Identification and Characterization of Verticillium longisporum Lineage A1/D1 from Brassica Crops in Manitoba, Canada
Abstract
:1. Introduction
2. Results
2.1. Pathogen Isolation
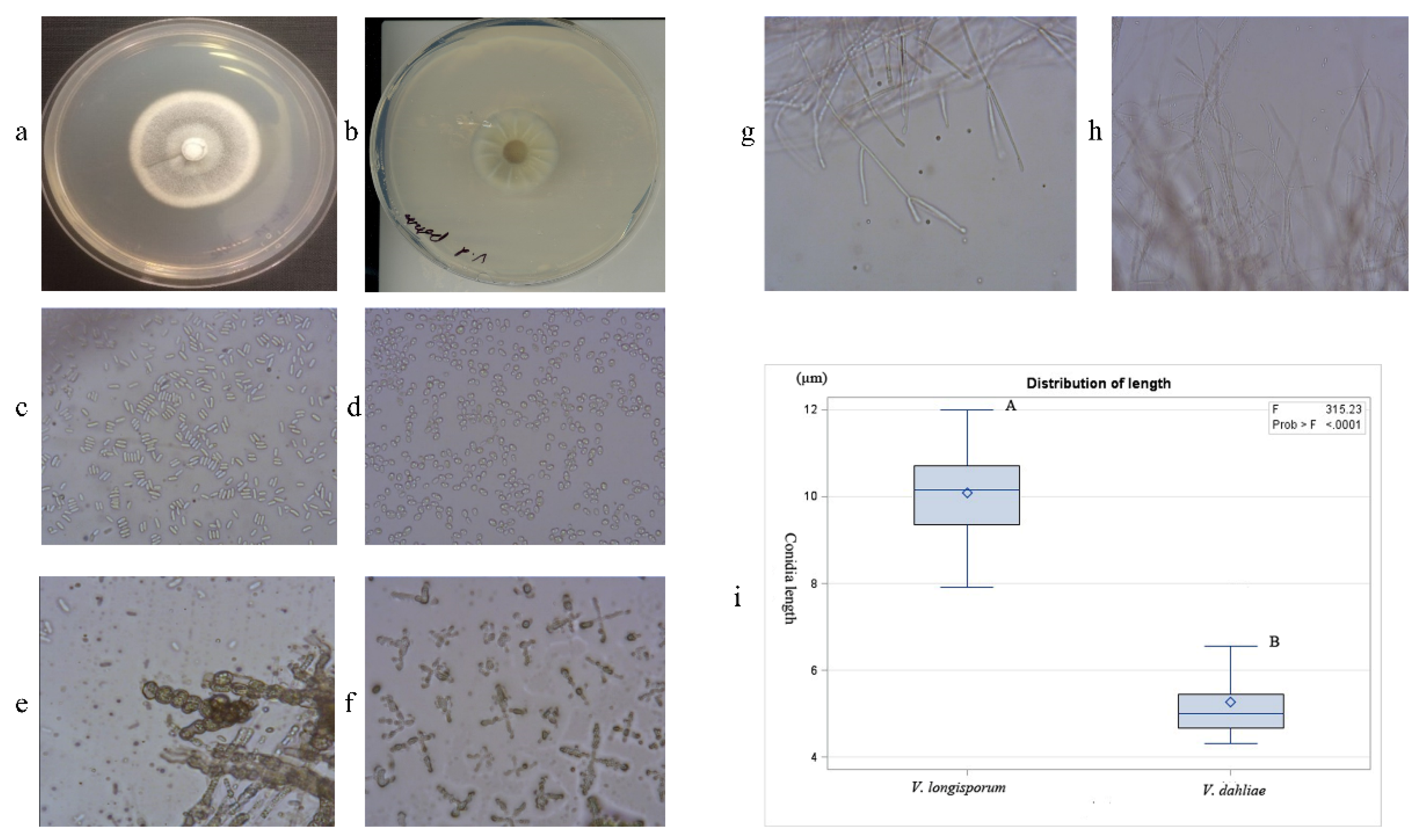
2.2. Morphological Identification
2.3. PCR Characterisation
2.4. Disease Assessment
3. Discussion
4. Materials and Methods
4.1. Sample Collection and Fungal Pathogen Isolation
4.2. Morphological Identification of Isolates
4.3. DNA extraction, PCR Amplification and PCR-Restriction Fragment Length Polymorphism
4.4. Pathogenicity Tests of V. longisporum on Canola
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nagaharu, U. Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. Jpn. J. Bot. 1935, 7, 389–452. [Google Scholar]
- Kaneko, Y.; Kimizuka-Takagi, C.; Bang, S.W.; Matsuzawa, Y. Genome Mapping and Molecular Breeding in Plants; kole, C., Ed.; Springer: New York, NY, USA, 2007; Volume 5, pp. 141–160. [Google Scholar]
- Isaac, I. Verticillium wilt of Brussels Sprout. Ann. Appl. Biol. 1957, 45, 276–283. [Google Scholar] [CrossRef]
- Stark, C. Das Auftreten der Verticillium-Tracheomykosen in Hamburger Gartenbau-kulturen. Gartenbauwissenschaft 1961, 26, 493–528. [Google Scholar]
- Karapapa, V.K.; Bainbridge, B.W.; Heale, J.B. Morphological and molecular characterization of Verticillium longisporum comb. nov., pathogenic to oilseed rape. Mycol. Res. 1997, 101, 1281–1294. [Google Scholar] [CrossRef]
- Steventon, L.; Fahleson, J.; Hu, Q.; Dixelius, C. Identification of the causal agent of Verticillium wilt of winter oilseed rape in Sweden as Verticillium longisporum. Mycol. Res. 2002, 106, 570–578. [Google Scholar] [CrossRef]
- Johansson, A.; Goud, J.K.C.; Dixelius, C. Plant host range of Verticillium longisporum and microsclerotia density in Swedish soils. Eur. J. Plant Pathol. 2006, 114, 139–149. [Google Scholar] [CrossRef]
- Inderbitzin, P.; Davis, R.M.; Bostock, R.M.; Subbarao, K.V. The ascomycete Verticillium longisporum is a hybrid and a plant pathogen with an expanded host range. PLoS ONE 2011, 6, e18260. [Google Scholar] [CrossRef] [Green Version]
- Ikeda, K.; Banno, S.; Watanabe, K.; Fujinaga, M.; Ogiso, H.; Sakai, H.; Tanaka, H.; Miki, S.; Shibata, S.; Shiraishi, T.; et al. Association of Verticillium dahliae and Verticillium longisporum with Chinese cabbage yellows and their distribution in the main production areas of Japan. J. Gen. Plant Pathol. 2012, 78, 331–337. [Google Scholar] [CrossRef]
