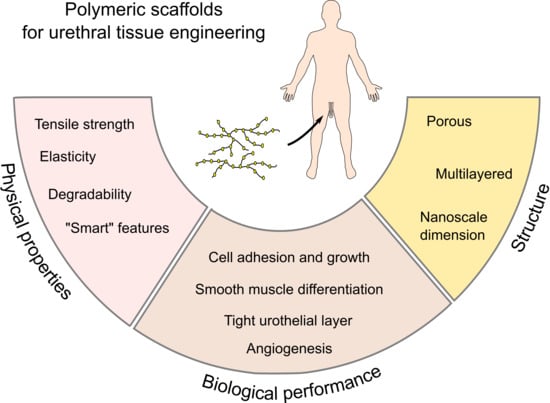
From Acellular Matrices to Smart Polymers: Degradable Scaffolds that are Transforming the Shape of Urethral Tissue Engineering
Abstract
1. Introduction
2. Structural and Functional Properties of the Male Urethra
3. Polymeric Biomaterials in Urethral Tissue Engineering
4. In Vivo Performance of Biodegradable Scaffolds for Urethral Reconstruction
5. Future Directions: Hybrid and Smart Polymers
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Lazzeri, M.; Sansalone, S.; Guazzoni, G.; Barbagli, G. Incidence, Causes, and Complications of Urethral Stricture Disease. Eur. Urol. Suppl. 2016, 15, 2–6. [Google Scholar] [CrossRef]
- Keays, M.A.; Dave, S. Current hypospadias management: Diagnosis, surgical management, and long-term patient-centred outcomes. Can. Urol. Assoc. J. 2017, 11, S48–S53. [Google Scholar] [CrossRef]
- Chapple, C.; Andrich, D.; Atala, A.; Barbagli, G.; Cavalcanti, A.; Kulkarni, S.; Mangera, A.; Nakajima, Y. SIU/ICUD Consultation on Urethral Strictures: The Management of Anterior Urethral Stricture Disease Using Substitution Urethroplasty. Urology 2014, 83, S31–S47. [Google Scholar] [CrossRef]
- Snodgrass, W.; Bush, N. Primary hypospadias repair techniques: A review of the evidence. Urol. Ann. 2016, 8, 403–408. [Google Scholar] [CrossRef]
- Fenton, A.S.; Morey, A.F.; Aviles, R.; Garcia, C.R. Anterior urethral strictures: Etiology and characteristics. Urology 2005, 65, 1055–1058. [Google Scholar] [CrossRef]
- Mundy, A.R.; Andrich, D.E. Urethral strictures. BJU Int. 2011, 107, 6–26. [Google Scholar] [CrossRef]
- Caldamone, A.A.; Edstrom, L.E.; Koyle, M.A.; Rabinowitz, R.; Hulbert, W.C. Buccal mucosal grafts for urethral reconstruction. Urology 1998, 51, 15–19. [Google Scholar] [CrossRef]
- Barbagli, G.; Palminteri, E.; Guazzoni, G.; Montorsi, F.; Turini, D.; Lazzeri, M. Bulbar urethroplasty using buccal mucosa grafts placed on the ventral, dorsal or lateral surface of the urethra: Are results affected by the surgical technique? J. Urol. 2005, 174, 955–958. [Google Scholar] [CrossRef]
- Ding, J.; Li, Q.; Li, S.; Li, F.; Zhou, C.; Zhou, Y.; Hu, J.; Xie, L.; Cao, Y.; Zhang, S. Ten years’ experience for hypospadias repair: Combined buccal mucosa graft and local flap for urethral reconstruction. Urol. Int. 2014, 93, 454–459. [Google Scholar] [CrossRef]
- Markiewicz, M.R.; DeSantis, J.L.; Margarone, J.E.; Pogrel, M.A.; Chuang, S.-K. Morbidity Associated with Oral Mucosa Harvest for Urological Reconstruction: An Overview. J. Oral Maxillofac. Surg. 2008, 66, 739–744. [Google Scholar] [CrossRef]
- Harris, C.R.; Osterberg, E.C.; Sanford, T.; Alwaal, A.; Gaither, T.W.; McAninch, J.W.; McCulloch, C.E.; Breyer, B.N. National Variation in Urethroplasty Cost and Predictors of Extreme Cost: A Cost Analysis with Policy Implications. Urology 2016, 94, 246–254. [Google Scholar] [CrossRef]
- Abbas, T.O.; Mahdi, E.; Hasan, A.; AlAnsari, A.; Pennisi, C.P. Current Status of Tissue Engineering in the Management of Severe Hypospadias. Front. Pediatr. 2018, 5, 283. [Google Scholar] [CrossRef]
