Apolipoprotein C1: Its Pleiotropic Effects in Lipid Metabolism and Beyond
Abstract
:1. Introduction
2. APOC1 Gene
2.1. APOC1 Gene and Pseudogene Localization
2.2. Gene Structure: Encoding and Regulatory Elements
2.3. Significant Polymorphisms
3. Protein Expression and Structure
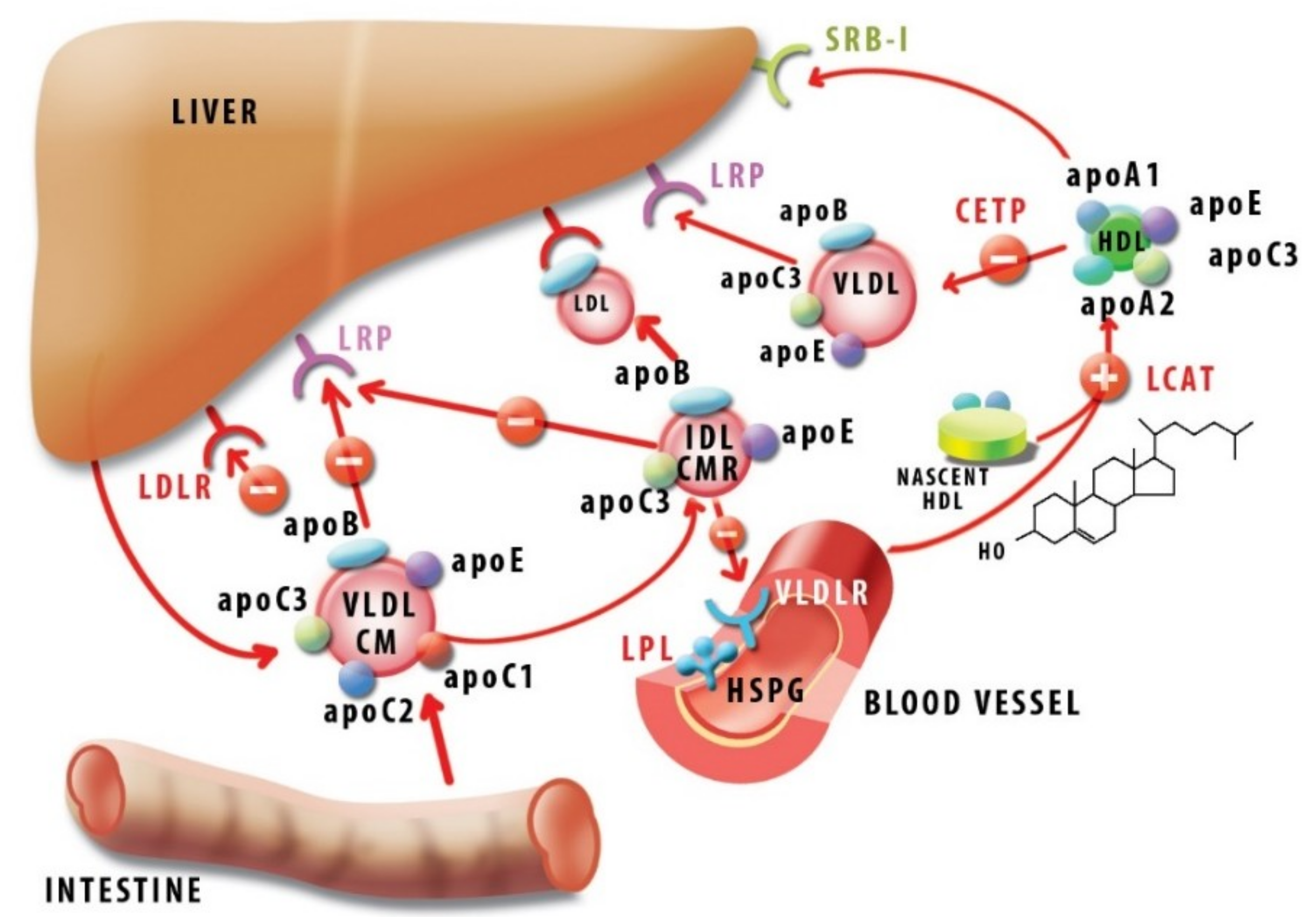
4. The Biochemistry of ApoC1: Modulating the Plasma Lipid Profile
5. ApoC1 in pathology
5.1. Atherosclerosis
5.2. Sepsis and Immunity
5.3. Cognitive Processes
5.4. Atopic Dermatitis
5.5. Viral Infectivity
5.6. Cancer
5.7. Diabetes
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| apo | apolipoprotein |
| CETP | cholesteryl ester transfer protein |
| CM | chylomicrons |
| CMR | chylomicron remnants |
| HDL | high-density lipoproteins |
| HL | hepatic lipase |
| HSPG | heparan-sulfate proteoglycans |
| IDL | intermediate density lipoproteins |
| kb | kilobase |
| LCAT | lecithin–cholesterol acyltransferase |
| LDL | low-density lipoprotein |
| LDLR | low-density lipoprotein receptor |
| LRP | LDLR-related protein |
| LPS | lipopolysaccharide |
| Lp(a) | lipoprotein a |
| PLaseA2 | phospholipase A2 |
| PON-1 | paraoxonase-1 |
| SNP | single nucleotide polymorphism |
| SRB-I | scavenger receptor BI |
| VLDL | very low-density lipoprotein |
| VLDLR | very low-density lipoprotein receptor |
References
- Feingold, K.R.; Grunfeld, C. Introduction to Lipids and Lipoproteins. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., Dungan, K., Grossman, A., Hershman, J.M., Kaltsas, G., Koch, C., Kopp, P., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Zannis, V.I.; Chroni, A.; Kardassis, D.; Zanni, E.E. Lipoproteins and atherogenesis. In Molecular Mechanisms of Atherosclerosis; Loscalzo, J., Ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 111–174. [Google Scholar]
- Kypreos, K.E.; Bitzur, R.; Karavia, E.A.; Xepapadaki, E.; Panayiotakopoulos, G.; Constantinou, C. Pharmacological Management of Dyslipidemia in Atherosclerosis: Limitations, Challenges, and New Therapeutic Opportunities. Angiology 2018, 70, 197–209. [Google Scholar] [CrossRef]
- Karathanasis, S.K. Apolipoprotein multigene family: tandem organization of human apolipoprotein AI, CIII, and AIV genes. Proc. Natl. Acad. Sci. USA 1985, 82, 6374–6378. [Google Scholar] [CrossRef] [PubMed]
- Pennacchio, L.A.; Olivier, M.; Hubacek, J.A.; Cohen, J.C.; Cox, D.R.; Fruchart, J.C.; Krauss, R.M.; Rubin, E.M. An apolipoprotein influencing triglycerides in humans and mice revealed by comparative sequencing. Science 2001, 294, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Eichenbaum-Voline, S.; Olivier, M.; Jones, E.L.; Naoumova, R.P.; Jones, B.; Gau, B.; Patel, H.N.; Seed, M.; Betteridge, D.J.; Galton, D.J.; et al. Linkage and association between distinct variants of the APOA1/C3/A4/A5 gene cluster and familial combined hyperlipidemia. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.; Knott, T.J.; Shaw, D.J.; Brook, J.D. Localization of genes encoding apolipoproteins CI, CII, and E to the p13—cen region of human chromosome 19. Hum. Genet. 1985, 71, 144–146. [Google Scholar] [CrossRef] [PubMed]
- Tudorache, I.F.; Trusca, V.G.; Gafencu, A.V. Apolipoprotein E—A Multifunctional Protein with Implications in Various Pathologies as a Result of Its Structural Features. Comput. Struct. Biotechnol. J. 2017, 15, 359–365. [Google Scholar] [CrossRef]
- Bennet, A.M.; Di Angelantonio, E.; Ye, Z.; Wensley, F.; Dahlin, A.; Ahlbom, A.; Keavney, B.; Collins, R.; Wiman, B.; de Faire, U.; et al. Association of apolipoprotein E genotypes with lipid levels and coronary risk. JAMA 2007, 298, 1300–1311. [Google Scholar] [CrossRef]
- Gautier, T.; Masson, D.; de Barros, J.P.; Athias, A.; Gambert, P.; Aunis, D.; Metz-Boutigue, M.H.; Lagrost, L. Human apolipoprotein C-I accounts for the ability of plasma high density lipoproteins to inhibit the cholesteryl ester transfer protein activity. J. Biol. Chem. 2000, 275, 37504–37509. [Google Scholar] [CrossRef]
- Pillois, X.; Gautier, T.; Bouillet, B.; Pais de Barros, J.P.; Jeannin, A.; Verges, B.; Bonnet, J.; Lagrost, L. Constitutive inhibition of plasma CETP by apolipoprotein C1 is blunted in dyslipidemic patients with coronary artery disease. J. Lipid Res. 2012, 53, 1200–1209. [Google Scholar] [CrossRef]
- Allan, C.M.; Taylor, J.M. Expression of a novel human apolipoprotein (apoC-IV) causes hypertriglyceridemia in transgenic mice. J. Lipid Res. 1996, 37, 1510–1518. [Google Scholar]
- Kim, E.; Li, K.; Lieu, C.; Tong, S.; Kawai, S.; Fukutomi, T.; Zhou, Y.; Wands, J.; Li, J. Expression of apolipoprotein C-IV is regulated by Ku antigen/peroxisome proliferator-activated receptor gamma complex and correlates with liver steatosis. J. Hepatol. 2008, 49, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, T.; Sudo, H.; Ishikawa, K.; Yamamoto, A. Familial type I hyperlipoproteinemia caused by apolipoprotein C-II deficiency. Atherosclerosis 1979, 34, 53–65. [Google Scholar] [CrossRef]
- Ken-Dror, G.; Talmud, P.J.; Humphries, S.E.; Drenos, F. APOE/C1/C4/C2 gene cluster genotypes, haplotypes and lipid levels in prospective coronary heart disease risk among UK healthy men. Mol. Med. 2010, 16, 389–399. [Google Scholar] [CrossRef]
- Pirim, D.; Radwan, Z.H.; Wang, X.; Niemsiri, V.; Hokanson, J.E.; Hamman, R.F.; Feingold, E.; Bunker, C.H.; Demirci, F.Y.; Kamboh, M.I. Apolipoprotein E-C1-C4-C2 gene cluster region and inter-individual variation in plasma lipoprotein levels: a comprehensive genetic association study in two ethnic groups. PLoS ONE 2019, 14, e0214060. [Google Scholar] [CrossRef] [PubMed]
- Lusis, A.J.; Heinzmann, C.; Sparkes, R.S.; Scott, J.; Knott, T.J.; Geller, R.; Sparkes, M.C.; Mohandas, T. Regional mapping of human chromosome 19: organization of genes for plasma lipid transport (APOC1, -C2, and -E and LDLR) and the genes C3, PEPD, and GPI. Proc. Natl. Acad. Sci. USA 1986, 83, 3929–3933. [Google Scholar] [CrossRef]
- Plump, A.S.; Smith, J.D.; Hayek, T.; Aalto-Setala, K.; Walsh, A.; Verstuyft, J.G.; Rubin, E.M.; Breslow, J.L. Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 1992, 71, 343–353. [Google Scholar] [CrossRef]
- Strittmatter, W.J.; Saunders, A.M.; Schmechel, D.; Pericak-Vance, M.; Enghild, J.; Salvesen, G.S.; Roses, A.D. Apolipoprotein E: high-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease. Proc. Natl. Acad. Sci. USA 1993, 90, 1977–1981. [Google Scholar] [CrossRef]
- Jong, M.C.; Hofker, M.H.; Havekes, L.M. Role of ApoCs in lipoprotein metabolism: functional differences between ApoC1, ApoC2, and ApoC3. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 472–484. [Google Scholar] [CrossRef]
- Jong, M.C.; Havekes, L.M. Insights into apolipoprotein C metabolism from transgenic and gene-targeted mice. Int. J. Tissue React. 2000, 22, 59–66. [Google Scholar]
- Myklebost, O.; Rogne, S. The gene for human apolipoprotein CI is located 4.3 kilobases away from the apolipoprotein E gene on chromosome 19. Hum. Genet. 1986, 73, 286–289. [Google Scholar] [CrossRef]
- Lauer, S.J.; Walker, D.; Elshourbagy, N.A.; Reardon, C.A.; Levy-Wilson, B.; Taylor, J.M. Two copies of the human apolipoprotein C-I gene are linked closely to the apolipoprotein E gene. J. Biol. Chem. 1988, 263, 7277–7286. [Google Scholar] [PubMed]
