Galectin-3 in Inflammasome Activation and Primary Biliary Cholangitis Development
Abstract
:1. Primary Biliary Cholangitis
2. Genetic and Environmental Factors in PBC
3. Immune Dysregulation in PBC
4. NLRP3 Inflammasome
5. Galectin-3
5.1. Functions of Gal-3
5.2. The Role and Importance of Galectin-3 in the Regulation of the Immune Response
5.3. Galectin-3 and Liver Diseases
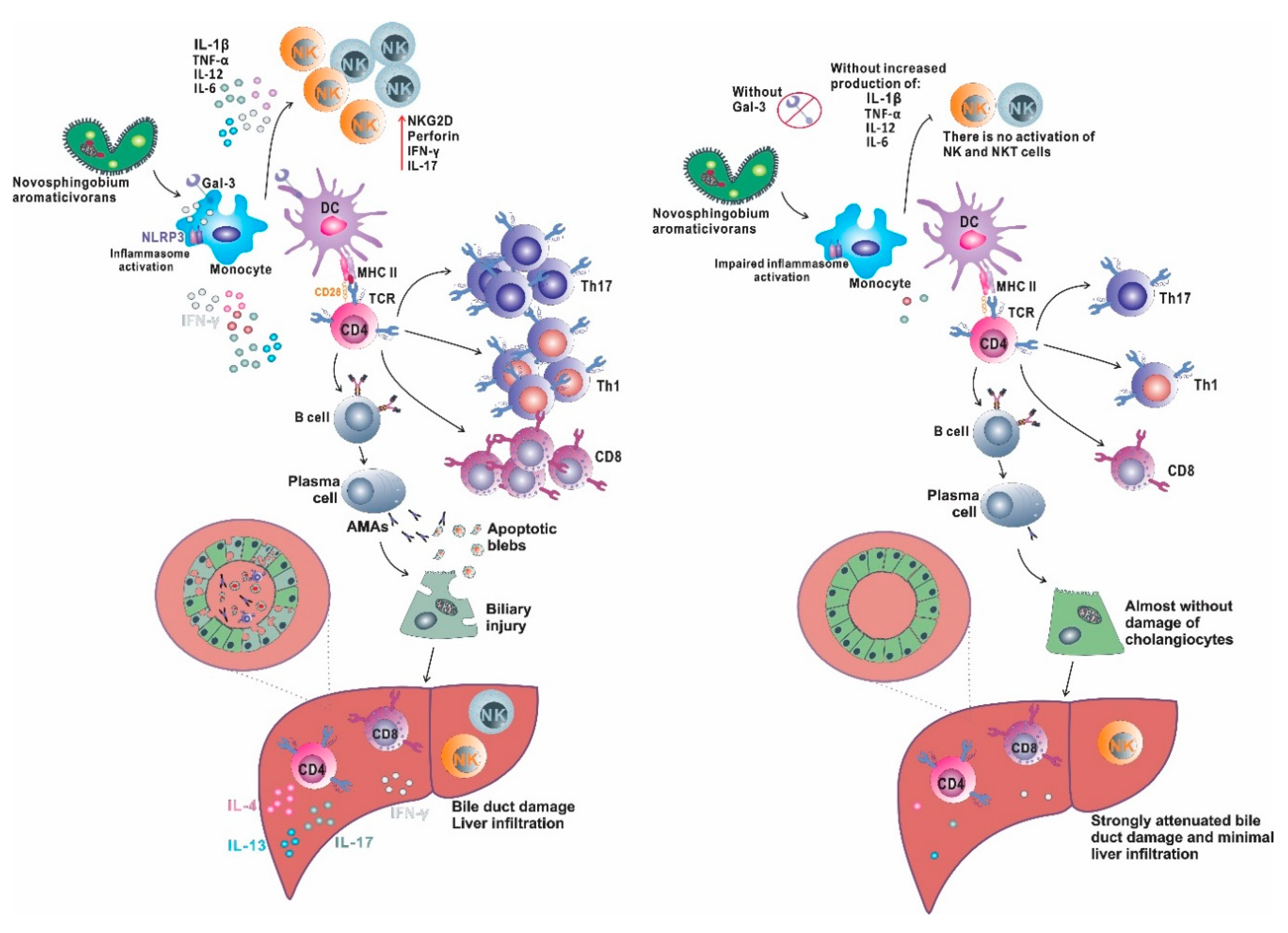
5.4. Galectin-3, Inflammasome and PBC
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 2OADC-E2 | 2-oxo-acid E2 dehydrogenase |
| AMA | anti-mitochondrial antibodies |
| ASC | apoptosis-associated speck-like protein |
| BCOADC-E2 | branched-chain 2-oxo-acid E2 dehydrogenase |
| CRD | carbohydrate recognition domain |
| DAMPs | damage-associated molecular patterns |
| Gal-3 | galectin-3 |
| MAIT cells | mucosal associated invariant T cells |
| NF-κB | nuclear factor kappa B |
| OGDC-E2 | 2-oxo-glutarate E2 dehydrogenase |
| PBC | primary biliary cholangitis |
| PDC-E2 | pyruvate E2 dehydrogenase |
| PAMPs | pathogen-associated molecular patterns of microorganisms |
| TLRs | toll like receptors |
| TNF | tumor necrosis factor |
References
- Hirschfield, G.M.; Gershwin, M.E. The immunobiology and pathophysiology of primary biliary cirrhosis. Annu. Rev. Pathol. 2014, 8, 303–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Tang, R.; Leung, P.-S.C.; Gershwin, M.E.; Ma, X. Bile acids and intestinal microbiota in autoimmune cholestatic liver diseases. Autoimmun. Rev. 2017, 16, 885–896. [Google Scholar] [CrossRef]
- Carey, E.J.; Ali, A.H.; Lindor, K.D. Primary biliary cirrhosis. Lancet 2015, 386, 1565–1575. [Google Scholar] [CrossRef]
- Shimoda, S.; Tanaka, A. It is time to change primary biliary cirrhosis (PBC): New nomenclature from “cirrhosis” to “cholangitis”, and upcoming treatment based on unveiling pathology. Hepatol. Res. 2016, 46, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Webb, G.J.; Hirschfield, G.M. Primary biliary cholangitis in 2016: High-definition PBC: Biology, models and therapeutic advances. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 76–78. [Google Scholar] [CrossRef] [PubMed]
- Gulamhusein, A.F.; Hirschfield, G.M. Primary biliary cholangitis: Pathogenesis and therapeutic opportunities. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 93–110. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.E.; Metcalf, J.V.; Collier, J.D.; Bassendine, M.F.; James, O.F. Hepatocellular carcinoma in primary biliary cirrhosis and its impact on outcomes. Hepatology 1997, 26, 1138–1142. [Google Scholar] [CrossRef]
- Lööf, L.; Adami, H.O.; Sparén, P.; Danielsson, A.; Eriksson, L.S.; Hultcrantz, R.; Lindgren, S.; Olsson, R.; Prytz, H.; Ryden, B.O. Cancer risk in primary biliary cirrhosis: A population-based study from Sweden. Hepatology 1994, 20, 101–104. [Google Scholar] [CrossRef]
- Ghidini, M.; Cascione, L.; Carotenuto, P.; Lampis, A.; Trevisan, F.; Previdi, M.C.; Hahne, J.C.; Said-Huntingford, I.; Raj, M.; Zerbi, A.; et al. Characterisation of the immune-related transcriptome in resected biliary tract cancers. Eur. J. Cancer 2017, 86, 158–165. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, M.; Furuta, K.; Kitamura, H.; Oguchi, K.; Arai, M.; Koike, S.; Nakazawa, K. A case of primary biliary cirrhosis that complicated with combined hepatocellular and cholangiocellular carcinoma. Clin. J. Gastroenterol. 2011, 4, 236–241. [Google Scholar] [CrossRef]
- Ide, R.; Oshita, A.; Nishisaka, T.; Nakahara, H.; Aimitsu, S.; Itamoto, T. Primary biliary cholangitis metachronously complicated with combined hepatocellular carcinoma-cholangiocellular carcinoma and hepatocellular carcinoma. World J. Hepatol. 2017, 9, 1378–1384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gershwin, M.E.; Leung, P.S.; Li, H.; Seldin, M.F. Primary biliary cirrhosis and autoimmunity: Evaluating the genetic risk. Isr. Med. Assoc. J. 2020, 2, 7–10. [Google Scholar]
- Webb, G.J.; Siminovitch, K.A.; Hirschfield, G.M. The immunogenetics of primary biliary cirrhosis: A comprehensive review. J. Autoimmun. 2015, 64, 42–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gulamhusein, A.F.; Lazaridis, K.N. Primary biliary cholangitis, DNA, and beyond: The Relative contribution of genes. Hepatology 2018, 68, 19–21. [Google Scholar] [CrossRef] [Green Version]
- Mells, G.F.; Floyd, J.A.; Morley, K.I.; Cordell, H.J.; Franklin, C.S.; Shin, S.Y.; Heneghan, M.A.; Neuberger, J.M.; Donaldson, P.T.; Day, D.B.; et al. Genome-wide association study identifies 12 new susceptibility loci for primary biliary cirrhosis. Nat. Gen. 2011, 43, 329–332. [Google Scholar] [CrossRef]
- Gulamhusein, A.F.; Juran, B.D.; Lazaridis, K.N. Genome-Wide Association Studies in Primary Biliary Cirrhosis. Semin. Liver Dis. 2015, 35, 392–401. [Google Scholar] [CrossRef] [Green Version]
- Cordell, H.J.; Han, Y.; Mells, G.F.; Li, Y.; Hirschfield, G.M.; Greene, C.S.; Xie, G.; Juran, B.D.; Zhu, D.; Qian, D.C.; et al. International genome-wide meta-analysis identifies new primary biliary cirrhosis risk loci and targetable pathogenic pathways. Nat. Commun. 2015, 6, 8019. [Google Scholar] [CrossRef] [Green Version]
- Hirschfield, G.M.; Liu, X.; Xu, C.; Lu, Y.; Xie, G.; Lu, Y.; Gu, X.; Walker, E.J.; Jing, K.; Juran, B.D.; et al. Primary biliary cirrhosis associated with HLA, IL12A, and IL12RB2 variants. N. Engl. J. Med. 2009, 360, 2544–2555. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Wang, F.S.; Chang, C.; Gershwin, M.E. Breach of tolerance: Primary biliary cirrhosis. Semin. Liver Dis. 2014, 34, 297–317. [Google Scholar] [CrossRef] [Green Version]
- Oo, Y.H.; Banz, V.; Kavanagh, D.; Liaskou, E.; Withers, D.R.; Humphreys, E.; Reynolds, G.M.; Lee-Turner, L.; Kalia, N.; Hubscher, S.G.; et al. CXCR3-dependent recruitment and CCR6-mediated positioning of Th-17 cells in the inflamed liver. J. Hepatol. 2012, 57, 1044–1051. [Google Scholar] [CrossRef] [Green Version]