- De Kemp, V.; De Graaf, P.; Fledderus, J.O.; Bosch, J.L.H.R.; De Kort, L.M.O. Tissue engineering for human urethral reconstruction: Systematic review of recent literature. PLoS ONE 2015, 10, e0118653. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Xu, Y.-M.; Fu, Q.; Zhu, W.-D.; Cui, L.; Chen, J. Evaluation of the biocompatibility and mechanical properties of naturally derived and synthetic scaffolds for urethral reconstruction. J. Biomed. Mater. Res. Part A 2010, 94A, 317–325. [Google Scholar] [CrossRef]
- Beiko, D.T.; Knudsen, B.E.; Watterson, J.D.; Cadieux, P.A.; Reid, G.; Denstedt, J.D. Urinary tract biomaterials. J. Urol. 2004, 171, 2438–2444. [Google Scholar] [CrossRef]
- de Graaf, P.; van der Linde, E.M.; Rosier, P.F.W.M.; Izeta, A.; Sievert, K.-D.; Bosch, J.L.H.R.; de Kort, L.M.O. Systematic Review to Compare Urothelium Differentiation with Urethral Epithelium Differentiation in Fetal Development, as a Basis for Tissue Engineering of the Male Urethra. Tissue Eng. Part B Rev. 2017, 23, 257–267. [Google Scholar] [CrossRef]
- Da Silva, E.A.; Sampaio, F.J.B.; Ortiz, V.; Cardoso, L.E.M. Regional differences in the extracellular matrix of the human spongy urethra as evidenced by the composition of glycosaminoglycans. J. Urol. 2002, 167, 2183–2187. [Google Scholar] [CrossRef]
- De Graaf, P.; Ramadan, R.; Linssen, E.C.; Staller, N.A.; Hendrickx, A.P.A.; Pigot, G.L.S.; Meuleman, E.J.H.; Bouman, M.; Özer, M.; Bosch, J.L.H.R.; et al. The multilayered structure of the human corpus spongiosum. Histol. Histopathol. 2018, 33, 1335–1345. [Google Scholar] [PubMed]
- Coenen, A.M.J.; Bernaerts, K.V.; Harings, J.A.W.; Jockenhoevel, S.; Ghazanfari, S. Elastic materials for tissue engineering applications: Natural, synthetic, and hybrid polymers. Acta Biomater. 2018, 79, 60–82. [Google Scholar] [CrossRef]
- Augsburger, H.R. Elastic fibre system of the female canine urethra. Histochemical identification of elastic, elaunin and oxytalan fibres. Anat. Histol. Embryol. 1997, 26, 297–302. [Google Scholar] [CrossRef]
- Kim, R.J.; Kerns, J.M.; Liu, S.; Nagel, T.; Zaszczurynski, P.; Lin, D.L.; Damaser, M.S. Striated muscle and nerve fascicle distribution in the female raturethral sphincter. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2007, 290, 145–154. [Google Scholar] [CrossRef]
- Lim, S.H.; Wang, T.-J.; Tseng, G.-F.; Lee, Y.F.; Huang, Y.-S.; Chen, J.-R.; Cheng, C.-L. The Distribution of Muscles Fibers and Their Types in the Female Rat Urethra: Cytoarchitecture and Three-Dimensional Reconstruction. Anat. Rec. 2013, 296, 1640–1649. [Google Scholar] [CrossRef]
- Jankowski, R.J.; Prantil, R.L.; Fraser, M.O.; Chancellor, M.B.; de Groat, W.C.; Huard, J.; Vorp, D.A. Development of an experimental system for the study of urethral biomechanical function. Am. J. Physiol. Physiol. 2004, 286, F225–F232. [Google Scholar] [CrossRef]
- Watanabe, H.; Yamamoto, T.Y. Autonomic innervation of the muscles in the wall of the bladder and proximal urethra of male rats. J. Anat. 1979, 128, 873–886. [Google Scholar]
- Male Reproductive System. Available online: http://histology.medicine.umich.edu/slides/male-reproductive-system (accessed on 20 December 2018).