- Puppione, D.L.; Ryan, C.M.; Bassilian, S.; Souda, P.; Xiao, X.; Ryder, O.A.; Whitelegge, J.P. Detection of two distinct forms of apoC-I in great apes. Comp. Biochem. Physiol. Part. D Genomics Proteomics 2010, 5, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Ransohoff, J.D.; Wei, Y.; Khavari, P.A. The functions and unique features of long intergenic non-coding RNA. Nat. Rev. Mol. Cell Biol. 2018, 19, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.H.; Wang, J.G.; Li, L.Y.; Zhou, D.M.; Ren, K.H.; Jin, Y.T.; Lv, L.; Yu, J.G.; Yang, J.Y.; Lu, Q.; et al. Long intergenic non-coding RNA APOC1P1-3 inhibits apoptosis by decreasing alpha-tubulin acetylation in breast cancer. Cell Death Dis 2016, 7, e2236. [Google Scholar] [CrossRef]
- Han, B.W.; Ye, H.; Wei, P.P.; He, B.; Han, C.; Chen, Z.H.; Chen, Y.Q.; Wang, W.T. Global identification and characterization of lncRNAs that control inflammation in malignant cholangiocytes. BMC Genomics 2018, 19, 735. [Google Scholar] [CrossRef]
- Dang, Q.; Walker, D.; Taylor, S.; Allan, C.; Chin, P.; Fan, J.; Taylor, J. Structure of the hepatic control region of the human apolipoprotein E/C-I gene locus. J. Biol. Chem. 1995, 270, 22577–22585. [Google Scholar] [CrossRef]
- Allan, C.M.; Walker, D.; Taylor, J.M. Evolutionary duplication of a hepatic control region in the human apolipoprotein E gene locus. Identification of a second region that confers high level and liver-specific expression of the human apolipoprotein E gene in transgenic mice. J. Biol. Chem. 1995, 270, 26278–26281. [Google Scholar] [CrossRef]
- Shih, S.J.; Allan, C.; Grehan, S.; Tse, E.; Moran, C.; Taylor, J.M. Duplicated downstream enhancers control expression of the human apolipoprotein E gene in macrophages and adipose tissue. J. Biol. Chem. 2000, 275, 31567–31572. [Google Scholar] [CrossRef]
- Freitas, E.M.; Gaudieri, S.; Zhang, W.J.; Kulski, J.K.; van Bockxmeer, F.M.; Christiansen, F.T.; Dawkins, R.L. Duplication and diversification of the apolipoprotein CI (APOCI) genomic segment in association with retroelements. J. Mol. Evol. 2000, 50, 391–396. [Google Scholar] [CrossRef]
- Medstrand, P.; Landry, J.R.; Mager, D.L. Long terminal repeats are used as alternative promoters for the endothelin B receptor and apolipoprotein C-I genes in humans. J. Biol. Chem. 2001, 276, 1896–1903. [Google Scholar] [CrossRef]
- Simonet, W.S.; Bucay, N.; Lauer, S.J.; Taylor, J.M. A far-downstream hepatocyte-specific control region directs expression of the linked human apolipoprotein E and C-I genes in transgenic mice. J. Biol. Chem. 1993, 268, 8221–8229. [Google Scholar] [PubMed]
- Zannis, V.I.; Kan, H.Y.; Kritis, A.; Zanni, E.; Kardassis, D. Transcriptional regulation of the human apolipoprotein genes. Front. Biosci 2001, 6, D456–504. [Google Scholar] [CrossRef] [PubMed]
- Allan, C.M.; Taylor, S.; Taylor, J.M. Two hepatic enhancers, HCR.1 and HCR.2, coordinate the liver expression of the entire human apolipoprotein E/C-I/C-IV/C-II gene cluster. J. Biol. Chem. 1997, 272, 29113–29119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, E.; Xie, S.; Yeh, S.D.; Lee, Y.F.; Collins, L.L.; Hu, Y.C.; Shyr, C.R.; Mu, X.M.; Liu, N.C.; Chen, Y.T.; et al. Disruption of TR4 orphan nuclear receptor reduces the expression of liver apolipoprotein E/C-I/C-II gene cluster. J. Biol. Chem. 2003, 278, 46919–46926. [Google Scholar] [CrossRef] [Green Version]
- Mak, P.A.; Laffitte, B.A.; Desrumaux, C.; Joseph, S.B.; Curtiss, L.K.; Mangelsdorf, D.J.; Tontonoz, P.; Edwards, P.A. Regulated expression of the apolipoprotein E/C-I/C-IV/C-II gene cluster in murine and human macrophages. A critical role for nuclear liver X receptors alpha and beta. J. Biol. Chem. 2002, 277, 31900–31908. [Google Scholar] [CrossRef] [Green Version]
- Trusca, V.G.; Fuior, E.V.; Florea, I.C.; Kardassis, D.; Simionescu, M.; Gafencu, A.V. Macrophage-specific up-regulation of apolipoprotein E gene expression by STAT1 is achieved via long range genomic interactions. J. Biol. Chem. 2011, 286, 13891–13904. [Google Scholar] [CrossRef] [Green Version]
- Galetto, R.; Albajar, M.; Polanco, J.I.; Zakin, M.M.; Rodriguez-Rey, J.C. Identification of a peroxisome-proliferator-activated-receptor response element in the apolipoprotein E gene control region. Biochem. J. 2001, 357, 521–527. [Google Scholar] [CrossRef]
- Dahabreh, D.F.; Medh, J.D. Activation of peroxisome proliferator activated receptor-gamma results in an atheroprotective apolipoprotein profile in HepG2 cells. Adv. Biol. Chem. 2012, 2, 218–225. [Google Scholar] [CrossRef] [Green Version]
- Subramanian, S.; Gottschalk, W.K.; Kim, S.Y.; Roses, A.D.; Chiba-Falek, O. The effects of PPARgamma on the regulation of the TOMM40-APOE-C1 genes cluster. Biochim. Biophys. Acta Mol. Basis. Dis. 2017, 1863, 810–816. [Google Scholar] [CrossRef]
- Fernandes, G.W.; Bocco, B.; Fonseca, T.L.; McAninch, E.A.; Jo, S.; Lartey, L.J.; I, O.S.; Unterman, T.G.; Preite, N.Z.; Voigt, R.M.; et al. The Foxo1-Inducible Transcriptional Repressor Zfp125 Causes Hepatic Steatosis and Hypercholesterolemia. Cell Rep. 2018, 22, 523–534. [Google Scholar] [CrossRef] [Green Version]
- Vrins, C.L.; Out, R.; van Santbrink, P.; van der Zee, A.; Mahmoudi, T.; Groenendijk, M.; Havekes, L.M.; van Berkel, T.J.; Willems van Dijk, K.; Biessen, E.A. Znf202 affects high density lipoprotein cholesterol levels and promotes hepatosteatosis in hyperlipidemic mice. PLoS ONE 2013, 8, e57492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smit, M.; van der Kooij-Meijs, E.; Woudt, L.P.; Havekes, L.M.; Frants, R.R. Exact localization of the familial dysbetalipoproteinemia associated HpaI restriction site in the promoter region of the APOC1 gene. Biochem. Biophys. Res. Commun. 1988, 152, 1282–1288. [Google Scholar] [CrossRef]
- Smit, M.; van der Kooij-Meijs, E.; Frants, R.R.; Havekes, L.; Klasen, E.C. Apolipoprotein gene cluster on chromosome 19. Definite localization of the APOC2 gene and the polymorphic Hpa I site associated with type III hyperlipoproteinemia. Hum. Genet. 1988, 78, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Olsson, B.; Gigante, B.; Mehlig, K.; Bergsten, A.; Leander, K.; de Faire, U.; Lissner, L.; Thelle, D.S.; Carlsson, L.M. Apolipoprotein C-I genotype and serum levels of triglycerides, C-reactive protein and coronary heart disease. Metabolism 2010, 59, 1736–1741. [Google Scholar] [CrossRef]
- Poduslo, S.E.; Neal, M.; Herring, K.; Shelly, J. The apolipoprotein CI A allele as a risk factor for Alzheimer’s disease. Neurochem Res. 1998, 23, 361–367. [Google Scholar] [CrossRef]
- Drigalenko, E.; Poduslo, S.; Elston, R. Interaction of the apolipoprotein E and CI loci in predisposing to late-onset Alzheimer’s disease. Neurology 1998, 51, 131–135. [Google Scholar] [CrossRef]
- Retz, W.; Thome, J.; Durany, N.; Harsanyi, A.; Retz-Junginger, P.; Kornhuber, J.; Riederer, P.; Rosler, M. Potential genetic markers of sporadic Alzheimer’s dementia. Psychiatr. Genet. 2001, 11, 115–122. [Google Scholar] [CrossRef]
- Ki, C.S.; Na, D.L.; Kim, D.K.; Kim, H.J.; Kim, J.W. Genetic association of an apolipoprotein C-I (APOC1) gene polymorphism with late-onset Alzheimer’s disease. Neurosci. Lett. 2002, 319, 75–78. [Google Scholar] [CrossRef]
- Bertram, L.; McQueen, M.B.; Mullin, K.; Blacker, D.; Tanzi, R.E. Systematic meta-analyses of Alzheimer disease genetic association studies: the AlzGene database. Nat. Genet. 2007, 39, 17–23. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhao, F.; Lv, Z.P.; Zheng, C.G.; Zheng, W.D.; Sun, L.; Wang, N.N.; Pang, S.; de Andrade, F.M.; Fu, M.; et al. Association between APOC1 polymorphism and Alzheimer’s disease: A case-control study and meta-analysis. PLoS ONE 2014, 9, e87017. [Google Scholar] [CrossRef] [Green Version]