- Williams, I.R. CCR6 and CCL20: Partners in intestinal immunity and lymphorganogenesis. Ann. N. Y. Acad. Sci. 2012, 1072, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.L.; Ip, P.P.; Liao, F. CCR6 Deficiency Impairs IgA Production and Dysregulates Antimicrobial Peptide Production, Altering the Intestinal Flora. Front. Immun. 2017, 8, 805. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Leung, P.; Gershwin, M.E. Pathogen infections and primary biliary cholangitis. Clin. Exp. Immunol. 2019, 195, 25–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, H.D.; Zhao, Z.B.; Ma, W.T.; Liu, Q.Z.; Gao, C.Y.; Li, L.; Wang, J.; Tsuneyama, K.; Liu, B.; Zhang, W.; et al. Gut microbiota translocation promotes autoimmune cholangitis. J. Autoimm. 2018, 95, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Leung, P.S.; Gershwin, M.E. Environmental basis of primary biliary cholangitis. Exp. Biol. Med. 2018, 243, 184–189. [Google Scholar] [CrossRef]
- Wakabayashi, K.; Lian, Z.X.; Leung, P.S.; Moritoki, Y.; Tsuneyama, K.; Kurth, M.J.; Lam, K.S.; Yoshida, K.; Yang, G.X.; Hibi, T.; et al. Loss of tolerance in C57BL/6 mice to the autoantigen E2 subunit of pyruvate dehydrogenase by a xenobiotic with ensuing biliary ductular disease. Hepatology 2008, 48, 531–540. [Google Scholar] [CrossRef] [Green Version]
- Sebode, M.; Weiler-Normann, C.; Liwinski, T.; Schramm, C. Autoantibodies in Autoimmune Liver Disease-Clinical and Diagnostic Relevance. Front. Immun. 2018, 9, 609. [Google Scholar] [CrossRef] [Green Version]
- Lleo, A.; Selmi, C.; Invernizzi, P.; Podda, M.; Coppel, R.L.; Mackay, I.R.; Gores, G.J.; Ansari, A.A.; Van de Water, J.; Gershwin, M.E. Apotopes and the biliary specificity of primary biliary cirrhosis. Hepatology 2009, 49, 871–879. [Google Scholar] [CrossRef] [Green Version]
- Yeaman, S.J.; Fussey, S.P.; Danner, D.J.; James, O.F.; Mutimer, D.J.; Bassendine, M.F. Primary biliary cirrhosis: Identification of two major M2 mitochondrial autoantigens. Lancet 1988, 1, 1067–1070. [Google Scholar] [CrossRef]
- Lleo, A.; Shimoda, S.; Ishibashi, H.; Gershwin, M.E. Primary biliary cirrhosis and autoimmune hepatitis: Apotopes and epitopes. J. Gastroenterol. 2011, 46 (Suppl. 1), 29–38. [Google Scholar] [CrossRef]
- Lleo, A.; Bowlus, C.L.; Yang, G.X.; Invernizzi, P.; Podda, M.; Van de Water, J.; Ansari, A.A.; Coppel, R.L.; Worman, H.J.; Gores, G.J.; et al. Biliary apotopes and anti-mitochondrial antibodies activate innate immune responses in primary biliary cirrhosis. Hepatology 2010, 52, 987–998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kourtzelis, I.; Hajishengallis, G.; Chavakis, T. Phagocytosis of Apoptotic Cells in Resolution of Inflammation. Front. Immun. 2020, 11, 553. [Google Scholar] [CrossRef] [PubMed]
- Kita, H.; Lian, Z.X.; Van de Water, J.; He, X.S.; Matsumura, S.; Kaplan, M.; Luketic, V.; Coppel, R.L.; Ansari, A.A.; Gershwin, M.E. Identification of HLA-A2-restricted CD8(+) cytotoxic T cell responses in primary biliary cirrhosis: T cell activation is augmented by immune complexes cross-presented by dendritic cells. J. Exp. Med. 2020, 195, 113–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shimoda, S.; Miyakawa, H.; Nakamura, M.; Ishibashi, H.; Kikuchi, K.; Kita, H.; Niiro, H.; Arinobu, Y.; Ono, N.; Mackay, I.R.; et al. CD4 T-cell autoreactivity to the mitochondrial autoantigen PDC-E2 in AMA-negative primary biliary cirrhosis. J. Autoimm. 2008, 31, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.D.; Ma, W.T.; Liu, Q.Z.; Zhao, Z.B.; Liu, M.Z.; Tsuneyama, K.; Gao, J.M.; Ridgway, W.M.; Ansari, A.A.; Gershwin, M.E.; et al. Chemokine receptor CXCR3 deficiency exacerbates murine autoimmune cholangitis by promoting pathogenic CD8+ T cell activation. J. Autoimm. 2017, 78, 19–28. [Google Scholar] [CrossRef] [Green Version]
- Katsumi, T.; Tomita, K.; Leung, P.S.; Yang, G.X.; Gershwin, M.E.; Ueno, Y. Animal models of primary biliary cirrhosis. Clin. Rev. Allergy Immunol. 2015, 48, 142–153. [Google Scholar] [CrossRef]
- Lee, G.R. The Balance of Th17 versus Treg Cells in Autoimmunity. Int. J. Mol. Sci. 2018, 19, 730. [Google Scholar] [CrossRef] [Green Version]
- Ueno, H. T follicular helper cells in human autoimmunity. Curr. Opin. Immunol. 2016, 43, 24–31. [Google Scholar] [CrossRef]
- Rong, G.; Zhou, Y.; Xiong, Y.; Zhou, L.; Geng, H.; Jiang, T.; Zhu, Y.; Lu, H.; Zhang, S.; Wang, P.; et al. Imbalance between T helper type 17 and T regulatory cells in patients with primary biliary cirrhosis: The serum cytokine profile and peripheral cell population. Clin. Exp. Immunol. 2009, 156, 217–225. [Google Scholar] [CrossRef]
- Kamali, A.N.; Noorbakhsh, S.M.; Hamedifar, H.; Jadidi-Niaragh, F.; Yazdani, R.; Bautista, J.M.; Azizi, G. A role for Th1-like Th17 cells in the pathogenesis of inflammatory and autoimmune disorders. Mol. Immunol. 2019, 105, 107–115. [Google Scholar] [CrossRef]
- Wang, L.; Sun, Y.; Zhang, Z.; Jia, Y.; Zou, Z.; Ding, J.; Li, Y.; Xu, X.; Jin, L.; Yang, T.; et al. CXCR5+ CD4+ T follicular helper cells participate in the pathogenesis of primary biliary cirrhosis. Hepatology 2015, 61, 627–638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, J.; Wang, T.; Zhang, L.; Cui, L. Dysregulation of Circulating Tfr/Tfh Ratio in Primary biliary cholangitis. Scand. J. Immunol. 2017, 86, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Strazzabosco, M.; Fiorotto, R.; Cadamuro, M.; Spirli, C.; Mariotti, V.; Kaffe, E.; Scirpo, R.; Fabris, L. Pathophysiologic implications of innate immunity and autoinflammation in the biliary epithelium. Biochim Biophys. Acta Mol. Basis Dis. 2018, 1864, 1374–1379. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, S.; Harada, K.; Niiro, H.; Taketomi, A.; Maehara, Y.; Tsuneyama, K.; Kikuchi, K.; Nakanuma, Y.; Mackay, I.R.; Gershwin, M.E.; et al. CX3CL1 (fractalkine): A signpost for biliary inflammation in primary biliary cirrhosis. Hepatology 2010, 51, 567–575. [Google Scholar] [CrossRef] [Green Version]
