Anti-Wrinkling and Anti-Melanogenic Effect of Pradosia mutisii Methanol Extract
Abstract
:1. Introduction
2. Results
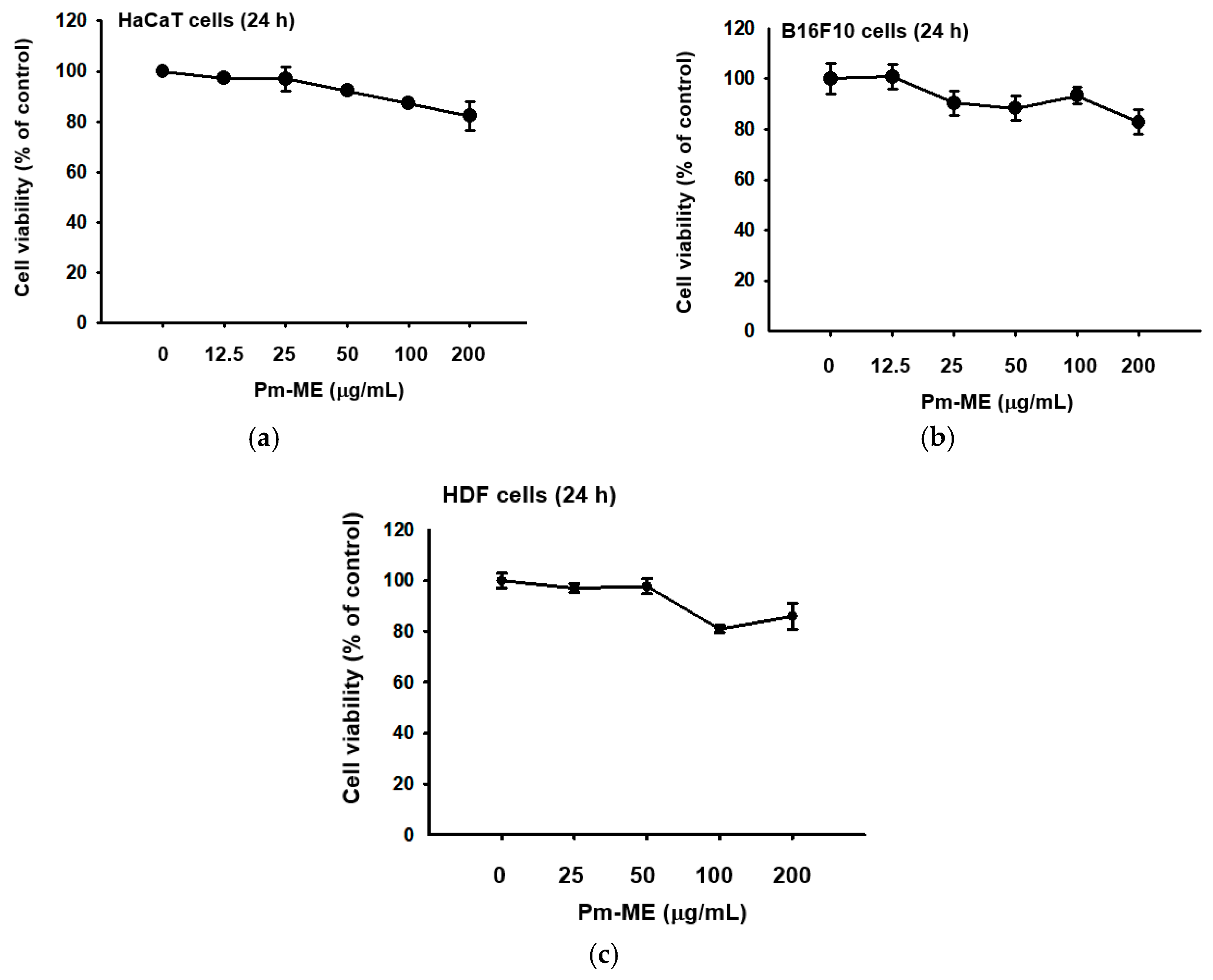
2.1. Pm-ME Characterization and its Effect on Cell Viability
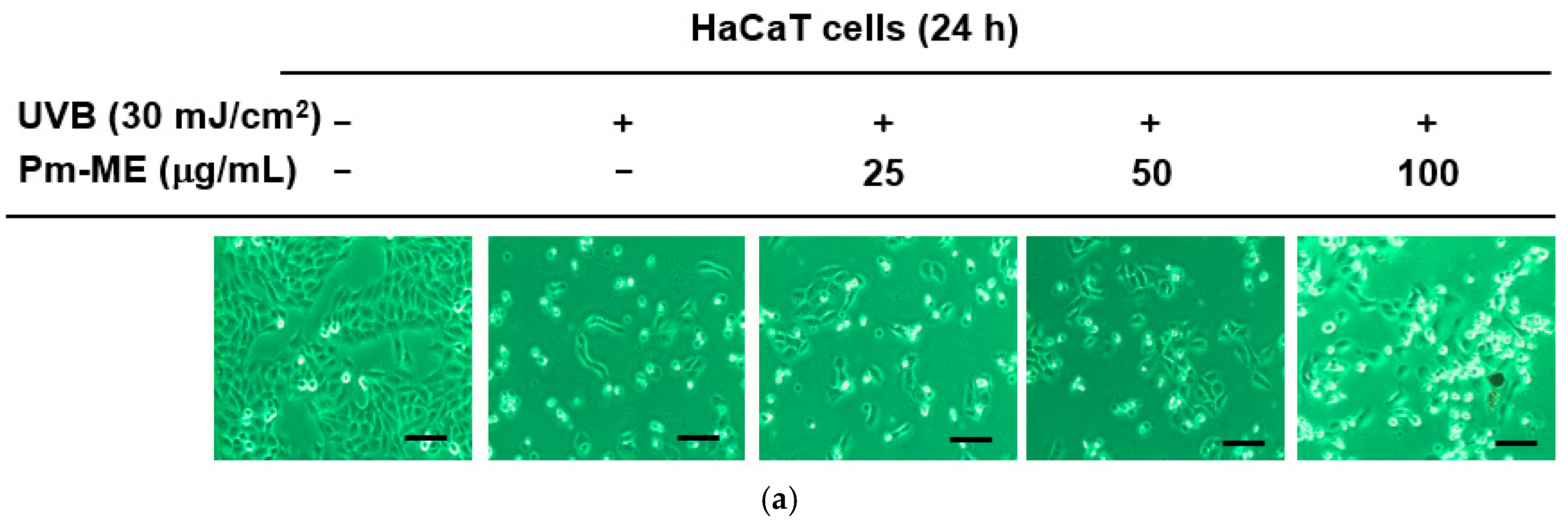
2.2. Protective Effect of Pm-ME against UVB and H2O2 damage
2.3. Moisturizing and Collagen-Increasing Effects of Pm-ME
2.4. Anti-Melanogenic Effect of Pm-ME
2.5. Molecular Mechanisms of Pm-ME-Mediated Antiphotoaging and Moisturizing Effects
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Compound Analysis from Pm-ME by UHPLC, Coupled to Negative Electrospray Ionization High-Resolution Tandem Mass Spectrometry (UPLC/HRMS)
4.3. Cell Culture
4.4. Cell Viability Assay
4.5. Free Radical Scavenging Activity
4.6. DAPI Staining
4.7. UVB Irradiation and the Morphological Change Assay
4.8. Semi-Quantitative RT-PCR Analysis
4.9. Plasmid Transfection and Luciferase Reporter Gene Assay
4.10. Melanogenesis and Melanin Secretion Assays
4.11. Tyrosinase Assay
4.12. Western Blot Analysis
4.13. Statistical Analysis
Data availability
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Pm-ME | P. mutisii methanol extract |
| MMPs | matrix metalloproteinases |
| α-MSH | α-melanocyte-stimulating hormone |
| ROS | reactive oxygen species |
| KA | kojic acid |
| L-DOPA | L-3,4-dihydroxyphenylalanine |
| UV | ultraviolet light |
| DAPI | 6-diamidino-2-phenylindole |
| H2O2 | hydrogen peroxide |
| MAPK | mitogen-activated protein kinases |
| ERK | extracellular signal-regulated kinase |
| JNK | c-Jun-N-terminal kinase |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| ABTS | 2,2’-Azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) diammonium salt |
| AA | ascorbic acid |
| RT-PCR | reverse transcription-polymerase chain reaction |
References
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev. 2004, 84, 1155–1228. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Zmijewski, M.A.; Zbytek, B.; Tobin, D.J.; Theoharides, T.C.; Rivier, J. Key role of CRF in the skin stress response system. Endocr. Rev. 2013, 34, 827–884. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Zmijewski, M.A.; Skobowiat, C.; Zbytek, B.; Slominski, R.M.; Steketee, J.D. Sensing the environment: Regulation of local and global homeostasis by the skin’s neuroendocrine system. Adv. Anat. Embryol. Cell Biol. 2012, 212, 1–115. [Google Scholar]
- Draelos, Z.D. New treatments for restoring impaired epidermal barrier permeability: Skin barrier repair creams. Clin. Dermatol. 2012, 30, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Zmijewski, M.A.; Plonka, P.M.; Szaflarski, J.P.; Paus, R. How UV light touches the brain and endocrine system through skin, and why. Endocrinology 2018, 159, 1992–2007. [Google Scholar] [CrossRef] [PubMed]
- Videira, I.F.d.S.; Moura, D.F.L.; Magina, S. Mechanisms regulating melanogenesis. Anais Brasil. Dermatol. 2013, 88, 76–83. [Google Scholar] [CrossRef]
