Sulforaphane Elicits Protective Effects in Intestinal Ischemia Reperfusion Injury
Abstract
:1. Introduction
2. Results
2.1. SFN Treated Mice were Less Susceptible to Intestinal IRI
2.2. SFN Ameliorated IRI-Elicited Leukocyte and Platelet Recruitment
2.3. SFN Treatment Decreased IRI-Elicited Neutrophil Infiltration in Intestinal Tissue without Altering Expression of Endothelial Adhesion Molecules
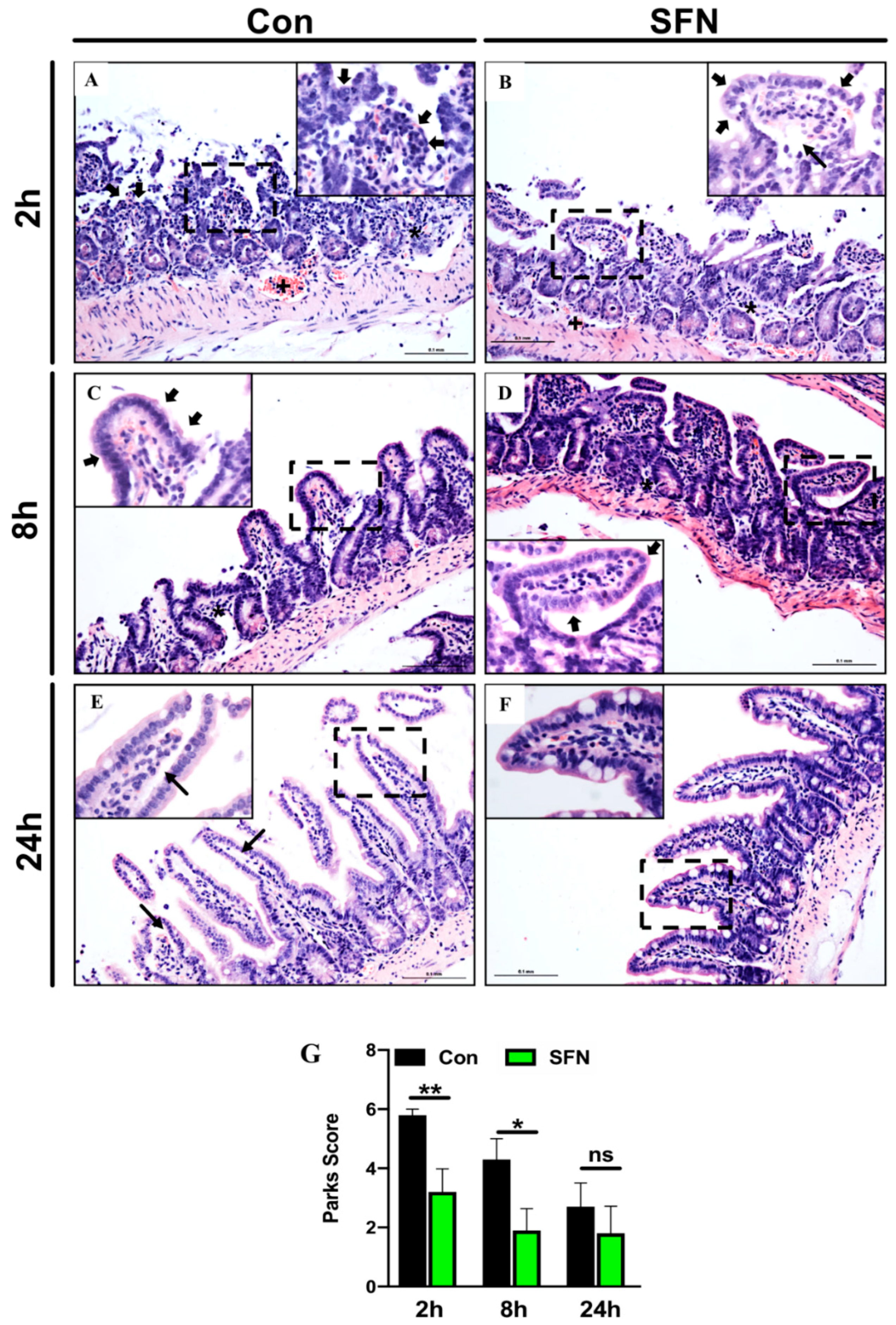
2.4. SFN Reduced IRI-Elicited Ultrastructural Damage in the Intestine Protect Intestinal Barrier Integrity
2.5. SFN Treated Mice were Less Prone to Altered Mucosal Permeability of the Small Bowel
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Animals
4.3. Experimental Groups and Treatment
4.4. In Vivo Model of Intestinal IRI
4.5. Ex Vivo Platelet Preparation
4.6. In Vivo Leukocyte Staining and Fluorescence Intravital Microscopy (IVM)
4.7. Offline Video Analysis
4.8. Intestinal Histology
4.9. Electron Microscope
4.10. Intestinal Tissue Myeloperoxidase (MPO) Activity
4.11. Intestinal Mucosal Permeability
4.12. Quantitative Polymerase Chain Reaction (qPCR)
4.13. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| IRI | Intestinal Reperfusion Injury |
| SFN | Sulforaphane |
| DAMPs | Damage Associated Molecular Patterns |
| PRRs | Pattern Recognition Receptors |
| TLRs | Toll Like Receptors |
| VEC | Vascular Endothelial Cell |
| ICAM-1 | Intracellular Adhesion Molecule-1 |
| VCAM-1 | Vascular Cell Adhesion Molecule-1 |
| PSGL-1 | P-selectin Glycoprotein Ligand-1 |
| Nrf2 | NF-erythroid 2 Related Factor 2 |
| ARE | Antioxidant Response Element |
| HO-1 | Heme Oxygenase-1 |
| GSH-Px | Glutathione Peroxidase |
| H&E | Hematoxylin & Eosin |
| IVM | Intravital Microscopy |
| MPO | Myeloperoxidase |
| OVA | Ovalbumine |
| GPIIb/IIIa | Glycoprotein IIb/IIIa |
| LPS | Lipopolysaccharide |
| ROS | Reactive Oxygen Species |
| BW | Body Weight |
| PBS | Phosphate Buffered Saline |
| Ip | Intraperitoneal |
| SMA | Superior Mesenteric Artery |
| ACD | Acid-Citrate-Dextrose Solution |
| CFSE | Carboxyfluorescein Diacetate Succinimidyl Ester |
| Pcvs | Postischemic Intestinal Postcapillary Venules |
| HETAB | Hexadecyltrimethylammonium Bromide |
| KPi | Potassium Phosphate Buffer |
| OD | o-Dianisidine Dihydrochloride |
References
- Lenaerts, K.; Ceulemans, L.J.; Hundscheid, I.H.; Grootjans, J.; Dejong, C.H.; Olde Damink, S.W. New insights in intestinal ischemia-reperfusion injury: Implications for intestinal transplantation. Curr. Opin. Organ Transplant. 2013, 18, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Eltzschig, H.K.; Eckle, T. Ischemia and reperfusion—From mechanism to translation. Nat. Med. 2011, 17, 1391–1401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clair, D.G.; Beach, J.M. Mesenteric Ischemia. N. Engl. J. Med. 2016, 374, 959–968. [Google Scholar] [CrossRef] [Green Version]
