Promoter Methylation of Selected Genes in Non-Small-Cell Lung Cancer Patients and Cell Lines
Abstract
1. Introduction
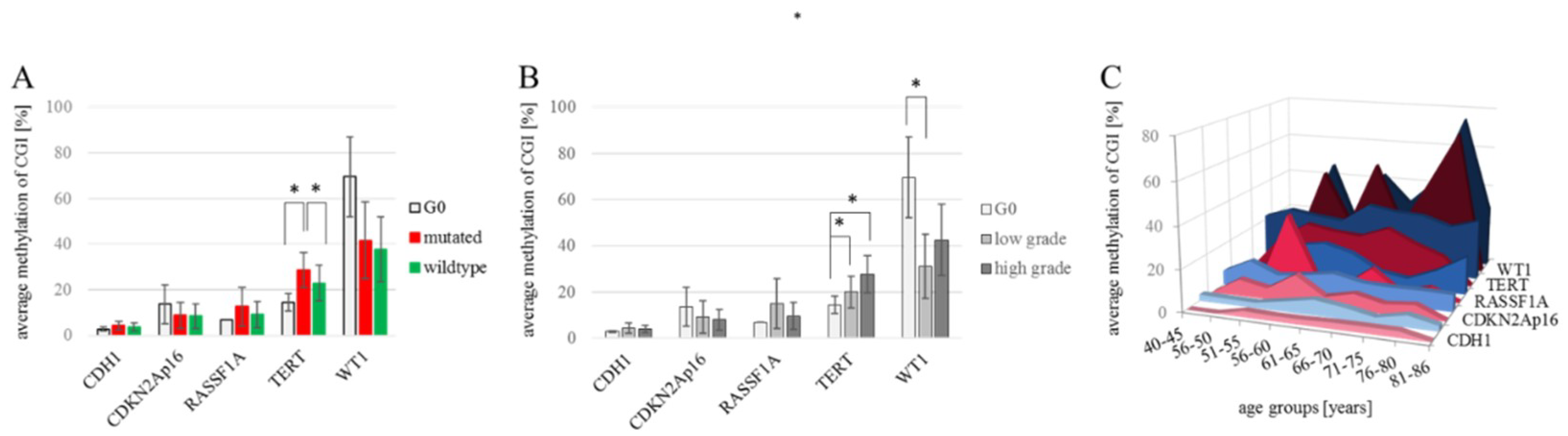
2. Results
2.1. FFPE Samples vs. Cell Lines
2.2. 2D vs. 3D Cell Culture and Methylation Level Stability
2.3. EC50 Values
3. Discussion
4. Materials and Methods
4.1. Assay Development and Validation
4.2. Sequencing Assays
4.3. Formalin-Fixed, Paraffin-Embedded (FFPE) Samples
4.4. Cell Culture
4.5. Zebularine/Decitabine Treatment and EC50 Determination
4.6. Statistical Methods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CDH1 | Cadherin 1 |
| CDKN2Ap16 | Cyclin-Dependent Kinase Inhibitor 2A/p16 |
| CGI | CpG-island |
| CLSI | Clinical and Laboratory Standards Institute |
| DMGs | differentially methylated genes |
| DNMTI | methyltransferase inhibitors |
| EGFR | epidermal growth factor receptor |
| EMT | Epithelial–mesenchymal transition |
| FFPE | formalin-fixed, paraffin-embedded |
| KRAS | KRAS proto-oncogene, GTPase |
| LC | Lung cancer |
| LINE-1 | global interspersed nuclear elements-1 |
| LoB | Limit of Blank |
| LoD | Limit of Detection |
| LoQ | Limit of Quantification |
| NSCLC | Non-small-cell lung cancer |
| RASSF1A | RAS-association domain family 1 isoform A |
| RFU | relative fluorescence units |
| SCLC | Small cell lung cancer |
| TERT | Telomerase Reverse Transcriptase |
| TKIs | tyrosine kinase inhibitors |
| WT1 | Wilms’ tumor 1 |
References
- Ferlay, J.E.M.; Lam, F.; Colombet, M.; Mery, L.; Piñeros, M.; Znaor, A.; Soerjomataram, I.; Bray, F. Global Cancer Observatory: Cancer Today. Available online: https://gco.iarc.fr/ (accessed on 24 September 2019).
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef]
- Barta, J.A.; Powell, C.A.; Wisnivesky, J.P. Global Epidemiology of Lung Cancer. Ann. Glob. Health 2019, 85. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Austria, S. Statistics Austria, Austrian Register of Cancer. 2017. Available online: http://www.statistik.at/web_de/statistiken/menschen_und_gesellschaft/gesundheit/krebserkrankungen/luftroehre_bronchien_lunge/index.html (accessed on 24 September 2019).
- Gridelli, C.; Rossi, A.; Carbone, D.P.; Guarize, J.; Karachaliou, N.; Mok, T.; Petrella, F.; Spaggiari, L.; Rosell, R. Non-small-cell lung cancer. Nat. Rev. Dis. Primers 2015, 1, 15009. [Google Scholar] [CrossRef]
- Midha, A.; Dearden, S.; McCormack, R. EGFR mutation incidence in non-small-cell lung cancer of adenocarcinoma histology: A systematic review and global map by ethnicity (mutMapII). Am. J. Cancer Res. 2015, 5, 2892–2911. [Google Scholar]
- Yoneda, K.; Imanishi, N.; Ichiki, Y.; Tanaka, F. Treatment of Non-small Cell Lung Cancer with EGFR-mutations. J. UOEH 2019, 41, 153–163. [Google Scholar] [CrossRef]
- Aran, V.; Omerovic, J. Current Approaches in NSCLC Targeting K-RAS and EGFR. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Smolle, E.; Leithner, K.; Olschewski, H. Oncogene addiction and tumor mutational burden in non-small-cell lung cancer: Clinical significance and limitations. Thorac. Cancer 2020, 11, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Jacobi, N.; Seeboeck, R.; Hofmann, E.; Eger, A. ErbB Family Signalling: A Paradigm for Oncogene Addiction and Personalized Oncology. Cancers 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Baxter, E.; Windloch, K.; Gannon, F.; Lee, J.S. Epigenetic regulation in cancer progression. Cell Biosci. 2014, 4, 45. [Google Scholar] [CrossRef]
- Greger, V.; Debus, N.; Lohmann, D.; Hopping, W.; Passarge, E.; Horsthemke, B. Frequency and parental origin of hypermethylated RB1 alleles in retinoblastoma. Hum. Genet. 1994, 94, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Ohtani-Fujita, N.; Fujita, T.; Aoike, A.; Osifchin, N.E.; Robbins, P.D.; Sakai, T. CpG methylation inactivates the promoter activity of the human retinoblastoma tumor-suppressor gene. Oncogene 1993, 8, 1063–1067. [Google Scholar] [PubMed]
- Makos, M.; Nelkin, B.D.; Lerman, M.I.; Latif, F.; Zbar, B.; Baylin, S.B. Distinct hypermethylation patterns occur at altered chromosome loci in human lung and colon cancer. Proc. Natl. Acad. Sci. USA 1992, 89, 1929–1933. [Google Scholar] [CrossRef]
- Eckhardt, F.; Lewin, J.; Cortese, R.; Rakyan, V.K.; Attwood, J.; Burger, M.; Burton, J.; Cox, T.V.; Davies, R.; Down, T.A.; et al. DNA methylation profiling of human chromosomes 6, 20 and 22. Nat. Genet. 2006, 38, 1378–1385. [Google Scholar] [CrossRef]