- Shirozu, H.; Koyanagi, T.; Takashima, T.; Horimoto, N.; Akazawa, K.; Nakano, H. Penile tumescence in the human fetus at term—A preliminary report. Early Hum. Dev. 1995, 41, 159–166. [Google Scholar] [CrossRef]
- Ohel, G.; Haddad, S.; Samueloff, A. Fetal Urine Production and Micturition and Fetal Behavioral State. Am. J. Perinatol. 1995, 12, 91–92. [Google Scholar] [CrossRef]
- Walter, J.S.; Wheeler, J.S.; Morgan, C.; Zaszczurynski, P.; Plishka, M. Measurement of total urethral compliance in females with stress incontinence. Neurourol. Urodyn. 1993, 12, 273–276. [Google Scholar] [CrossRef]
- Mijailovich, S.M.; Sullivan, M.P.; Yalla, S.V.; Venegas, J.G. Effect of urethral compliance on the steady state p-Q relationships assessed with a mechanical analog of the male lower urinary tract. Neurourol. Urodyn. 2007, 26, 234–246. [Google Scholar] [CrossRef]
- Thind, P.; Lose, G.; Colstrup, H. How to measure urethral elastance in a simple way. Elastance: Definition, determination and implications. Urol. Res. 1991, 19, 241–244. [Google Scholar] [CrossRef]
- Yalla, S.V.; Burros, H.M. Conduit and regional compliance of female canine urethra. Urology 1974, 4, 155–161. [Google Scholar] [CrossRef]
- Husmann, D.A.; Rathbun, S.R. Long-Term Followup of Visual Internal Urethrotomy for Management of Short (Less Than 1 Cm) Penile Urethral Strictures Following Hypospadias Repair. J. Urol. 2006, 176, 1738–1741. [Google Scholar] [CrossRef]
- Lalla, M.; Gregersen, H.; Olsen, L.H.; Jørgensen, T.M. In Vivo Biomechanical Assessment of Anterior Rabbit Urethra After Repair of Surgically Created Hypospadias. J. Urol. 2010, 184, 675–682. [Google Scholar] [CrossRef]
- Hammouda, H.M.; El-Ghoneimi, A.; Bagli, D.J.; McLorie, G.A.; Khoury, A.E. Tubularized Incised Plate Repair: Functional Outcome After Intermediate Followup. J. Urol. 2003, 169, 331–333. [Google Scholar] [CrossRef]
- Eassa, W.; Brzezinski, A.; Capolicchio, J.P.; Jednak, R.; El-Sherbiny, M. How do asymptomatic toilet-trained children void following tubularized incised-plate hypospadias repair? Can. Urol. Assoc. J. 2012, 6, 238–242. [Google Scholar] [CrossRef]
- Lalla, M.; Riis, C.; Jørgensen, C.S.; Danielsen, C.C.; Jørgensen, T.M. A biomechanical, histological and biochemical study in an experimental rabbit hypospadias repair model using scanning acoustic microscopy. J. Pediatr. Urol. 2011, 7, 404–411. [Google Scholar] [CrossRef]
- Gabrich, P.N.; Vasconcelos, J.S.P.; Damião, R.; Silva, E.A. Penile anthropometry in Brazilian children and adolescents. J. Pediatr. 2007, 83, 441–446. [Google Scholar] [CrossRef]
- Simsek, A.; Aldamanhori, R.; Chapple, C.R.; MacNeil, S. Overcoming scarring in the urethra: Challenges for tissue engineering. Asian J. Urol. 2018, 5, 69–77. [Google Scholar] [CrossRef]
- Liu, J.S.; Bury, M.I.; Fuller, N.J.; Sturm, R.M.; Ahmad, N.; Sharma, A.K. Bone Marrow Stem/Progenitor Cells Attenuate the Inflammatory Milieu Following Substitution Urethroplasty. Sci. Rep. 2016, 6, 35638. [Google Scholar] [CrossRef]
- Birder, L.; Andersson, K.-E. Urothelial Signaling. Physiol. Rev. 2013, 93, 653–680. [Google Scholar] [CrossRef]
- Akkad, T.; Pelzer, A.E.; Mitterberger, M.; Rehder, P.; Leonhartsberger, N.; Bartsch, G.; Strasser, H. Influence of intravesical potassium on pelvic floor activity in women with recurrent urinary tract infections: Comparative urodynamics might lead to enhanced detection of dysfunctional voiding. BJU Int. 2007, 100, 1071–1074. [Google Scholar] [CrossRef]