- Vollmar, B.; Menger, M.D. Intestinal ischemia/reperfusion: Microcirculatory pathology and functional consequences. Langenbeck’s Arch. Surg. 2011, 396, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kobata, A.; Tanigawa, T.; Nadatani, Y.; Yamagami, H.; Watanabe, K.; Tominaga, K.; Fujiwara, Y.; Takeuchi, K.; Arakawa, T. Activation of the MyD88 signaling pathway inhibits ischemia-reperfusion injury in the small intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G324–G334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nourshargh, S.; Alon, R. Leukocyte migration into inflamed tissues. Immunity 2014, 41, 694–707. [Google Scholar] [CrossRef] [Green Version]
- Cooper, D.; Russell, J.; Chitman, K.D.; Williams, M.C.; Wolf, R.E.; Granger, D.N. Leukocyte dependence of platelet adhesion in postcapillary venules. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H1895–H1900. [Google Scholar] [CrossRef] [Green Version]
- Van Gils, J.M.; Zwaginga, J.J.; Hordijk, P.L. Molecular and functional interactions among monocytes, platelets, and endothelial cells and their relevance for cardiovascular diseases. J. Leukoc. Biol. 2009, 85, 195–204. [Google Scholar] [CrossRef]
- Ghasemzadeh, M.; Hosseini, E. Platelet-leukocyte crosstalk: Linking proinflammatory responses to procoagulant state. Thromb. Res. 2013, 131, 191–197. [Google Scholar] [CrossRef]
- Rossaint, J.; Margraf, A.; Zarbock, A. Role of Platelets in Leukocyte Recruitment and Resolution of Inflammation. Front. Immunol. 2018, 9, 2712. [Google Scholar] [CrossRef]
- Salter, J.W.; Krieglstein, C.F.; Issekutz, A.C.; Granger, D.N. Platelets modulate ischemia/reperfusion-induced leukocyte recruitment in the mesenteric circulation. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 281, G1432–G1439. [Google Scholar] [CrossRef] [PubMed]
- Ed Rainger, G.; Chimen, M.; Harrison, M.J.; Yates, C.M.; Harrison, P.; Watson, S.P.; Lordkipanidzé, M.; Nash, G.B. The role of platelets in the recruitment of leukocytes during vascular disease. Platelets 2015, 26, 507–520. [Google Scholar] [CrossRef] [PubMed]
- Jurk, K.; Kehrel, B.E. Platelets: Physiology and biochemistry. Semin. Thromb. Hemost. 2005, 31, 381–392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Talalay, P.; Cho, C.G.; Posner, G.H. A major inducer of anticarcinogenic protective enzymes from broccoli: Isolation and elucidation of structure. Proc. Natl. Acad. Sci. USA 1992, 89, 2399–2403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.D.; Zhang, F.; Shen, G.; Li, Y.B.; Li, Y.H.; Jing, H.R.; Ma, L.F.; Yao, J.H.; Tian, X.F. Sulforaphane protects liver injury induced by intestinal ischemia reperfusion through Nrf2-ARE pathway. World J. Gastroenterol. 2010, 16, 3002–3010. [Google Scholar] [CrossRef]