- Sarter, B.; Long, T.I.; Tsong, W.H.; Koh, W.P.; Yu, M.C.; Laird, P.W. Sex differential in methylation patterns of selected genes in Singapore Chinese. Hum. Genet. 2005, 117, 402–403. [Google Scholar] [CrossRef]
- Sandovici, I.; Kassovska-Bratinova, S.; Loredo-Osti, J.C.; Leppert, M.; Suarez, A.; Stewart, R.; Bautista, F.D.; Schiraldi, M.; Sapienza, C. Interindividual variability and parent of origin DNA methylation differences at specific human Alu elements. Hum. Mol. Genet. 2005, 14, 2135–2143. [Google Scholar] [CrossRef] [PubMed]
- Fuke, C.; Shimabukuro, M.; Petronis, A.; Sugimoto, J.; Oda, T.; Miura, K.; Miyazaki, T.; Ogura, C.; Okazaki, Y.; Jinno, Y. Age related changes in 5-methylcytosine content in human peripheral leukocytes and placentas: An HPLC-based study. Ann. Hum. Genet. 2004, 68, 196–204. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, N.S.; Melton, P.E.; Cadby, G.; Yazar, S.; Franchina, M.; Moses, E.K.; Mackey, D.A.; Hewitt, A.W. Meta-analysis of human methylation data for evidence of sex-specific autosomal patterns. BMC Genom. 2014, 15, 981. [Google Scholar] [CrossRef]
- Zhang, F.F.; Cardarelli, R.; Carroll, J.; Fulda, K.G.; Kaur, M.; Gonzalez, K.; Vishwanatha, J.K.; Santella, R.M.; Morabia, A. Significant differences in global genomic DNA methylation by gender and race/ethnicity in peripheral blood. Epigenetics 2011, 6, 623–629. [Google Scholar] [CrossRef]
- Kautzky, A. Gendermedizin: Prävention, diagnose, therapie. Querelles-Net 2012. [Google Scholar] [CrossRef]
- Baggio, G.; Corsini, A.; Floreani, A.; Giannini, S.; Zagonel, V. Gender medicine: A task for the third millennium. Clin. Chem. Lab. Med. 2013, 51, 713–727. [Google Scholar] [CrossRef] [PubMed]
- Scandlyn, M.J.; Stuart, E.C.; Rosengren, R.J. Sex-specific differences in CYP450 isoforms in humans. Expert Opin. Drug Metab. Toxicol. 2008. [Google Scholar] [CrossRef] [PubMed]
- Mittal, B.; Tulsyan, S.; Kumar, S.; Mittal, R.D.; Agarwal, G. Cytochrome P450 in Cancer Susceptibility and Treatment. Adv. Clin. Chem. 2015, 71, 77–139. [Google Scholar] [CrossRef] [PubMed]
- El-Maarri, O.; Becker, T.; Junen, J.; Manzoor, S.S.; Diaz-Lacava, A.; Schwaab, R.; Wienker, T.; Oldenburg, J. Gender specific differences in levels of DNA methylation at selected loci from human total blood: A tendency toward higher methylation levels in males. Hum. Genet. 2007, 122, 505–514. [Google Scholar] [CrossRef] [PubMed]
- El-Maarri, O.; Walier, M.; Behne, F.; van Uum, J.; Singer, H.; Diaz-Lacava, A.; Nusgen, N.; Niemann, B.; Watzka, M.; Reinsberg, J.; et al. Methylation at global LINE-1 repeats in human blood are affected by gender but not by age or natural hormone cycles. PLoS ONE 2011, 6, e16252. [Google Scholar] [CrossRef]
- Yousefi, P.; Huen, K.; Dave, V.; Barcellos, L.; Eskenazi, B.; Holland, N. Sex differences in DNA methylation assessed by 450 K BeadChip in newborns. BMC Genom. 2015, 16, 911. [Google Scholar] [CrossRef]
- Maschietto, M.; Bastos, L.C.; Tahira, A.C.; Bastos, E.P.; Euclydes, V.L.; Brentani, A.; Fink, G.; de Baumont, A.; Felipe-Silva, A.; Francisco, R.P.; et al. Sex differences in DNA methylation of the cord blood are related to sex-bias psychiatric diseases. Sci. Rep. 2017, 7, 44547. [Google Scholar] [CrossRef]
- Dong, Y.; Huang, Y.; Gutin, B.; Raed, A.; Zhu, H. Associations between Global DNA Methylation and Telomere Length in Healthy Adolescents. Sci. Rep. 2017, 7, 4210. [Google Scholar] [CrossRef]
- Reizel, Y.; Spiro, A.; Sabag, O.; Skversky, Y.; Hecht, M.; Keshet, I.; Berman, B.P.; Cedar, H. Gender-specific postnatal demethylation and establishment of epigenetic memory. Genes Dev. 2015, 29, 923–933. [Google Scholar] [CrossRef]
- Zochbauer-Muller, S.; Fong, K.M.; Virmani, A.K.; Geradts, J.; Gazdar, A.F.; Minna, J.D. Aberrant promoter methylation of multiple genes in non-small cell lung cancers. Cancer Res. 2001, 61, 249–255. [Google Scholar]
- Belinsky, S.A.; Nikula, K.J.; Palmisano, W.A.; Michels, R.; Saccomanno, G.; Gabrielson, E.; Baylin, S.B.; Herman, J.G. Aberrant methylation of p16(INK4a) is an early event in lung cancer and a potential biomarker for early diagnosis. Proc. Natl. Acad. Sci. USA 1998, 95, 11891–11896. [Google Scholar] [CrossRef] [PubMed]
