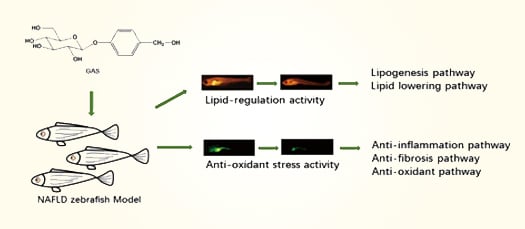
Lipid Modulating Anti-oxidant Stress Activity of Gastrodin on Nonalcoholic Fatty Liver Disease Larval Zebrafish Model
Abstract
:1. Introduction
2. Results
2.1. Effects GAS on HCD Induced Larval Zebrafish Model
2.2. Lipid Regulating Effect GAS on the Larval Zebrafish Model
2.3. Anti-oxidant Stress Effect GAS on the Larval Zebrafish Model
2.4. mRNA Expression Changes GAS on the Larval Zebrafish Model
3. Discussion
4. Materials and Method
4.1. Reagents
4.2. Preparation High Cholesterol Diet and Drug Solutions
4.3. Maintenance Larval Zebrafish and Treatment
4.4. Biochemical Measurement
4.5. Fluorescence Photography
4.6. Histopathological examination
4.7. Real-time Quantitative PCR (qRT-PCR) Analysis
4.8. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Goh, B.B.; Mccullough, A.J. Natural History of Nonalcoholic Fatty Liver Disease. Dig. Dis. Sci. 2016, 61, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Diehl, A.M.; Day, C. Cause, Pathogenesis, and Treatment of Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2017, 377, 2063–2072. [Google Scholar] [CrossRef]
- Ying, L.; Yan, F.; Zhao, Y.; Gao, H.; Williams, B.R.; Hu, Y.; Li, X.; Tian, R.; Xu, P.; Wang, Y. (−)–Epigallocatechin-3-gallate and atorvastatin treatment down-regulates liver fibrosis-related genes in non-alcoholic fatty liver disease. Clin. Exp. Pharmacol. Physiol. 2017, 44, 1180. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Rosa, M.D.; Nicoletti, F.; Malaguarnera, L. Molecular mechanisms involved in NAFLD progression. J. Mol. Med. 2009, 87, 679. [Google Scholar] [CrossRef]
- Qin, G.; Ma, J.; Huang, Q.; Yin, H.; Han, J.; Li, M.; Deng, Y.; Wang, B.; Hassan, W.; Shang, J. Isoquercetin Improves Hepatic Lipid Accumulation by Activating AMPK Pathway and Suppressing TGF-beta Signaling on an HFD-Induced Nonalcoholic Fatty Liver Disease Rat Model. Int. J. Mol. Sci. 2018, 19, 4126. [Google Scholar] [CrossRef]
- Li, N.; Wang, K.J.; Chen, J.J.; Zhou, J. Phenolic compounds from the rhizomes of Gastrodia elata. J. Asian Nat. Prod. Res. 2007, 9, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gao, J.; Peng, M.; Meng, H.; Ma, H.; Cai, P.; Xu, Y.; Zhao, Q.; Si, G. A Review on Central Nervous System Effects of Gastrodin. Front. Pharmacol. 2018, 9, 24. [Google Scholar] [CrossRef]
- Geng, Y.; Bin, Y.U.; Zuo, Z.; Kong, W. Experimental study of Gastrodia Powder in improving hepatic steatosis in rats intragastrically administered with fat emulsion. China Med. Her. 2013, 10, 11–15. [Google Scholar]
- Geng, Y.N.; Bin, Y.U.; Kong, W.J. Gastrodin ameliorates oleic acid-induced fat accumulation through activation of AMPK pathway in HL-7702 cells. Chin. Pharmacol. Bull. 2015, 1, 39–44. [Google Scholar]
- Peng, Z.; Wang, S.; Chen, G.; Cai, M.; Liu, R.; Deng, J.; Liu, J.; Zhang, T.; Tan, Q.; Hai, C. Gastrodin Alleviates Cerebral Ischemic Damage in Mice by Improving Anti-oxidant and Anti-inflammation Activities and Inhibiting Apoptosis Pathway. Neurochem. Res. 2015, 40, 661–673. [Google Scholar] [CrossRef]
- Yin, H.; Wang, S.; Zhang, Y.; Wu, M.; Wang, J.; Ma, Y. Zuogui Pill improves the dexamethasone-induced osteoporosis progression in zebrafish larvae. Biomed. Pharmacother. 2017, 97, 995–999. [Google Scholar] [CrossRef] [PubMed]
