L-Arginine Inhibited Inflammatory Response and Oxidative Stress Induced by Lipopolysaccharide via Arginase-1 Signaling in IPEC-J2 Cells
Abstract
:1. Introduction
2. Results
2.1. Effect of LPS Stimulation on TLR4 Expression and p38 in IPEC-J2 Cells
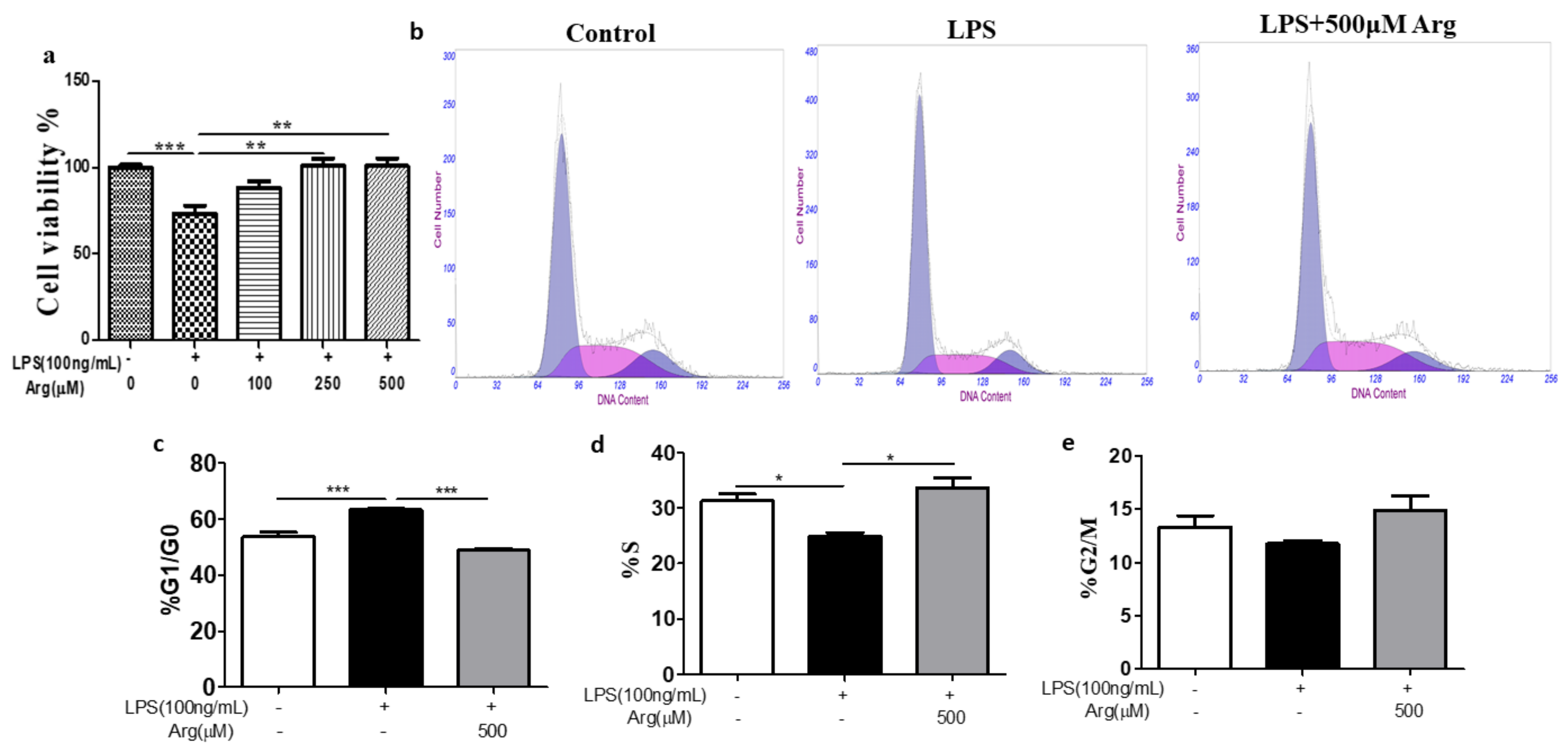
2.2. Effect of L-Arginine on Cell Survival and Cell Cycle
2.3. Effect of L-Arginine on TLR4, MyD88, CD14, and Pro-Inflammatory Cytokines in LPS-Treated IPEC-J2 Cells
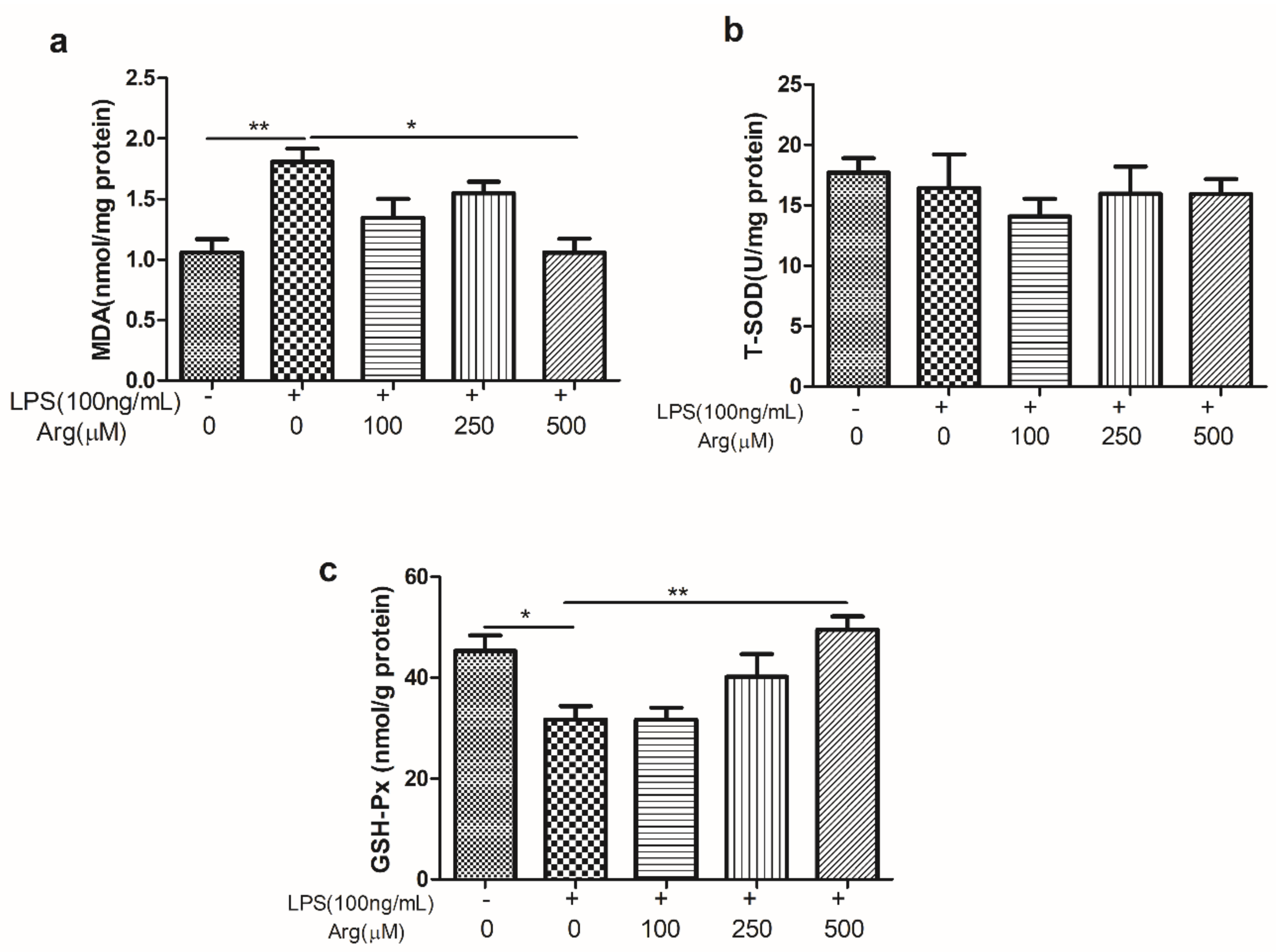
2.4. The Effect of L-Arginine on ROS Production and Contents of MDA, Total Superoxide Dismutase (T-SOD) and GSH-Px in LPS-Stimulated IPEC-J2 Cells
2.5. L-Arginine Enhanced the Activity of Arg-1, While Arg-1 Expression Inhibition Abrogated the Protection of L-Arginine against LPS-Mediated Damage
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Culture
4.3. Cell Viability
4.4. Cell Cycle Distribution
4.5. TLR4 Expression was Analyzed by Flow Cytometry
4.6. Real-Time Quantitative PCR Analysis
4.7. Determination of ROS Superoxide Levels
4.8. Evaluating the Role of Arg-1 Signaling
4.9. Western Blot Analysis of Protein Levels
4.10. Antioxidant/Oxidant Indices Analysis
4.11. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Anderson, K.V. Toll signaling pathways in the innate immune respons. Curr. Opin. Immunol. 2000, 12, 13–19. [Google Scholar] [CrossRef]
- Takeuchi, O.; Akira, S. Pattern recognition receptors and inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef] [PubMed]
- Bonham, K.S.; Orzalli, M.H.; Hayashi, K.; Wolf, A.I.; Glanemann, C.; Weninger, W.; Iwasaki, A.; Knipe, D.M.; Kagan, J.C. A promiscuous lipid-binding protein diversifies the subcellular sites of toll-like receptor signal transduction. Cell 2014, 156, 705–716. [Google Scholar] [CrossRef]
- Botos, I.; Segal, D.M.; Davies, D.R. The structural biology of Toll-like receptors. Structure 2011, 19, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Spiljar, M.; Merkler, D.; Trajkovski, M. The Immune System Bridges the Gut Microbiota with Systemic Energy Homeostasis: Focus on TLRs, Mucosal Barrier, and SCFAs. Front. Immunol. 2017, 8, 1353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandraju, R.; Murray, S.; Forman, J.; Pasare, C. Differential ability of surface and endosomal TLRs to induce CD8 T cell responses in vivo. J. Immunol. 2014, 192, 4303–4315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergman, I.M.; Rosengren, J.K.; Edman, K.; Edfors, I. European wild boars and domestic pigs display different polymorphic patterns in the Toll-like receptor (TLR) 1, TLR2, and TLR6 genes. Immunogenetics 2010, 62, 49–58. [Google Scholar] [CrossRef]