- Yokoyama, T.; Komori, A.; Nakamura, M.; Takii, Y.; Kamihira, T.; Shimoda, S.; Mori, T.; Fujiwara, S.; Koyabu, M.; Taniguchi, K.; et al. Human intrahepatic biliary epithelial cells function in innate immunity by producing IL-6 and IL-8 via the TLR4-NF-kappaB and -MAPK signaling pathways. Liver Int. 2006, 26, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Lian, M.; Zhang, J.; Bian, Z.; Tang, R.; Miao, Q.; Peng, Y.; Fang, J.; You, Z.; Invernizzi, P.; et al. A functional characteristic of cysteine-rich protein 61: Modulation of myeloid-derived suppressor cells in liver inflammation. Hepatology 2018, 67, 232–246. [Google Scholar] [CrossRef]
- Lleo, A.; Invernizzi, P. Apotopes and innate immune system: Novel players in the primary biliary cirrhosis scenario. Dig. Liver Dis. 2013, 45, 630–636. [Google Scholar] [CrossRef]
- Chuang, Y.H.; Lian, Z.X.; Tsuneyama, K.; Chiang, B.L.; Ansari, A.A.; Coppel, R.L.; Gershwin, M.E. Increased killing activity and decreased cytokine production in NK cells in patients with primary biliary cirrhosis. J. Autoimmun. 2006, 26, 232–240. [Google Scholar] [CrossRef]
- Shimoda, S.; Hisamoto, S.; Harada, K.; Iwasaka, S.; Chong, Y.; Nakamura, M.; Bekki, Y.; Yoshizumi, T.; Shirabe, K.; Ikegami, T.; et al. Natural killer cells regulate T cell immune responses in primary biliary cirrhosis. Hepatology 2015, 62, 1817–1827. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Kong, D.; Wang, H. Mucosal-Associated Invariant T cell in liver diseases. Int. J. Biol. Sci. 2020, 16, 460–470. [Google Scholar] [CrossRef]
- Al-Dury, S.; Wahlström, A.; Wahlin, S.; Langedijk, J.; Elferink, R.O.; Ståhlman, M.; Marschall, H.U. Pilot study with IBAT inhibitor A4250 for the treatment of cholestatic pruritus in primary biliary cholangitis. Sci. Rep. 2018, 8, 6658. [Google Scholar] [CrossRef] [PubMed]
- Acharya, C.; Sahingur, S.E.; Bajaj, J.S. Microbiota, cirrhosis, and the emerging oral-gut-liver axis. JCI Insight 2017, 2, e94416. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, P.; Hochrath, K.; Horvath, A.; Chen, P.; Seebauer, C.T.; Llorente, C.; Wang, L.; Alnouti, Y.; Fouts, D.E.; Stärkel, P.; et al. Modulation of the intestinal bile acid/farnesoid X receptor/fibroblast growth factor 15 axis improves alcoholic liver disease in mice. Hepatology 2018, 67, 2150–2166. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wan, Y.Y. The role of gut microbiota in liver disease development and treatment. Liver Res. 2019, 3, 3–18. [Google Scholar] [CrossRef]
- Inamine, T.; Schnabl, B. Immunoglobulin A and liver diseases. J. Gastroenterol. 2018, 53, 691–700. [Google Scholar] [CrossRef]
- Emamat, H.; Ghalandari, H.; Tangestani, H.; Abdollahi, A.; Hekmatdoost, A. Artificial sweeteners are related to non-alcoholic fatty liver disease: Microbiota dysbiosis as a novel potential mechanism. EXCLI J. 2020, 19, 620–626. [Google Scholar]
- Ohtani, N.; Kawada, N. Role of the Gut-Liver Axis in Liver Inflammation, Fibrosis, and Cancer: A Special Focus on the Gut Microbiota Relationship. Hepatol. Commun. 2019, 3, 456–470. [Google Scholar] [CrossRef] [Green Version]
- Hamoud, A.R.; Weaver, L.; Stec, D.E.; Hinds, T.D. Bilirubin in the Liver-Gut Signaling Axis. Trends Endocrinol. Metab. 2018, 29, 140–150. [Google Scholar] [CrossRef]
- Sato, K.; Hall, C.; Glaser, S.; Francis, H.; Meng, F.; Alpini, G. Pathogenesis of Kupffer Cells in Cholestatic Liver Injury. Am. J. Pathol. 2016, 186, 2238–2247. [Google Scholar] [CrossRef] [Green Version]
- Jeffery, H.C.; van Wilgenburg, B.; Kurioka, A.; Parekh, K.; Stirling, K.; Roberts, S.; Dutton, E.E.; Hunter, S.; Geh, D.; Braitch, M.K.; et al. Biliary epithelium and liver B cells exposed to bacteria activate intrahepatic MAIT cells through MR1. J. Hepatol. 2016, 64, 1118–1127. [Google Scholar] [CrossRef] [Green Version]
- Böttcher, K.; Rombouts, K.; Saffioti, F.; Roccarina, D.; Rosselli, M.; Hall, A.; Luong, T.; Tsochatzis, E.A.; Thorburn, D.; Pinzani, M. MAIT cells are chronically activated in patients with autoimmune liver disease and promote profibrogenic hepatic stellate cell activation. Hepatology 2018, 68, 172–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atif, M.; Warner, S.; Oo, Y.H. Linking the gut and liver: Crosstalk between regulatory T cells and mucosa-associated invariant T cells. Hepatol. Int. 2018, 12, 305–314. [Google Scholar] [CrossRef] [Green Version]
- Wiest, R.; Albillos, A.; Trauner, M.; Bajaj, J.S.; Jalan, R. Targeting the gut-liver axis in liver disease. J. Hepatol. 2017, 67, 1084–1103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Floreani, A.; Baragiotta, A.; Pizzuti, D.; Martines, D.; Cecchetto, A.; Chiarelli, S. Mucosal IgA defect in primary biliary cirrhosis. Am. J. Gastroenterol. 2002, 97, 508–510. [Google Scholar] [CrossRef] [PubMed]
- Campisi, L.; Barbet, G.; Ding, Y.; Esplugues, E.; Flavell, R.A.; Blander, J.M. Apoptosis in response to microbial infection induces autoreactive TH17 cells. Nat. Immunol. 2016, 17, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Haruta, I.; Kikuchi, K.; Hashimoto, E.; Nakamura, M.; Miyakawa, H.; Hirota, K.; Shibata, N.; Kato, H.; Arimura, Y.; Kato, Y.; et al. Long-term bacterial exposure can trigger nonsuppurative destructive cholangitis associated with multifocal epithelial inflammation. Lab. Investig. 2010, 90, 577–588. [Google Scholar] [CrossRef] [Green Version]
- Padgett, K.A.; Selmi, C.; Kenny, T.P.; Leung, P.S.; Balkwill, D.L.; Ansari, A.A.; Coppel, R.L.; Gershwin, M.E. Phylogenetic and immunological definition of four lipoylated proteins from Novosphingobium aromaticivorans, implications for primary biliary cirrhosis. J. Autoimmun. 2005, 24, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Mattner, J.; Savage, P.B.; Leung, P.; Oertelt, S.S.; Wang, V.; Trivedi, O.; Scanlon, S.T.; Pendem, K.; Teyton, L.; Hart, J.; et al. Liver autoimmunity triggered by microbial activation of natural killer T cells. Cell Host Microbe 2008, 3, 304–315. [Google Scholar] [CrossRef] [Green Version]
- Broz, P.; Dixit, V.M. Inflammasomes: Mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 2016, 16, 407–420. [Google Scholar] [CrossRef]
- Schroder, K.; Tschopp, J. The inflammasomes. Cell 2010, 140, 821–832. [Google Scholar] [CrossRef] [Green Version]
- Moretti, J.; Blander, J.M. Increasing complexity of NLRP3 inflammasome regulation. J. Leukoc. Biol. 2020. [Google Scholar] [CrossRef]