- Dixit, M.; Choudhuri, G.; Mittal, B. Association of APOE-C1 gene cluster polymorphisms with gallstone disease. Dig. Liver Dis 2006, 38, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Willer, C.J.; Sanna, S.; Jackson, A.U.; Scuteri, A.; Bonnycastle, L.L.; Clarke, R.; Heath, S.C.; Timpson, N.J.; Najjar, S.S.; Stringham, H.M.; et al. Newly identified loci that influence lipid concentrations and risk of coronary artery disease. Nat. Genet. 2008, 40, 161–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.; Ye, H.D.; Gao, X.; Nie, S.; Hong, Q.X.; Ji, H.H.; Sun, J.; Zhou, S.J.; Fei, B.; Li, K.Q.; et al. Significant interaction of APOE rs4420638 polymorphism with HDL-C and APOA-I levels in coronary heart disease in Han Chinese men. Genet. Mol. Res. 2015, 14, 13414–13424. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, T.; Lam, K.; Chen, Y.; Xia, Y.; Liu, C. A decade in psychiatric GWAS research. Mol. Psychiatry 2019, 24, 378–389. [Google Scholar] [CrossRef]
- Wroblewski, M.S.; Wilson-Grady, J.T.; Martinez, M.B.; Kasthuri, R.S.; McMillan, K.R.; Flood-Urdangarin, C.; Nelsestuen, G.L. A functional polymorphism of apolipoprotein C1 detected by mass spectrometry. FEBS J. 2006, 273, 4707–4715. [Google Scholar] [CrossRef]
- Lahiry, P.; Cao, H.; Ban, M.R.; Pollex, R.L.; Mamakeesick, M.; Zinman, B.; Harris, S.B.; Hanley, A.J.; Huff, M.W.; Connelly, P.W.; et al. APOC1 T45S polymorphism is associated with reduced obesity indices and lower plasma concentrations of leptin and apolipoprotein C-I in aboriginal Canadians. J. Lipid Res. 2010, 51, 843–848. [Google Scholar] [CrossRef] [Green Version]
- Kasthuri, R.S.; McMillan, K.R.; Flood-Urdangarin, C.; Harvey, S.B.; Wilson-Grady, J.T.; Nelsestuen, G.L. Correlation of a T45S variant of apolipoprotein C1 with elevated BMI in persons of American Indian and Mexican ancestries. Int J. Obes (Lond) 2007, 31, 1334–1336. [Google Scholar] [CrossRef] [Green Version]
- Marcel, Y.L.; Bergseth, M.; Nestruck, A.C. Preparative isoelectric focussing of apolipoproteins C and E from human very low density lipoproteins. Biochim Biophys Acta 1979, 573, 175–183. [Google Scholar] [CrossRef]
- Barker, W.C.; Dayhoff, M.O. Evolution of lipoproteins deduced from protein sequence data. Comp. Biochem. Physiol B 1977, 57, 309–315. [Google Scholar] [CrossRef]
- Rozek, A.; Sparrow, J.T.; Weisgraber, K.H.; Cushley, R.J. Conformation of human apolipoprotein C-I in a lipid-mimetic environment determined by CD and NMR spectroscopy. Biochemistry 1999, 38, 14475–14484. [Google Scholar] [CrossRef]
- Segrest, J.P.; Jones, M.K.; De Loof, H.; Brouillette, C.G.; Venkatachalapathi, Y.V.; Anantharamaiah, G.M. The amphipathic helix in the exchangeable apolipoproteins: a review of secondary structure and function. J. Lipid Res. 1992, 33, 141–166. [Google Scholar] [PubMed]
- Mendoza-Espinosa, P.; Moreno, A.; Castillo, R.; Mas-Oliva, J. Lipid dependant disorder-to-order conformational transitions in apolipoprotein CI derived peptides. Biochem. Biophys Res. Commun 2008, 365, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Buchko, G.W.; Rozek, A.; Zhong, Q.; Cushley, R.J. Sequence-specific 1H NMR assignments and secondary structure of a lipid-associating peptide from human ApoC-I: an NMR study of an amphipathic helix motif. Pept Res. 1995, 8, 86–94. [Google Scholar] [PubMed]
- Rozek, A.; Buchko, G.W.; Kanda, P.; Cushley, R.J. Conformational studies of the N-terminal lipid-associating domain of human apolipoprotein C-I by CD and 1H NMR spectroscopy. Protein Sci. 1997, 6, 1858–1868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buchko, G.W.; Rozek, A.; Hoyt, D.W.; Cushley, R.J.; Kennedy, M.A. The use of sodium dodecyl sulfate to model the apolipoprotein environment. Evidence for peptide-SDS complexes using pulsed-field-gradient NMR spectroscopy. Biochim. Biophys. Acta 1998, 1392, 101–108. [Google Scholar] [CrossRef]
- Rozek, A.; Sparrow, J.T.; Weisgraber, K.H.; Cushley, R.J. Sequence-specific 1H NMR resonance assignments and secondary structure of human apolipoprotein C-I in the presence of sodium dodecyl sulfate. Biochem. Cell Biol. 1998, 76, 267–275. [Google Scholar] [CrossRef]
- Rozek, A.; Buchko, G.W.; Cushley, R.J. Conformation of two peptides corresponding to human apolipoprotein C-I residues 7-24 and 35-53 in the presence of sodium dodecyl sulfate by CD and NMR spectroscopy. Biochemistry 1995, 34, 7401–7408. [Google Scholar] [CrossRef]
- Soutar, A.K.; Sigler, G.F.; Smith, L.C.; Gotto, A.M., Jr.; Sparrow, J.T. Lecithin:cholesterol acyltransferase activation and lipid binding by synthetic fragments of apolipoprotein C-I. Scand. J. Clin. Lab. Invest. Suppl. 1978, 150, 53–58. [Google Scholar] [CrossRef]
- James, P.F.; Dogovski, C.; Dobson, R.C.; Bailey, M.F.; Goldie, K.N.; Karas, J.A.; Scanlon, D.B.; O’Hair, R.A.; Perugini, M.A. Aromatic residues in the C-terminal helix of human apoC-I mediate phospholipid interactions and particle morphology. J. Lipid Res. 2009, 50, 1384–1394. [Google Scholar] [CrossRef] [Green Version]
- Smith, L.E.; Segrest, J.P.; Davidson, W.S. Helical domains that mediate lipid solubilization and ABCA1-specific cholesterol efflux in apolipoproteins C-I and A-II. J. Lipid Res. 2013, 54, 1939–1948. [Google Scholar] [CrossRef] [Green Version]
- Gursky, O. Solution conformation of human apolipoprotein C-1 inferred from proline mutagenesis: far- and near-UV CD study. Biochemistry 2001, 40, 12178–12185. [Google Scholar] [CrossRef]
- Pastorcic, M.; Birnbaum, S.; Hixson, J.E. Baboon apolipoprotein C-I: cDNA and gene structure and evolution. Genomics 1992, 13, 368–374. [Google Scholar] [CrossRef]
- McPherson, A.; Larson, S.B. The structure of human apolipoprotein C-1 in four different crystal forms. J. Lipid Res. 2019, 60, 400–411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gursky, O.; Atkinson, D. Thermodynamic analysis of human plasma apolipoprotein C-1: high-temperature unfolding and low-temperature oligomer dissociation. Biochemistry 1998, 37, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Berbee, J.F.; van der Hoogt, C.C.; Kleemann, R.; Schippers, E.F.; Kitchens, R.L.; van Dissel, J.T.; Bakker-Woudenberg, I.A.; Havekes, L.M.; Rensen, P.C. Apolipoprotein CI stimulates the response to lipopolysaccharide and reduces mortality in gram-negative sepsis. FASEB J. 2006, 20, 2162–2164. [Google Scholar] [CrossRef]
- Kushwaha, R.S.; Hasan, S.Q.; McGill, H.C., Jr.; Getz, G.S.; Dunham, R.G.; Kanda, P. Characterization of cholesteryl ester transfer protein inhibitor from plasma of baboons (Papio sp.). J. Lipid Res. 1993, 34, 1285–1297. [Google Scholar]
- Dumont, L.; Gautier, T.; de Barros, J.P.; Laplanche, H.; Blache, D.; Ducoroy, P.; Fruchart, J.; Fruchart, J.C.; Gambert, P.; Masson, D.; et al. Molecular mechanism of the blockade of plasma cholesteryl ester transfer protein by its physiological inhibitor apolipoprotein CI. J. Biol. Chem. 2005, 280, 38108–38116. [Google Scholar] [CrossRef] [Green Version]
- Segrest, J.P.; Jackson, R.L.; Morrisett, J.D.; Gotto, A.M., Jr. A molecular theory of lipid-protein interactions in the plasma lipoproteins. FEBS Lett 1974, 38, 247–258. [Google Scholar] [CrossRef] [Green Version]