- Cao, L.; Ge, X.; Gao, Y.; Ren, Y.; Ren, X.; Li, G. Porcine epidemic diarrhea virus infection induces NF-kappaB activation through the TLR2, TLR3, and TLR9 pathways in porcine intestinal epithelial cells. J. Gen. Virol. 2015, 96, 1757–1767. [Google Scholar] [CrossRef] [PubMed]
- Zanoni, I.; Ostuni, R.; Marek, L.R.; Barresi, S.; Barbalat, R.; Barton, G.M.; Granucci, F.; Kagan, J.C. CD14 controls the LPS-induced endocytosis of Toll-like receptor 4. Cell 2011, 147, 868–880. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.T.; Bian, C.; Yuan, J.C.; Chu, W.H.; Xiang, X.; Chen, F.; Wang, C.S.; Feng, H.; Lin, J.K. Curcumin attenuates acute inflammatory injury by inhibiting the TLR4/MyD88/NF-κB signaling pathway in experimental traumatic brain injury. J. Neuroinflamm. 2014, 11, 59. [Google Scholar] [CrossRef]
- Zhou, J.; Deng, Y.; Li, F.; Yin, C.; Shi, J.; Gong, Q. Icariside II attenuates lipopolysaccharide-induced neuroinflammation through inhibiting TLR4/MyD88/NF-κB pathway in rats. Biomed. Pharmacother. 2019, 111, 315–324. [Google Scholar] [CrossRef]
- Ryu, J.K.; Kim, S.J.; Rah, S.H.; Kang, J.I.; Jung, H.E.; Lee, D.; Lee, H.K.; Lee, J.O.; Park, B.S.; Yoon, T.Y.; et al. Reconstruction of LPS Transer Cascade Reveals Structural Determinants within LBP, CD14, and TLR4-MD2 for Efficient LPS Recognition and Transfer. Immunity 2017, 46, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, N.; Saegusa, M.; Yoshida, T.; Okayasu, I. Squamouscell carcinoma arising from a seminal vesicular mcyst: Possible relationship between chronic inflammation and tumor development. Pathol. Int. 2002, 52, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Serrano, D.; Mayer, L. The role of epithelial cells in immune regulation in the gut. Semin. Immunol. 2001, 131, 63–76. [Google Scholar] [CrossRef]