- Jarmalaite, S.; Kannio, A.; Anttila, S.; Lazutka, J.R.; Husgafvel-Pursiainen, K. Aberrant p16 promoter methylation in smokers and former smokers with nonsmall cell lung cancer. Int. J. Cancer 2003, 106, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Romagosa, C.; Simonetti, S.; Lopez-Vicente, L.; Mazo, A.; Lleonart, M.E.; Castellvi, J.; Ramon y Cajal, S. p16(Ink4a) overexpression in cancer: A tumor suppressor gene associated with senescence and high-grade tumors. Oncogene 2011, 30, 2087–2097. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Yuan, X.; Xu, D. Cancer-Specific Telomerase Reverse Transcriptase (TERT) Promoter Mutations: Biological and Clinical Implications. Genes 2016, 7. [Google Scholar] [CrossRef]
- Shin, K.H.; Kang, M.K.; Dicterow, E.; Park, N.H. Hypermethylation of the hTERT promoter inhibits the expression of telomerase activity in normal oral fibroblasts and senescent normal oral keratinocytes. Br. J. Cancer 2003, 89, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Agathanggelou, A.; Cooper, W.N.; Latif, F. Role of the Ras-association domain family 1 tumor suppressor gene in human cancers. Cancer Res. 2005, 65, 3497–3508. [Google Scholar] [CrossRef]
- Dammann, R.; Li, C.; Yoon, J.H.; Chin, P.L.; Bates, S.; Pfeifer, G.P. Epigenetic inactivation of a RAS association domain family protein from the lung tumor suppressor locus 3p21.3. Nat. Genet. 2000, 25, 315–319. [Google Scholar] [CrossRef]
- Wei, H.; Fang, N.; Guo, L.; Wu, Z.; Zhou, Q. Meta-analysis of the Association between RASSF1A Gene Promoter Methylation and Non-small Cell Lung Cancer. Zhongguo Fei Ai Za Zhi 2015, 18, 443–450. [Google Scholar] [CrossRef]
- Hesson, L.B.; Cooper, W.N.; Latif, F. The role of RASSF1A methylation in cancer. Dis. Markers 2007, 23, 73–87. [Google Scholar] [CrossRef]
- Gheldof, A.; Berx, G. Cadherins and epithelial-to-mesenchymal transition. Prog. Mol. Biol. Transl. Sci. 2013, 116, 317–336. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Han, J.Y.; Lin, S.C.; Liu, Z.Y.; Jiang, W.T. Effect of CDH1 gene methylation on transforming growth factor (TGF-beta)-induced epithelial-mesenchymal transition in alveolar epithelial cell line A549. Genet. Mol. Res. 2014, 13, 8568–8576. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Han, Y.; Suarez Saiz, F.; Minden, M.D. A tumor suppressor and oncogene: The WT1 story. Leukemia 2007, 21, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Bruno, P.; Gentile, G.; Mancini, R.; De Vitis, C.; Esposito, M.C.; Scozzi, D.; Mastrangelo, M.; Ricci, A.; Mohsen, I.; Ciliberto, G.; et al. WT1 CpG islands methylation in human lung cancer: A pilot study. Biochem. Biophys. Res. Commun. 2012, 426, 306–309. [Google Scholar] [CrossRef] [PubMed]
- van der Drift, M.A.; Prinsen, C.F.M.; Knuiman, G.J.; Janssen, J.P.; Dekhuijzen, P.N.R.; Thunnissen, F. Diagnosing peripheral lung cancer: The additional value of the Ras-association domain family 1A gene methylation and Kirsten rat sarcoma 2 viral oncogene homolog mutation analyses in washings in nondiagnostic bronchoscopy. Chest 2012, 141, 169–175. [Google Scholar] [CrossRef]
- Halliday, P.R.; Blakely, C.M.; Bivona, T.G. Emerging Targeted Therapies for the Treatment of Non-small Cell Lung Cancer. Curr. Oncol. Rep. 2019, 21, 21. [Google Scholar] [CrossRef]
- Schwartzberg, L.; Kim, E.S.; Liu, D.; Schrag, D. Precision Oncology: Who, How, What, When, and When Not? Am. Soc. Clin. Oncol. Educ. Book 2017, 37, 160–169. [Google Scholar] [CrossRef]
- Burney, I.A.; Lakhtakia, R. Precision Medicine: Where have we reached and where are we headed? Sultan Qaboos Univ. Med. J. 2017, 17, e255–e258. [Google Scholar] [CrossRef]
- Bjaanaes, M.M.; Fleischer, T.; Halvorsen, A.R.; Daunay, A.; Busato, F.; Solberg, S.; Jorgensen, L.; Kure, E.; Edvardsen, H.; Borresen-Dale, A.L.; et al. Genome-wide DNA methylation analyses in lung adenocarcinomas: Association with EGFR, KRAS and TP53 mutation status, gene expression and prognosis. Mol. Oncol. 2016, 10, 330–343. [Google Scholar] [CrossRef]
- Masramon, L. Genetic instability and divergence of clonal populations in colon cancer cells in vitro. J. Cell Sci. 2006, 119, 1477–1482. [Google Scholar] [CrossRef]
- Hernandez-Rosas, F.; Lopez-Rosas, C.A.; Saavedra-Velez, M.V. Disruption of the Molecular Circadian Clock and Cancer: An Epigenetic Link. Biochem. Genet. 2019. [Google Scholar] [CrossRef]
- Karthi, N.; Karthiga, A.; Kalaiyarasu, T.; Stalin, A.; Manju, V.; Singh, S.K.; Cyril, R.; Lee, S.M. Exploration of cell cycle regulation and modulation of the DNA methylation mechanism of pelargonidin: Insights from the molecular modeling approach. Comput. Biol. Chem. 2017, 70, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Şükrüoğlu Erdoğan, Ö.; Kılıç Erciyas, S.; Bilir, A.; Buğra Tunçer, Ş.; Akdeniz Ödemiş, D.; Kurul, S.; Karanlık, H.; Cabıoğlu, N.; Yazıcı, H. Methylation Changes of Primary Tumors, Monolayer, and Spheroid Tissue Culture Environments in Malignant Melanoma and Breast Carcinoma. BioMed. Res. Int. 2019, 2019, 1–9. [Google Scholar] [CrossRef]