- Meyers, N.L.; Wang, L.; Small, D.M. Apolipoprotein C-I binds more strongly to phospholipid/triolein/water than triolein/water interfaces: a possible model for inhibiting cholesterol ester transfer protein activity and triacylglycerol-rich lipoprotein uptake. Biochemistry 2012, 51, 1238–1248. [Google Scholar] [CrossRef] [Green Version]
- Meyers, N.L.; Wang, L.; Gursky, O.; Small, D.M. Changes in helical content or net charge of apolipoprotein C-I alter its affinity for lipid/water interfaces. J. Lipid Res. 2013, 54, 1927–1938. [Google Scholar] [CrossRef] [Green Version]
- Beesley, C.E.; Young, E.P.; Finnegan, N.; Jackson, M.; Mills, K.; Vellodi, A.; Cleary, M.; Winchester, B.G. Discovery of a new biomarker for the mucopolysaccharidoses (MPS), dipeptidyl peptidase IV (DPP-IV; CD26), by SELDI-TOF mass spectrometry. Mol. Genet. Metab 2009, 96, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Gursky, O. Probing the conformation of a human apolipoprotein C-1 by amino acid substitutions and trimethylamine-N-oxide. Protein Sci 1999, 8, 2055–2064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bjorkegren, J.; Hamsten, A.; Milne, R.W.; Karpe, F. Alterations of VLDL composition during alimentary lipemia. J. Lipid Res. 1997, 38, 301–314. [Google Scholar] [PubMed]
- Bjorkegren, J.; Karpe, F.; Milne, R.W.; Hamsten, A. Differences in apolipoprotein and lipid composition between human chylomicron remnants and very low density lipoproteins isolated from fasting and postprandial plasma. J. Lipid Res. 1998, 39, 1412–1420. [Google Scholar]
- Marcel, Y.L.; Weech, P.K.; Nguyen, T.D.; Milne, R.W.; McConathy, W.J. Apolipoproteins as the basis for heterogeneity in high-density lipoprotein2 and high-density lipoprotein3. Studies by isoelectric focusing on agarose films. Eur J. Biochem 1984, 143, 467–476. [Google Scholar] [CrossRef]
- Hsieh, J.Y.; Chang, C.T.; Huang, M.T.; Chang, C.M.; Chen, C.Y.; Shen, M.Y.; Liao, H.Y.; Wang, G.J.; Chen, C.H.; Chen, C.J.; et al. Biochemical and functional characterization of charge-defined subfractions of high-density lipoprotein from normal adults. Anal. Chem 2013, 85, 11440–11448. [Google Scholar] [CrossRef] [Green Version]
- Curry, M.D.; McConathy, W.J.; Fesmire, J.D.; Alaupovic, P. Quantitative determination of apolipoproteins C-I and C-II in human plasma by separate electroimmunoassays. Clin. Chem 1981, 27, 543–548. [Google Scholar]
- Sehayek, E.; Eisenberg, S. Mechanisms of inhibition by apolipoprotein C of apolipoprotein E-dependent cellular metabolism of human triglyceride-rich lipoproteins through the low density lipoprotein receptor pathway. J. Biol. Chem. 1991, 266, 18259–18267. [Google Scholar]
- Weisgraber, K.H.; Mahley, R.W.; Kowal, R.C.; Herz, J.; Goldstein, J.L.; Brown, M.S. Apolipoprotein C-I modulates the interaction of apolipoprotein E with beta-migrating very low density lipoproteins (beta-VLDL) and inhibits binding of beta-VLDL to low density lipoprotein receptor-related protein. J. Biol. Chem. 1990, 265, 22453–22459. [Google Scholar]
- Shachter, N.S.; Ebara, T.; Ramakrishnan, R.; Steiner, G.; Breslow, J.L.; Ginsberg, H.N.; Smith, J.D. Combined hyperlipidemia in transgenic mice overexpressing human apolipoprotein Cl. J. Clin. Invest. 1996, 98, 846–855. [Google Scholar] [CrossRef] [Green Version]
- Jong, M.C.; van Dijk, K.W.; Dahlmans, V.E.; Van der Boom, H.; Kobayashi, K.; Oka, K.; Siest, G.; Chan, L.; Hofker, M.H.; Havekes, L.M. Reversal of hyperlipidaemia in apolipoprotein C1 transgenic mice by adenovirus-mediated gene delivery of the low-density-lipoprotein receptor, but not by the very-low-density-lipoprotein receptor. Biochem. J. 1999, 338((Pt. 2)), 281–287. [Google Scholar] [CrossRef]
- de Haan, W.; Out, R.; Berbee, J.F.; van der Hoogt, C.C.; van Dijk, K.W.; van Berkel, T.J.; Romijn, J.A.; Jukema, J.W.; Havekes, L.M.; Rensen, P.C. Apolipoprotein CI inhibits scavenger receptor BI and increases plasma HDL levels in vivo. Biochem. Biophys Res. Commun. 2008, 377, 1294–1298. [Google Scholar] [CrossRef] [PubMed]
- Soutar, A.K.; Garner, C.W.; Baker, H.N.; Sparrow, J.T.; Jackson, R.L.; Gotto, A.M.; Smith, L.C. Effect of the human plasma apolipoproteins and phosphatidylcholine acyl donor on the activity of lecithin: cholesterol acyltransferase. Biochemistry 1975, 14, 3057–3064. [Google Scholar] [CrossRef] [PubMed]
- Albers, J.J.; Lin, J.; Roberts, G.P. Effect of human plasma apolipoproteins on the activity of purified lecithin: cholesterol acyltransferase. Artery 1979, 5, 61–75. [Google Scholar] [PubMed]
- Verine, A.; Benkirane, M.; Meignen, J.M.; Boyer, J. Lipoprotein lipase in rat heart--II. Influence of apolipoproteins and nutritional factors on tri-, di- and monoacylglycerol lipase activities in post-heparin effluents. Comp. Biochem. Physiol. B 1989, 94, 19–25. [Google Scholar] [CrossRef]
- Westerterp, M.; de Haan, W.; Berbee, J.F.; Havekes, L.M.; Rensen, P.C. Endogenous apoC-I increases hyperlipidemia in apoE-knockout mice by stimulating VLDL production and inhibiting LPL. J. Lipid Res. 2006, 47, 1203–1211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dautin, G.; Soltani, Z.; Ducloux, D.; Gautier, T.; Pais de Barros, J.P.; Gambert, P.; Lagrost, L.; Masson, D. Hemodialysis reduces plasma apolipoprotein C-I concentration making VLDL a better substrate for lipoprotein lipase. Kidney Int. 2007, 72, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M.; Vorrsjo, E.; Talmud, P.; Lookene, A.; Olivecrona, G. Apolipoproteins C-I and C-III inhibit lipoprotein lipase activity by displacement of the enzyme from lipid droplets. J. Biol. Chem. 2013, 288, 33997–34008. [Google Scholar] [CrossRef] [Green Version]
- Aryal, B.; Price, N.L.; Suarez, Y.; Fernandez-Hernando, C. ANGPTL4 in Metabolic and Cardiovascular Disease. Trends Mol. Med. 2019, 25, 723–734. [Google Scholar] [CrossRef]
- Theofilatos, D.; Fotakis, P.; Valanti, E.; Sanoudou, D.; Zannis, V.; Kardassis, D. HDL-apoA-I induces the expression of angiopoietin like 4 (ANGPTL4) in endothelial cells via a PI3K/AKT/FOXO1 signaling pathway. Metabolism 2018, 87, 36–47. [Google Scholar] [CrossRef]
- Shinomiya, M.; Sasaki, N.; Barnhart, R.L.; Shirai, K.; Jackson, R.L. Effect of apolipoproteins on the hepatic lipase-catalyzed hydrolysis of human plasma high density lipoprotein2-triacylglycerols. Biochim. Biophys. Acta 1982, 713, 292–299. [Google Scholar] [PubMed]
- Conde-Knape, K.; Bensadoun, A.; Sobel, J.H.; Cohn, J.S.; Shachter, N.S. Overexpression of apoC-I in apoE-null mice: severe hypertriglyceridemia due to inhibition of hepatic lipase. J. Lipid Res. 2002, 43, 2136–2145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poensgen, J. Apolipoprotein C-1 inhibits the hydrolysis by phospholipase A2 of phospholipids in liposomes and cell membranes. Biochim Biophys Acta 1990, 1042, 188–192. [Google Scholar] [CrossRef]
- Westerterp, M.; Berbee, J.F.; Delsing, D.J.; Jong, M.C.; Gijbels, M.J.; Dahlmans, V.E.; Offerman, E.H.; Romijn, J.A.; Havekes, L.M.; Rensen, P.C. Apolipoprotein C-I binds free fatty acids and reduces their intracellular esterification. J. Lipid Res. 2007, 48, 1353–1361. [Google Scholar] [CrossRef] [Green Version]
