungal resistance. Plant Methods 2017, 13, 41. [Google Scholar] [CrossRef] [PubMed]
- Schlumbaum, A.; Mauch, F.; Vögeli, U.; Boller, T. Plant chitinases are potent inhibitors of fungal growth. Nature 1986, 324, 365–367. [Google Scholar] [CrossRef]
- Broglie, K.; Chet, I.; Holliday, M.; Cressman, R.; Biddle, P.; Knowlton, S.; Mauvais, J.; Broglie, R. Transgenic Plants with Enhanced Resistance to the Fungal Pathogen Rhizoctonia solani. Science 1991, 254, 1194–1197. [Google Scholar] [CrossRef]
- Dong, X.; Zhao, Y.; Ran, X.; Guo, L.; Zhao, D.-G. Overexpression of a New Chitinase Gene EuCHIT2 Enhances Resistance to Erysiphe cichoracearum DC. in Tobacco Plants. Int. J. Mol. Sci. 2017, 18, 2361. [Google Scholar] [CrossRef]
- Bravo, A.; Likitvivatanavong, S.; Gill, S.S.; Soberón, M. Bacillus thuringiensis: A story of a successful bioinsecticide. Insect Biochem. Mol. Biol. 2011, 41, 423–431. [Google Scholar] [CrossRef]
- Bravo, A.; Gill, S.S.; Soberón, M. Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon 2007, 49, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Tabashnik, B.E.; Carrière, Y. Surge in insect resistance to transgenic crops and prospects for sustainability. Nat. Biotechnol. 2017, 35, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.H.; Ali, S.; Ren, S.X. Evaluation of Chitinase from Metarhizium anisopliae as Biopesticide Against Plutella xylostella. Pak. J. Zool. 2010, 42, 9. [Google Scholar]
- Bidari, F.; Shams-Bakhsh, M.; Mehrabadi, M. Isolation and characterization of a Serratia marcescens with insecticidal activity from Polyphylla olivieri (Col.: Scarabaeidae). J. Appl. Entomol. 2018, 142, 162–172. [Google Scholar] [CrossRef]
- Ding, X.; Gopalakrishnan, B.; Johnson, L.B.; White, F.F.; Wang, X.; Morgan, T.D.; Kramer, K.J.; Muthukrishnan, S. Insect resistance of transgenic tobacco expressing an insect chitinase gene. Transgenic Res. 1998, 7, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, C.; Paul, S.; Tripathi, V.; Paul, B.; Khan, M.A. Chitinolytic activity in Serratia marcescens (strain SEN) and potency against different larval instars of Spodoptera litura with effect of sublethal doses on insect development. BioControl 2015, 60, 631–640. [Google Scholar] [CrossRef]
- Brurberg, M.B.; Nes, I.F.; Eijsink, V.G.H. Comparative studies of chitinases A and B from Serratia marcescens. Microbiology 1996, 142, 1581–1589. [Google Scholar] [CrossRef]
- Osman, G.H.; Assem, S.K.; Alreedy, R.M.; El-Ghareeb, D.K.; Basry, M.A.; Rastogi, A.; Kalaji, H.M. Development of insect resistant maize plants expressing a chitinase gene from the cotton leaf worm. Spodoptera littoralis. Sci. Rep. 2015, 5, 18067. [Google Scholar] [CrossRef]
- Rao, R.; Fiandra, L.; Giordana, B.; de Eguileor, M.; Congiu, T.; Burlini, N.; Arcielloa, S.; Corrado, G.; Pennacchio, F. AcMNPVChiA protein disrupts the peritrophic membrane and alters midgut physiology of Bombyx mori larvae. Insect Biochem. Mol. Biol. 2004, 34, 1205. [Google Scholar] [CrossRef]
- Horsch, R.B.; Fry, J.E.; Hoffmann, N.L.; Rogers, S.G.; Fraley, R.T. A simple method for transferring genes into plants. Science 1985, 227, 1229–1231. [Google Scholar]
- Folders, J.; Algra, J.; Roelofs, M.S.; van Loon, L.C.; Tommassen, J.; Bitter, W. Characterization of Pseudomonas aeruginosa Chitinase, a Gradually Secreted Protein. J. Bacteriol. 2001, 183, 7044–7052. [Google Scholar] [CrossRef] [PubMed]