- Hwang, S.; Strelkov, S.E.; Ahmed, H.U.; Zhou, Q.; Fu, H.; Fredua-Agyeman, R.; Turnbull, G.D. First report of Verticillium dahliae Kleb. Causing wilt symptoms in canola (Brassica napus L.) in North America. Can. J. Plant Pathol. 2017, 39, 514–526. [Google Scholar] [CrossRef]
- Pegg, G.F.; Brady, B.L. Verticillium Wilts; CABI Publishing: Wallingford, Oxfordshire, UK, 2002. [Google Scholar]
- Rashid, K.Y.; Desjardins, M.L. Diseases of sunflower in Manitoba in 2014. Can. Plant Dis. Surv. 2015, 95, 185–187. [Google Scholar]
- Alkher, H.; El Hadrami, A.; Rashid, K.Y.; Adam, L.R.; Daayf, F. Pathogenic variation of Verticillium dahliae after serial passes through potato and sunflower. Can. J. Plant Pathol. 2009, 31, 427–438. [Google Scholar] [CrossRef]
- Inderbitzin, P.; Subbarao, K.V. Verticillium systematics and evolution: How confucion impedes Verticillium wilt management and how to resolve it. Phytopathology 2014, 104, 564–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banno, S.; Saito, H.; Sakai, H.; Urushibara, T.; Ikeda, K.; Kabe, T.; Kemmoch, I.; Fujimura, M. Quantitative nested real-time PCR detection of Verticillium longisporum and V. dahliae in the soil of cabbage fields. J. Gen. Plant Pathol. 2011, 77, 282–291. [Google Scholar] [CrossRef]
- Banno, S.; Ikeda, K.; Saito, H.; Sakai, H.; Urushibara, T.; Shiraishi, T.; Fujimura, M. Characterization and distribution of two subtypes of Verticillium longisporum isolated from cabbage fields in Japan. J. Gen. Plant Pathol. 2015, 81, 118–126. [Google Scholar] [CrossRef]
- Eynck, C.; Koopmann, B.; Karlovsky, P.; von Tiedemann, A. Internal resistance in winter oilseed rape inhibits systemic spread of vascular pathogen Verticillium longisporum. Phytopathol 2009, 99, 802–811. [Google Scholar] [CrossRef] [Green Version]
- Paun, O.; Fay, M.F.; Soltis, D.E.; Chase, M.W. Genetic and epigenetic alterations after hybridization and genome doubling. Taxon 2007, 56, 649–656. [Google Scholar] [CrossRef]
- Tran, V.T.; Braus-Stromeyer, S.A.; Timpner, C.; Braus, G.H. Molecular diagnosis to discriminate pathogen and a pathogen species of the hybrid Verticillium longisporum on the oilseed crop Brassica napus. Appl. Microbiol. Biotechnol. 2013, 97, 4467–4483. [Google Scholar] [CrossRef] [Green Version]
- Novakazi, F.; Inderbitzin, P.; Sandoya, G.; Hayes, R.J.; von Tiedemann, A.; Subbarao, K.V. The three lineages of the diploid hybrid Verticillium longisporum differ in virulence and pathogenicity. Phytopathology 2015, 105, 662–673. [Google Scholar] [CrossRef] [Green Version]