- Jong, M.C.; Dahlmans, V.E.; van Gorp, P.J.; van Dijk, K.W.; Breuer, M.L.; Hofker, M.H.; Havekes, L.M. In the absence of the low density lipoprotein receptor, human apolipoprotein C1 overexpression in transgenic mice inhibits the hepatic uptake of very low density lipoproteins via a receptor-associated protein-sensitive pathway. J. Clin. Invest. 1996, 98, 2259–2267. [Google Scholar] [CrossRef] [Green Version]
- Simonet, W.S.; Bucay, N.; Pitas, R.E.; Lauer, S.J.; Taylor, J.M. Multiple tissue-specific elements control the apolipoprotein E/C-I gene locus in transgenic mice. J. Biol. Chem. 1991, 266, 8651–8654. [Google Scholar]
- Berbee, J.F.; van der Hoogt, C.C.; Sundararaman, D.; Havekes, L.M.; Rensen, P.C. Severe hypertriglyceridemia in human APOC1 transgenic mice is caused by apoC-I-induced inhibition of LPL. J. Lipid Res. 2005, 46, 297–306. [Google Scholar] [CrossRef] [Green Version]
- Gautier, T.; Masson, D.; Lagrost, L. The potential of cholesteryl ester transfer protein as a therapeutic target. Expert. Opin. Ther. Targets 2016, 20, 47–59. [Google Scholar] [CrossRef]
- Armitage, J.; Holmes, M.V.; Preiss, D. Cholesteryl Ester Transfer Protein Inhibition for Preventing Cardiovascular Events: JACC Review Topic of the Week. J. Am. Coll Cardiol 2019, 73, 477–487. [Google Scholar] [CrossRef]
- Bouillet, B.; Gautier, T.; Blache, D.; Pais de Barros, J.P.; Duvillard, L.; Petit, J.M.; Lagrost, L.; Verges, B. Glycation of apolipoprotein C1 impairs its CETP inhibitory property: pathophysiological relevance in patients with type 1 and type 2 diabetes. Diabetes Care 2014, 37, 1148–1156. [Google Scholar] [CrossRef] [Green Version]
- De Barros, J.P.; Boualam, A.; Gautier, T.; Dumont, L.; Verges, B.; Masson, D.; Lagrost, L. Apolipoprotein CI is a physiological regulator of cholesteryl ester transfer protein activity in human plasma but not in rabbit plasma. J. Lipid Res. 2009, 50, 1842–1851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiao, S.; Cole, T.G.; Kitchens, R.T.; Pfleger, B.; Schonfeld, G. Genetic heterogeneity of lipoproteins in inbred strains of mice: analysis by gel-permeation chromatography. Metabolism 1990, 39, 155–160. [Google Scholar] [CrossRef]
- Gautier, T.; Masson, D.; Jong, M.C.; Duverneuil, L.; Le Guern, N.; Deckert, V.; Pais de Barros, J.P.; Dumont, L.; Bataille, A.; Zak, Z.; et al. Apolipoprotein CI deficiency markedly augments plasma lipoprotein changes mediated by human cholesteryl ester transfer protein (CETP) in CETP transgenic/ApoCI-knocked out mice. J. Biol. Chem. 2002, 277, 31354–31363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gautier, T.; Masson, D.; Jong, M.C.; Pais de Barros, J.P.; Duverneuil, L.; Le Guern, N.; Deckert, V.; Dumont, L.; Bataille, A.; Zak, Z.; et al. Apolipoprotein CI overexpression is not a relevant strategy to block cholesteryl ester transfer protein (CETP) activity in CETP transgenic mice. Biochem. J. 2005, 385, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.H.; Fu, Y.C.; Zhang, D.W.; Yin, K.; Tang, C.K. Foam cells in atherosclerosis. Clin. Chim. Acta 2013, 424, 245–252. [Google Scholar] [CrossRef] [Green Version]
- Domschke, G.; Linden, F.; Pawig, L.; Hafner, A.; Akhavanpoor, M.; Reymann, J.; Doesch, A.O.; Erbel, C.; Weber, C.; Katus, H.A.; et al. Systematic RNA-interference in primary human monocyte-derived macrophages: A high-throughput platform to study foam cell formation. Sci. Rep. 2018, 8, 10516. [Google Scholar] [CrossRef] [Green Version]
- Kolmakova, A.; Kwiterovich, P.; Virgil, D.; Alaupovic, P.; Knight-Gibson, C.; Martin, S.F.; Chatterjee, S. Apolipoprotein C-I induces apoptosis in human aortic smooth muscle cells via recruiting neutral sphingomyelinase. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 264–269. [Google Scholar] [CrossRef] [Green Version]
- Ference, B.A.; Ginsberg, H.N.; Graham, I.; Ray, K.K.; Packard, C.J.; Bruckert, E.; Hegele, R.A.; Krauss, R.M.; Raal, F.J.; Schunkert, H.; et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2017, 38, 2459–2472. [Google Scholar] [CrossRef] [Green Version]
- Peng, J.; Luo, F.; Ruan, G.; Peng, R.; Li, X. Hypertriglyceridemia and atherosclerosis. Lipids Health Dis. 2017, 16, 233. [Google Scholar] [CrossRef] [Green Version]
- Sandesara, P.B.; Virani, S.S.; Fazio, S.; Shapiro, M.D. The Forgotten Lipids: Triglycerides, Remnant Cholesterol, and Atherosclerotic Cardiovascular Disease Risk. Endocr. Rev. 2019, 40, 537–557. [Google Scholar] [CrossRef] [Green Version]
- Bjorkegren, J.; Silveira, A.; Boquist, S.; Tang, R.; Karpe, F.; Bond, M.G.; de Faire, U.; Hamsten, A. Postprandial enrichment of remnant lipoproteins with apoC-I in healthy normolipidemic men with early asymptomatic atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1470–1474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamsten, A.; Silveira, A.; Boquist, S.; Tang, R.; Bond, M.G.; de Faire, U.; Bjorkegren, J. The apolipoprotein CI content of triglyceride-rich lipoproteins independently predicts early atherosclerosis in healthy middle-aged men. J. Am. Coll Cardiol. 2005, 45, 1013–1017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hansen, J.B.; Fernandez, J.A.; Noto, A.T.; Deguchi, H.; Bjorkegren, J.; Mathiesen, E.B. The apolipoprotein C-I content of very-low-density lipoproteins is associated with fasting triglycerides, postprandial lipemia, and carotid atherosclerosis. J. Lipids 2011, 2011, 271062. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noto, A.T.; Mathiesen, E.B.; Brox, J.; Bjorkegren, J.; Hansen, J.B. The ApoC-I content of VLDL particles is associated with plaque size in persons with carotid atherosclerosis. Lipids 2008, 43, 673–679. [Google Scholar] [CrossRef] [PubMed]
- van der Ham, R.L.; Alizadeh Dehnavi, R.; Berbee, J.F.; Putter, H.; de Roos, A.; Romijn, J.A.; Rensen, P.C.; Tamsma, J.T. Plasma apolipoprotein CI and CIII levels are associated with increased plasma triglyceride levels and decreased fat mass in men with the metabolic syndrome. Diabetes Care 2009, 32, 184–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Ham, R.L.; Dehnavi, R.A.; van den Berg, G.A.; Putter, H.; de Roos, A.; Berbee, J.F.; Romijn, J.A.; Rensen, P.C.; Tamsma, J.T. Apolipoprotein CI levels are associated with atherosclerosis in men with the metabolic syndrome and systemic inflammation. Atherosclerosis 2009, 203, 355–357. [Google Scholar] [CrossRef] [PubMed]
- Wild, R.A. Dyslipidemia in PCOS. Steroids 2012, 77, 295–299. [Google Scholar] [CrossRef]
- Huang, S.; Qiao, J.; Li, R.; Wang, L.; Li, M. Can serum apolipoprotein C-I demonstrate metabolic abnormality early in women with polycystic ovary syndrome? Fertil Steril 2010, 94, 205–210. [Google Scholar] [CrossRef]
- Wassef, H.; Bissonnette, S.; Dufour, R.; Davignon, J.; Faraj, M. Enrichment of Triglyceride-Rich Lipoproteins with Apolipoprotein C-I Is Positively Associated with Their Delayed Plasma Clearance Independently of Other Transferable Apolipoproteins in Postmenopausal Overweight and Obese Women. J. Nutr. 2017, 147, 754–762. [Google Scholar] [CrossRef]
- Eo, H.S.; Lee, K.B.; Kim, A.K.; Kim, M.H.; Kim, D.H.; Kim, D.I. Association with inflammatory cells and apolipoproteins to the progression of atherosclerosis. J. Korean Surg. Soc. 2011, 80, 289–296. [Google Scholar] [CrossRef]