- Amali, A.A.; Rekha, R.D.; Lin, J.F.; Wang, W.L.; Gong, H.Y.; Her, G.M.; Wu, J.L. Thioacetamide induced liver damage in zebrafish embryo as a disease model for steatohepatitis. J. Biomed. Sci. 2006, 13, 225–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matthews, R.P.; Lorent, K.; Mañoral-Mobias, R.; Huang, Y.; Gong, W.; Murray, I.V.; Blair, I.A.; Pack, M. TNFalpha-dependent hepatic steatosis and liver degeneration caused by mutation of zebrafish S-adenosylhomocysteine hydrolase. Development 2009, 136, 865. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Li, M.; Kalavagunta, P.K.; Li, J.; He, Q.; Zhang, Y.; Ahmad, O.; Yin, H.; Wang, T.; Shang, J. Protective effects of cichoric acid on H2O2-induced oxidative injury in hepatocytes and larval zebrafish models. Biomed. Pharmacother. 2018, 104, 679–685. [Google Scholar] [CrossRef]
- Ma, J.; Yin, H.; Li, M.; Deng, Y.; Ahmad, O.; Qin, G.; He, Q.; Li, J.; Gao, K.; Zhu, J.; et al. A Comprehensive Study of High Cholesterol Diet-Induced Larval Zebrafish Model: A Short-Time In Vivo Screening Method for Non-Alcoholic Fatty Liver Disease Drugs. Int. J. Biol. Sci. 2019, 15, 973–983. [Google Scholar] [CrossRef]
- Greenspan, P.; Fowler, S.D. Spectrofluorometric studies of the lipid probe, nile red. J. Lipid Res. 1985, 26, 781–798. [Google Scholar]
- Bagatto, B.; Pelster, B.; Burggren, W.W. Growth and metabolism of larval zebrafish: Effects of swim training. J. Exp. Biol. 2001, 204, 4335–4343. [Google Scholar]
- Kobyliak, N.; Virchenko, O.; Falalyeyeva, T.; Kondro, M.; Beregova, T.; Bodnar, P.; Shcherbakov, O.; Bubnov, R.; Caprnda, M.; Delev, D. Cerium dioxide nanoparticles possess anti-inflammatory properties in the conditions of the obesity-associated NAFLD in rats. Biomed. Pharmacother. 2017, 90, 608. [Google Scholar] [CrossRef]
- Koek, G.H.; Liedorp, P.R.; Bast, A. The role of oxidative stress in non-alcoholic steatohepatitis. Clin. Chim. Acta 2011, 412, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Rolo, A.P.; Teodoro, J.S.; Palmeira, C.M. Role of oxidative stress in the pathogenesis of nonalcoholic steatohepatitis. Free Radic. Biol. Med. 2012, 52, 59–69. [Google Scholar] [CrossRef]
- Sumida, Y.; Naito, Y.; Yoshikawa, T.; Niki, E. Special issue on “Oxidative stress and redox signaling in the;gastrointestinal tract and related organs” for Free Radical Research;Involvement of free radicals and oxidative stress in NAFLD/NASH. Free Radic. Res. 2013, 47, 869–880. [Google Scholar] [CrossRef]
- Sprague, J.; Doerry, E.; Douglas, S.; Westerfield, M. The Zebrafish Information Network (ZFIN): A resource for genetic, genomic and developmental research. Nucleic Acids Res. 2001, 29, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Sim, W.C.; Kim, D.G.; Lee, K.J.; Choi, Y.J.; Choi, Y.J.; Shin, K.J.; Jun, D.W.; Park, S.J.; Park, H.J.; Kim, J. Cinnamamides, Novel Liver X Receptor Antagonists that Inhibit Ligand-Induced Lipogenesis and Fatty Liver. J. Pharmacol. Exp. Ther. 2015, 355, 362. [Google Scholar] [CrossRef]
- Montagner, A.; Polizzi, A.; Fouché, E.; Ducheix, S.; Lippi, Y.; Lasserre, F.; Barquissau, V.; Régnier, M.; Lukowicz, C.; Benhamed, F. Original article: Liver PPARα is crucial for whole-body fatty acid homeostasis and is protective against NAFLD. Gut 2016, 65, 1202–1214. [Google Scholar] [CrossRef]
- Arrese, M.; Cabrera, D.; Kalergis, A.M.; Feldstein, A.E. Innate Immunity and Inflammation in NAFLD/NASH. Dig. Dis. Sci. 2016, 61, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Zhou, C.; Yang, X.; Li, L. Down-regulation of microRNA-375 regulates adipokines and inhibits inflammatory cytokines by targeting AdipoR2 in non-alcoholic fatty liver disease. Clin. Exp. Pharmacol. Physiol. 2018, 45, 819–831. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.J.; Liu, J. Actein ameliorates hepatic steatosis and fibrosis in high fat diet-induced NAFLD by regulation of insulin and leptin resistant. Biomed. Pharmacother. 2017, 97, 1386. [Google Scholar] [CrossRef]