- Rajasekaran, M.; Stein, P.; Parsons, C.L. Toxic factors in human urine that injure urothelium. Int. J. Urol. 2006, 13, 409–414. [Google Scholar] [CrossRef]
- Adamowicz, J.; Kloskowski, T.; Tworkiewicz, J.; Pokrywczyńska, M.; Drewa, T. Urine Is a Highly Cytotoxic Agent: Does It Influence Stem Cell Therapies in Urology? Transplant. Proc. 2012, 44, 1439–1441. [Google Scholar] [CrossRef]
- Li, H.; Xu, Y.; Xie, H.; Li, C.; Song, L.; Feng, C.; Zhang, Q.; Xie, M.; Wang, Y.; Lv, X. Epithelial-differentiated adipose-derived stem cells seeded bladder acellular matrix grafts for urethral reconstruction: An animal model. Tissue Eng. Part A 2014, 20, 774–784. [Google Scholar] [CrossRef]
- Ribeiro-Filho, L.A.; Sievert, K.-D. Acellular matrix in urethral reconstruction. Adv. Drug Deliv. Rev. 2015, 82–83, 38–46. [Google Scholar] [CrossRef]
- Denstedt, J.; Atala, A. Biomaterials and Tissue Engineering in Urology; Woodhead Publishing: Sawston, UK, 2009; ISBN 9781845696375. [Google Scholar]
- Bisson, I.; Hilborn, J.; Wurm, F.; Meyrat, B.; Frey, P. Human urothelial cells grown on collagen adsorbed to surface-modified polymers. Urology 2002, 60, 176–180. [Google Scholar] [CrossRef]
- Azevedo, H.S.; Reis, R.L. Understanding the Enzymatic Degradation of Biodegradable Polymers and Strategies to Control Their Degradation Rate; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Babensee, J.E.; Anderson, J.M.; McIntire, L.V.; Mikos, A.G. Host response to tissue engineered devices. Adv. Drug Deliv. Rev. 1998, 33, 111–139. [Google Scholar] [CrossRef]
- Knecht, S.; Erggelet, C.; Endres, M.; Sittinger, M.; Kaps, C.; Stüssi, E. Mechanical testing of fixation techniques for scaffold-based tissue-engineered grafts. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 83, 50–57. [Google Scholar] [CrossRef]
- Perkins, L.E.L.E.L.; Kossuth, M.B.B.; Fox, J.C.C.; Rapoza, R.J.J. Paving the way to a bioresorbable technology: Development of the absorb BRS program. Catheter. Cardiovasc. Interv. 2016, 88, 1–9. [Google Scholar] [CrossRef]
- Moon, J.J.; West, J.L. Vascularization of engineered tissues: Approaches to promote angio-genesis in biomaterials. Curr. Top. Med. Chem. 2008, 8, 300–310. [Google Scholar]
- Nooeaid, P.; Salih, V.; Beier, J.P.; Boccaccini, A.R. Osteochondral tissue engineering: Scaffolds, stem cells and applications. J. Cell. Mol. Med. 2012, 16, 2247–2270. [Google Scholar] [CrossRef]
- Carmagnola, I.; Ranzato, E.; Chiono, V. Scaffold functionalization to support a tissue biocompatibility. In Functional 3D Tissue Engineering Scaffolds; Elsevier: Amsterdam, The Netherlands, 2018; pp. 255–277. [Google Scholar]
- Versteegden, L.R.M.; de Jonge, P.K.J.D.; IntHout, J.; van Kuppevelt, T.H.; Oosterwijk, E.; Feitz, W.F.J.; de Vries, R.B.M.; Daamen, W.F. Tissue Engineering of the Urethra: A Systematic Review and Meta-analysis of Preclinical and Clinical Studies. Eur. Urol. 2017, 72, 594–606. [Google Scholar] [CrossRef]
- Qi, N.; Li, W.; Tian, H. A Systematic Review of Animal and Clinical Studies on the Use of Scaffolds for Urethral Repair. J. Huazhong Univ. Sci. Technol. Med. Sci. 2016, 36, 111–117. [Google Scholar] [CrossRef]