- Rezeli, M.; Vegvari, A.; Donnarumma, F.; Gidlof, O.; Smith, J.G.; Erlinge, D.; Marko-Varga, G. Development of an MRM assay panel with application to biobank samples from patients with myocardial infarction. J. Proteomics 2013, 87, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.; McNeal, C.; Macfarlane, R. Isoforms of apolipoprotein C-I associated with individuals with coronary artery disease. Biochem. Biophys. Res. Commun. 2011, 404, 1034–1038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, C.T.; Liao, H.Y.; Chang, C.M.; Chen, C.Y.; Chen, C.H.; Yang, C.Y.; Tsai, F.J.; Chen, C.J. Oxidized ApoC1 on MALDI-TOF and glycated-ApoA1 band on gradient gel as potential diagnostic tools for atherosclerotic vascular disease. Clin. Chim. Acta 2013, 420, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Manduteanu, I.; Simionescu, M. Inflammation in atherosclerosis: a cause or a result of vascular disorders? J. Cell Mol. Med. 2012, 16, 1978–1990. [Google Scholar] [CrossRef] [Green Version]
- Libby, P.; Hansson, G.K. From Focal Lipid Storage to Systemic Inflammation: JACC Review Topic of the Week. J. Am. Coll Cardiol. 2019, 74, 1594–1607. [Google Scholar] [CrossRef]
- Westerterp, M.; Berbee, J.F.; Pires, N.M.; van Mierlo, G.J.; Kleemann, R.; Romijn, J.A.; Havekes, L.M.; Rensen, P.C. Apolipoprotein C-I is crucially involved in lipopolysaccharide-induced atherosclerosis development in apolipoprotein E-knockout mice. Circulation 2007, 116, 2173–2181. [Google Scholar] [CrossRef] [Green Version]
- Chen, R.; Zhang, F.; Song, L.; Shu, Y.; Lin, Y.; Dong, L.; Nie, X.; Zhang, D.; Chen, P.; Han, M. Transcriptome profiling reveals that the SM22alpha-regulated molecular pathways contribute to vascular pathology. J. Mol. Cell Cardiol. 2014, 72, 263–272. [Google Scholar] [CrossRef] [Green Version]
- Yin, C.; Ackermann, S.; Ma, Z.; Mohanta, S.K.; Zhang, C.; Li, Y.; Nietzsche, S.; Westermann, M.; Peng, L.; Hu, D.; et al. ApoE attenuates unresolvable inflammation by complex formation with activated C1q. Nat. Med. 2019, 25, 496–506. [Google Scholar] [CrossRef]
- Dittrich, J.; Beutner, F.; Teren, A.; Thiery, J.; Burkhardt, R.; Scholz, M.; Ceglarek, U. Plasma levels of apolipoproteins C-III, A-IV, and E are independently associated with stable atherosclerotic cardiovascular disease. Atherosclerosis 2019, 281, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Bouchard, C.; Dubuc, G.; Davignon, J.; Bernier, L.; Cohn, J.S. Post-transcriptional regulation of apoC-I synthesis and secretion in human HepG2 cells. Atherosclerosis 2005, 178, 257–264. [Google Scholar] [CrossRef]
- Castilho, L.N.; Chamberland, A.; Boulet, L.; Davignon, J.; Cohn, J.S.; Bernier, L. Effect of atorvastatin on ApoE and ApoC-I synthesis and secretion by THP-1 macrophages. J. Cardiovasc. Pharmacol. 2003, 42, 251–257. [Google Scholar] [CrossRef]
- Barber, M.J.; Mangravite, L.M.; Hyde, C.L.; Chasman, D.I.; Smith, J.D.; McCarty, C.A.; Li, X.; Wilke, R.A.; Rieder, M.J.; Williams, P.T.; et al. Genome-wide association of lipid-lowering response to statins in combined study populations. PLoS ONE 2010, 5, e9763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aslibekyan, S.; Goodarzi, M.O.; Frazier-Wood, A.C.; Yan, X.; Irvin, M.R.; Kim, E.; Tiwari, H.K.; Guo, X.; Straka, R.J.; Taylor, K.D.; et al. Variants identified in a GWAS meta-analysis for blood lipids are associated with the lipid response to fenofibrate. PLoS ONE 2012, 7, e48663. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trompet, S.; de Craen, A.J.; Postmus, I.; Ford, I.; Sattar, N.; Caslake, M.; Stott, D.J.; Buckley, B.M.; Sacks, F.; Devlin, J.J.; et al. Replication of LDL GWAs hits in PROSPER/PHASE as validation for future (pharmaco)genetic analyses. BMC Med. Genet. 2011, 12, 131. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Zhou, X.; Ye, S.; Han, D.; Tan, X.; Zheng, F.; Shi, Q. Combined effects of apoE-CI-CII cluster and LDL-R gene polymorphisms on chromosome 19 and coronary artery disease risk. Int. J. Hyg. Environ. Health 2006, 209, 265–273. [Google Scholar] [CrossRef]
- Remaley, A.T.; Stonik, J.A.; Demosky, S.J.; Neufeld, E.B.; Bocharov, A.V.; Vishnyakova, T.G.; Eggerman, T.L.; Patterson, A.P.; Duverger, N.J.; Santamarina-Fojo, S.; et al. Apolipoprotein specificity for lipid efflux by the human ABCAI transporter. Biochem. Biophys. Res. Commun. 2001, 280, 818–823. [Google Scholar] [CrossRef]
- Swertfeger, D.K.; Li, H.; Rebholz, S.; Zhu, X.; Shah, A.S.; Davidson, W.S.; Lu, L.J. Mapping Atheroprotective Functions and Related Proteins/Lipoproteins in Size Fractionated Human Plasma. Mol. Cell Proteomics 2017, 16, 680–693. [Google Scholar] [CrossRef] [Green Version]
- Yan, L.R.; Wang, D.X.; Liu, H.; Zhang, X.X.; Zhao, H.; Hua, L.; Xu, P.; Li, Y.S. A pro-atherogenic HDL profile in coronary heart disease patients: An iTRAQ labelling-based proteomic approach. PLoS ONE 2014, 9, e98368. [Google Scholar] [CrossRef]
- Westerterp, M.; Van Eck, M.; de Haan, W.; Offerman, E.H.; Van Berkel, T.J.; Havekes, L.M.; Rensen, P.C. Apolipoprotein CI aggravates atherosclerosis development in ApoE-knockout mice despite mediating cholesterol efflux from macrophages. Atherosclerosis 2007, 195, e9–e16. [Google Scholar] [CrossRef]
- Diez, D.; Wheelock, A.M.; Goto, S.; Haeggstrom, J.Z.; Paulsson-Berne, G.; Hansson, G.K.; Hedin, U.; Gabrielsen, A.; Wheelock, C.E. The use of network analyses for elucidating mechanisms in cardiovascular disease. Mol. Biosyst. 2010, 6, 289–304. [Google Scholar] [CrossRef] [Green Version]
- Rouhani, N.; Young, E.; Chatterjee, C.; Sparks, D.L. HDL composition regulates displacement of cell surface-bound hepatic lipase. Lipids 2008, 43, 793–804. [Google Scholar] [CrossRef]
- Jansen, H.; Verhoeven, A.J.; Sijbrands, E.J. Hepatic lipase: A pro- or anti-atherogenic protein? J. Lipid Res. 2002, 43, 1352–1362. [Google Scholar] [CrossRef] [Green Version]
- Berbee, J.F.; Havekes, L.M.; Rensen, P.C. Apolipoproteins modulate the inflammatory response to lipopolysaccharide. J. Endotoxin Res. 2005, 11, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Barlage, S.; Frohlich, D.; Bottcher, A.; Jauhiainen, M.; Muller, H.P.; Noetzel, F.; Rothe, G.; Schutt, C.; Linke, R.P.; Lackner, K.J.; et al. ApoE-containing high density lipoproteins and phospholipid transfer protein activity increase in patients with a systemic inflammatory response. J. Lipid Res. 2001, 42, 281–290. [Google Scholar]
- Berbee, J.F.; van der Hoogt, C.C.; de Haas, C.J.; van Kessel, K.P.; Dallinga-Thie, G.M.; Romijn, J.A.; Havekes, L.M.; van Leeuwen, H.J.; Rensen, P.C. Plasma apolipoprotein CI correlates with increased survival in patients with severe sepsis. Intensive Care Med. 2008, 34, 907–911. [Google Scholar] [CrossRef] [PubMed]
- Berbee, J.F.; Mooijaart, S.P.; de Craen, A.J.; Havekes, L.M.; van Heemst, D.; Rensen, P.C.; Westendorp, R.G. Plasma apolipoprotein CI protects against mortality from infection in old age. J. Gerontol. A Biol. Sci. Med. Sci. 2008, 63, 122–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, N.K.; Ferreira, B.L.; Tashima, A.K.; Brunialti, M.K.C.; Torquato, R.J.S.; Bafi, A.; Assuncao, M.; Azevedo, L.C.P.; Salomao, R. Lipid metabolism impairment in patients with sepsis secondary to hospital acquired pneumonia, a proteomic analysis. Clin. Proteomics 2019, 16, 29. [Google Scholar] [CrossRef]
- Schippers, E.F.; Berbee, J.F.; van Disseldorp, I.M.; Versteegh, M.I.; Havekes, L.M.; Rensen, P.C.; van Dissel, J.T. Preoperative apolipoprotein CI levels correlate positively with the proinflammatory response in patients experiencing endotoxemia following elective cardiac surgery. Intensive Care Med. 2008, 34, 1492–1497. [Google Scholar] [CrossRef] [Green Version]