- Yoon, H.Y.; Kang, N.I.; Lee, H.K.; Jang, K.Y.; Park, J.W.; Park, B.H. Sulforaphane protects kidneys against ischemia-reperfusion injury through induction of the Nrf2-dependent phase 2 enzyme. Biochem. Pharmacol. 2008, 75, 2214–2223. [Google Scholar] [CrossRef]
- Lee, J.M.; Johnson, J.A. An important role of Nrf2-ARE pathway in the cellular defense mechanism. J. Biochem. Mol. Biol. 2004, 37, 139–143. [Google Scholar] [CrossRef] [Green Version]
- Itoh, K.; Chiba, T.; Takahashi, S.; Ishii, T.; Igarashi, K.; Katoh, Y.; Oyake, T.; Hayashi, N.; Satoh, K.; Hatayama, I.; et al. An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem. Biophys. Res. Commun. 1997, 236, 313–322. [Google Scholar] [CrossRef]
- Venugopal, R.; Jaiswal, A.K. Nrf1 and Nrf2 positively and c-Fos and Fra1 negatively regulate the human antioxidant response element-mediated expression of NAD(P)H:quinone oxidoreductase1 gene. Proc. Natl. Acad. Sci. USA 1996, 93, 14960–14965. [Google Scholar] [CrossRef] [Green Version]
- Chuang, W.Y.; Kung, P.H.; Kuo, C.Y.; Wu, C.C. Sulforaphane prevents human platelet aggregation through inhibiting the phosphatidylinositol 3-kinase/Akt pathway. Thromb. Haemost. 2013, 109, 1120–1130. [Google Scholar] [CrossRef]
- Jayakumar, T.; Chen, W.F.; Lu, W.J.; Chou, D.S.; Hsiao, G.; Hsu, C.Y.; Sheu, J.R.; Hsieh, C.Y. A novel antithrombotic effect of sulforaphane via activation of platelet adenylate cyclase: Ex Vivo and In Vivo studies. J. Nutr. Biochem. 2013, 24, 1086–1095. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, S.; Holloway, P.M.; Becker, F.; Rauzi, F.; Vital, S.A.; Taylor, K.A.; Stokes, K.Y.; Emerson, M.; Gavins, F.N.E. The isothiocyanate sulforaphane modulates platelet function and protects against cerebral thrombotic dysfunction. Br. J. Pharmacol. 2018, 175, 3333–3346. [Google Scholar] [CrossRef] [PubMed]
- Subedi, L.; Lee, J.H.; Yumnam, S.; Ji, E.; Kim, S.Y. Anti-Inflammatory Effect of Sulforaphane on LPS-Activated Microglia Potentially through JNK/AP-1/NF-κB Inhibition and Nrf2/HO-1 Activation. Cells 2019, 8, 194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, J.; Jahraus, B.; Balta, E.; Ziegler, J.D.; Hübner, K.; Blank, N.; Niesler, B.; Wabnitz, G.H.; Samstag, Y. Sulforaphane Inhibits Inflammatory Responses of Primary Human T-Cells by Increasing ROS and Depleting Glutathione. Front. Immunol. 2018, 9, 2584. [Google Scholar] [CrossRef] [PubMed]