- Davis, N.F.; Cunnane, E.M.; O’Brien, F.J.; Mulvihill, J.J.; Walsh, M.T. Tissue engineered extracellular matrices (ECMs) in urology: Evolution and future directions. Surgeon 2018, 16, 55–65. [Google Scholar] [CrossRef]
- Badylak, S.F.; Lantz, G.C.; Coffey, A.; Geddes, L.A. Small intestinal submucosa as a large diameter vascular graft in the dog. J. Surg. Res. 1989, 47, 74–80. [Google Scholar] [CrossRef]
- Zhang, Y.; Kropp, B.P.; Moore, P.; Cowan, R.; Furness, P.D.; Kolligian, M.E.; Frey, P.; Cheng, E.Y. Coculture of bladder urothelial and smooth muscle cells on small intestinal submucosa: Potential applications for tissue engineering technology. J. Urol. 2000, 164, 928–934, discussion 934–935. [Google Scholar] [CrossRef]
- Fiala, R.; Vidlar, A.; Vrtal, R.; Belej, K.; Student, V. Porcine Small Intestinal Submucosa Graft for Repair of Anterior Urethral Strictures. Eur. Urol. 2007, 51, 1702–1708. [Google Scholar] [CrossRef]
- Palminteri, E.; Berdondini, E.; Colombo, F.; Austoni, E. Small Intestinal Submucosa (SIS) Graft Urethroplasty: Short-term Results. Eur. Urol. 2007, 51, 1695–1701. [Google Scholar] [CrossRef]
- Palminteri, E.; Berdondini, E.; Fusco, F.; De Nunzio, C.; Salonia, A. Long-term Results of Small Intestinal Submucosa Graft in Bulbar Urethral Reconstruction. Urology 2012, 79, 695–701. [Google Scholar] [CrossRef]
- Campodonico, F.; Benelli, R.; Michelazzi, A.; Ognio, E.; Toncini, C.; Maffezzini, M. Bladder Cell Culture on Small Intestinal Submucosa as Bioscaffold: Experimental Study on Engineered Urothelial Grafts. Eur. Urol. 2004, 46, 531–537. [Google Scholar] [CrossRef]
- Badylak, S.F.; Kropp, B.; McPherson, T.; Liang, H.; Snyder, P.W. Small Intestinal Submucosa: A Rapidly Resorbed Bioscaffold for Augmentation Cystoplasty in a Dog Model. Tissue Eng. 1998, 4, 379–387. [Google Scholar] [CrossRef]
- Kubricht, W.S.; Williams, B.J.; Eastham, J.A.; Venable, D.D. Tensile strength of cadaveric fascia lata compared to small intestinal submucosa using suture pull through analysis. J. Urol. 2001, 165, 486–490. [Google Scholar] [CrossRef]
- Weiser, A.C.; Franco, I.; Herz, D.B.; Silver, R.I.; Reda, E.F. Single Layered Small Intestinal Submucosa in the Repair of Severe Chordee and Complicated Hypospadias. J. Urol. 2003, 170, 1593–1595. [Google Scholar] [CrossRef]
- Hayn, M.H.; Bellinger, M.F.; Schneck, F.X. Small Intestine Submucosa as a Corporal Body Graft in the Repair of Severe Chordee. Urology 2009, 73, 277–279. [Google Scholar] [CrossRef]
- Zhang, L.; Du, A.; Li, J.; Pan, M.; Han, W.; Xiao, Y. Development of a cell-seeded modified small intestinal submucosa for urethroplasty. Heliyon 2016, 2, e00087. [Google Scholar] [CrossRef]
- Yoo, J.J.; Meng, J.; Oberpenning, F.; Atala, A. Bladder augmentation using allogenic bladder submucosa seeded with cells. Urology 1998, 51, 221–225. [Google Scholar] [CrossRef]
- Orabi, H.; Safwat, A.S.; Shahat, A.; Hammouda, H.M. The use of small intestinal submucosa graft for hypospadias repair: Pilot study. Arab J. Urol. 2013, 11, 415–420. [Google Scholar] [CrossRef]
- Hauser, S.; Bastian, P.J.; Fechner, G.; Müller, S.C. Small intestine submucosa in urethral stricture repair in a consecutive series. Urology 2006, 68, 263–266. [Google Scholar] [CrossRef]