- Bauernfeind, F.G.; Horvath, G.; Stutz, A.; Alnemri, E.S.; MacDonald, K.; Speert, D.; Fernandes-Alnemri, T.; Wu, J.; Monks, B.G.; Fitzgerald, K.A.; et al. Cutting edge: NF-kappaB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J. Immunol. 2009, 183, 787–791. [Google Scholar] [CrossRef] [PubMed]
- Christgen, S.; Place, D.E.; Kanneganti, T.D. Toward targeting inflammasomes: Insights into their regulation and activation. Cell Res. 2020, 30, 315–327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Hara, H.; Nunez, G. Mechanism and Regulation of NLRP3 Inflammasome Activation. Trends Biochem. Sci. 2016, 41, 1012–1021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elliott, E.I.; Sutterwala, F.S. Initiation and perpetuation of NLRP3 inflammasome activation and assembly. Immunol. Rev. 2015, 265, 35–52. [Google Scholar] [CrossRef] [Green Version]
- Szabo, G.; Csak, T. Inflammasomes in liver diseases. J. Hepatol. 2012, 57, 642–654. [Google Scholar] [CrossRef] [Green Version]
- Kahlenberg, J.M.; Dubyak, G.R. Mechanisms of caspase-1 activation by P2 × 7 receptor-mediated K+ release. Am. J. Physiol. Cell Physiol. 2004, 286, C1100–C1108. [Google Scholar] [CrossRef] [Green Version]
- Hornung, V.; Bauernfeind, F.; Halle, A.; Samstad, E.O.; Kono, H.; Rock, K.L.; Fitzgerald, K.A.; Latz, E. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat. Immunol. 2008, 9, 847–856. [Google Scholar] [CrossRef]
- Zhou, R.; Tardivel, A.; Thorens, B.; Choi, I.; Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 2010, 11, 136–140. [Google Scholar] [CrossRef]
- Duewell, P.; Kono, H.; Rayner, K.J.; Sirois, C.M.; Vladimer, G.; Bauernfeind, F.G.; Abela, G.S.; Franchi, L.; Nunez, G.; Schnurr, M.; et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 2010, 464, 1357–1361. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Yin, Y.; Zhou, Z.; He, M.; Dai, Y. OxLDL-induced IL-1 beta secretion promoting foam cells formation was mainly via CD36 mediated ROS production leading to NLRP3 inflammasome activation. Inflamm. Res. 2014, 63, 33–43. [Google Scholar] [CrossRef]
- Sayan, M.; Mossman, B.T. The NLRP3 inflammasome in pathogenic particle and fibre-associated lung inflammation and diseases. Part. Fibre Toxicol. 2016, 13, 51. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Viollet, B.; Terkeltaub, R.; Liu-Bryan, R. AMP-activated protein kinase suppresses urate crystalinduced inflammation and transduces colchicine effects in macrophages. Ann. Rheum. Dis. 2016, 75, 286–294. [Google Scholar] [CrossRef] [Green Version]
- Pellegrini, C.; Antonioli, L.; Lopez-Castejon, G.; Blandizzi, C.; Fornai, M. Canonical and Non-Canonical Activation of NLRP3 Inflammasome at the Crossroad between Immune Tolerance and Intestinal Inflammation. Front. Immunol. 2017, 8, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antonopoulos, C.; Russo, H.M.; el Sanadi, C.; Martin, B.N.; Li, X.; Kaiser, W.J.; Mocarski, E.S.; George, R. DubyakCaspase-8 as an Effector and Regulator of NLRP3 Inflammasome Signaling. J. Biol. Chem. 2015, 290, 20167–20184. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.; Xing, Y.; Lu, A.; Fang, W.; Sun, B.; Chen, C.; Liao, W.; Meng, G. Internalized Cryptococcus neoformans Activates the Canonical Caspase-1 and the Noncanonical Caspase-8 Inflammasomes. J. Immunol. 2015, 195, 4962–4972. [Google Scholar] [CrossRef] [Green Version]
- Chang, T.H.; Huang, J.H.; Lin, H.C.; Chen, W.Y.; Lee, Y.; Hsu, L.; Netea, M.G.; Ting, J.P.; Wu-Hsieh, B. Dectin-2 is a primary receptor for NLRP3 inflammasome activation in dendritic cell response to Histoplasma capsulatum. PLoS Pathog. 2017, 13, e1006485. [Google Scholar] [CrossRef]
- Chung, H.; Vilaysane, A.; Lau, A.; Stahl, M.; Morampudi, V.; Bondzi-Simpson, A.; Platnich, J.M.; Bracey, N.A.; French, M.C.; Beck, P.L.; et al. NLRP3 regulates a non-canonical platform for caspase-8 activation during epithelial cell apoptosis. Cell Death Differ. 2016, 23, 1331–1346. [Google Scholar] [CrossRef]
- Gurung, P.; Anand, P.K.; Malireddi, R.K.; Vande Walle, L.; Van Opdenbosch, N.; Dillon, C.P.; Weinlich, R.; Green, D.R.; Lamkanfi, M.; Kanneganti, T.D. FADD and caspase-8 mediate priming and activation of the canonical and noncanonical Nlrp3 inflammasomes. J. Immunol. 2014, 192, 1835–1846. [Google Scholar] [CrossRef] [Green Version]
- Lamkanfi, M.; Dixit, V.M. Mechanisms and functions of inflammasomes. Cell 2014, 157, 1013–1022. [Google Scholar] [CrossRef]
- Maroni, L.; Agostinelli, L.; Saccomanno, S.; Pinto, C.; Giordano, D.M.; Rychlicki, C.; De Minicis, S.; Trozzi, L.; Banales, J.M.; Melum, E.; et al. Nlrp3 Activation Induces Il-18 Synthesis and Affects the Epithelial Barrier Function in Reactive Cholangiocytes. Am. J. Pathol. 2017, 187, 366–376. [Google Scholar] [CrossRef] [Green Version]
- Johannes, L.; Jacob, R.; Leffler, H. Galectins at a glance. J. Cell Sci. 2018, 131, jcs208884. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Popa, S.J.; Stewart, S.E.; Moreau, K. Unconventional secretion of annexins and galectins. Semin. Cell Dev. Biol. 2018, 83, 42–50. [Google Scholar] [CrossRef]
- Kim, B.W.; Hong, S.B.; Kim, J.H.; Kwon, D.H.; Song, H.K. Structural basis for recognition of autophagic receptor NDP52 by the sugar receptor galectin-8. Nat. Commun. 2013, 4, 1613. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Wandel, M.P.; Li, F.; Liu, Z.; He, C.; Wu, J.; Shi, Y.; Randow, F. Sterical hindrance promotes selectivity of the autophagy cargo receptor NDP52 for the danger receptor galectin-8 in antibacterial autophagy. Sci. Signal 2013, 6, ra9. [Google Scholar] [CrossRef] [Green Version]
- Yang, R.Y.; Rabinovich, G.A.; Liu, F.T. Galectins: Structure, function and therapeutic potential. Expert Rev. Mol. Med. 2008, 10, e17. [Google Scholar] [CrossRef]
- Cooper, D.N.; Barondes, S.H. God must love galectins; he made so many of them. Glycobiology 1999, 9, 979–984. [Google Scholar] [CrossRef]