- Berbee, J.F.; Coomans, C.P.; Westerterp, M.; Romijn, J.A.; Havekes, L.M.; Rensen, P.C. Apolipoprotein CI enhances the biological response to LPS via the CD14/TLR4 pathway by LPS-binding elements in both its N- and C-terminal helix. J. Lipid Res. 2010, 51, 1943–1952. [Google Scholar] [CrossRef] [Green Version]
- Achkar, J.M.; Cortes, L.; Croteau, P.; Yanofsky, C.; Mentinova, M.; Rajotte, I.; Schirm, M.; Zhou, Y.; Junqueira-Kipnis, A.P.; Kasprowicz, V.O.; et al. Host Protein Biomarkers Identify Active Tuberculosis in HIV Uninfected and Co-infected Individuals. EBioMedicine 2015, 2, 1160–1168. [Google Scholar] [CrossRef] [Green Version]
- Devadas, K.; Biswas, S.; Haleyurgirisetty, M.; Wood, O.; Ragupathy, V.; Lee, S.; Hewlett, I. Analysis of Host Gene Expression Profile in HIV-1 and HIV-2 Infected T-Cells. PLoS ONE 2016, 11, e0147421. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, L.; Xu, F.; Qi, H.; Shen, C.; Jiao, W.; Xiao, J.; Li, Q.; Xu, B.; Shen, A. Screening and Identification of APOC1 as a Novel Potential Biomarker for Differentiate of Mycoplasma pneumoniae in Children. Front. Microbiol. 2016, 7, 1961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, H.; Fei, N.; Wu, G.; Zhang, C.; Zhao, L.; Zhang, M. Regulated Inflammation and Lipid Metabolism in Colon mRNA Expressions of Obese Germfree Mice Responding to Enterobacter cloacae B29 Combined with the High Fat Diet. Front. Microbiol. 2016, 7, 1786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cortes-Selva, D.; Elvington, A.F.; Ready, A.; Rajwa, B.; Pearce, E.J.; Randolph, G.J.; Fairfax, K.C. Schistosoma mansoni Infection-Induced Transcriptional Changes in Hepatic Macrophage Metabolism Correlate With an Athero-Protective Phenotype. Front. Immunol. 2018, 9, 2580. [Google Scholar] [CrossRef]
- Wu, D.; Qi, G.; Wang, X.; Xu, M.; Rong, R.; Wang, X.; Zhu, T. Hematopoietic stem cell transplantation induces immunologic tolerance in renal transplant patients via modulation of inflammatory and repair processes. J. Transl. Med. 2012, 10, 182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serra-Grabulosa, J.M.; Salgado-Pineda, P.; Junque, C.; Sole-Padulles, C.; Moral, P.; Lopez-Alomar, A.; Lopez, T.; Lopez-Guillen, A.; Bargallo, N.; Mercader, J.M.; et al. Apolipoproteins E and C1 and brain morphology in memory impaired elders. Neurogenetics 2003, 4, 141–146. [Google Scholar] [CrossRef]
- Abildayeva, K.; Berbee, J.F.; Blokland, A.; Jansen, P.J.; Hoek, F.J.; Meijer, O.; Lutjohann, D.; Gautier, T.; Pillot, T.; De Vente, J.; et al. Human apolipoprotein C-I expression in mice impairs learning and memory functions. J. Lipid Res. 2008, 49, 856–869. [Google Scholar] [CrossRef] [Green Version]
- Berbee, J.F.; Vanmierlo, T.; Abildayeva, K.; Blokland, A.; Jansen, P.J.; Lutjohann, D.; Gautier, T.; Sijbrands, E.; Prickaerts, J.; Hadfoune, M.; et al. Apolipoprotein CI knock-out mice display impaired memory functions. J. Alzheimers Dis. 2011, 23, 737–747. [Google Scholar] [CrossRef]
- Cudaback, E.; Li, X.; Yang, Y.; Yoo, T.; Montine, K.S.; Craft, S.; Montine, T.J.; Keene, C.D. Apolipoprotein C-I is an APOE genotype-dependent suppressor of glial activation. J. Neuroinflammation 2012, 9, 192. [Google Scholar] [CrossRef]
- Zhou, X.; Chen, Y.; Mok, K.Y.; Kwok, T.C.Y.; Mok, V.C.T.; Guo, Q.; Ip, F.C.; Chen, Y.; Mullapudi, N.; Alzheimer’s Disease Neuroimaging, I.; et al. Non-coding variability at the APOE locus contributes to the Alzheimer’s risk. Nat. Commun. 2019, 10, 3310. [Google Scholar] [CrossRef] [Green Version]
- Prendecki, M.; Florczak-Wyspianska, J.; Kowalska, M.; Ilkowski, J.; Grzelak, T.; Bialas, K.; Wiszniewska, M.; Kozubski, W.; Dorszewska, J. Biothiols and oxidative stress markers and polymorphisms of TOMM40 and APOC1 genes in Alzheimer’s disease patients. Oncotarget 2018, 9, 35207–35225. [Google Scholar] [CrossRef] [PubMed]
- Kulminski, A.M.; Huang, J.; Wang, J.; He, L.; Loika, Y.; Culminskaya, I. Apolipoprotein E region molecular signatures of Alzheimer’s disease. Aging Cell 2018, 17, e12779. [Google Scholar] [CrossRef] [PubMed]
- Yashin, A.I.; Fang, F.; Kovtun, M.; Wu, D.; Duan, M.; Arbeev, K.; Akushevich, I.; Kulminski, A.; Culminskaya, I.; Zhbannikov, I.; et al. Hidden heterogeneity in Alzheimer’s disease: Insights from genetic association studies and other analyses. Exp. Gerontol. 2018, 107, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Cui, Z.; Shen, L.; Ji, H.F. Shared Genetic Etiology between Type 2 Diabetes and Alzheimer’s Disease Identified by Bioinformatics Analysis. J. Alzheimers. Dis. 2016, 50, 13–17. [Google Scholar] [CrossRef]
- Shao, Y.; Shaw, M.; Todd, K.; Khrestian, M.; D’Aleo, G.; Barnard, P.J.; Zahratka, J.; Pillai, J.; Yu, C.E.; Keene, C.D.; et al. DNA methylation of TOMM40-APOE-APOC2 in Alzheimer’s disease. J. Hum. Genet. 2018, 63, 459–471. [Google Scholar] [CrossRef]
- Jong, M.C.; Gijbels, M.J.; Dahlmans, V.E.; Gorp, P.J.; Koopman, S.J.; Ponec, M.; Hofker, M.H.; Havekes, L.M. Hyperlipidemia and cutaneous abnormalities in transgenic mice overexpressing human apolipoprotein C1. J. Clin. Invest. 1998, 101, 145–152. [Google Scholar] [CrossRef] [Green Version]
- Nagelkerken, L.; Verzaal, P.; Lagerweij, T.; Persoon-Deen, C.; Berbee, J.F.; Prens, E.P.; Havekes, L.M.; Oranje, A.P. Development of atopic dermatitis in mice transgenic for human apolipoprotein C1. J. Invest. Dermatol 2008, 128, 1165–1172. [Google Scholar] [CrossRef] [Green Version]
- Oranje, A.P.; Verbeek, R.; Verzaal, P.; Haspels, I.; Prens, E.; Nagelkerken, L. Wet-wrap treatment using dilutions of tacrolimus ointment and fluticasone propionate cream in human APOC1 (+/+) mice with atopic dermatitis. Br. J. Dermatol. 2009, 160, 54–61. [Google Scholar] [CrossRef]
- Mariman, R.; Reefman, E.; Tielen, F.; Persoon-Deen, C.; van de Mark, K.; Worms, N.; Koning, F.; Nagelkerken, L. Lactobacillus plantarum NCIMB8826 ameliorates inflammation of colon and skin in human APOC1 transgenic mice. Benef. Microbes. 2016, 7, 215–225. [Google Scholar] [CrossRef]
- Scharschmidt, T.C.; Segre, J.A. Modeling atopic dermatitis with increasingly complex mouse models. J. Invest. Dermatol. 2008, 128, 1061–1064. [Google Scholar] [CrossRef] [Green Version]
- MacManes, M.D.; Eisen, M.B. Characterization of the transcriptome, nucleotide sequence polymorphism, and natural selection in the desert adapted mouse Peromyscus eremicus. PeerJ 2014, 2, e642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fukuhara, T.; Wada, M.; Nakamura, S.; Ono, C.; Shiokawa, M.; Yamamoto, S.; Motomura, T.; Okamoto, T.; Okuzaki, D.; Yamamoto, M.; et al. Amphipathic alpha-helices in apolipoproteins are crucial to the formation of infectious hepatitis C virus particles. PLoS Pathog. 2014, 10, e1004534. [Google Scholar] [CrossRef] [PubMed]