- Yoo, I.H.; Kim, M.J.; Kim, J.; Sung, J.J.; Park, S.T.; Ahn, S.W. The Anti-Inflammatory Effect of Sulforaphane in Mice with Experimental Autoimmune Encephalomyelitis. J. Korean Med. Sci. 2019, 34, e197. [Google Scholar] [CrossRef]
- Huang, C.S.; Lin, A.H.; Liu, C.T.; Tsai, C.W.; Chang, I.S.; Chen, H.W.; Lii, C.K. Isothiocyanates protect against oxidized LDL-induced endothelial dysfunction by upregulating Nrf2-dependent antioxidation and suppressing NFkappaB activation. Mol. Nutr. Food Res. 2013, 57, 1918–1930. [Google Scholar] [CrossRef]
- Pan, H.; He, M.; Liu, R.; Brecha, N.C.; Yu, A.C.; Pu, M. Sulforaphane protects rodent retinas against ischemia-reperfusion injury through the activation of the Nrf2/HO-1 antioxidant pathway. PLoS ONE 2014, 9, e114186. [Google Scholar] [CrossRef]
- Chi, X.; Zhang, R.; Shen, N.; Jin, Y.; Alina, A.; Yang, S.; Lin, S. Sulforaphane reduces apoptosis and oncosis along with protecting liver injury-induced ischemic reperfusion by activating the Nrf2/ARE pathway. Hepatol. Int. 2015, 9, 321–329. [Google Scholar] [CrossRef]
- Mao, L.; Yang, T.; Li, X.; Lei, X.; Sun, Y.; Zhao, Y.; Zhang, W.; Gao, Y.; Sun, B.; Zhang, F. Protective effects of sulforaphane in experimental vascular cognitive impairment: Contribution of the Nrf2 pathway. J. Cereb. Blood Flow Metab. 2019, 39, 352–366. [Google Scholar] [CrossRef]
- Ping, Z.; Liu, W.; Kang, Z.; Cai, J.; Wang, Q.; Cheng, N.; Wang, S.; Wang, S.; Zhang, J.H.; Sun, X. Sulforaphane protects brains against hypoxic-ischemic injury through induction of Nrf2-dependent phase 2 enzyme. Brain Res. 2010, 1343, 178–185. [Google Scholar] [CrossRef]
- Li, Z.; Galli, U.; Becker, L.E.; Bruns, H.; Nickkolgh, A.; Hoffmann, K.; Karck, M.; Schemmer, P. Sulforaphane protects hearts from early injury after experimental transplantation. Ann. Transplant. 2013, 18, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Cekauskas, A.; Bruns, H.; Manikas, M.; Herr, I.; Gross, M.L.; Zorn, M.; Jankevicius, F.; Strupas, K.; Schemmer, P. Sulforaphane decreases kidney injury after transplantation in rats: Role of mitochondrial damage. Ann. Transplant. 2013, 18, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Wang, X.; Deng, X.; Lasson, A.; Wallén, R.; Hallberg, E.; Andersson, R. The influence of intestinal ischemia and reperfusion on bidirectional intestinal barrier permeability, cellular membrane integrity, proteinase inhibitors, and cell death in rats. Shock 1998, 10, 203–212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurose, I.; Anderson, D.C.; Miyasaka, M.; Tamatani, T.; Paulson, J.C.; Todd, R.F.; Rusche, J.R.; Granger, D.N. Molecular determinants of reperfusion-induced leukocyte adhesion and vascular protein leakage. Circ. Res. 1994, 74, 336–343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooper, D.; Chitman, K.D.; Williams, M.C.; Granger, D.N. Time-dependent platelet-vessel wall interactions induced by intestinal ischemia-reperfusion. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 284, G1027–G1033. [Google Scholar] [CrossRef] [Green Version]