- Herrmann, J.; Turck, C.W.; Atchison, R.E.; Huflejt, M.E.; Poulter, L.; Gitt, M.A.; Burlingame, A.L.; Barondes, S.H.; Leffler, H. Primary structure of the soluble lactose binding lectin L-29 from rat and dog and interaction of its non-collagenous praline-, glycine-, tyrosine-rich sequence with bacteria and tissue collagenase. J. Biol. Chem. 1993, 268, 26704–26711. [Google Scholar]
- Flores-Ibarra, A.; Vértesy, S.; Medrano, F.J.; Gabius, H.J.; Romero, A. Crystallization of a human galectin-3 variant with two ordered segments in the shortened N-terminal tail. Sci. Rep. 2018, 8, 9835. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.H.; Qiu, D.C.; Chang, W.H.; Yeh, Y.Q.; Jeng, U.-S.; Liu, F.T.; Huang, J.R. The intrinsically disordered N-terminal domain of galectin-3 dynamically mediates multisite self-association of the protein through fuzzy interactions. J. Biol. Chem. 2017, 292, 17845–17856. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.T.; Rabinovich, G.A. Galectins as modulators of tumour progression. Nat. Rev. Cancer 2005, 5, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Ochieng, J.; Furtak, V.; Lukyanov, P. Extracellular functions of galectin-3. Glycoconj. J. 2002, 19, 527–535. [Google Scholar] [CrossRef]
- Brinchmann, M.F.; Patel, D.M.; Iversen, M.H. The Role of Galectins as Modulators of Metabolism and Inflammation. Mediat. Inflamm. 2018, 2018, 9186940. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, F.; Finley, R.L., Jr.; Raz, A.; Kim, H.R. Galectin-3 translocates to the perinuclear membranes and inhibits cytochrome c release from the mitochondria. A role for synexin in galectin-3 translocation. J. Biol. Chem. 2002, 277, 15819–15827. [Google Scholar] [CrossRef] [Green Version]
- Chou, F.C.; Chen, H.Y.; Kuo, C.C.; Sytwu, H.K. Role of Galectins in Tumors and in Clinical Immunotherapy. Int. J. Mol. Sci. 2018, 19, 430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thijssen, V.L.; Poirier, F.; Baum, L.G.; Griffioen, A.W. Galectins in the tumor endothelium: Opportunities for combined cancer therapy. Blood 2007, 110, 2819–2827. [Google Scholar] [CrossRef]
- Moutsatsos, I.K.; Wade, M.; Schindler, M.; Wang, J.L. Endogenous lectins from cultured cells: Nuclear localization of carbohydrate-binding protein 35 in proliferating 3T3 fibroblasts. Proc. Natl. Acad. Sci. USA 1987, 84, 6452–6456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Openo, K.P.; Kadrofske, M.M.; Patterson, R.J.; Wang, J.L. Galectin-3 expression and subcellular localization in senescent human fibroblasts. Exp. Cell Res. 2000, 255, 278–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, F.T.; Patterson, R.J.; Wang, J.L. Intracellular functions of galectins. Biochim. Biophys. Acta 2002, 1572, 263–273. [Google Scholar] [CrossRef]
- Paz, A.; Haklai, R.; Elad-Sfadia, G.; Ballan, E.; Kloog, Y. Galectin-1 binds oncogenic H-Ras to mediate Ras membrane anchorage and cell transformation. Oncogene 2001, 20, 7486–7493. [Google Scholar] [CrossRef] [Green Version]
- Shalom-Feuerstein, R.; Cooks, T.; Raz, A.; Kloog, Y. Galectin-3 regulates a molecular switch from N-Ras to K-Ras usage in human breast carcinoma cells. Cancer Res. 2005, 65, 7292–7300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, A.; Poirier, F.; Jacob, R.; Delacour, D. Galectin-3, a novel centrosome-associated protein, required for epithelial morphogenesis. Mol. Biol. Cell 2010, 21, 219–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clare, D.K.; Magescas, J.; Piolot, T.; Dumoux, M.; Vesque, C.; Pichard, E.; Dang, T.; Duvauchelle, B.; Poirier, F.; Delacour, D. Basal foot MTOC organizes pillar MTs required for coordination of beating cilia. Nat. Commun. 2014, 5, 4888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magescas, J.; Sengmanivong, L.; Viau, A.; Mayeux, A.; Dang, T.; Burtin, M.; Nilsson, U.J.; Leffler, H.; Poirier, F.; Terzi, F.; et al. Spindle pole cohesion requires glycosylation-mediated localization of NuMA. Sci. Rep. 2017, 7, 1474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gilson, R.C.; Gunasinghe, S.D.; Johannes, L.; Gaus, K. Galectin-3 modulation of T-cell activation: Mechanisms of membrane remodelling. Prog. Lipid Res. 2019, 76, 101010. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Hsu, D.K.; Chen, H.Y.; Yang, R.Y.; Carraway, K.L., 3rd; Isseroff, R.R.; Liu, F.T. Galectin-3 regulates intracellular trafficking of EGFR through Alix and promotes keratinocyte migration. J. Invest. Dermatol. 2012, 132, 2828–2837. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.F.; Tsao, C.H.; Lin, Y.T.; Hsu, D.K.; Chiang, M.L.; Lo, C.H.; Chien, F.C.; Chen, P.; Arthur Chen, Y.M.; Chen, H.Y.; et al. Galectin-3 promotes HIV-1 budding via association with Alix and Gag p6. Glycobiology 2014, 24, 1022–1035. [Google Scholar] [CrossRef] [Green Version]
- Shimura, T.; Takenaka, Y.; Fukumori, T.; Tsutsumi, S.; Okada, K.; Hogan, V.; Kikuchi, A.; Kuwano, H.; Raz, A. Implication of galectin-3 in Wnt signaling. Cancer Res. 2005, 65, 3535–3537. [Google Scholar] [CrossRef] [Green Version]
- Harazono, Y.; Nakajima, K.; Raz, A. Why anti-Bcl-2 clinical trials fail: A solution. Cancer Metastasis Rev. 2014, 33, 285–294. [Google Scholar] [CrossRef] [Green Version]
- Nangia-Makker, P.; Hogan, V.; Raz, A. Galectin-3 and cancer stemness. Glycobiology 2018, 28, 172–181. [Google Scholar] [CrossRef] [Green Version]
- Dagher, S.F.; Wang, J.L.; Patterson, R.J. Identification of galectin-3 as a factor in pre-mRNA splicing. Proc. Natl. Acad. Sci. USA 1995, 92, 1213–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laing, J.G.; Wang, J.L. Identification of carbohydrate binding protein 35 in heterogeneous nuclear ribonucleoprotein complex. Biochemistry 1988, 27, 5329–5334. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.M.; Pestell, R.G.; Raz, A.; Kim, H.R. Galectin-3 enhances cyclin D1 promoter activity through SP1 and a cAMP-responsive element in human breast epithelial cells. Oncogene 2002, 21, 8001–8010. [Google Scholar] [CrossRef] [Green Version]
- Paz, I.; Sachse, M.; Dupont, N.; Mounier, J.; Cederfur, C.; Enninga, J.; Leffler, H.; Poirier, F.; Prevost, M.C.; Lafont, F.; et al. Galectin-3, a marker for vacuole lysis by invasive pathogens. Cell Microbiol. 2010, 12, 530–544. [Google Scholar] [CrossRef] [PubMed]