- Cejkova, J.; Stipek, S.; Crkovska, J.; Ardan, T.; Midelfart, A. Reactive oxygen species (ROS)-generating oxidases in the normal rabbit cornea and their involvement in the corneal damage evoked by UVB rays. Histol. Histopathol. 2001, 16, 523–533. [Google Scholar] [PubMed]
- Glady, A.; Tanaka, M.; Moniaga, C.S.; Yasui, M.; Hara-Chikuma, M. Involvement of NADPH oxidase 1 in UVB-induced cell signaling and cytotoxicity in human keratinocytes. Biochem. Biophys. Rep. 2018, 14, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Valacchi, G.; Sticozzi, C.; Pecorelli, A.; Cervellati, F.; Cervellati, C.; Maioli, E. Cutaneous responses to environmental stressors. Ann. N. Y. Acad. Sci. 2012, 1271, 75–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong, Y.H.; Lee, H.S.; Jung, E.Y.; Han, S.H.; Park, Y.; Suh, H.J. Photoprotective effects of topical ginseng leaf extract using Ultraflo L against UVB-induced skin damage in hairless mice. J. Ginseng Res. 2017, 41, 456–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McDaniel, D.; Farris, P.; Valacchi, G. Atmospheric skin aging-contributors and inhibitors. J. Cosmet. Dermatol. 2018, 17, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.V.; Pal, S.; Mohammed, A.; Farooqui, M.; Doescher, M.P.; Asch, A.S.; Yamada, H.Y. Biological effects and epidemiological consequences of arsenic exposure, and reagents that can ameliorate arsenic damage in vivo. Oncotarget 2017, 8, 57605–57621. [Google Scholar] [CrossRef] [PubMed]
- Augereau, P.; Patsouris, A.; Bourbouloux, E.; Gourmelon, C.; Abadie Lacourtoisie, S.; Berton Rigaud, D.; Soulie, P.; Frenel, J.S.; Campone, M. Hormonoresistance in advanced breast cancer: A new revolution in endocrine therapy. Ther. Adv. Med. Oncol. 2017, 9, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Bickers, D.R.; Athar, M. Oxidative stress in the pathogenesis of skin disease. J. Invest. Dermatol. 2006, 126, 2565–2575. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Shin, C.M.; Park, C.H.; Kim, K.H.; Cho, K.H.; Eun, H.C.; Chung, J.H. Eicosapentaenoic acid inhibits UV-induced MMP-1 expression in human dermal fibroblasts. J. Lipid Res. 2005, 46, 1712–1720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chae, S.; Piao, M.J.; Kang, K.A.; Zhang, R.; Kim, K.C.; Youn, U.J.; Nam, K.W.; Lee, J.H.; Hyun, J.W. Inhibition of matrix metalloproteinase-1 induced by oxidative stress in human keratinocytes by mangiferin isolated from Anemarrhena asphodeloides. Biosci. Biotechnol. Biochem. 2011, 75, 2321–2325. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkar, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Nanni, V.; Canuti, L.; Gismondi, A.; Canini, A. Hydroalcoholic extract of Spartium junceum L. flowers inhibits growth and melanogenesis in B16-F10 cells by inducing senescence. Phytomedicine 2018, 46, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Dzialo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F. Potential role of natural compounds against skin aging. Curr. Med. Chem. 2015, 22, 1515–1538. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Cho, S.Y.; Kim, S.H.; Cho, D.; Kim, S.; Park, C.W.; Shimizu, T.; Cho, J.Y.; Seo, D.B.; Shin, S.S. Effects of Korean ginseng berry on skin antipigmentation and antiaging via FoxO3a activation. J. Ginseng Res. 2017, 41, 277–283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Yi, Y.S.; Kim, M.Y.; Cho, J.Y. Role of ginsenosides, the main active components of Panax ginseng, in inflammatory responses and diseases. J. Ginseng Res. 2017, 41, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Rundhaug, J.E.; Mikulec, C.; Pavone, A.; Fischer, S.M. A role for cyclooxygenase-2 in ultraviolet light-induced skin carcinogenesis. Mol. Carcinog 2007, 46, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Tripp, C.S.; Blomme, E.A.; Chinn, K.S.; Hardy, M.M.; LaCelle, P.; Pentland, A.P. Epidermal COX-2 induction following ultraviolet irradiation: Suggested mechanism for the role of COX-2 inhibition in photoprotection. J. Invest. Dermatol. 2003, 121, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Ming, M.; Soltani, K.; Shea, C.R.; Li, X.; He, Y.Y. Dual role of SIRT1 in UVB-induced skin tumorigenesis. Oncogene 2015, 34, 357–363. [Google Scholar] [CrossRef] [PubMed]
- D’Mello, S.A.; Finlay, G.J.; Baguley, B.C.; Askarian-Amiri, M.E. Signaling pathways in melanogenesis. Int. J. Mol. Sci. 2016, 17, 1144. [Google Scholar] [CrossRef] [PubMed]
- Kameyama, K.; Vieira, W.D.; Tsukamoto, K.; Law, L.W.; Hearing, V.J. Differentiation and the tumorigenic and metastatic phenotype of murine melanoma cells. Int. J. Cancer 1990, 45, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Smit, N.; Vicanova, J.; Pavel, S. The hunt for natural skin whitening agents. Int. J. Mol. Sci. 2009, 10, 5326–5349. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.J.; Choi, B.R.; Lee, E.K.; Kim, S.H.; Yi, H.Y.; Park, H.R.; Song, C.H.; Lee, Y.J.; Ku, S.K. Inhibitory effect of dried pomegranate concentration powder on melanogenesis in B16F10 melanoma cells; involvement of p38 and PKA signaling pathways. Int. J. Mol. Sci. 2015, 16, 24219–24242. [Google Scholar] [CrossRef] [PubMed]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermatoendocrinol 2012, 4, 253–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, M.; Visscher, M.; Laruffa, A.; Wickett, R. Natural moisturizing factors (NMF) in the stratum corneum (SC). II. Regeneration of NMF over time after soaking. J. Cosmet. Sci. 2010, 61, 23–29. [Google Scholar] [PubMed]