- Hueging, K.; Doepke, M.; Vieyres, G.; Bankwitz, D.; Frentzen, A.; Doerrbecker, J.; Gumz, F.; Haid, S.; Wolk, B.; Kaderali, L.; et al. Apolipoprotein E codetermines tissue tropism of hepatitis C virus and is crucial for viral cell-to-cell transmission by contributing to a postenvelopment step of assembly. J. Virol. 2014, 88, 1433–1446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hueging, K.; Weller, R.; Doepke, M.; Vieyres, G.; Todt, D.; Wolk, B.; Vondran, F.W.; Geffers, R.; Lauber, C.; Kaderali, L.; et al. Several Human Liver Cell Expressed Apolipoproteins Complement HCV Virus Production with Varying Efficacy Conferring Differential Specific Infectivity to Released Viruses. PLoS ONE 2015, 10, e0134529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meunier, J.C.; Russell, R.S.; Engle, R.E.; Faulk, K.N.; Purcell, R.H.; Emerson, S.U. Apolipoprotein c1 association with hepatitis C virus. J. Virol. 2008, 82, 9647–9656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tournier, J.F.; Bayard, F.; Tauber, J.P. Rapid purification and activity of apolipoprotein C1 on the proliferation of bovine vascular endothelial cells in vitro. Biochim. Biophys. Acta 1984, 804, 216–220. [Google Scholar] [CrossRef]
- Su, W.P.; Sun, L.N.; Yang, S.L.; Zhao, H.; Zeng, T.Y.; Wu, W.Z.; Wang, D. Apolipoprotein C1 promotes prostate cancer cell proliferation in vitro. J. Biochem. Mol. Toxicol. 2018, 32, e22158. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto-Ishikawa, K.; Suzuki, H.; Nezu, M.; Kamiya, N.; Imamoto, T.; Komiya, A.; Sogawa, K.; Tomonaga, T.; Nomura, F.; Ichikawa, T. The isolation and identification of apolipoprotein C-I in hormone-refractory prostate cancer using surface-enhanced laser desorption/ionization time-of-flight mass spectrometry. Asian J. Androl. 2009, 11, 299–307. [Google Scholar] [CrossRef] [Green Version]
- Ren, H.; Chen, Z.; Yang, L.; Xiong, W.; Yang, H.; Xu, K.; Zhai, E.; Ding, L.; He, Y.; Song, X. Apolipoprotein C1 (APOC1) promotes tumor progression via MAPK signaling pathways in colorectal cancer. Cancer Manag. Res. 2019, 11, 4917–4930. [Google Scholar] [CrossRef] [Green Version]
- Takano, S.; Yoshitomi, H.; Togawa, A.; Sogawa, K.; Shida, T.; Kimura, F.; Shimizu, H.; Tomonaga, T.; Nomura, F.; Miyazaki, M. Apolipoprotein C-1 maintains cell survival by preventing from apoptosis in pancreatic cancer cells. Oncogene 2008, 27, 2810–2822. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, J.; Guo, F.; Zhao, W.; Zhan, Y.; Liu, C.; Fan, Y.; Wang, J. Identification of Apolipoprotein C-I Peptides as a Potential Biomarker and its Biological Roles in Breast Cancer. Med. Sci. Monit. 2016, 22, 1152–1160. [Google Scholar] [CrossRef] [Green Version]
- Song, D.; Yue, L.; Zhang, J.; Ma, S.; Zhao, W.; Guo, F.; Fan, Y.; Yang, H.; Liu, Q.; Zhang, D.; et al. Diagnostic and prognostic significance of serum apolipoprotein C-I in triple-negative breast cancer based on mass spectrometry. Cancer Biol. Ther. 2016, 17, 635–647. [Google Scholar] [CrossRef] [PubMed]
- Chung, L.; Moore, K.; Phillips, L.; Boyle, F.M.; Marsh, D.J.; Baxter, R.C. Novel serum protein biomarker panel revealed by mass spectrometry and its prognostic value in breast cancer. Breast Cancer Res. 2014, 16, R63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, H.L.; Wang, Y.S.; Fong, W.L.; Chi, M.S.; Chi, K.H.; Kao, S.J. Apolipoprotein C1 (APOC1) as a novel diagnostic and prognostic biomarker for lung cancer: A marker phase I trial. Thorac. Cancer 2014, 5, 500–508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Guo, F.; Wang, L.; Zhao, W.; Zhang, D.; Yang, H.; Yu, J.; Niu, L.; Yang, F.; Zheng, S.; et al. Identification of apolipoprotein C-I as a potential Wilms’ tumor marker after excluding inflammatory factors. Int. J. Mol. Sci. 2014, 15, 16186–16195. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Pan, C.; Liang, Y. Screening and identification of serum proteomic biomarkers for gastric adenocarcinoma. Exp. Ther. Med. 2012, 3, 1005–1009. [Google Scholar] [CrossRef]
- Luczak, M.; Kubicki, T.; Rzetelska, Z.; Szczepaniak, T.; Przybylowicz-Chalecka, A.; Ratajczak, B.; Czerwinska-Rybak, J.; Nowicki, A.; Joks, M.; Jakubowiak, A.; et al. Comparative proteomic profiling of sera from patients with refractory multiple myeloma reveals potential biomarkers predicting response to bortezomib-based therapy. Pol. Arch. Intern. Med. 2017, 127, 392–400. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Gong, Y.; Deng, T.; Zhang, L.; Liao, X.; Han, C.; Yang, C.; Huang, J.; Wang, Q.; Song, X.; et al. Diagnostic and prognostic significance of mRNA expressions of apolipoprotein A and C family genes in hepatitis B virus-related hepatocellular carcinoma. J. Cell Biochem. 2019, 120, 18246–18265. [Google Scholar] [CrossRef]
- Tomkin, G.H. The intestine as a regulator of cholesterol homeostasis in diabetes. Atheroscler. Suppl. 2008, 9, 27–32. [Google Scholar] [CrossRef]
- Bouillet, B.; Gautier, T.; Rouland, A.; Duvillard, L.; Petit, J.M.; Lagrost, L.; Verges, B. Plasma apolipoprotein C1 concentration is associated with plasma triglyceride concentration but not with visceral fat and liver fat content in people with type 1 diabetes. Acta. Diabetol. 2019, 56, 1155–1157. [Google Scholar] [CrossRef]
- Bouillet, B.; Gautier, T.; Aho, L.S.; Duvillard, L.; Petit, J.M.; Lagrost, L.; Verges, B. Plasma apolipoprotein C1 concentration is associated with plasma triglyceride concentration, but not visceral fat, in patients with type 2 diabetes. Diabetes Metab. 2016, 42, 263–266. [Google Scholar] [CrossRef]
- Jong, M.C.; Voshol, P.J.; Muurling, M.; Dahlmans, V.E.; Romijn, J.A.; Pijl, H.; Havekes, L.M. Protection from obesity and insulin resistance in mice overexpressing human apolipoprotein C1. Diabetes 2001, 50, 2779–2785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bus, P.; Pierneef, L.; Bor, R.; Wolterbeek, R.; van Es, L.A.; Rensen, P.C.; de Heer, E.; Havekes, L.M.; Bruijn, J.A.; Berbee, J.F.; et al. Apolipoprotein C-I plays a role in the pathogenesis of glomerulosclerosis. J. Pathol. 2017, 241, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Overgaard, A.J.; Hansen, H.G.; Lajer, M.; Pedersen, L.; Tarnow, L.; Rossing, P.; McGuire, J.N.; Pociot, F. Plasma proteome analysis of patients with type 1 diabetes with diabetic nephropathy. Proteom. Sci. 2010, 8, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKay, G.J.; Savage, D.A.; Patterson, C.C.; Lewis, G.; McKnight, A.J.; Maxwell, A.P.; Warren 3/UK GoKinD Study Group. Association analysis of dyslipidemia-related genes in diabetic nephropathy. PLoS ONE 2013, 8, e58472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mooyaart, A.L.; Valk, E.J.; van Es, L.A.; Bruijn, J.A.; de Heer, E.; Freedman, B.I.; Dekkers, O.M.; Baelde, H.J. Genetic associations in diabetic nephropathy: A meta-analysis. Diabetologia 2011, 54, 544–553. [Google Scholar] [CrossRef] [Green Version]
- Curtiss, L.K.; Witztum, J.L. Plasma apolipoproteins AI, AII, B, CI, and E are glucosylated in hyperglycemic diabetic subjects. Diabetes 1985, 34, 452–461. [Google Scholar] [CrossRef]
- Skinner, N.E.; Wroblewski, M.S.; Kirihara, J.A.; Nelsestuen, G.L.; Seaquist, E.R. Sitagliptin Results in a Decrease of Truncated Apolipoprotein C1. Diabetes Ther. 2015, 6, 395–401. [Google Scholar] [CrossRef] [Green Version]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuior, E.V.; Gafencu, A.V. Apolipoprotein C1: Its Pleiotropic Effects in Lipid Metabolism and Beyond. Int. J. Mol. Sci. 2019, 20, 5939. https://doi.org/10.3390/ijms20235939
Fuior EV, Gafencu AV. Apolipoprotein C1: Its Pleiotropic Effects in Lipid Metabolism and Beyond. International Journal of Molecular Sciences. 2019; 20(23):5939. https://doi.org/10.3390/ijms20235939
Chicago/Turabian StyleFuior, Elena V., and Anca V. Gafencu. 2019. "Apolipoprotein C1: Its Pleiotropic Effects in Lipid Metabolism and Beyond" International Journal of Molecular Sciences 20, no. 23: 5939. https://doi.org/10.3390/ijms20235939