- Jacobi, N.; Seeboeck, R.; Hofmann, E.; Schweiger, H.; Smolinska, V.; Mohr, T.; Boyer, A.; Sommergruber, W.; Lechner, P.; Pichler-Huebschmann, C.; et al. Organotypic three-dimensional cancer cell cultures mirror drug responses in vivo: Lessons learned from the inhibition of EGFR signaling. Oncotarget 2017, 8, 107432–107440. [Google Scholar] [CrossRef] [PubMed]
- Brock, M.V.; Hooker, C.M.; Ota-Machida, E.; Han, Y.; Guo, M.; Ames, S.; Glockner, S.; Piantadosi, S.; Gabrielson, E.; Pridham, G.; et al. DNA methylation markers and early recurrence in stage I lung cancer. N. Engl. J. Med. 2008, 358, 1118–1128. [Google Scholar] [CrossRef] [PubMed]
- Pesek, M.; Kopeckova, M.; Benesova, L.; Meszarosova, A.; Mukensnabl, P.; Bruha, F.; Minarik, M. Clinical significance of hypermethylation status in NSCLC: Evaluation of a 30-gene panel in patients with advanced disease. Anticancer Res. 2011, 31, 4647–4652. [Google Scholar]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef]
- Chen, J.; Gao, S.; Wang, C.; Wang, Z.; Zhang, H.; Huang, K.; Zhou, B.; Li, H.; Yu, Z.; Wu, J.; et al. Pathologically decreased expression of miR-193a contributes to metastasis by targeting WT1-E-cadherin axis in non-small cell lung cancers. J. Exp. Clin. Cancer Res. 2016, 35. [Google Scholar] [CrossRef]
- Wong, S.H.M.; Fang, C.M.; Chuah, L.-H.; Leong, C.O.; Ngai, S.C. E-cadherin: Its dysregulation in carcinogenesis and clinical implications. Crit. Rev. Oncol./Hematol. 2018, 121, 11–22. [Google Scholar] [CrossRef]
- Wu, C.; Zhu, W.; Qian, J.; He, S.; Chen, Y.; Shu, Y. WT1 promotes invasion of NSCLC via suppression of CDH1. J. Thorac. Oncol. 2013, 8, 1163–1169. [Google Scholar] [CrossRef]
- Brett, A.; Pandey, S.; Fraizer, G. The Wilms’ tumor gene (WT1) regulates E-cadherin expression and migration of prostate cancer cells. Mol. Cancer 2013, 12, 3. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Protocols for Determination of Limits of Detection and Limits of Quantitation, Approved Guideline; CLSI document EP17; CLSI: Wayne, PA, USA, 2004. [Google Scholar]
- Sarne, V.; Braunmueller, S.; Rakob, L.; Seeboeck, R. The Relevance of Gender in Tumor-Influencing Epigenetic Traits. Epigenomes 2019, 3, 6. [Google Scholar] [CrossRef]
- Wang, W.; Feng, X.; Duan, X.; Tan, S.; Wang, S.; Wang, T.; Feng, F.; Wu, Y.; Wu, Y. Establishment of two data mining models of lung cancer screening based on three gene promoter methylations combined with telomere damage. Int. J. Biol. Markers 2017, 32, e141–e146. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.L.; Hobbing, K.R.; Donninger, H.; Clark, G.J. RASSF1A Deficiency Enhances RAS-Driven Lung Tumorigenesis. Cancer Res. 2018, 78, 2614–2623. [Google Scholar] [CrossRef] [PubMed]
- Nunes, S.P.; Diniz, F.; Moreira-Barbosa, C.; Constancio, V.; Silva, A.V.; Oliveira, J.; Soares, M.; Paulino, S.; Cunha, A.L.; Rodrigues, J.; et al. Subtyping Lung Cancer Using DNA Methylation in Liquid Biopsies. J. Clin. Med. 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zeng, Y.; Huang, J. Signaling pathways and clinical application of RASSF1A and SHOX2 in lung cancer. J. Cancer Res. Clin. Oncol. 2020, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Din Shah, N.U.; Ali, M.N.; Ganai, B.A.; Mudassar, S.; Khan, M.S.; Kour, J.; Waza, A.A.; Rasool, M.T.; Lone, A.M. Association of promoter methylation of RASSF1A and KRAS mutations in non-small cell lung carcinoma in Kashmiri population (India). Heliyon 2020, 6, e03488. [Google Scholar] [CrossRef]
- Deng, Q.; Su, B.; Ji, X.; Fang, Q.; Zhou, S.; Zhou, C. Predictive value of unmethylated RASSF1A on disease progression in non-small cell lung cancer patients receiving pemetrexed-based chemotherapy. Cancer Biomark 2020, 27, 313–323. [Google Scholar] [CrossRef]
- Hu, H.; Zhou, Y.; Zhang, M.; Ding, R. Prognostic value of RASSF1A methylation status in non-small cell lung cancer (NSCLC) patients: A meta-analysis of prospective studies. Biomarkers 2019, 24, 207–216. [Google Scholar] [CrossRef]