- Wang, A.S.; Dreesen, O. Biomarkers of cellular senescence and skin aging. Front. Gen. 2018, 9, 247. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Qin, Z.; Xia, W.; Shao, Y.; Voorhees, J.J.; Fisher, G.J. Matrix-degrading metalloproteinases in photoaging. J. Invest. Dermatol. 2009, 14, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Kim, D.; Yoo, S.; Hong, Y.H.; Han, S.Y.; Jeong, S.; Jeong, D.; Kim, J.H.; Cho, J.Y.; Park, J. The skin protective effects of compound K, a metabolite of ginsenoside Rb1 from Panax ginseng. J. Ginseng Res. 2018, 42, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Hwang, K.; Lee, J.; Han, S.Y.; Kim, E.M.; Park, J.; Cho, J.Y. Skin protective effect of epigallocatechin gallate. Int. J. Mol. Sci. 2018, 19, 173. [Google Scholar] [CrossRef] [PubMed]
- Arranz-Solis, D.; Benavides, J.; Regidor-Cerrillo, J.; Fuertes, M.; Ferre, I.; Ferreras Mdel, C.; Collantes-Fernandez, E.; Hemphill, A.; Perez, V.; Ortega-Mora, L.M. Influence of the gestational stage on the clinical course, lesional development and parasite distribution in experimental ovine neosporosis. Vet. Res. 2015, 46, 19. [Google Scholar] [CrossRef] [PubMed]
- de la Torre, L.; Nieto, R.; Noguerol, M.; Anel, J.A.; Gimeno, L. A climatology based on reanalysis of baroclinic developmental regions in the extratropical northern hemisphere. Ann. N. Y. Acad. Sci. 2008, 1146, 235–255. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.; Semo, L.; Morales, M.; Noza, Z.; Nunez, H.; Cayuba, A.; Noza, M.; Humaday, N.; Vaya, J.; Van Damme, P. Ethnomedicinal practices and medicinal plant knowledge of the Yuracares and Trinitarios from Indigenous Territory and National Park Isiboro-Secure, Bolivian Amazon. J. Ethnopharmacol. 2011, 133, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Niles, A.L.; Moravec, R.A.; Riss, T.L. In vitro viability and cytotoxicity testing and same-well multi-parametric combinations for high throughput screening. Curr. Chem. Genom. 2009, 3, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Liang, Q.H.; Xiong, X.G.; Wang, Y.; Zhang, Z.H.; Sun, M.J.; Lu, X.; Wu, D. Anti-Inflammatory Effects of p-coumaric acid, a natural compound of Oldenlandia diffusa, on arthritis model rats. Evid. Based Complement. Alternat. Med. 2018, 2018, 5198594. [Google Scholar] [CrossRef] [PubMed]
- Etoh, H.; Murakami, K.; Yogoh, T.; Ishikawa, H.; Fukuyama, Y.; Tanaka, H. Anti-oxidative compounds in barley tea. Biosci. Biotechnol. Biochem. 2004, 68, 2616–2618. [Google Scholar] [CrossRef] [PubMed]
- Benbettaieb, N.; Nyagaya, J.; Seuvre, A.M.; Debeaufort, F. Antioxidant activity and release kinetics of caffeic and p-Coumaric acids from hydrocolloid-based active films for healthy packaged food. J. Agric. Food Chem. 2018, 66, 6906–6916. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.S.; Pravda, E.A.; Li, D.; Shih, S.C.; Dallabrida, S.M. Angiopoietin-1 reduces H(2)O(2)-induced increases in reactive oxygen species and oxidative damage to skin cells. J. Invest. Dermatol. 2010, 130, 1307–1317. [Google Scholar] [CrossRef] [PubMed]