- Massberg, S.; Enders, G.; Matos, F.C.; Tomic, L.I.; Leiderer, R.; Eisenmenger, S.; Messmer, K.; Krombach, F. Fibrinogen deposition at the postischemic vessel wall promotes platelet adhesion during ischemia-reperfusion in vivo. Blood 1999, 94, 3829–3838. [Google Scholar] [CrossRef]
- Massberg, S.; Enders, G.; Leiderer, R.; Eisenmenger, S.; Vestweber, D.; Krombach, F.; Messmer, K. Platelet-endothelial cell interactions during ischemia/reperfusion: The role of P-selectin. Blood 1998, 92, 507–515. [Google Scholar] [CrossRef]
- Da Silva, F.P.; Soriano, F.G. Neutrophils recruitment during sepsis: Critical points and crossroads. Front. Biosci. 2009, 14, 4464–4476. [Google Scholar] [CrossRef]
- Rossaint, J.; Zarbock, A. Tissue-specific neutrophil recruitment into the lung, liver, and kidney. J. Innate Immun. 2013, 5, 348–357. [Google Scholar] [CrossRef]
- Helou, D.G.; Braham, S.; De Chaisemartin, L.; Granger, V.; Damien, M.H.; Pallardy, M.; Kerdine-Römer, S.; Chollet-Martin, S. Nrf2 downregulates zymosan-induced neutrophil activation and modulates migration. PLoS ONE 2019, 14, e0216465. [Google Scholar] [CrossRef] [Green Version]
- Holloway, P.M.; Gillespie, S.; Becker, F.; Vital, S.A.; Nguyen, V.; Alexander, J.S.; Evans, P.C.; Gavins, F.N.E. Sulforaphane induces neurovascular protection against a systemic inflammatory challenge via both Nrf2-dependent and independent pathways. Vascul. Pharmacol. 2016, 85, 29–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pal, S.; Konkimalla, V.B. Sulforaphane regulates phenotypic and functional switching of both induced and spontaneously differentiating human monocytes. Int. Immunopharmacol. 2016, 35, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Geisel, J.; Brück, J.; Glocova, I.; Dengler, K.; Sinnberg, T.; Rothfuss, O.; Walter, M.; Schulze-Osthoff, K.; Röcken, M.; Ghoreschi, K. Sulforaphane protects from T cell-mediated autoimmune disease by inhibition of IL-23 and IL-12 in dendritic cells. J. Immunol. 2014, 192, 3530–3539. [Google Scholar] [CrossRef] [PubMed]
- Foell, D.; Becker, F.; Hadrian, R.; Palmes, D.; Kebschull, L. A practical guide for small bowel transplantation in rats-review of techniques and models. J. Surg. Res. 2017, 213, 115–130. [Google Scholar] [CrossRef]