- Nuininga, J.E.; van Moerkerk, H.; Hanssen, A.; Hulsbergen, C.A.; Oosterwijk-Wakka, J.; Oosterwijk, E.; de Gier, R.P.E.; Schalken, J.A.; van Kuppevelt, T.; Feitz, W.F.J. Rabbit urethra replacement with a defined biomatrix or small intestinal submucosa. Eur. Urol. 2003, 44, 266–271. [Google Scholar] [CrossRef]
- Chun, S.Y.; Kim, B.S.; Kwon, S.Y.; Park, S.I.; Song, P.H.; Yoo, E.S.; Kim, B.W.; Kwon, T.G.; Kim, H.T. Urethroplasty using autologous urethral tissue-embedded acellular porcine bladder submucosa matrix grafts for the management of long-segment urethral stricture in a rabbit model. J. Korean Med. Sci. 2015, 30, 301–307. [Google Scholar] [CrossRef]
- Huang, J.-W.; Xie, M.-K.; Zhang, Y.; Wei, G.-J.; Li, X.; Li, H.-B.; Wang, J.-H.; Zhu, W.-D.; Li, C.; Xu, Y.-M.; et al. Reconstruction of Penile Urethra With the 3-Dimensional Porous Bladder Acellular Matrix in a Rabbit Model. Urology 2014, 84, 1499–1505. [Google Scholar] [CrossRef]
- Li, C.; Xu, Y.-M.; Song, L.-J.; Fu, Q.; Cui, L.; Yin, S. Urethral Reconstruction Using Oral Keratinocyte Seeded Bladder Acellular Matrix Grafts. J. Urol. 2008, 180, 1538–1542. [Google Scholar] [CrossRef]
- Atala, A.; Guzman, L.; Retik, A.B. A novel inert collagen matrix for hypospadias repair. J. Urol. 1999, 162, 1148–1151. [Google Scholar] [CrossRef]
- Gu, G.-L.; Xia, S.-J.; Zhang, J.; Liu, G.-H.; Yan, L.; Xu, Z.-H.; Zhu, Y.-J. Tubularized Urethral Replacement Using Tissue-Engineered Peritoneum-Like Tissue in a Rabbit Model. Urol. Int. 2012, 89, 358–364. [Google Scholar] [CrossRef]
- Roth, C.C.; Kropp, B.P. Recent advances in urologic tissue engineering. Curr. Urol. Rep. 2009, 10, 119–125. [Google Scholar] [CrossRef]
- Xie, M.; Song, L.; Wang, J.; Fan, S.; Zhang, Y.; Xu, Y. Evaluation of stretched electrospun silk fibroin matrices seeded with urothelial cells for urethra reconstruction. J. Surg. Res. 2013, 184, 774–781. [Google Scholar] [CrossRef]
- Xie, M.; Xu, Y.; Song, L.; Wang, J.; Lv, X.; Zhang, Y. Tissue-engineered buccal mucosa using silk fibroin matrices for urethral reconstruction in a canine model. J. Surg. Res. 2014, 188, 1–7. [Google Scholar] [CrossRef]
- Hudson, S.M. Silk polymers: Material science and biotechnology, D. Kaplan, W. Adams, B. Farmer and C. Viney, eds. American Chemical Society, ACS Symposium Series 544 (1944). 370 pp. Polym. Adv. Technol. 1995, 6, 717. [Google Scholar] [CrossRef]
- Cunniff, P.M.; Fossey, S.A.; Auerbach, M.A.; Song, J.W.; Kaplan, D.L.; Adams, W.W.; Eby, R.K.; Mahoney, D.; Vezie, D.L. Mechanical and thermal properties of dragline silk from the spider Nephila clavipes. Polym. Adv. Technol. 1994, 5, 401–410. [Google Scholar] [CrossRef]
- Wang, Y.; Rudym, D.D.; Walsh, A.; Abrahamsen, L.; Kim, H.-J.; Kim, H.S.; Kirker-Head, C.; Kaplan, D.L. In vivo degradation of three-dimensional silk fibroin scaffolds. Biomaterials 2008, 29, 3415–3428. [Google Scholar] [CrossRef]
- Altman, G.H.; Diaz, F.; Jakuba, C.; Calabro, T.; Horan, R.L.; Chen, J.; Lu, H.; Richmond, J.; Kaplan, D.L. Silk-based biomaterials. Biomaterials 2003, 24, 401–416. [Google Scholar] [CrossRef]
- Panilaitis, B.; Altman, G.H.; Chen, J.; Jin, H.J.; Karageorgiou, V.; Kaplan, D.L. Macrophage responses to silk. Biomaterials 2003, 24, 3079–3085. [Google Scholar] [CrossRef]