- Maier, O.; Marvin, S.A.; Wodrich, H.; Campbell, E.M.; Wiethoff, C.M. Spatiotemporal dynamics of adenovirus membrane rupture and endosomal escape. J. Virol. 2012, 86, 10821–10828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flavin, W.P.; Bousset, L.; Green, Z.C.; Chu, Y.; Skarpathiotis, S.; Chaney, M.J.; Kordower, J.H.; Melki, R.; Campbell, E.M. Endocytic vesicle rupture is a conserved mechanism of cellular invasion by amyloid proteins. Acta Neuropathol. 2017, 134, 629–653. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Gan, M.; Yen, S.H.; McLean, P.J.; Dickson, D.W. Impaired endo-lysosomal membrane integrity accelerates the seeding progression of α-synuclein aggregates. Sci. Rep. 2017, 7, 7690. [Google Scholar] [CrossRef]
- Vasta, G.R. Galectins in Host-Pathogen Interactions: Structural, Functional and Evolutionary Aspects. Adv. Exp. Med. Biol. 2020, 1204, 169–196. [Google Scholar]
- Furtak, V.; Hatcher, F.; Ochieng, J. Galectin-3 mediates the endocytosis of beta-1 integrins by breast carcinoma cells. Biochem. Biophys. Res. Commun. 2001, 289, 845–850. [Google Scholar] [CrossRef]
- Stechly, L.; Morelle, W.; Dessein, A.F.; André, S.; Grard, G.; Trinel, D.; Dejonghe, M.J.; Leteurtre, E.; Drobecq, H.; Trugnan, G.; et al. Galectin-4-regulated delivery of glycoproteins to the brush border membrane of enterocyte-like cells. Traffic 2009, 10, 438–450. [Google Scholar] [CrossRef]
- Straube, T.; von Mach, T.; Hönig, E.; Greb, C.; Schneider, D.; Jacob, R. pH-dependent recycling of galectin-3 at the apical membrane of epithelial cells. Traffic 2013, 14, 1014–1027. [Google Scholar] [CrossRef] [PubMed]
- Arthur, C.M.; Baruffi, M.D.; Cummings, R.D.; Stowell, S.R. Evolving mechanistic insights into galectin functions. Methods Mol. Biol. 2015, 1207, 1–35. [Google Scholar] [PubMed] [Green Version]
- Gordon-Alonso, M.; Hirsch, T.; Wildmann, C.; van der Bruggen, P. Galectin-3 captures interferon-gamma in the tumor matrix reducing chemokine gradient production and T-cell tumor infiltration. Nat. Commun. 2017, 8, 793. [Google Scholar] [CrossRef]
- Thiemann, S.; Baum, L.G. Galectins and Immune Responses-Just How Do They Do Those Things They Do? Annu. Rev. Immunol. 2016, 34, 243–264. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Nieminen, J. Seeing strangers or announcing “danger”: Galectin-3 in two models of innate immunity. Glycoconj. J. 2002, 19, 583–591. [Google Scholar] [CrossRef]
- Blidner, A.G.; Méndez-Huergo, S.P.; Cagnoni, A.J.; Rabinovich, G.A. Re-wiring regulatory cell networks in immunity by galectin-glycan interactions. FEBS Lett. 2015, 589, 3407–3418. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.T.; Hsu, D.K. The role of galectin-3 in promotion of the inflammatory response. Drug News Perspec. 2007, 20, 455–460. [Google Scholar] [CrossRef]
- Liu, F.T.; Hsu, D.K.; Zuberi, R.I.; Kuwabara, I.; Chi, E.Y.; Henderson, W.R., Jr. Expression and function of galectin-3, a beta-galactoside-binding lectin, in human monocytes and macrophages. Am. J. Pathol. 1995, 147, 1016–1028. [Google Scholar]
- Acosta-Rodríguez, E.V.; Montes, C.L.; Motrán, C.C.; Zuniga, E.I.; Liu, F.T.; Rabinovich, G.A.; Gruppi, A. Galectin-3 Mediates IL-4-Induced Survival and Differentiation of B Cells: Functional Cross-Talk and Implications during Trypanosoma cruzi Infection. J. Immunol. 2004, 172, 493–502. [Google Scholar] [CrossRef] [Green Version]
- Dietz, A.B.; Bulur, P.A.; Knutson, G.J.; Matasić, R.; Vuk-Pavlović, S. Maturation of Human Monocyte-Derived Dendritic Cells Studied by Microarray Hybridization. Biochem. Biophys. Res. Commun. 2000, 275, 731–738. [Google Scholar] [CrossRef]
- Cerliani, J.P.; Stowell, S.R.; Mascanfroni, I.D.; Arthur, C.M.; Cummings, R.D.; Rabinovich, G.A. Expanding the Universe of Cytokines and Pattern Recognition Receptors: Galectins and Glycans in Innate Immunity. J. Clin. Immunol. 2011, 31, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, T.K.; Honing, H.; Franke, N.; van Remoortere, A.; Schiphorst, W.E.; Liu, F.T.; Deelder, A.M.; Cummings, R.D.; Hokke, C.H.; van Die, I. LacdiNAc-glycans constitute a parasite pattern for galectin-3-mediated immune recognition. J. Immunol. 2004, 173, 1902–1907. [Google Scholar] [CrossRef] [Green Version]
- John, C.M.; Jarvis, G.A.; Swanson, K.V.; Leffler, H.; Cooper, M.D.; Huflejt, M.E.; Griffiss, J.M. Galectin-3 binds lactosaminylated lipooligosaccharides from Neisseria gonorrhoeae and is selectively expressed by mucosal epithelial cells that are infected. Cell Microbiol. 2002, 4, 649–662. [Google Scholar] [CrossRef]
- Pelletier, I.; Sato, S. Specific recognition and cleavage of galectin-3 by Leishmania major through species-specific polygalactose epitope. J. Biol. Chem. 2002, 277, 17663–17670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Souza, B.; da Silva, K.N.; Silva, D.N.; Rocha, V.; Paredes, B.D.; Azevedo, C.M.; Nonaka, C.K.; Carvalho, G.B.; Vasconcelos, J.F.; Dos Santos, R.R.; et al. Galectin-3 Knockdown Impairs Survival, Migration, and Immunomodulatory Actions of Mesenchymal Stromal Cells in a Mouse Model of Chagas Disease Cardiomyopathy. Stem Cells Int. 2017, 2017, 3282656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radosavljevic, G.; Volarevic, V.; Jovanovic, I.; Milovanovic, M.; Pejnovic, N.; Arsenijevic, N.; Hsu, D.K.; Lukic, M.L. The roles of Galectin-3 in autoimmunity and tumor progression. Immunol. Res. 2012, 52, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Wongkham, S.; Junking, M.; Wongkham, C.; Sripa, B.; Chur-In, S.; Araki, N. Suppression of galectin-3 expression enhances apoptosis and chemosensitivity in liver fluke-associated cholangiocarcinoma. Cancer Sci. 2009, 100, 2077–2084. [Google Scholar] [CrossRef]
- Arsenijevic, A.; Milovanovic, M.; Milovanovic, J.; Stojanovic, B.; Zdravkovic, N.; Leung, P.S.; Liu, F.T.; Gershwin, M.E.; Lukic, M.L. Deletion of Galectin-3 enhances xenobiotic induced murine primary biliary cholangitis by facilitating apoptosis of BECs and release of autoantigens. Sci. Rep. 2016, 6, 23348. [Google Scholar] [CrossRef] [Green Version]