- Schierack, P.; Nordhoff, M.; Pollmann, M.; Weyrauch, K.D.; Amasheh, S.; Lodemann, U.; Jores, J.; Tachu, B.; Kleta, S.; Blikslager, A.; et al. Characterization of a porcine intestinal epithelial cell line for in vitro studies of microbial pathogenesis in swine. Histochem. Cell Biol. 2006, 125, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Circu, M.L.; Aw, T.Y. Reactive oxygen species, cellular redox systems, and apoptosi. Free Radic. Biol. Med. 2010, 48, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, J.; Goichon, A.; Déchelotte, P.; Coëffier, M. Regulation of intestinal protein metabolism by amino acids. Amino Acids 2013, 45, 443–450. [Google Scholar] [CrossRef]
- Wu, G.; Morris, S.M. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 336, 1–17. [Google Scholar] [CrossRef]
- Tsikas, D.; Wu, G. Homoarginine, arginine, and relatives: Analysis, metabolism, transport, physiology, and pathology. Amino Acids 2015, 47, 1697–1702. [Google Scholar] [CrossRef]
- Anggard, E. Nitric oxide: Mediator, murderer, and medicine. Lancet 1994, 14, 1199–1206. [Google Scholar] [CrossRef]
- Valentová, K.; Vidlář, A.; Zatloukalová, M.; Stuchlík, M.; Vacek, J.; Šimánek, V.; Ulrichová, J. Biosafety and antioxidant effects of a beverage containing silymarin and arginine. A pilot, human intervention cross-over trial. Food Chem. Toxicol. 2013, 56, 178–183. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Hou, Y.; Yin, Y.; Qiu, Y.; Wu, G.; Hu, C.A.A. Roles of amino acids in preventing and treating intestinal diseases: Recent studies with pig models. Amino Acids 2017, 49, 1277–1291. [Google Scholar] [CrossRef]
- Ruth, M.R.; Field, C.J. The immune modifying effects of amino acids on gutassociated lymphoid tissue. J. Anim. Sci. Biotechnol. 2013, 4, 27. [Google Scholar] [CrossRef]
- Coburn, L.A.; Gong, X.; Singh, K.; Asim, M.; Scull, B.P.; Allaman, M.M. L-arginine supplementation improves responses to injury and inflammation in dextran sulfate sodium colitis. PLoS ONE 2012, 7, e33546. [Google Scholar] [CrossRef]
- Meng, Q.H.; Cooney, M.; Yepuri, N.; Cooney, R.N. L-arginine attenuates Interleukin-1β (IL-1β) induced Nuclear Factor Kappa-Beta (NF-κB) activation in Caco-2 cells. PLoS ONE 2017, 12, e0174441. [Google Scholar] [CrossRef]
- Zhang, H.; Peng, A.; Yu, Y.; Guo, S.; Wang, M.Z.; Wang, H.R. L-Arginine protects ovine intestinal epithelial cells from lipopolysaccharides-induced apoptosis through alleviating oxidative stress. J. Agric. Food Chem. 2019, 67, 1683–1690. [Google Scholar] [CrossRef]
- Wang, X.P.; Chen, Y.G.; Qin, W.D.; Zhang, W.; Wei, S.J.; Wang, J.; Liu, F.Q.; Gong, L.; An, F.S.; Zhang, Y.; et al. Arginase I attenuates inflammatory cytokine secretion induced by lipopolysaccharide in vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1853–1860. [Google Scholar] [CrossRef]