- Chung, Y.G.; Tu, D.; Franck, D.; Gil, E.S.; Algarrahi, K.; Adam, R.M.; Kaplan, D.L.; Estrada, C.R.; Mauney, J.R. Acellular bi-layer silk fibroin scaffolds support tissue regeneration in a rabbit model of onlay urethroplasty. PLoS ONE 2014, 9, e91592. [Google Scholar] [CrossRef]
- Ribeiro, C.; Sencadas, V.; Correia, D.M.; Lanceros-Méndez, S. Piezoelectric polymers as biomaterials for tissue engineering applications. Colloids Surf. B Biointerfaces 2015, 136, 46–55. [Google Scholar] [CrossRef]
- Aguilar, M.R.; San Román, J. Introduction to smart polymers and their applications. In Smart Polymers and their Applications; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1–11. [Google Scholar]
- Knipe, J.M.; Peppas, N.A. Multi-responsive hydrogels for drug delivery and tissue engineering applications. Regen. Biomater. 2014, 1, 57–65. [Google Scholar] [CrossRef]
- Kowalski, P.S.; Bhattacharya, C.; Afewerki, S.; Langer, R. Smart Biomaterials: Recent Advances and Future Directions. ACS Biomater. Sci. Eng. 2018, 4, 3809–3817. [Google Scholar] [CrossRef]
- Anderson, D.G.; Burdick, J.A.; Langer, R. MATERIALS SCIENCE: Smart Biomaterials. Science 2004, 305, 1923–1924. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Matsunaga, Y.T. Thermo-responsive polymers and their application as smart biomaterials. J. Mater. Chem. B 2017, 5, 4307–4321. [Google Scholar] [CrossRef]
- Jochum, F.D.; Theato, P. Temperature- and light-responsive smart polymer materials. Chem. Soc. Rev. 2013, 42, 7468–7483. [Google Scholar] [CrossRef]
- Tsuda, Y.; Shimizu, T.; Yamato, M.; Kikuchi, A.; Sasagawa, T.; Sekiya, S.; Kobayashi, J.; Chen, G.; Okano, T. Cellular control of tissue architectures using a three-dimensional tissue fabrication technique. Biomaterials 2007, 28, 4939–4946. [Google Scholar] [CrossRef]
- Shiroyanagi, Y.; Yamato, M.; Yamazaki, Y.; Toma, H.; Okano, T. Transplantable Urothelial Cell Sheets Harvested Noninvasively from Temperature-Responsive Culture Surfaces by Reducing Temperature. Tissue Eng. 2003, 9, 1005–1012. [Google Scholar] [CrossRef]
- Rickert, D.; Moses, M.A.; Lendlein, A.; Kelch, S.; Franke, R.-P. The importance of angiogenesis in the interaction between polymeric biomaterials and surrounding tissue. Clin. Hemorheol. Microcirc. 2003, 28, 175–181. [Google Scholar]
- Correia, C.O.; Leite, Á.J.; Mano, J.F. Chitosan/bioactive glass nanoparticles scaffolds with shape memory properties. Carbohydr. Polym. 2015, 123, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Tzoneva, R.; Seifert, B.; Behl, M.; Lendlein, A. Elastic multiblock copolymers for vascular regeneration: Protein adsorption and hemocompatibility. Clin. Hemorheol. Microcirc. 2012, 52, 337–348. [Google Scholar]
- Hardy, J.G.; Palma, M.; Wind, S.J.; Biggs, M.J. Responsive Biomaterials: Advances in Materials Based on Shape-Memory Polymers. Adv. Mater. 2016, 28, 5717–5724. [Google Scholar] [CrossRef] [PubMed]
- Roelofs, L.A.J.; Oosterwijk, E.; Kortmann, B.B.M.; Daamen, W.F.; Tiemessen, D.M.; Brouwer, K.M.; Eggink, A.J.; Crevels, A.J.; Wijnen, R.M.H.; van Kuppevelt, T.H.; et al. Bladder Regeneration Using a Smart Acellular Collagen Scaffold with Growth Factors VEGF, FGF2 and HB-EGF. Tissue Eng. Part A 2016, 22, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Yuan, B.; Jin, Y.; Sun, Y.; Wang, D.; Sun, J.; Wang, Z.; Zhang, W.; Jiang, X. A strategy for depositing different types of cells in three dimensions to mimic tubular structures in tissues. Adv. Mater. 2012, 24, 890–896. [Google Scholar] [CrossRef]