- Pastuszak-Lewandoska, D.; Kordiak, J.; Migdalska-Sek, M.; Czarnecka, K.H.; Antczak, A.; Gorski, P.; Nawrot, E.; Kiszalkiewicz, J.M.; Domanska, D.; Brzezianska-Lasota, E. Quantitative analysis of mRNA expression levels and DNA methylation profiles of three neighboring genes: FUS1, NPRL2/G21 and RASSF1A in non-small cell lung cancer patients. Respir Res. 2015, 16, 76. [Google Scholar] [CrossRef]
- Exner, R.; Pulverer, W.; Diem, M.; Spaller, L.; Woltering, L.; Schreiber, M.; Wolf, B.; Sonntagbauer, M.; Schröder, F.; Stift, J.; et al. Potential of DNA methylation in rectal cancer as diagnostic and prognostic biomarkers. Br. J. Cancer 2015, 113, 1035–1045. [Google Scholar] [CrossRef]
- Guo, M.; Alumkal, J.; Drachova, T.; Gao, D.; Marina, S.S.; Jen, J.; Herman, J.G. CHFR methylation strongly correlates with methylation of DNA damage repair and apoptotic pathway genes in non-small cell lung cancer. Discov. Med. 2015, 19, 151–158. [Google Scholar] [PubMed]
- Tuo, L.; Sha, S.; Huayu, Z.; Du, K. P16(INK4a) gene promoter methylation as a biomarker for the diagnosis of non-small cell lung cancer: An updated meta-analysis. Thorac. Cancer 2018, 9, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Lin, H.; Gan, Y.; Cui, C.; Zhang, B.; Gu, L.; Zhou, J.; Zhu, G.; Deng, D. P16 Methylation Leads to Paclitaxel Resistance of Advanced Non-Small Cell Lung Cancer. J. Cancer 2019, 10, 1726–1733. [Google Scholar] [CrossRef]
- Li, P.; Zhang, X.; Gu, L.; Zhou, J.; Deng, D. P16 methylation increases the sensitivity of cancer cells to the CDK4/6 inhibitor palbociclib. PLoS ONE 2019, 14, e0223084. [Google Scholar] [CrossRef]
- Zhong, K.; Chen, W.; Xiao, N.; Zhao, J. The clinicopathological significance and potential drug target of E-cadherin in NSCLC. Tumor Biol. 2015, 36, 6139–6148. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Guo, Q.; Chen, L.; Liu, S. Clinicopathological significance and potential drug targeting of CDH1 in lung cancer: A meta-analysis and literature review. Drug Des. Devel. Ther. 2015, 9, 2171–2178. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, K.; Mishra, T.K.; Saxena, A.; Daga, M.K.; Khurana, N.; Masroor, M.; Jamatia, E. Evaluating NISCH and CDH1 Promoter Hypermethylation in Nonsmokers, Cancer Free Smokers and Lung Cancer Patients: A Case Control Study. Indian J. Clin. Biochem. 2019, 34, 458–464. [Google Scholar] [CrossRef]
- Zhang, X.; Gao, C.; Liu, L.; Zhou, C.; Liu, C.; Li, J.; Zhuang, J.; Sun, C. DNA methylation-based diagnostic and prognostic biomarkers of nonsmoking lung adenocarcinoma patients. J. Cell Biochem. 2019, 120, 13520–13530. [Google Scholar] [CrossRef]
- Liu, Z.L.; Wang, Q.; Huang, L.N. E-cadherin gene methylation in lung cancer. Tumor Biol. 2014, 35, 9027–9033. [Google Scholar] [CrossRef]
- Sun, Z.; Liu, G.; Xu, N. Does hypermethylation of CpG island in the promoter region of the E-cadherin gene increase the risk of lung cancer? A meta-analysis. Thorac. Cancer 2019, 10, 54–59. [Google Scholar] [CrossRef]
- Battram, T.; Richmond, R.C.; Baglietto, L.; Haycock, P.C.; Perduca, V.; Bojesen, S.E.; Gaunt, T.R.; Hemani, G.; Guida, F.; Carreras-Torres, R.; et al. Appraising the causal relevance of DNA methylation for risk of lung cancer. Int. J. Epidemiol. 2019, 48, 1493–1504. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Wang, S.; Xu, C.; Tyler, A.; Li, X.; Andersson, C.; Oji, Y.; Sugiyama, H.; Chen, Y.; Li, A. WT1 enhances proliferation and impedes apoptosis in KRAS mutant NSCLC via targeting cMyc. Cell Physiol. Biochem. 2015, 35, 647–662. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Zhang, Y.; Breitling, L.P.; Brenner, H. Tobacco smoking and methylation of genes related to lung cancer development. Oncotarget 2016, 7, 59017–59028. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.D.; Komosa, M.; Nunes, N.M.; Tabori, U. DNA methylation of the TERT promoter and its impact on human cancer. Curr. Opin. Genet. Dev. 2020, 60, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Chen, X.; Hong, Q.; Deng, Z.; Ma, H.; Xin, Y.; Fang, Y.; Ye, H.; Wang, R.; Zhang, C.; et al. Meta-analyses of gene methylation and smoking behavior in non-small cell lung cancer patients. Sci. Rep. 2015, 5, 8897. [Google Scholar] [CrossRef] [PubMed]
- Han, J.C.; Xu, F.; Chen, N.; Qi, G.B.; Wei, Y.J.; Li, H.B.; Zhang, Y.J.; Li, J.H.; Wang, X.L.; Xu, W.; et al. Promoter methylations of RASSF1A and p16 is associated with clinicopathological features in lung cancers. J. Cancer Res. Ther. 2016, 12, 340–349. [Google Scholar] [CrossRef]