- Subbarao, K.V.; Hubbard, J.C.; Greathead, A.S.; Spencer, G.A. Compendium of Lettuce Diseases; Davis, R.M., Subbarao, K.V., Raid, R.N., Kurtz, E.A., von Tiedemann, A., Subbarao, K.V., Eds.; The American Phytopathological Society: Saint Paul, MN, USA, 1997; pp. 26–27. [Google Scholar]
- Depotter, J.R.L.; Seidl, M.F.; van de Berg, G.C.M.; Thommma, B.P.H.L. A distinct and genetically diverse lineage of the hybrid fungal pathogen Verticillium longiporum population causes stem striping in British oilseed rape. Environ. Microbiol. 2017, 19, 3997–4009. [Google Scholar] [CrossRef] [Green Version]
- Zeise, K.; Buchmülle, M. Studies on the susceptibility to Verticillium dahliae KLEB. var. longisporum STARK of six related Brassica species. Z. Pflanzenkrankh. Pflanzenschutz 1997, 104, 501–505. [Google Scholar]
- Rygulla, W.; Snowdon, R.J.; Eynck, C.; Koopmann, B.; von Tiedemann, A.; Lühs, W.; Friedt, W. Broadening the genetic basis of Verticillium longisporum resistance in Brassica napus by interspecific hybridization. Phytopathology 2007, 97, 1391–1396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liban, S.H.; Cross, D.J.; Kutcher, H.R.; Peng, G.; Fernando, W.G.D. Race structure and frequency of avirulence genes in the western Canadian Leptosphaeria maculans pathogen population, the causal agent of blackleg in brassica species. Plant Pathol. 2016, 65, 1161–1169. [Google Scholar] [CrossRef] [Green Version]
- Inderbitzin, P.; Davis, R.M.; Bostock, R.M.; Subbarao, K.V. Identification and differentiation of Verticillium species and V. longisporum lineages by simplex and multiplex PCR assays. PLoS ONE 2013, 6, e65990. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, K.N.; Rouse, D.I.; German, T.L. PCR primers that allow intergeneric differentiation of ascomycetes and their application to Verticillium spp. Appl. Environ. Microbio. 1994, 60, 4324–4331. [Google Scholar] [CrossRef] [Green Version]
- Zeise, K. Gewächshaustest zur Resistenzprüfung von Winterraps (Brassica napus L. var. oleifera Metzger) gegen den Erreger der Rapswelke Verticillium dahliae Kleb. Nachrichtenbl Deutscher Pflanzenschutzd 1992, 44, 125–128. [Google Scholar]
- Eynck, C.; Koopmann, B.; Gruenewaldt-Stoecker, G.; Karlovsky, P.; von Tiedemann, A. Differential interactions of Verticillium longisporum and V. dahliae with Brassica napus detected with molecular and histological techniques. Eur. J. Plant Pathol. 2007, 118, 259–274. [Google Scholar] [CrossRef]