- Inzaugarat, M.E.; Johnson, C.D.; Holtmann, T.M.; McGeough, M.D.; Trautwein, C.; Papouchado, B.G.; Schwabe, R.; Hoffman, H.M.; Wree, A.; Feldstein, A.E. NLR Family Pyrin Domain-Containing 3 Inflammasome Activation in Hepatic Stellate Cells Induces Liver Fibrosis in Mice. Hepatology 2019, 69, 845–859. [Google Scholar] [CrossRef] [Green Version]
- Szabo, G.; Petrasek, J. Inflammasome activation and function in liver disease. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 387–400. [Google Scholar] [CrossRef]
- Tian, J.; Yang, G.; Chen, H.Y.; Hsu, D.K.; Tomilov, A.; Olson, K.A.; Dehnad, A.; Fish, S.R.; Cortopassi, G.; Zhao, B.; et al. Galectin-3 regulates inflammasome activation in cholestatic liver injury. FASEB J. 2016, 30, 4202–4213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, B.K.; Wen, H.; Ting, J.P. The inflammasome NLRs in immunity, inflammation, and associated diseases. Annu. Rev. Immunol. 2011, 29, 707–735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sato, S.; St-Pierre, C.; Bhaumik, P.; Nieminen, J. Galectins in innate immunity: Dual functions of host soluble beta-galactoside-binding lectins as damage-associated molecular patterns (DAMPs) and as receptors for pathogen-associated molecular patterns (PAMPs). Immunol. Rev. 2009, 230, 172–187. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.X.; Chen, X.; Hsu, D.K.; Baghy, K.; Serizawa, N.; Scott, F.; Takada, Y.; Takada, Y.; Fukada, H.; Chen, J.; et al. Galectin-3 modulates phagocytosis-induced stellate cell activation and liver fibrosis in vivo. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, G439–G446. [Google Scholar]
- Arsenijevic, A.; Milovanovic, J.; Stojanovic, B.; Djordjevic, D.; Stanojevic, I.; Jankovic, N.; Vojvodic, D.; Arsenijevic, N.; Lukic, M.L.; Milovanovic, M. Gal-3 Deficiency Suppresses Novosphyngobium aromaticivorans Inflammasome Activation and IL-17 Driven Autoimmune Cholangitis in Mice. Front. Immunol. 2019, 10, 1309. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Han, C.; Liu, J. The Role of Toll-Like Receptors in Oncotherapy. Oncol. Res. 2019, 27, 965–978. [Google Scholar] [CrossRef]
- Kawahara, K.; Moll, H.; Knirel, Y.A.; Seydel, U.; Zähringer, U. Structural analysis of two glycosphingolipids from the lipopolysaccharide-lacking bacterium Sphingomonas capsulata. Eur. J. Biochem. 2000, 267, 1837–1846. [Google Scholar] [CrossRef] [Green Version]
- Simovic Markovic, B.; Nikolic, A.; Gazdic, M.; Bojic, S.; Vucicevic, L.; Kosic, M.; Mitrovic, S.; Milosavljevic, M.; Besra, G.; Trajkovic, V.; et al. Galectin-3 Plays an important pro-inflammatory role in the induction phase of acute colitis by promoting activation of NLRP3 inflammasome and production of IL-1β in macrophages. J. Crohns Colitis 2016, 10, 593–606. [Google Scholar] [CrossRef] [Green Version]
- Matsushita, H.; Miyake, Y.; Takaki, A.; Yasunaka, T.; Koike, K.; Ikeda, F.; Shiraha, H.; Nouso, K.; Yamamoto, K. TLR4, TLR9, and NLRP3 in biliary epithelial cells of primary sclerosing cholangitis: Relationship with clinical characteristics. J. Gastroenterol. Hepatol. 2015, 30, 600–608. [Google Scholar] [CrossRef]
- Shimonishi, T.; Miyazaki, K.; Kono, N.; Sabit, H.; Tuneyama, K.; Harada, K.; Hirabayashi, J.; Kasai, K.; Nakanuma, Y. Expression of endogenous galectin-1 and galectin-3 in intrahepatic cholangiocarcinoma. Hum. Pathol. 2001, 32, 302–310. [Google Scholar] [CrossRef]
- Shimura, T.; Kofunato, Y.; Okada, R.; Yashima, R.; Koyama, Y.; Araki, K.; Kuwano, H.; Takenoshita, S. Intranuclear accumulation of galectin-3 is an independent prognostic factor for patients with distal cholangiocarcinoma. Oncol. Lett. 2017, 14, 819–829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stojanovic, B.; Milovanovic, J.; Arsenijevic, A.; Geljic, I.S.; Arsenijevic, N.; Jonjic, S.; Lukic, M.L.; Milovanovic, M.; Stojanovic, B. Galectin-3 Deficiency Facilitates TNF-α-Dependent Hepatocyte Death and Liver Inflammation in MCMV Infection. Front. Microbiol. 2019, 10, 185. [Google Scholar] [CrossRef]
- Volarevic, V.; Milovanovic, M.; Ljujic, B.; Pejnovic, N.; Arsenijevic, N.; Nilsson, U.J.; Leffler, H.; Lukic, M.L. Gal-3 regulates the capacity of dendritic cells to promote NKT-cell-induced liver injury. Eur. J. Immunol. 2015, 45, 531–543. [Google Scholar] [CrossRef]
- Volarevic, V.; Markovic, B.S.; Bojic, S.; Stojanović, M.; Nilsson, U.J.; Leffler, H.; Besra, G.S.; Arsenijevic, N.; Paunovic, V.; Trajkovic, V.; et al. Galectin-3 deficiency prevents concanavalin A-induced hepatitis in mice. Hepatology 2012, 55, 1954–1964. [Google Scholar] [CrossRef] [PubMed]
- Iacobini, C.; Menini, S.; Ricci, C.; Fantauzzi, C.B.; Scipioni, A.; Salvi, L.; Cordone, S.; Delucchi, F.; Serino, M.; Federici, M.; et al. Galectin-3 ablation protects mice from diet-induced NASH: A major scavenging role for galectin-3 in liver. J. Hepatol. 2011, 54, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Jeftic, I.; Jovicic, N.; Pantic, J.; Arsenijević, N.N.; Lukic, M.L.; Pejnovic, N. Galectin-3 Ablation Enhances Liver Steatosis, but Attenuates Inflammation and IL-33-Dependent Fibrosis in Obesogenic Mouse Model of Nonalcoholic Steatohepatitis. Mol. Med. 2015, 21, 453–465. [Google Scholar] [CrossRef] [PubMed]
- Serizawa, N.; Tian, J.; Fukada, H.; Baghy, K.; Scott, F.; Chen, X.; Kiss, Z.; Olson, K.; Hsu, D.; Liu, F.-T.; et al. Galectin 3 regulates HCC cell invasion by RhoA and MLCK activation. Lab. Invest. 2015, 95, 1145–1156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanbe, A.; Ito, H.; Omori, Y.; Hara, A.; Seishima, M. The inhibition of NLRP3 signaling attenuates liver injury in an α-galactosylceramide-induced hepatitis model. Biochem. Biophys. Res. Commun. 2017, 490, 364–370. [Google Scholar] [CrossRef]
- Luan, J.; Zhang, X.; Wang, S.; Li, Y.; Fan, J.; Chen, W.; Zai, W.; Wang, S.; Wang, Y.; Chen, M.; et al. NOD-Like Receptor Protein 3 Inflammasome-Dependent IL-1β Accelerated ConA-Induced Hepatitis. Front. Immunol. 2018, 9, 758. [Google Scholar] [CrossRef]