- Stempin, C.C.; Dulgerian, L.R.; Garrido, V.V.; Cerban, F.M. Arginase in parasitic infections: Macrophage activation, immunosuppression, and intracellular signals. J. Biomed. Biotechnol. 2010, 2010, 683485. [Google Scholar] [CrossRef]
- Mariani, V.; Palermo, S.; Fiorentini, S.; Lanubile, A.; Giuffra, E. Gene expression study of two widely used pig intestinal epithelial cell lines: IPEC-J2 and IPI-2I. Vet. Immunol. Immunopathol. 2009, 131, 278–284. [Google Scholar] [CrossRef]
- Sargeant, H.R.; Miller, H.M.; Shaw, M.A. Inflammatory response of porcine epithelial IPEC J2 cells to enterotoxigenic E. coli infection is modulated by zinc supplementation. Mol. Immunol. 2011, 48, 2113–2121. [Google Scholar] [CrossRef]
- Hermes, R.G.; Manzanilla, E.G.; Martín-Orúe, S.M.; Pérez, J.F.; Klasing, K.C. Influence of dietary ingredients on in vitro inflammatory response of intestinal porcine epithelial cells challenged by an enterotoxigenic Escherichia coli (K88). Comp. Immunol. Microbiol. Infect. Dis. 2011, 34, 479–488. [Google Scholar] [CrossRef]
- Burkey, T.E.; Skjolaas, K.A.; Dritz, S.S.; Minton, J.E. Expression of porcine Toll-like receptor 2, 4 and 9 gene transcripts in the presence of lipopolysaccharide and Salmonella enterica serovars Typhimurium and Choleraesuis. Vet. Immunol. Immunopathol. 2009, 130, 96–101. [Google Scholar] [CrossRef]
- Burkey, T.E.; Skjolaas, K.A.; Dritz, S.S.; Minton, J.E. Expression of Toll-like receptors, interleukin 8, macrophage migration inhibitory factor, and osteopontin in tissues from pigs challenged with Salmonella enterica serovar Typhimurium or serovar Choleraesuis. Vet. Immunol. Immunopathol. 2007, 115, 309–319. [Google Scholar] [CrossRef]
- Skjolaas, K.A.; Burkey, T.E.; Dritz, S.S.; Minton, J.E. Effects of Salmonella enterica serovar Typhimurium, or serovar Choleraesuis, Lactobacillus reuteri and Bacillus licheniformis on chemokine and cytokine expression in the swine jejunal epithelial cell line, IPEC-J2. Vet. Immunol. Immunopathol. 2007, 115, 299–308. [Google Scholar] [CrossRef]
- Devriendt, B.; Stuyven, E.; Verdonck, F.; Goddeeris, B.M.; Cox, E. Enterotoxigenic Escherichia coli (K88) induce proinflammatory responses in porcine intestinal epithelial cells. Dev. Comp. Immunol. 2010, 34, 1175–1182. [Google Scholar] [CrossRef]