- Nikolaidis, G.; Raji, O.Y.; Markopoulou, S.; Gosney, J.R.; Bryan, J.; Warburton, C.; Walshaw, M.; Sheard, J.; Field, J.K.; Liloglou, T. DNA methylation biomarkers offer improved diagnostic efficiency in lung cancer. Cancer Res. 2012, 72, 5692–5701. [Google Scholar] [CrossRef]



| Mutations | Methylation [%] | |||||||
|---|---|---|---|---|---|---|---|---|
| FFPE Sample Tumorgrade gender | EGFR | KRAS | CDH1 | CDKN2Ap16 | RASSF1A | TERT | WT1 | Combined Methylation Score |
| G0 f | wt | Q61H | 3.5 | 1.7 | 6.9 | 10.5 | 94.4 | 1.4 |
| G0 f | wt | wt | 2.2 | 49.5 | 14.3 | 1.3 | ||
| G0 f | wt | wt | 17.3 | 10.8 | 1.0 | |||
| G0 f | wt | wt | 6.9 | 21.7 | 1.5 | |||
| G0 m | wt | wt | 2.3 | 4.6 | 29.5 | 1.3 | ||
| G0 m | wt | wt | 14.3 | 26.9 | 1.5 | |||
| G0 m | wt | wt | 3.9 | 11.7 | 84.8 | 1.7 | ||
| G1 f | wt | wt | 7.2 | 67.6 | 22.8 | 1.8 | 1.8 | |
| G1 m | wt | G12V | 11.9 | 37.7 | 1.5 | |||
| G1 m | wt | wt | 20.5 | 37.1 | 1.5 | |||
| G2 f | G719C; S768I | wt | 7.3 | 13.8 | 1.0 | |||
| G2 f | E746_A750del (#) | wt | 42.9 | 38.6 | 2.0 | |||
| G2 f | L858R | wt | 2.2 | 2.4 | 1.0 | |||
| G2 f | L861Q | wt | 2.3 | 1.9 | 17.2 | 1.0 | ||
| G2 f | wt | G12C | 6.3 | 4.1 | 1.0 | |||
| G2 f | wt | G12C | 12.8 | 1.0 | ||||
| G2 f | wt | G12C | 5.7 | 23.5 | 1.5 | |||
| G2 f | wt | G12C | 3.2 | 5.0 | 1.7 | 33.2 | 22.5 | 1.4 |
| G2 f | wt | G12D | 3.0 | 1.0 | ||||
| G2 f | wt | G12F | 0.9 | 86.9 | 2.0 | |||
| G2 f | wt | G12V | 3.0 | 6.1 | 1.0 | |||
| G2 f | wt | wt | 3.1 | 5.3 | 1.0 | |||
| G2 f | wt | wt | 1.7 | 14.5 | 62.3 | 1.7 | ||
| G2 f | wt | wt | 46.6 | 40.3 | 2.0 | |||
| G2 f | wt | wt | 1.5 | 17.6 | 1.0 | |||
| G2 f | wt | wt | 4.1 | 25.3 | 1.5 | |||
| G2 f | wt | wt | 3.3 | 5.9 | 90.2 | 1.7 | ||
| G2 f | wt | wt | 2.1 | 1.0 | ||||
| G2 f | wt | wt | 8.5 | 7.6 | 1.0 | |||
| G2 f | wt | 4.5 | 1.0 | |||||
| G2 f | wt | wt | 6.9 | 7.1 | 6.5 | 1.0 | ||
| G2 f | wt | wt | 2.2 | 0.6 | 1.2 | 3.8 | 17.5 | 1.0 |
| G2 f | wt | wt | 2.5 | 1.7 | 6.0 | 21.9 | 24.3 | 1.4 |
| G2 f | wt | wt | 4.2 | 6.3 | 20.7 | 60.6 | 1.5 | |
| G2 m | P733L | wt | 1.3 | 1.0 | ||||
| G2 m | wt | G12C | 2.0 | 8.6 | 6.5 | 7.9 | 35.9 | 1.2 |
| G2 m | wt | G12D | 1.2 | 7.9 | 1.0 | |||
| G2 m | wt | G12D | 19.0 | 6.6 | 5.0 | 4.2 | 13.9 | 1.0 |
| G2 m | wt | G12D | 8.8 | 3.3 | 6.2 | 16.0 | 44.1 | 1.2 |
| G2 m | wt | G12V | 2.2 | 3.3 | 5.5 | 1.0 | ||
| G2 m | wt | G12V | 38.6 | 45.3 | 40.4 | 2.0 | ||
| G2 m | wt | G12V | 3.3 | 22.1 | 2.8 | 27.8 | 1.5 | |
| G2 m | wt | wt | 61.2 | 56.9 | 3.0 | |||
| G2 m | wt | wt | 2.5 | 10.9 | 26.1 | 1.3 | ||
| G2 m | wt | wt | 1.0 | 1.0 | ||||
| G2 m | wt | wt | 2.5 | 2.7 | 16.8 | 1.0 | ||
| G2 m | wt | wt | 0.0 | |||||
| G2 m | wt | wt | 2.3 | 3.1 | 1.0 | |||
| G2 m | wt | wt | 29.0 | 29.3 | 2.0 | |||
| G2 m | wt | wt | 3.2 | 1.9 | 10.8 | 13.4 | 1.0 | |
| G2 m | wt | wt | 0.0 | |||||
| G2 m | wt | wt | 5.0 | 1.2 | 1.0 | |||
| G2 m | wt | wt | 0.9 | 34.2 | 1.5 | |||
| G2 m | wt | wt | 3.4 | 1.8 | 36.0 | 1.3 | ||
| G3 f | E709K; L858R | wt | 3.5 | 51.5 | 2.0 | |||
| G3 f | E746_# | wt | 4.5 | 3.8 | 5.4 | 6.6 | 21.9 | 1.2 |
| G3 f | T790M; T751ind | wt | 3.0 | 2.9 | 1.7 | 38.8 | 1.3 | |
| G3 f | wt | G12A | 8.1 | 15.0 | 9.8 | 1.0 | ||
| G3 f | wt | G12C | 0.9 | 28.6 | 59.5 | 2.0 | ||
| G3 f | wt | G12C | 2.4 | 44.5 | 1.5 | |||
| G3 f | wt | G12C | 2.7 | 3.3 | 1.7 | 7.6 | 1.0 | |
| G3 f | wt | G12C | 3.0 | 17.4 | 0.6 | 22.6 | 1.3 | |
| G3 f | wt | G12V | 1.8 | 46.8 | 1.5 | |||
| G3 f | wt | wt | 10.1 | 2.7 | 40.1 | 1.0 | ||
| G3 f | wt | wt | 13.3 | 1.0 | ||||
| G3 f | wt | wt | 0.9 | 2.4 | 1.0 | |||
| G3 f | wt | wt | 2.2 | 18.7 | 1.0 | |||
| G3 f | wt | wt | 1.9 | 22.0 | 1.5 | |||
| G3 f | wt | wt | 2.2 | 12.0 | 1.0 | |||
| G3 f | wt | wt | 3.5 | 8.8 | 1.0 | |||
| G3 f | wt | wt | 1.1 | 1.0 | ||||
| G3 f | wt | wt | 16.4 | 1.0 | ||||
| G3 f | wt | wt | 1.8 | 8.7 | 10.3 | 1.0 | ||
| G3 f | wt | wt | 2.5 | 2.8 | 1.0 | |||
| G3 f | wt | wt | 1.8 | 4.1 | 10.4 | 1.0 | ||
| G3 f | wt | wt | 5.6 | 20.6 | 1.0 | |||
| G3 f | wt | wt | 18.4 | 8.4 | 27.4 | 1.3 | ||
| G3 f | wt | wt | 15.3 | 30.2 | 1.5 | |||
| G3 f | wt | wt | 7.2 | 37.6 | 31.8 | 71.6 | 37.1 | 2.0 |
| G3 f | wt | wt | 2.3 | 5.9 | 3.7 | 9.8 | 1.0 | |
| G3 f | wt | wt | 2.2 | 2.0 | 4.5 | 1.0 | ||
| G3 f | wt | wt | 6.9 | 36.9 | 1.5 | |||
| G3 f | wt | wt | 4.5 | 24.4 | 1.5 | |||
| G3 f | wt | wt | 2.3 | 3.6 | 16.8 | 1.0 | ||
| G3 f | wt | wt | 19.4 | 6.9 | 21.7 | 92.5 | 1.8 | |
| G3 m | E746K | wt | 3.7 | 11.1 | 2.4 | 27.1 | 1.3 | |
| G3 m | L858R | wt | 1.1 | 9.9 | 38.8 | 1.3 | ||
| G3 m | wt | G12A | 2.6 | 25.8 | 1.5 | |||
| G3 m | wt | G12A | 2.0 | 1.9 | 42.0 | 1.3 | ||