- Crawford, S. Anti-inflammatory/antioxidant use in long-term maintenance cancer therapy: A new therapeutic approach to disease progression and recurrence. Ther. Adv. Med. Oncol. 2014, 6, 52–68. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Zhang, T.; Chen, Z.; Wang, Y.; Ma, S.; Liu, J. The beneficial effect of ginsenosides extracted by pulsed electric field against hydrogen peroxide-induced oxidative stress in HEK-293 cells. J. Ginseng Res. 2017, 41, 169–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuch, A.P.; Moreno, N.C.; Schuch, N.J.; Menck, C.F.M.; Garcia, C.C.M. Sunlight damage to cellular DNA: Focus on oxidatively generated lesions. Free Radic. Biol. Med. 2017, 107, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.H.; Kim, D.; Nam, G.; Yoo, S.; Han, S.Y.; Jeong, S.G.; Kim, E.; Jeong, D.; Yoon, K.; Kim, S.; et al. Photoaging protective effects of BIOGF1K, a compound-K-rich fraction prepared from Panax ginseng. J. Ginseng Res. 2018, 42, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Hardeland, R.; Zmijewski, M.A.; Slominski, R.M.; Reiter, R.J.; Paus, R. Melatonin: A cutaneous perspective on its production, metabolism, and functions. J. Invest. Dermatol. 2018, 138, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Skobowiat, C.; Brozyna, A.A.; Janjetovic, Z.; Jeayeng, S.; Oak, A.S.W.; Kim, T.K.; Panich, U.; Reiter, R.J.; Slominski, A.T. Melatonin and its derivatives counteract the ultraviolet B radiation-induced damage in human and porcine skin ex vivo. J. Pineal Res. 2018, 65, e12501. [Google Scholar] [CrossRef] [PubMed]
- Janjetovic, Z.; Jarrett, S.G.; Lee, E.F.; Duprey, C.; Reiter, R.J.; Slominski, A.T. Melatonin and its metabolites protect human melanocytes against UVB-induced damage: Involvement of NRF2-mediated pathways. Sci. Rep. 2017, 7, 1274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slominski, A.T.; Semak, I.; Fischer, T.W.; Kim, T.K.; Kleszczynski, K.; Hardeland, R.; Reiter, R.J. Metabolism of melatonin in the skin: Why is it important? Exp. Dermatol. 2017, 26, 563–568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.Y.; Park, S.C. Physiological antioxidative network of the bilirubin system in aging and age-related diseases. Front. Pharmacol. 2012, 3, 45. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Franco, M.; Planells, E.; Quintero, B.; Acuna-Castroviejo, D.; Rusanova, I.; Escames, G.; Molina-Lopez, J. Effect of melatonin supplementation on antioxidant status and DNA damage in high intensity trained athletes. Int. J. Sports Med. 2017, 38, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Hossen, M.J.; Hong, Y.D.; Baek, K.S.; Yoo, S.; Hong, Y.H.; Kim, J.H.; Lee, J.O.; Kim, D.; Park, J.; Cho, J.Y. In vitro antioxidative and anti-inflammatory effects of the compound K-rich fraction BIOGF1K, prepared from Panax ginseng. J. Ginseng Res. 2017, 41, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Han, S.Y.; Kim, E.; Hwang, K.; Ratan, Z.A.; Hwang, H.; Kim, E.M.; Kim, D.; Park, J.; Cho, J.Y. Cytoprotective effect of epigallocatechin gallate (EGCG)-5’-O-alpha-glucopyranoside, a novel EGCG derivative. Int. J. Mol. Sci. 2018, 19, 1466. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.L.; Li, C.H.; Chang, C.Y.; Lu, S.R.; Lin, C.H.; Tse, L.S.; Cheng, Y.W. Anti-ageing effects of alpha-naphthoflavone on normal and UVB-irradiated human skin fibroblasts. Exp. Dermatol. 2012, 21, 546–548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muthusamy, V.; Piva, T.J. The UV response of the skin: A review of the MAPK, NFkappaB and TNFalpha signal transduction pathways. Arch. Dermatol. Res. 2010, 302, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.L.; Lee, C.L.; Korivi, M.; Liao, J.W.; Rajendran, P.; Wu, J.J.; Hseu, Y.C. Zerumbone protects human skin keratinocytes against UVA-irradiated damages through Nrf2 induction. Biochem. Pharmacol. 2018, 148, 130–146. [Google Scholar] [CrossRef] [PubMed]
- Bhogal, R.K.; Bona, C.A. Regulatory effect of extracellular signal-regulated kinases (ERK) on type I collagen synthesis in human dermal fibroblasts stimulated by IL-4 and IL-13. Int. Rev. Immunol. 2008, 27, 472–496. [Google Scholar] [CrossRef] [PubMed]
- Vigetti, D.; Karousou, E.; Viola, M.; Deleonibus, S.; De Luca, G.; Passi, A. Hyaluronan: Biosynthesis and signaling. Biochim. Biophys. Acta 2014, 1840, 2452–2459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Becatti, M.; Barygina, V.; Mannucci, A.; Emmi, G.; Prisco, D.; Lotti, T.; Fiorillo, C.; Taddei, N. Sirt1 protects against oxidative stress-induced apoptosis in fibroblasts from psoriatic patients: A new insight into the pathogenetic mechanisms of psoriasis. Int. J. Mol. Sci. 2018, 19, 1466. [Google Scholar] [CrossRef] [PubMed]
- Efimova, T. p38 delta mitogen-activated protein kinase regulates skin homeostasis and tumorigenesis. Cell Cycle 2010, 9, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Powell, B.S.; Dhaher, Y.Y.; Szleifer, I.G. Review of the multiscale effects of female sex hormones on Matrix Metalloproteinase-Mediated collagen degradation. Crit Rev. Biomed. Eng. 2015, 43, 401–428. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Park, Y.G.; Lee, H.J.; Lim, S.J.; Nho, C.W. Youngiasides A and C Isolated from Youngia denticulatum Inhibit UVB-Induced MMP expression and promote type I procollagen production via repression of MAPK/AP-1/NF-kappaB and activation of AMPK/Nrf2 in HaCaT cells and human dermal fibroblasts. J. Agric. Food Chem. 2015, 63, 5428–5438. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Man, Y.; Liu, S. Long non-coding RNA HULC promotes UVB-induced injury by up-regulation of BNIP3 in keratinocytes. Biomed. Pharmacother. 2018, 104, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Spörl, F.; Schellenberg, K.; Blatt, T.; Wenck, H.; Wittern, K.-P.; Schrader, A.; Kramer, A. A circadian clock in HaCaT keratinocytes. J. Invest. Dermatol. 