- Martínez-Cardona, C.; Lozano-Ruiz, B.; Bachiller, V.; Peiró, G.; Algaba-Chueca, F.; Gomez-Hurtado, I.; Such, J.; Zapater, P.; Francés, R.; González-Navajas, J. AIM2 deficiency reduces the development of hepatocellular carcinoma in mice. Int. J. Cancer 2018, 143, 2997–3007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pierantonelli, I.; Rychlicki, C.; Agostinelli, L.; Giordano, D.M.; Gaggini, M.; Fraumene, C.; Saponaro, C.; Mangina, V.; Sartini, L.; Mingarelli, E.; et al. Lack of NLRP3-inflammasome leads to gut-liver axis derangement, gut dysbiosis and a worsened phenotype in a mouse model of NAFLD. Sci. Rep. 2017, 7, 12200. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Zhu, X.; Li, P.; Li, J.; Gong, J.P.; Shen, W.; He, K. NLRP3 Deletion Inhibits the Non-alcoholic Steatohepatitis Development and Inflammation in Kupffer Cells Induced by Palmitic Acid. Inflammation 2017, 40, 1875–1883. [Google Scholar] [CrossRef] [PubMed]
- Wree, A.; McGeough, M.D.; Pena, C.A.; Schlattjan, M.; Li, H.; Inzaugarat, M.E.; Messer, K.; Canbay, A.; Hoffman, H.M.; Feldstein, A.E. NLRP3 inflammasome activation is required for fibrosis development in NAFLD. J. Mol. Med. 2014, 92, 1069–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Disease | Model | Role of Gal-3 | Mechanisms | References |
|---|---|---|---|---|
| Experimental Primary Biliary Cholangitis (PBC) | Xenobiotic induced PBC in Gal-3 deficient mice | Protective | Gal-3 protects biliary epithelial cells from apoptosis and thus attenuates autoantigen release | [148] |
| Novosphingobium aromaticivorans induced PBC in Gal-3 deficient mice | Proinflammatory | Gal-3 is necessary for the activation of NLRP3 inflammasome in liver macrophages and subsequent IL-17 mediated inflammatory process | [155] | |
| Spontaneously developed autoimmune cholangitis in dnTGF-βRII mice and Gal-3 deficient mice | Proinflammatory | Gal-3 is an initiator of inflammatory signaling and mediation of the activation of NLRP3 inflammasome and IL-17 proinflammatory cascades | [151] | |
| Experimental Hepatitis | MCMV induced hepatitis Gal-3 deficient mice | Antiinflammatory, protective | Gal-3 attenuates TNF-α-mediated death of hepatocytes | [162] |
| α-galactosylceramide induced hepatitis in Gal-3 deficient mice | Proinflammatory | Gal-3 regulates the capacity of DCs to support NKT-cell-mediated liver injury | [163] | |
| Con A-induced hepatitis in Gal-3 deficient mice and Gal-3 inhibitor treated mice | Proinflammatory | Gal-3 promotes the influx of mononuclear cells and proinflammatory CD4+ cells in the liver and decreases influx of IL-10 expressing CD4+ T cells and F4/80+ macrophages | [164] | |
| Experimental Non-Alcoholic Fatty Liver Disease | High-fat diet nonalcoholic steatohepatitis induced in Gal-3 deficient mice | Proinflammatory | Gal-3 increases hepatic accumulation of advanced lipoxidation endproducts, up-regulates lipid synthesis and oxidation causing more fat deposition, oxidative stress, and inflammation | [165] |
| High-fat diet induced steatohepatitis and liver steatosis in Gal-3 deficient mice | Proinflammatory | Increased mRNA expression of CCL2, F4/80, CD11c, TLR4, CD14, NLRP3 inflammasome, IL-1β, IL-13 and IL-33 in the liver | [166] | |
| Experimental Hepatocellular Carcinoma | N-diethylnitrosamine induced hepatocellular carcinoma in Gal-3 deficient mice | Pro-tumor effects | Gal-3 promotes motility and invasion of hepatoma cells by an autocrine pathway | [167] |
| Disease | Model | Role of Inflammasome | Mechanisms | References |
|---|---|---|---|---|
| Experimental Hepatitis | α-galactosylceramide induced hepatitis in NALP3 deficient mice | Proinflammatory | NALP3 promotes expression of proinflammatory cytokines (IL-6, and TNF-α) | [168] |
| Con A-induced hepatitis in Nlrp3 deficient mice | Proinflammatory | NLRP3 inflammasome dependent IL-1β production is crucial for hepatitis development | [169] | |
| Experimental Hepatocellular Carcinoma | N-diethylnitrosamine induced hepatocellular carcinoma in AIM2 deficient mice | Pro-tumor effects | AIM2 inflammasome component promotes inflammation during carcinogenic liver injury and contributes to genotoxic HCC development in mice | [170] |
| Experimental Non-Alcoholic Fatty Liver Disease | Western-lifestyle diet with fructose in drinking water induced Non-Alcoholic Fatty Liver Disease in Nlrp3 deficient mice | Antiinflammatory | NLRP3 maintains normal gut immune response and attenuates adipose tissue inflammation | [171] |
| Experimental Non-Alcoholic Fatty Liver Disease | Methionine-choline-deficient diet induced Non-Alcoholic Fatty Liver Disease in Nlrp3 deficient mice | Proinflammatory | NLRP3 stimulates IL-1β and IL-18 secretion induced by palmitic acid stimulation and promotes liver inflammation | [172] |
| Experimental Non-Alcoholic Fatty Liver Disease | Short-term choline-deficient amino acid-defined diet, to induce isolated hepatic steatosis or long-term exposure, to induce severe steatohepatitis and fibrosis in Nlrp3 deficient mice | Proinflammatory and profibrotic | NLRP3 stimulates IL-1β secretion and promotes liver inflammation and fibrosis | [173] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arsenijevic, A.; Stojanovic, B.; Milovanovic, J.; Arsenijevic, D.; Arsenijevic, N.; Milovanovic, M. Galectin-3 in Inflammasome Activation and Primary Biliary Cholangitis Development. Int. J. Mol. Sci. 2020, 21, 5097. https://doi.org/10.3390/ijms21145097
Arsenijevic A, Stojanovic B, Milovanovic J, Arsenijevic D, Arsenijevic N, Milovanovic M. Galectin-3 in Inflammasome Activation and Primary Biliary Cholangitis Development. International Journal of Molecular Sciences. 2020; 21(14):5097. https://doi.org/10.3390/ijms21145097
Chicago/Turabian StyleArsenijevic, Aleksandar, Bojana Stojanovic, Jelena Milovanovic, Dragana Arsenijevic, Nebojsa Arsenijevic, and Marija Milovanovic. 2020. "Galectin-3 in Inflammasome Activation and Primary Biliary Cholangitis Development" International Journal of Molecular Sciences 21, no. 14: 5097. https://doi.org/10.3390/ijms21145097