- Babashpour, S.; Aminzadeh, S.; Farrokhi, N.; Karkhane, A.; Haghbeen, K. Characterization of a chitinase (Chit62) from Serratia marcescens B4A and its efficacy as a bioshield against plant fungal pathogens. Biochem. Genet. 2012, 50, 722–735. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Maher, E.A.; Masoud, S.; Dixon, R.A.; Lamb, C.J. Enhanced Protection Against Fungal Attack by Constitutive Co–expression of Chitinase and Glucanase Genes in Transgenic Tobacco. Tob. Nat. Biotechnol. 1994, 12, 807–812. [Google Scholar] [CrossRef]
- Lin, W.; Anuratha, C.S.; Potrykus, I.; Muthukrishnan, S.; Datta, S.K. Genetic Engineering of Rice for Resistance to Sheath Blight. Nat. Biotechnol. 1995, 13, 686–691. [Google Scholar] [CrossRef]
- Datta, K.; Tu, J.; Oliva, N.; Ona, I.; Velazhahan, R.; Mew, T.W.; Muthukrishnan, S.; Datta, S.K. Enhanced resistance to sheath blight by constitutive expression of infection-related rice chitinase in transgenic elite indica rice cultivars. Plant Sci. 2001, 160, 405–414. [Google Scholar] [CrossRef]
- Mehmood, M.A.; Xiao, X.; Hafeez, F.Y.; Gai, Y.; Wang, F. Purification and characterization of a chitinase from Serratia proteamaculans. World J. Microbiol. Biotechnol. 2009, 25, 1955–1961. [Google Scholar] [CrossRef]
- Javed, S.; Ahmad, M.M.; Ahmad, M.; Abdin, M.Z.; Hamid, R.; Khan, M.A.; Musarrat, J. Chitinases: An update. J. Pharm. Bioallied Sci. 2013, 5, 21–29. [Google Scholar] [CrossRef]
- Parani, K.; Shetty, G.P.; Saha, B.K. Isolation of Serratia marcescens SR1 as a Source of Chitinase Having Potentiality of Using as a Biocontrol Agent. Indian J. Microbiol. 2011, 51, 247–250. [Google Scholar] [CrossRef]
- Gutiérrez-Román, M.I.; Dunn, M.F.; Tinoco-Valencia, R.; Holguín-Meléndez, F.; Huerta-Palacios, G.; Guillén-Navarro, K. Potentiation of the synergistic activities of chitinases ChiA, ChiB and ChiC from Serratia marcescens CFFSUR-B2 by chitobiase (Chb) and chitin binding protein (CBP). World J. Microbiol. Biotechnol. 2014, 30, 33–42. [Google Scholar] [CrossRef]
- Liu, Z.; Shi, L.; Yang, S.; Lin, Y.; Weng, Y.; Li, X.; Hussain, A.; Noman, A.; He, S. Functional and Promoter Analysis of ChiIV3, a Chitinase of Pepper Plant, in Response to Phytophthora capsici Infection. Int. J. Mol. Sci. 2017, 18, 1661. [Google Scholar] [CrossRef]
- Emani, C.; Garcia, J.M.; Lopata-Finch, E.; Pozo, M.J.; Uribe, P.; Kim, D.-J.; Sunilkumar, G.; Cook, D.R.; Kenerley, C.M.; Rathore, K.S. Enhanced fungal resistance in transgenic cotton expressing an endochitinase gene from Trichoderma virens: Fungal-resistant transgenic cotton. Plant Biotechnol. J. 2003, 1, 321–336. [Google Scholar] [CrossRef] [PubMed]
- Carstens, M.; Vivier, M.A.; Pretorius, I.S. The Saccharomyces cerevisiae chitinase, encoded by the CTS1-2 gene, confers antifungal activity against Botrytis cinerea to transgenic tobacco. Transgenic Res. 2003, 12, 497–508. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.R.; Mukherjee, P.K.; Eapen, S. Expression of a fungal endochitinase gene in transgenic tomato and tobacco results in enhanced tolerance to fungal pathogens. Physiol. Mol. Biol. Plants 2010, 16, 39–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zagorchev, L.; Kamenova, P.; Odjakova, M. The Role of Plant Cell Wall Proteins in Response to Salt Stress. Sci. World J. 2014, 2014, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fry, S.C.; Aldington, S.; Hetherington, P.R.; Aitken, J. Oligosaccharides as Signals and Substrates in the Plant Cell Wall. Plant Physiol. 1993, 103, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, R.; Revathi, K.; Nisha, S.; Kirubakaran, S.A.; Sathish-Narayanan, S.; Senthil-Nathan, S. Physiological effect of chitinase purified from Bacillus subtilis against the tobacco cutworm Spodoptera litura Fab. Pestic. Biochem. Physiol. 2012, 104, 65–71. [Google Scholar] [CrossRef]