| Inoculum. | Days Post-Inoculation (dpi) | ||||
|---|---|---|---|---|---|
| 7 | 14 | 21 | 28 | 35 | |
| Westar-water | 1.00(±0.00) 1a 2 | 1.00(±0.00)a | 1.00(±0.00)a | 1.85(±0.00)a | 2.15(±0.00)a |
| Westar-VL-H1 | 1.10(±0.00)a | 2.05(±0.00)a | 3.65(±0.00)b | 4.85(±0.25)b | 5.95(±0.21)b |
| Westar-VL-H2 | 1.00(±0.00)a | 2.25(±0.00)a | 2.95(±0.00)b | 4.75(±0.22)b | 6.15(±0.23)b |
| Westar-VL-H3 | 1.25(±0.00)a | 2.15(±0.00)a | 3.25(±0.10)b | 4.20(±0.20)b | 5.25(±0.18)b |
| Westar-VL-H4 | 1.15(±0.00)a | 2.00(±0.00)a | 3.75(±0.15)b | 4.45(±0.17)b | 5.75(±0.23)b |
| Westar-VL-H5 | 1.05(±0.00)a | 2.25(±0.10)b | 4.05(±0.20)b | 4.95(±0.21)b | 5.75(±0.24)b |
| Westar-VL-H6 | 1.00(±0.00)a | 2.35(±0.12)b | 3.85(±0.10)b | 4.75(±0.22)b | 6.05(±0.18)b |
| Westar-VL-H7 | 1.10(±0.00)a | 2.00(±0.00)a | 3.45(±0.00)b | 4.95(±0.21)b | 5.75(±0.20)b |
| Westar-VL-H8 | 1.15(±0.00)a | 2.05(±0.10)a | 3.35(±0.00)b | 4.50(±0.15)b | 5.75(±0.20)b |
| Westar-VL-H9 | 1.25(±0.00)a | 2.00(±0.00)a | 3.65(±0.10)b | 4.70(±0.21)b | 5.65(±0.16)b |
| Westar-VL-H10 | 1.20(±0.00)a | 2.15(±0.10)a | 3.75(±0.12)b | 4.95(±0.24)b | 5.85(±0.18)b |
| Westar-VL-H11 | 1.10(±0.00)a | 3.05(±0.10)b | 3.95(±0.12)b | 4.65(±0.20)b | 5.25(±0.15)b |
| Westar-VL-H12 | 1.05(±0.00)a | 2.15(±0.09)a | 3.75(±0.15)b | 4.80(±0.22)b | 5.75(±0.18)b |
| Westar-VL-H13 | 1.25(±0.00)a | 2.25(±0.10)a | 3.95(±0.10)b | 5.15(±0.20)b | 6.15(±0.18)b |
| Westar-VL-H14 | 1.00(±0.00)a | 2.00(±0.00)a | 3.45(±0.10)b | 4.75(±0.19)b | 5.75(±0.20)b |
| Westar-VL-H15 | 1.15(±0.00)a | 2.25(±0.00)a | 3.70(±0.10)b | 4.95(±0.20)b | 5.90(±0.20)b |
| Westar-VL-H16 | 1.00(±0.00)a | 1.95(±0.00)a | 3.45(±0.10)b | 4.50(±0.15)b | 5.65(±0.15)b |
| Westar-VL-H17 | 1.10(±0.00)a | 2.25(±0.00)a | 3.50(±0.10)b | 4.75(±0.21)b | 6.05(±0.22)b |
| Westar-VL-H18 | 1.20(±0.00)a | 2.00(±0.00)a | 3.50(±0.00)b | 4.90(±0.25)b | 5.85(±0.20)b |
| Westar-VL-H19 | 1.00(±0.00)a | 2.15(±0.08)a | 3.45(±0.20)b | 4.75(±0.24)b | 5.75(±0.20)b |
| Westar-VL-H20 | 1.20(±0.00)a | 2.25(±0.00)a | 3.75(±0.10)b | 5.05(±0.22)b | 5.45(±0.15)b |
| Westar-VL-H21 | 1.05(±0.00)a | 2.45(±0.00)b | 3.80(±0.20)b | 4.95(±0.20)b | 5.70(±0.23)b |
| Westar-VL-H22 | 1.20(±0.00)a | 2.00(±0.00)a | 3.50(±0.15)b | 4.85(±0.25)b | 5.85(±0.21)b |
| Westar-VL-H23 | 1.00(±0.00)a | 2.05(±0.00)a | 3.55(±0.11)b | 4.45(±0.20)b | 5.40(±0.15)b |
| Westar-VL-H24 | 1.50(±0.00)a | 2.55(±0.05)b | 3.75(±0.10)b | 4.95(±0.19)b | 6.15(±0.25)b |
| Westar-VL-H25 | 1.15(±0.00)a | 2.25(±0.00)a | 3.55(±0.10)b | 4.55(±0.15)b | 5.65(±0.20)b |