- Rui, W.; Zou, Y.; Lee, J.; Nambiar, S.M.; Lin, J.; Zhang, L.; Yang, Y.; Dai, G. Nuclear Factor Erythroid 2–Related Factor 2 Deficiency Results in Amplification of the Liver Fat-Lowering Effect of Estrogen. J. Pharmacol. Exp. Ther. 2016, 358, 14. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, W.; Liu, X.; Xue, P.; Jiang, S.; Lu, D.; Zhang, Q.; He, G.; Pi, J.; Andersen, M.E. Iodoacetic acid activates Nrf2-mediated antioxidant response in vitro and in vivo. Environ. Sci. Technol. 2014, 48, 13478–13488. [Google Scholar] [CrossRef]
- Holze, C.; Michaudel, C.; Mackowiak, C.; Haas, D.A.; Benda, C.; Hubel, P.; Pennemann, F.L.; Schnepf, D.; Wettmarshausen, J.; Braun, M. Oxeiptosis, a ROS-induced caspase-independent apoptosis-like cell-death pathway. Nat. Immunol. 2017, 19, 130. [Google Scholar] [CrossRef]
- Liu, D.; He, Z.; Wu, L.; Fang, Y. Effects of induction/inhibition of endogenous heme oxygenase-1 on lipid metabolism, endothelial function, and atherosclerosis in rabbits on a high fat diet. J. Pharmacol. Sci. 2012, 118, 14. [Google Scholar] [CrossRef] [PubMed]





| Gene Name | Acceccion Number (Danio rerio) | Forward Primer (5′->3′) | Reverse Primer (5′->3′) |
|---|---|---|---|
| Danio Rerio | |||
| srebf1 | NM_001105129 | CATCCACATGGCTCTGAGTG | CTCATCCACAAAGAAGCGGT |
| fasn | XM_005169478 | ATCTGTTCCTGTTCGATGGC | AGCATATCTCGGCTGACGTT |
| pparab | NM_001102567 | CGTCGTCAGGTGTTTACGGT | AGGCACTTCTGGAATCGACA |
| pparg | NM_131467 | CTGCCGCATACACAAGAAGA | TCACGTCACTGGAGAACTCG |
| tnfa | NM_212859 | GCTTATGAGCCATGCAGTGA | TGCCCAGTCTGTCTCCTTCT |
| il1b | NM_212844 | TGGCGAACGTCATCCAAG | GGAGCACTGGGCGACGCATA |
| il6 | NM_001261449 | AGACCGCTGCCTGTCTAAAA | TTTGATGTCGTTCACCAGGA |
| mmp9 | NM213123.1 | GAAGCGTTACGGCTACGT | TTCCATGTCTGGCGAATAG |
| tgfb | NM_182873.1 | CATAAGAGCCACAGACAGAAG | GTAGAGCGAGCGTAAACAG |
| keap1 | NM_182864.2 | CCAACGGCATAGAGGTAGTTAT | CCTGTATGTGGTAGGAGGGTT |
| nrf2 | NM_182889.1 | TTGTCTTTGGTGAACGGAGGT | CTCGGAGGAGATGGAAGGAAG |
| HO-1 | NM_001127516.1 | GCT CAA CAT CCA GCT CTT TGA GG | GAC AAA GTT CAT GGC CCTGGG A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, O.; Wang, B.; Ma, K.; Deng, Y.; Li, M.; Yang, L.; Yang, Y.; Zhao, J.; Cheng, L.; Zhou, Q.; et al. Lipid Modulating Anti-oxidant Stress Activity of Gastrodin on Nonalcoholic Fatty Liver Disease Larval Zebrafish Model. Int. J. Mol. Sci. 2019, 20, 1984. https://doi.org/10.3390/ijms20081984
Ahmad O, Wang B, Ma K, Deng Y, Li M, Yang L, Yang Y, Zhao J, Cheng L, Zhou Q, et al. Lipid Modulating Anti-oxidant Stress Activity of Gastrodin on Nonalcoholic Fatty Liver Disease Larval Zebrafish Model. International Journal of Molecular Sciences. 2019; 20(8):1984. https://doi.org/10.3390/ijms20081984
Chicago/Turabian StyleAhmad, Owais, Bing Wang, Kejian Ma, Yang Deng, Maoru Li, Liping Yang, Yuqi Yang, Jingyun Zhao, Lijun Cheng, Qinyang Zhou, and et al. 2019. "Lipid Modulating Anti-oxidant Stress Activity of Gastrodin on Nonalcoholic Fatty Liver Disease Larval Zebrafish Model" International Journal of Molecular Sciences 20, no. 8: 1984. https://doi.org/10.3390/ijms20081984
APA StyleAhmad, O., Wang, B., Ma, K., Deng, Y., Li, M., Yang, L., Yang, Y., Zhao, J., Cheng, L., Zhou, Q., & Shang, J. (2019). Lipid Modulating Anti-oxidant Stress Activity of Gastrodin on Nonalcoholic Fatty Liver Disease Larval Zebrafish Model. International Journal of Molecular Sciences, 20(8), 1984. https://doi.org/10.3390/ijms20081984