- Roberts, W.K.; Selitrennikoff, C.P. Plant and Bacterial Chitinases Differ in Antifungal Activity. Microbiology 1988, 134, 169–176. [Google Scholar] [CrossRef] [Green Version]
- Marchesi, J.R.; Sato, T.; Weightman, A.J.; Martin, T.A.; Fry, J.C.; Hiom, S.J.; Wade, W.G. Design and Evaluation of Useful Bacterium-Specific PCR Primers That Amplify Genes Coding for Bacterial 16S rRNA. Appl. Environ. Microbiol. 1998, 64, 795–799. [Google Scholar] [PubMed]
- Nübel, U.; Engelen, B.; Felske, A.; Snaidr, J.; Wieshuber, A.; Amann, R.I.; Ludwig, W.; Backhaus, H. Sequence heterogeneities of genes encoding 16S rRNAs in Paenibacillus polymyxa detected by temperature gradient gel electrophoresis. J. Bacteriol. 1996, 178, 5636–5643. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugars. Anal. Chem. 1959, 31, 426–430. [Google Scholar] [CrossRef]
- Fenice, M.; Leuba, J.-L.; Federici, F. Chitinolytic enzyme activity of Penicillium janthinellum P9 in bench-top bioreactor. J. Ferment. Bioeng. 1998, 86, 620–623. [Google Scholar] [CrossRef]
- Bradford, M.M. Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Fiddaman, P.J.; O’Neill, T.M.; Rossall, S. Screening of bacteria for the suppression of Botrytis cinerea and Rhizoctonia solani on lettuce (Lactuca sativa) using leaf disc bioassays. Ann. Appl. Biol. 2000, 137, 223–235. [Google Scholar] [CrossRef]
- Ludwig, A.; Boller, T. A method for the study of fungal growth inhibition by plant proteins. FEMS Microbiol. Lett. 1990, 69, 61–66. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro-González, S.S.; Ramírez-Trujillo, J.A.; Peña-Chora, G.; Gaytán, P.; Roldán-Salgado, A.; Corzo, G.; Lina-García, L.P.; Hernández-Velázquez, V.M.; Suárez-Rodríguez, R. Enhanced Tolerance against a Fungal Pathogen and Insect Resistance in Transgenic Tobacco Plants Overexpressing an Endochitinase Gene from Serratia marcescens. Int. J. Mol. Sci. 2019, 20, 3482. https://doi.org/10.3390/ijms20143482
Navarro-González SS, Ramírez-Trujillo JA, Peña-Chora G, Gaytán P, Roldán-Salgado A, Corzo G, Lina-García LP, Hernández-Velázquez VM, Suárez-Rodríguez R. Enhanced Tolerance against a Fungal Pathogen and Insect Resistance in Transgenic Tobacco Plants Overexpressing an Endochitinase Gene from Serratia marcescens. International Journal of Molecular Sciences. 2019; 20(14):3482. https://doi.org/10.3390/ijms20143482
Chicago/Turabian StyleNavarro-González, Samantha Sarai, José Augusto Ramírez-Trujillo, Guadalupe Peña-Chora, Paul Gaytán, Abigail Roldán-Salgado, Gerardo Corzo, Laura Patricia Lina-García, Víctor Manuel Hernández-Velázquez, and Ramón Suárez-Rodríguez. 2019. "Enhanced Tolerance against a Fungal Pathogen and Insect Resistance in Transgenic Tobacco Plants Overexpressing an Endochitinase Gene from Serratia marcescens" International Journal of Molecular Sciences 20, no. 14: 3482. https://doi.org/10.3390/ijms20143482