- Brosnahan, A.J.; Brown, D.R. Porcine IPEC-J2 intestinal epithelial cells in microbiological investigations. Vet. Microbiol. 2012, 156, 229–237. [Google Scholar] [CrossRef]
- Farkas, O.; Mátis, G.; Pászti-Gere, E.; Palócz, O.; Kulcsár, A.; Petrilla, J.; Csikó, G.; Neogrády, Z.; Gálfi, P. Effects of Lactobacillus plantarum 2142 and sodium n-butyrate in lipopolysaccharide-triggered inflammation: Comparison of a porcine intestinal epithelial cell line and primary hepatocyte monocultures with a porcine enterohepatic co-culture system. J. Anim. Sci. 2014, 92, 3835–3845. [Google Scholar] [CrossRef]
- Paszti-Gere, E.; Matis, G.; Farkas, O.; Kulcsar, A.; Palocz, O.; Csiko, G.; Neogrady, Z.; Galfi, P. The Effects of Intestinal LPS Exposure on Inflamatory Responses in a Porcine Enterohepatic Coculture System. Inflammation 2014, 37, 247–260. [Google Scholar] [CrossRef]
- Geens, M.M.; Niewold, T.A. Preliminary Characterization of the Transcriptional Response of the Porcine Intestinal Cell Line IPEC-J2 to Enterotoxigenic Escherichia coli, Escherichia coli, and E. coli Lipopolysaccharide. Comp. Funct. Genom. 2010, 10, 469–583. [Google Scholar]
- Skjolaas, K.A.; Burkey, T.E.; Dritz, S.S.; Minton, J.E. Effects of Salmonella entericaserovars Typhimurium (ST) and Cholerasuis (SC) on chemokine and cytokine expression in swine ileum and jejunal epithelial cells. Vet. Immunol. Immunopathol. 2006, 111, 199–209. [Google Scholar] [CrossRef]
- Arce, C.; Ramı’rez-Boo, M.; Lucena, C.; Garrido, J.J. Innate immune activation of swine intestinal epithelial cell lines (IPEC-J2 and IPI-2I) in response to LPS from Salmonella typhimurium.Comparative Immunology. Microbiol. Infect. Dis. 2010, 33, 161–174. [Google Scholar] [CrossRef]
- Akira, S.; Takeda, K. Toll-like receptor signaling. Nat. Rev. Immunol. 2004, 4, 499–511. [Google Scholar] [CrossRef]
- Zheng, S.L.; Augustsson-Bälter, K.; Chang, B.; Hedelin, M.; Li, L.; Adami, H.O. Sequence variants of Toll-like receptor4 are associated with prostate cancer risk: Results from the cancer prostate in Sweden study. Cancer Res. 2004, 64, 2918–2922. [Google Scholar] [CrossRef]
- Kelly, M.G.; Alvero, A.B.; Chen, R.; Silasi, D.A.; Abrahams, V.M.; Chan, S. TLR-4 signaling promotes tumor growth and paclitaxel chemo resistance in ovarian cancer. Cancer Res. 2006, 66, 3859–3868. [Google Scholar] [CrossRef]