| G3 m | wt | G12A | 9.6 | 27.1 | 1.5 | |||
| G3 m | wt | G12C | 24.7 | 3.2 | 35.2 | 1.7 | ||
| G3 m | wt | G12C | 1.8 | 2.5 | 1.0 | |||
| G3 m | wt | G12C | 13.0 | 56.2 | 2.0 | |||
| G3 m | wt | G12C | 5.0 | 5.3 | 35.4 | 44.4 | 1.5 | |
| G3 m | wt | G12C | 2.2 | 1.7 | 2.8 | 5.4 | 11.5 | 1.0 |
| G3 m | wt | G12C | 6.5 | 48.8 | 21.3 | 1.7 | ||
| G3 m | wt | G12D | 1.3 | 54.3 | 2.0 | |||
| G3 m | wt | G12D | 2.9 | 34.3 | 1.5 | |||
| G3 m | wt | G12V | 2.7 | 5.9 | 28.9 | 93.4 | 1.8 | |
| G3 m | wt | G12V | 2.2 | 2.4 | 30.1 | 19.4 | 1.3 | |
| G3 m | wt | G13C | 2.7 | 6.1 | 37.2 | 1.3 | ||
| G3 m | wt | G13C | 4.8 | 25.5 | 27.5 | 63.4 | 2.0 | |
| G3 m | wt | G13C | 4.2 | 4.6 | 39.1 | 86.8 | 1.8 | |
| G3 m | wt | G13D | 7.7 | 32.2 | 4.3 | 25.7 | 1.5 | |
| G3 m | wt | wt | 12.8 | 2.0 | 19.2 | 1.0 | ||
| G3 m | wt | wt | 11.3 | 24.9 | 37.6 | 1.7 | ||
| G3 m | wt | wt | 0.0 | |||||
| G3 m | wt | wt | 4.0 | 23.2 | 1.5 | |||
| G3 m | wt | wt | 3.3 | 1.4 | 3.6 | 1.0 | ||
| G3 m | wt | wt | 33.2 | 3.0 | 16.0 | 1.3 | ||
| G3 m | wt | wt | 1.5 | 49.2 | 1.5 | |||
| G3 m | wt | wt | 0.0 | |||||
| G3 m | wt | wt | 5.1 | 36.9 | 1.5 | |||
| G3 m | wt | wt | 8.6 | 34.4 | 87.0 | 2.0 | ||
| G3 m | wt | wt | 4.2 | 27.2 | 19.9 | 1.7 | ||
| G3 m | wt | wt | 5.8 | 30.4 | 1.5 | |||
| G3 m | wt | wt | 3.6 | 8.3 | 1.0 | |||
| G3 m | wt | wt | 4.2 | 0.9 | 1.0 | |||
| G3 m | wt | wt | 3.5 | 2.4 | 76.6 | 1.7 | ||
| G3 m | wt | wt | 1.6 | 1.0 | ||||
| G3 m | wt | wt | 4.4 | 22.0 | 1.5 | |||
| G3 m | wt | wt | 28.6 | 15.6 | 1.5 | |||
| G3 m | wt | wt | 8.0 | 1.0 | ||||
| G3 m | wt | wt | 10.2 | 16.4 | 20.3 | 8.4 | 7.2 | 1.0 |
| G3 m | wt | wt | 3.0 | 3.1 | 24.8 | 20.1 | 1.3 | |
| G3 m | wt | wt | 2.7 | 17.1 | 48.9 | 24.3 | 1.5 | |
| G3 m | wt | wt | 13.5 | 8.9 | 1.0 | |||
| G3 m | wt | wt | 2.3 | 3.6 | 1.0 | 5.5 | 7.2 | 1.0 |
| G3 m | wt | wt | 17.2 | 3.2 | 2.9 | 13.2 | 1.0 | |
| G3 m | wt | wt | 2.0 | 14.8 | 6.5 | 38.3 | 1.3 | |
| G3 m | wt | wt | 1.1 | 1.0 | ||||
| G3 m | wt | wt | 2.2 | 5.6 | 26.3 | 1.3 | ||
| G3 m | wt | wt | 1.1 | 12.1 | 1.0 | |||
| G3 m | wt | wt | 2.0 | 5.5 | 39.9 | 7.2 | 1.3 | |
| G3 m | wt | wt | 2.0 | 3.4 | 63.2 | 1.7 | ||
| G3 m | wt | wt | 15.3 | 25.9 | 1.5 | |||
| G3 m | wt | wt | 15.8 | 26.9 | 1.5 | |||
| G3 m | wt | wt | 2.0 | 35.3 | 1.5 | |||
| G3 m | wt | wt | 3.8 | 2.1 | 3.7 | 36.0 | 1.3 | |
| G3 m | wt | wt | 4.1 | 24.4 | 1.5 | |||
| G3 m | wt | wt | 2.2 | 1.6 | 45.8 | 1.3 | ||
| G3 m | wt | wt | 3.2 | 1.3 | 6.8 | 38.0 | 1.3 | |
| G3 m | wt | wt | 3.2 | 8.0 | 22.9 | 1.3 | ||
| G3 m | wt | wt | 2.0 | 8.0 | 21.7 | 1.3 | ||
| G3 m | wt | wt | 3.2 | 10.1 | 21.8 | 1.3 | ||
| G3 m | wt | wt | 3.4 | 46.0 | 48.9 | 1.7 | ||
| G3 m | wt | wt | 2.7 | 5.9 | 6.8 | 60.1 | 57.4 | 1.8 |
| G3 m | wt | wt | 2.3 | 3.7 | 18.5 | 72.2 | 1.8 | |
| G4 m | wt | wt | 3.0 | 18.2 | 30.3 | 1.3 | ||
| G4 m | wt | wt | 3.3 | 25.0 | 3.7 | 47.6 | 1.5 | |
| Mutations | Methylation [%] | Combined Methylation Score a,b,c,d | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cell Line | Gender | Source Site | EGFR | KRAS | CDH1 | CDKN2Ap16 | RASSF1A | TERT | WT1 | ||
| H1975 | f | primary | MUT (L858R) | WT | 15.8 | 1.4 | 24.7 | 43.5 | 88.0 | 9 | |
| H1993 | f | metastasis | WT amplified | WT | 5.2 | 1.6 | 80.3 | 21.3 | 22.0 | 9 | |
| HCC827 | f | primary | MUT (Glu746_Ala750del) | WT | 10.2 | 1.1 | 91.4 | 37.9 | 79.9 | 10 | |
| H2347 | f | primary | WT | MUT (L19F) | 3.8 | 91.6 | 2.4 | 43.8 | 91.3 | 10 | |
| IMR90 | f | benign ctrl | WT | WT | 5.7 | 1.2 | 3.4 | 9.1 | 65.9 | 7 | |
| HCC2935 | m | primary | MUT (E746_T751 del, S752I) | WT | 3.5 | 1.9 | 24.2 | 48.9 | 83.9 | 9 | |
| HCC4006 | m | metastasis | MUT (L747_E749 del, A750P) | WT | 12.7 | 1.0 | 5.9 | 8.3 | 25.1 | 6 | |
| Calu1 | m | metastasis | WT | MUT (G12C) | 7.0 | 97.2 | 9.4 | 56.7 | 80.1 | 10 | |
| Calu3 | m | metastasis | WT | MUT (G13D) | 4.5 | 1.6 | 3.6 | 71.3 | 73.9 | 9 | |
| H441 | m | primary | WT | MUT (G12V) | 6.2 | 95.3 | 93.6 | 21.9 | 91.1 | 12 | |
| H647 | m | metastasis | WT | MUT (G12L) | 3.7 | 1.6 | 1.4 | 12.0 | 37.9 | 6 | |
| H1437 | m | metastasis | WT | WT | 7.0 | 2.1 | 2.2 | 11.7 | 45.1 | 6 | |
| H2087 | m | metastasis | WT | WT | 3.8 | 1.1 | 57.3 | 84.3 | 52.9 | 9 | |
| MRC-5 | m | benign ctrl | WT | WT | 16.8 | 3.4 | 6.5 | 18.3 | 4.9 | 5 | |
| Cell Line | Gender | 2D CDH1 | 3D CDH1 | 2D CDKN2Ap16 | 3D CDKN2Ap16 | 2D RASSF1A | 3D RASSF1A | 2D TERT | 3D TERT | 2D WT1 | 3D WT1 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| H1975 | f | 15.8 | 4.8 | 1.4 | 2.6 | 24.7 | 28.7 | 43.5 | 39.2 | 88.0 | 79.3 |