2011, 131, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zuo, L.; Sun, T.; Tang, J.; Ding, D.; Zhou, L.; Kang, J.; Zhang, X. Chemical profiling and quantification of XueBiJing injection, a systematic quality control strategy using UHPLC-Q Exactive hybrid quadrupole-orbitrap high-resolution mass spectrometry. Sci. Rep. 2017, 7, 16921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pavlovic, I.; Petrik, I.; Tarkowska, D.; Lepedus, H.; Vujcic Bok, V.; Radic Brkanac, S.; Novak, O.; Salopek-Sondi, B. Correlations between phytohormones and drought tolerance in selected Brassica crops: Chinese cabbage, white cabbage and kale. Int. J. Mol. Sci. 2018, 19, 2866. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Atale, N.; Gupta, S.; Yadav, U.C.; Rani, V. Cell-death assessment by fluorescent and nonfluorescent cytosolic and nuclear staining techniques. J. Microsc. 2014, 255, 7–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dash, R.; Mandal, M.; Ghosh, S.K.; Kundu, S.C. Silk sericin protein of tropical tasar silkworm inhibits UVB-induced apoptosis in human skin keratinocytes. Mol. Cell Biochem. 2008, 311, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Yi, Y.S.; Hong, Y.H.; Yoo, S.; Han, S.Y.; Kim, E.; Jeong, S.G.; Aravinthan, A.; Baik, K.S.; Choi, S.Y.; et al. Tabetri (Tabebuia avellanedae Ethanol Extract) ameliorates osteoarthritis symptoms induced by monoiodoacetate through its anti-inflammatory and chondroprotective activities. Mediators Inflamm. 2017, 2017, 3619879. [Google Scholar] [CrossRef] [PubMed]






| Name | Primer | Sequence (5′ to 3′) |
|---|---|---|
| MMP-1 | Forward | TCTGACGTTGATCCCAGAGAGCAG |
| Reverse | CAGGGTGACACCAGTGACTGCAC | |
| MMP-9 | Forward | GCCACTTGTCGGCGATAAGG |
| Reverse | CACTGTCCACCCCTCAGAGC | |
| Col-1A1 | Forward | CAGGTACCATGACCGAGACG |
| Reverse | AGCACCATCATTTCCACGAG | |
| COX2 | Forward | CAAAAGCTGGGAAGCCTTCT |
| Reverse | CCATCCTTCAAAAGGCGCAG | |
| HAS-2 | Forward | CCACCCAGTACAGCGTCAAC |
| Reverse | CATGGTGCTTCTGTCGCTCT | |
| Sirt-1 | Forward | CAGTGTCATGGTTCCTTTGC |
| Reverse | CACCGAGGAACTACCTGAT | |
| TGM-1 | Forward | AGGGAAGATCCAAGAGCCCA |
| Reverse | ACTCTGGATCCCCTACGCTT | |
| GAPDH | Forward | CACTCACGGCAAATTCAACGGCAC |
| Reverse | GACTCCACGACATACTCAGCAC |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lorz, L.R.; Yoo, B.C.; Kim, M.-Y.; Cho, J.Y. Anti-Wrinkling and Anti-Melanogenic Effect of Pradosia mutisii Methanol Extract. Int. J. Mol. Sci. 2019, 20, 1043. https://doi.org/10.3390/ijms20051043
Lorz LR, Yoo BC, Kim M-Y, Cho JY. Anti-Wrinkling and Anti-Melanogenic Effect of Pradosia mutisii Methanol Extract. International Journal of Molecular Sciences. 2019; 20(5):1043. https://doi.org/10.3390/ijms20051043
Chicago/Turabian StyleLorz, Laura Rojas, Byong Chul Yoo, Mi-Yeon Kim, and Jae Youl Cho. 2019. "Anti-Wrinkling and Anti-Melanogenic Effect of Pradosia mutisii Methanol Extract" International Journal of Molecular Sciences 20, no. 5: 1043. https://doi.org/10.3390/ijms20051043