- He, W.; Liu, Q.; Wang, L.; Chen, W.; Li, N.; Cao, X. TLR4 signaling promotes immuneescape of human lung cancercells by inducing immunosuppressive cytokines and apoptosis resistance. Mol. Immunol. 2007, 44, 2850–2859. [Google Scholar] [CrossRef]
- Yin, J.; Duan, J.; Cui, Z.; Ren, W.; Li, T.; Yin, Y. Hydrogen peroxide-induced oxidative stress activates NF-κB and Nrf2/Keap1 signals and triggers autophagy in piglets. RSC Adv. 2015, 5, 15479–15486. [Google Scholar] [CrossRef]
- Yin, J.; Ren, W.K.; Wu, X.S.; Yang, G.; Wang, J.; Li, T.J.; Su, D.D. Oxidative stress-mediated signaling pathways: A review. J. Food Agric. Environ. 2013, 11, 132–139. [Google Scholar]
- Yin, J.; Wu, M.M.; Xiao, H.; Ren, W.K.; Duan, J.L.; Yang, G.; Yin, Y.L. Development of an antioxidant system after early weaning in piglets. J. Anim. Sci. 2014, 92, 612–619. [Google Scholar] [CrossRef]
- Gou, Z.; Jiang, S.; Zheng, C.; Tian, Z.; Lin, X. Equol Inhibits LPS-Induced Oxidative Stress and Enhances the Immune Response in Chicken HD11 Macrophages. Cell Physiol. Biochem. 2015, 36, 611–621. [Google Scholar] [CrossRef]
- Babior, B.M. Phagocytes and oxidative stress. Am. J. Med. 2000, 109, 33–44. [Google Scholar] [CrossRef]
- Yuan, X.; Zhou, Y.; Wang, W.; Li, J.; Xie, G.; Zhao, Y.; Xu, D.; Shen, L. Activation of TLR4 signaling promotes gastric cancer progression by inducing mitochondrial ROS production. Cell Death Dis. 2013, 4, e794. [Google Scholar] [CrossRef] [Green Version]
- Pirinccioglu, A.G.; Gokalp, D.; Pirinccioglu, M.; Kizil, G.; Kizil, M. Malondialdehyde (MDA) and protein carbonyl (PCO) levels as biomarkers of oxidative stress in subjects with familial hypercholesterolemia. Clin. Biochem. 2010, 43, 1220–1224. [Google Scholar] [CrossRef]
- Qiao, Y.; Bai, X.F.; Du, Y.G. Chitosan oligosaccharides protect mice from lps challenge by attenuation of inflammation and oxidative stress. Int. Immunopharmacol. 2011, 11, 121–127. [Google Scholar] [CrossRef]
- Liang, M.; Wang, Z.; Li, H.; Cai, L.; Pan, J.; He, H.; Wu, Q.; Tang, Y.; Ma, J.; Yang, L. L Arginine induces antioxidant response to prevent oxidative stress viastimulation of glutathione synthesis and activation of Nrf2 pathway. Food Chem. Toxicol. 2018, 115, 315–328. [Google Scholar] [CrossRef]
- Burkitt, M.D.; Sutton, R.; Hughes, K.R.; Hall, L.J.; Caamano, J.H. A mouse model of pathological small intestinal epithelial cell apoptosis and shedding induced by systemic administration of lipopolysaccharide. Dis. Mod. Mech. 2013, 6, 1388–1399. [Google Scholar] [Green Version]