- Smolar, J.; Salemi, S.; Horst, M.; Sulser, T.; Eberli, D. Stem Cells in Functional Bladder Engineering. Transfus. Med. Hemother. 2016, 43, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.G.; Cornelison, R.C.; Sukhavasi, R.C.; Saballos, R.J.; Vu, P.; Kaplan, D.L.; Schmidt, C.E. Electroactive Tissue Scaffolds with Aligned Pores as Instructive Platforms for Biomimetic Tissue Engineering. Bioengineering 2015, 2, 15–34. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, L.; Guo, B.; Shao, Y.; Ma, P.X. Electroactive biodegradable polyurethane significantly enhanced Schwann cells myelin gene expression and neurotrophin secretion for peripheral nerve tissue engineering. Biomaterials 2016, 87, 18–31. [Google Scholar] [CrossRef]
- StÖlting, M.N.L.; Arnold, A.S.; Haralampieva, D.; Handschin, C.; Sulser, T.; Eberli, D. Magnetic stimulation supports muscle and nerve regeneration after trauma in mice. Muscle Nerve 2016, 53, 598–607. [Google Scholar] [CrossRef]
- Orabi, H.; Bouhout, S.; Morissette, A.; Rousseau, A.; Chabaud, S.; Bolduc, S. Tissue engineering of urinary bladder and urethra: Advances from bench to patients. Sci. World J. 2013, 2013, 154564. [Google Scholar] [CrossRef] [PubMed]
- Dodat, H.; Landry, J.-L.; Szwarc, C.; Culem, S.; Murat, F.-J.; Dubois, R. Spongioplasty and separation of the corpora cavernosa for hypospadias repair. BJU Int. 2003, 91, 528–531. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.; Sabharwal, K.; Bhat, M.; Saran, R.; Singla, M.; Kumar, V. Outcome of tubularized incised plate urethroplasty with spongioplasty alone as additional tissue cover: A prospective study. Indian J. Urol. 2014, 30, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Beaudoin, S.; Delaage, P.-H.; Bargy, F. Anatomical basis of surgical repair of hypospadias by spongioplasty. Surg. Radiol. Anat. 2000, 22, 139–141. [Google Scholar] [CrossRef] [PubMed]
- Mauney, J.R.; Adam, R.M. Dynamic reciprocity in cell–scaffold interactions. Adv. Drug Deliv. Rev. 2015, 82–83, 77–85. [Google Scholar] [CrossRef] [PubMed]






© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbas, T.O.; Yalcin, H.C.; Pennisi, C.P. From Acellular Matrices to Smart Polymers: Degradable Scaffolds that are Transforming the Shape of Urethral Tissue Engineering. Int. J. Mol. Sci. 2019, 20, 1763. https://doi.org/10.3390/ijms20071763
Abbas TO, Yalcin HC, Pennisi CP. From Acellular Matrices to Smart Polymers: Degradable Scaffolds that are Transforming the Shape of Urethral Tissue Engineering. International Journal of Molecular Sciences. 2019; 20(7):1763. https://doi.org/10.3390/ijms20071763
Chicago/Turabian StyleAbbas, Tariq O., Huseyin C. Yalcin, and Cristian P. Pennisi. 2019. "From Acellular Matrices to Smart Polymers: Degradable Scaffolds that are Transforming the Shape of Urethral Tissue Engineering" International Journal of Molecular Sciences 20, no. 7: 1763. https://doi.org/10.3390/ijms20071763
APA StyleAbbas, T. O., Yalcin, H. C., & Pennisi, C. P. (2019). From Acellular Matrices to Smart Polymers: Degradable Scaffolds that are Transforming the Shape of Urethral Tissue Engineering. International Journal of Molecular Sciences, 20(7), 1763. https://doi.org/10.3390/ijms20071763