- Houghton, C.A. Sulforaphane: Its “Coming of Age” as a Clinically Relevant Nutraceutical in the Prevention and Treatment of Chronic Disease. Oxid. Med. Cell. Longev. 2019, 2019, 2716870. [Google Scholar] [CrossRef] [Green Version]
- Becker, F.; Holthoff, C.; Anthoni, C.; Rijcken, E.; Alexander, J.S.; Gavins, F.N.; Spiegel, H.U.; Senninger, N.; Vowinkel, T. Downregulation of CX(3)CR1 ameliorates experimental colitis: Evidence for CX(3)CL1-CX(3)CR1-mediated immune cell recruitment. Int. J. Colorectal. Dis. 2017, 32, 315–324. [Google Scholar] [CrossRef]
- Moolenbeek, C.; Ruitenberg, E.J. The “Swiss roll”: A simple technique for histological studies of the rodent intestine. Lab. Anim. 1981, 15, 57–59. [Google Scholar] [CrossRef]
- Nowacki, T.M.; Remaley, A.T.; Bettenworth, D.; Eisenblätter, M.; Vowinkel, T.; Becker, F.; Vogl, T.; Roth, J.; Tietge, U.J.; Lügering, A.; et al. The 5A apolipoprotein A-I (apoA-I) mimetic peptide ameliorates experimental colitis by regulating monocyte infiltration. Br. J. Pharmacol. 2016, 173, 2780–2792. [Google Scholar] [CrossRef] [Green Version]
- Chiu, C.J.; McArdle, A.H.; Brown, R.; Scott, H.J.; Gurd, F.N. Intestinal mucosal lesion in low-flow states. I. A morphological, hemodynamic, and metabolic reappraisal. Arch. Surg. 1970, 101, 478–483. [Google Scholar] [CrossRef]
- Park, P.O.; Haglund, U.; Bulkley, G.B.; Fält, K. The sequence of development of intestinal tissue injury after strangulation ischemia and reperfusion. Surgery 1990, 107, 574–580. [Google Scholar]
- Becker, F.; Potepalov, S.; Shehzahdi, R.; Bernas, M.; Witte, M.; Abreo, F.; Traylor, J.; Orr, W.A.; Tsunoda, I.; Alexander, J.S. Downregulation of FoxC2 Increased Susceptibility to Experimental Colitis: Influence of Lymphatic Drainage Function? Inflamm. Bowel Dis. 2015, 21, 1282–1296. [Google Scholar] [CrossRef] [PubMed]





© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Z.; Mohr, A.; Heitplatz, B.; Hansen, U.; Pascher, A.; Brockmann, J.G.; Becker, F. Sulforaphane Elicits Protective Effects in Intestinal Ischemia Reperfusion Injury. Int. J. Mol. Sci. 2020, 21, 5189. https://doi.org/10.3390/ijms21155189
Chen Z, Mohr A, Heitplatz B, Hansen U, Pascher A, Brockmann JG, Becker F. Sulforaphane Elicits Protective Effects in Intestinal Ischemia Reperfusion Injury. International Journal of Molecular Sciences. 2020; 21(15):5189. https://doi.org/10.3390/ijms21155189
Chicago/Turabian StyleChen, Zhiquan, Annika Mohr, Barbara Heitplatz, Uwe Hansen, Andreas Pascher, Jens G. Brockmann, and Felix Becker. 2020. "Sulforaphane Elicits Protective Effects in Intestinal Ischemia Reperfusion Injury" International Journal of Molecular Sciences 21, no. 15: 5189. https://doi.org/10.3390/ijms21155189