- Choi, S.; Park, C.; Ahn, M.; Lee, J.H.; Shin, T. Immunohistochemical study of arginase 1 and 2 in various tissues of rats. Acta Histochem. 2012, 114, 487–494. [Google Scholar] [CrossRef]
- Qiu, Y.Q.; Jiang, Z.Y.; Hu, S.L.; Wang, L.; Ma, X.Y.; Yang, X.F. Lactobacillus plantarum Enhanced IL-22 Production in Natural Killer (NK) Cells That Protect the Integrity of Intestinal Epithelial Cell Barrier Damaged by Enterotoxigenic Escherichia coli. Int. J. Mol. Sci. 2017, 18, 2409. [Google Scholar] [CrossRef]





| Gene | Sequences (5′–3′) | Product Size (bp) | GenBank Accession | |
|---|---|---|---|---|
| TLR4 | Forward Reverse | TGACGCCTTTGTTATCTACTCC GGTCTGGGCAATCTCATACTC | 246 | NM_001113039 |
| Myd88 | Forward Reverse | CCCCAGCGATACCCAGTTTGT ATCCGACGGCACCTCTTTTCA | 152 | NM_001099923 |
| CD14 | Forward Reverse | GAGTGAGGACAGATAGCGTTTG CTGCGGATGCGTGAAGTT | 242 | NM_001097445 |
| IL-8 | Forward Reverse | CTCATTCCTGTGCTGGTCA GCAAGTTGAGGCAAGAAGAC | 270 | NM_213867 |
| NF-kappa Bp65 | Forward Reverse | ACCCCTTCCAAGTTCCC CCCGAGTTCCGATTCAC | 195 | NM_001114281 |
| IL-6 | Forward Reverse | TCAGTCCAGTCGCCTTCT TACCTCCTTGCTGTTTTCAC | 227 | NM_214399.1 |
| TNF-ɑ | Forward Reverse | CACGCTCTTCTGCCTACTGC GTCCCTCGGCTTTGACATT | 164 | NM_214022.1 |
| Arginase-1 | Forward Reverse | CTTTCTCCAAGGGTCAGC TCCCCGTAATCTTTCACAT | 115 | NM_214048.2 |
| β-actin | Forward Reverse | CACGCCATCCTGCGTCTGGA AGCACCGTGTTGGCGTAGAG | 210 | NC_010445 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, Y.; Yang, X.; Wang, L.; Gao, K.; Jiang, Z. L-Arginine Inhibited Inflammatory Response and Oxidative Stress Induced by Lipopolysaccharide via Arginase-1 Signaling in IPEC-J2 Cells. Int. J. Mol. Sci. 2019, 20, 1800. https://doi.org/10.3390/ijms20071800
Qiu Y, Yang X, Wang L, Gao K, Jiang Z. L-Arginine Inhibited Inflammatory Response and Oxidative Stress Induced by Lipopolysaccharide via Arginase-1 Signaling in IPEC-J2 Cells. International Journal of Molecular Sciences. 2019; 20(7):1800. https://doi.org/10.3390/ijms20071800
Chicago/Turabian StyleQiu, Yueqin, Xuefen Yang, Li Wang, Kaiguo Gao, and Zongyong Jiang. 2019. "L-Arginine Inhibited Inflammatory Response and Oxidative Stress Induced by Lipopolysaccharide via Arginase-1 Signaling in IPEC-J2 Cells" International Journal of Molecular Sciences 20, no. 7: 1800. https://doi.org/10.3390/ijms20071800