| Westar-VL-H26 | 1.15(±0.00)a | 2.15(±0.00)a | 3.50(±0.10)b | 4.95(±0.22)b | 5.80(±0.16)b |
| Westar-VL-H27 | 1.20(±0.00)a | 2.05(±0.00)a | 3.75(±0.10)b | 4.75(±0.25)b | 5.40(±0.25)b |
| Westar-VL-H28 | 1.00(±0.00)a | 2.00(±0.00)a | 3.25(±0.10)b | 4.50(±0.20)b | 5.05(±0.15)b |
| Westar-VL-H29 | 1.20(±0.00)a | 2.15(±0.00)a | 3.45(±0.00)b | 4.80(±0.25)b | 5.25(±0.11)b |
| Westar-VL-R 3 | 1.25(±0.00)a | 2.45(±0.00)a | 3.80(±0.11)b | 5.05(±0.21)b | 6.45(±0.25)b |
| Westar-Vd-M 4 | 1.00(±0.00)a | 2.00(±0.00)a | 3.25(±0.10)b | 4.50(±0.21)b | 5.25(±0.14)b |
| Primer. | Sequence | Amplicon/Locus | Reference |
|---|---|---|---|
| VlspF1 | AGCCTGAGTCACGAGAGATATGGG | 18S intron | Banno et al. 2011 [15] |
| VlspR4 | CAAACCACGCCACTGCATTCTCGT | ||
| VeruniF2 | TCGTAGTAGAAGCTCGGCCTCCGGTC | 18S rDNA | Ikeda et al. 2012 [9] |
| VeruniR3 | TAAGAAGTCGGCGTACTACCGGGGT | ||
| VdMAT1-1-1F01 | CAACTCCTGGACTCTCATCG | MAT1-1-1 | Banno et al. 2015 [16] |
| VdMAT1-1-1R01 | AAATGGTCTGAGTCGGGGGT | ||
| VdMAT1-2-1F02-2 | CTAAGGCTCCAAGTCAATCC | MAT1-2-1 | Banno et al. 2015 [16] |
| VdMAT1-2-1R02-2 | TGCTGCCACTTGTTCAAACG | ||
| D1f | CCCCGGCCTTGGTCTGAT | GPD gene1 | Inderbitzin et al. 2013 [26] |
| AlfD1r | TGCCGGCATCGACCTTGG | ||
| A1f | AAGTGGAGCCCCGTATCTTGAAT | EF-1α2 | Inderbitzin et al. 2013 [26] |
| A1r | CAACTGGCAACAGGGCTTGAAT | ||
| Df | CCGGTCCATCAGTCTCTCTG | ITS | Inderbitzin et al. 2013 [26] |
| Dr | CTGTTGCCGCTTCACTCG | ||
| NMS1 | CAGCAGTGAGGAATATTGGTCAATG | mtSSU-rDNA | Li et al. 1994 [27] |
| NMS2 | GCGGATCATCGAATTAAATAACAT | ||
| cobF1 | GTTGAATTTATCTGAGGAGG | Cytochrome b | Ikeda et al. 2012 [9] |
| cobR1 | GGAGGAGTTTGCATTGGATTAGCC |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zou, Z.; Bisht, V.; Fernando, W.G.D. Identification and Characterization of Verticillium longisporum Lineage A1/D1 from Brassica Crops in Manitoba, Canada. Int. J. Mol. Sci. 2020, 21, 3499. https://doi.org/10.3390/ijms21103499
Zou Z, Bisht V, Fernando WGD. Identification and Characterization of Verticillium longisporum Lineage A1/D1 from Brassica Crops in Manitoba, Canada. International Journal of Molecular Sciences. 2020; 21(10):3499. https://doi.org/10.3390/ijms21103499
Chicago/Turabian StyleZou, Zhongwei, Vikram Bisht, and W. G. Dilantha Fernando. 2020. "Identification and Characterization of Verticillium longisporum Lineage A1/D1 from Brassica Crops in Manitoba, Canada" International Journal of Molecular Sciences 21, no. 10: 3499. https://doi.org/10.3390/ijms21103499