| HCC827 | f | 10.2 | 6.2 | 1.1 | 11.2 | 91.4 | 88.1 | 37.9 | 33.9 | 79.9 | 94.1 |
| HCC2935 | m | 3.5 | 2.8 | 1.9 | 8.1 | 24.2 | 76.4 | 48.9 | 59.5 | 83.9 | 71.2 |
| HCC4006 | m | 12.7 | 7.0 | 1.0 | 5.5 | 5.9 | 30.4 | 8.3 | 47.8 | 25.1 | 89.6 |
| Calu1 | m | 7.0 | 3.8 | 97.2 | 98.4 | 9.4 | 33.7 | 56.7 | 43.5 | 80.1 | 84.3 |
| H1437 | m | 7.0 | 3.7 | 2.1 | 16.9 | 2.1 | 3.9 | 2.1 | 10.7 | 2.1 | 39.1 |
| Cell Line | Mutational Status | Methylation Score | EC50 Decitabine [µM] | EC50 Zebularine [µM] | Viability Reduction Decitabine | Viability Reduction Zebularine | EC50 Gefitinib | EC50 Erlotinib |
|---|---|---|---|---|---|---|---|---|
| H1975 | EGFR mut | 9 | 24.42 | 97.68 | 60% | 42% | >10 µM | >10 µM |
| H1993 | EGFR amp | 9 | 83.5 | 493.8 | 36% | 40% | ||
| HCC827 | EGFR mut | 10 | 63.72 | 267.9 | 37% | 62% | 2.39 nM | 1.29 nM |
| H2347 | KRAS mut | 10 | 46.85 | 378.1 | 34% | 21% | ||
| IMR90 | wt | 7 | 146.8 | 230.3 | 58% | 54% | ||
| H4006 | EGFR mut | 6 | 33 | 117.9 | 36% | 77% | 276 nM | 259 nM |
| H647 | KRAS mut | 6 | 63.43 | 184.3 | 55% | 33% | ||
| H1437 | wt | 6 | 132.6 | 275.6 | 41% | 21% | >10 µM | >10 µM |
| H2087 | wt | 9 | 187 | 319.9 | 32% | 39% | ||
| average female LC a | 54.6 | 309.4 | 42% | 41% | ||||
| average male LC b | 104.0 | 224.4 | 41% | 43% | ||||
| average mutated LC c | 46.3 | 209.2 | 44% | 47% | ||||
| average wildtype LC d | 134.4 | 363.1 | 36% | 33% | ||||
| average < 8 methylation LC e | 76.3 | 192.6 | 44% | 44% | ||||
| average > 8 methylation LC f | 81.1 | 311.5 | 40% | 41% | ||||
| Gene | LoB | LoQ | LoD |
|---|---|---|---|
| CDH1 | 0.55 RFU | 2.89 RFU | 69.0 pg/µL |
| CDKN2Ap16 | 0.50 RFU | 2.78 RFU | 11 pg/µL |
| RASSF1A | 0.81 RFU | 3.38 RFU | 54.75 pg/µL |
| TERT | 1.00 RFU | 3.96 RFU | 0.94 pg/µL |
| WT1 | 0.56 RFU | 2.57 RFU | 1.45 pg/µL |
| Gene | Forward Primer | Reverse Primer | Sequencing Primer | Sequence to Be Analyzed | Amplicon Length |
|---|---|---|---|---|---|
| CDH1 | B_GATTTTAGTAATTTTAGGTTAGAGGGTTAT | ACTAACTTCCCCAAACTCACAAATACTT | TCCCCAAACTCACAAATACTTTAC | AATTCCTACTCCACTAAAAAAAAATACRTTT | 236 bp |
| CDKN2Ap16 | AGAGGATTTGAGGGATAGGG | B_TACCTACTCTCCCCCTCT | GGTTGGTTGGTTATTAGA | GGGTGGGGYGGATYGYGTGYGTTYGGYGGTTGYGGAGAGG | 135 bp |
| RASSF1A | AGGGAAGGAAGGGTAAGG | B_ACTCCCCCAACTCAATAAACTCAAACTC | GGGGTTAGTTTTGTGG | TTTYGTTYGGTTYGYGTTTGTTAGYGTTTAAAGTTAG | 265 bp |
| TERT | GGTTAGGTAGGGTTTTTAGTGGA | B_ATACCCCAATCCCCAATCCCTC | GGTAGGGTTTTTAGTGGAT | TYGYGGGTATAGAYGTTTAGGATYGYGTTTTTTAYGTGG | 87 bp |
| WT1 | TGGGGTAAGGAGTTTAAGGT | B_AACTCCCTACTACTCTAACTACTATA | GAGTAGGGAAGGTAGTTTAG | GYGTTYGGGTTTYGTYGTTTTTTYGTYGYGATT | 285 bp |
| Gene | Annealing Temperature | PCR Cycles | MasterMix | Additives |
|---|---|---|---|---|
| CDH1 | 59 °C | 35 | HotStarTaq Plus MM * | 1 µL CoralLoad * |
| CDKN2Ap16 | 59 °C | 35 | HotStarTaq Plus MM * | 1 µL CoralLoad * |
| RASSF1A | 57 °C | 37 | HotStarTaq Plus MM * | 1 µL CoralLoad * |
| TERT | 57 °C | 37 | HotStarTaq Plus MM * | 1 µL CoralLoad * |
| WT1 | 57 °C | 37 | HotStarTaq Plus MM * | 1 µL CoralLoad * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarne, V.; Huter, S.; Braunmueller, S.; Rakob, L.; Jacobi, N.; Kitzwögerer, M.; Wiesner, C.; Obrist, P.; Seeboeck, R. Promoter Methylation of Selected Genes in Non-Small-Cell Lung Cancer Patients and Cell Lines. Int. J. Mol. Sci. 2020, 21, 4595. https://doi.org/10.3390/ijms21134595
Sarne V, Huter S, Braunmueller S, Rakob L, Jacobi N, Kitzwögerer M, Wiesner C, Obrist P, Seeboeck R. Promoter Methylation of Selected Genes in Non-Small-Cell Lung Cancer Patients and Cell Lines. International Journal of Molecular Sciences. 2020; 21(13):4595. https://doi.org/10.3390/ijms21134595
Chicago/Turabian StyleSarne, Victoria, Samuel Huter, Sandrina Braunmueller, Lisa Rakob, Nico Jacobi, Melitta Kitzwögerer, Christoph Wiesner, Peter Obrist, and Rita Seeboeck. 2020. "Promoter Methylation of Selected Genes in Non-Small-Cell Lung Cancer Patients and Cell Lines" International Journal of Molecular Sciences 21, no. 13: 4595. https://doi.org/10.3390/ijms21134595
APA StyleSarne, V., Huter, S., Braunmueller, S., Rakob, L., Jacobi, N., Kitzwögerer, M., Wiesner, C., Obrist, P., & Seeboeck, R. (2020). Promoter Methylation of Selected Genes in Non-Small-Cell Lung Cancer Patients and Cell Lines. International Journal of Molecular Sciences, 21(13), 4595. https://doi.org/10.3390/ijms21134595






