Role of Bacterial and Host DNases on Host-Pathogen Interaction during Streptococcus suis Meningitis
Abstract
:1. Introduction
2. Results
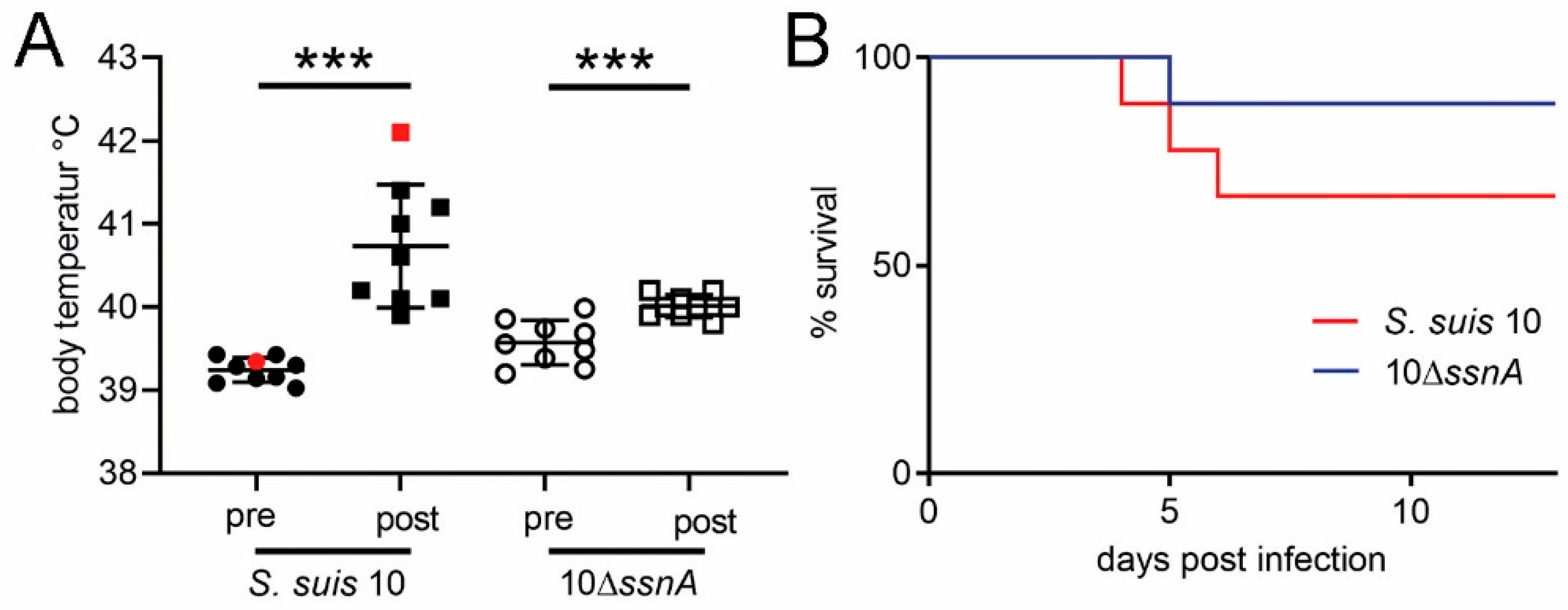
2.1. SsnA Only Slightly Increases S. suis Virulence in Pigs
2.2. NETs Are Formed in CSF and Consist of PR-39 at the Onset of Meningitis
2.3. NET-Markers Are Present in Meninges, but No NET-Fibers Are Detectable in Brain Tissue
2.4. Host DNases Are Active during S. suis Meningitis in Brain Tissue, CSF and Serum
2.5. DNase 1 Has No Impact on Transmigration of Neutrophils through a Choroid Plexus Epithelial Cell Layer
2.6. DNase 1 Partially Improves Killing of S. suis by Neutrophils of Individual Animals in CSF
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Growth Conditions
4.2. Experimental Infection of Piglets
4.3. Survival Experiment
4.4. Early Phase Experiment
4.5. Sample Collection
4.6. Re-Isolation of Infection Strains
4.7. Blood Survival Assay
4.8. Pico Green Quantification Assay
4.9. Histology of the Brain
4.10. Cytospin of CSF
4.11. Staining and Microscopy
4.11.1. Staining of CSF in CellView® Slides
- Staining of NETs and S. suis: CSF was fixed with 4% paraformaldehyde (Science Services, E15710-25, Munich, Germany). Staining was performed as previously described [2] using as first antibodies mouse IgG2a anti-DNA/histone (Millipore MAB3864; 0.55 mg; 1:1000, Billerica, MA, USA) and rabbit IgG anti-PR39 (1:75) or rabbit IgG anti-S. suis [19] (1:500). After washing, the DNA was stained with aqueous Hoechst 33,342 (Stock 50mg/mL, Sigma B-2261, St. Louis, MO, USA) and covered with ProLong™ Gold Antifade Mountant (Invitrogen, P36930, Carlsbad, CA, USA). The samples were examined microscopically on a Leica TCS SP5 AOBS confocal inverted-base fluorescence microscope with a HCX PL APO 40 × 0.75–1.25 oil immersion objective with an Argon, 405 nm and 633 nm laser. The settings were adjusted using isotype control antibodies in separate preparations.
- Staining of NETs and intra- and extracellular S. suis: staining was performed as previously described [2] using first antibodies mouse IgG2a anti-DNA/histone (Millipore MAB3864, Billerica, MA, USA; 0.55 mg; 1:1000) and rabbit IgG anti-S. suis [19] (1:500). After washing the DNA was stained with aqueous Hoechst 33,342 [(Stock 50 mg/mL, Sigma B-2261, St. Louis, MO, USA) and covered with ProLong™ Gold Antifade Mountant (Invitrogen, P36930, Carlsbad, CA, USA). The samples were examined microscopically on a Leica TCS SP5 AOBS confocal inverted-base fluorescence microscope with a HCX PL APO 40 × 0.75–1.25 oil immersion objective with an Argon, 405 nm, 561 nm and 633 nm laser. The settings were adjusted using isotype control antibodies in separate preparations.
4.11.2. Staining of Slides
4.11.3. Staining of 2–4 µm Paraffin Embedded Brain Sections
- Hematoxylin-Eosin (HE) Staining: Automated dying in LEICA ST 4040 with 0.1 % hematoxylin (Roth, Karlsruhe, Germany) and 1 % eosin (Roth, Karlsruhe, Germany).
- Immunofluorescence Staining: The staining was performed as previously described [12] with the following changes. First antibodies mouse IgG2a anti-DNA/histone (Millipore MAB3864, Billerica, MA, USA; 0.55 mg; 1:100) or mouse anti-PR-39 (Lionex, Braunschweig, Germany; 1:100) and rabbit anti-elastase (Abcam, ab1876, Cambridge, UK; 1:50) or rabbit IgG anti-S. suis [19] (1:500) or rabbit anti-DNase 1 (Invitrogen; PA5-22017, Carlsbad, CA, USA; 1:100) dissolved in blocking buffer were incubated overnight 4 °C, while gently shaking. The samples were examined microscopically on a Leica TCS SP5 AOBS confocal inverted-base fluorescence microscope with HCX PL APO 40 × 0.75–1.25 and HCX PL APO lambda blue 63 × 1.40 oil immersion objectives with an Argon, 405 nm and 633 nm laser. The settings were adjusted using isotype control antibodies in separate preparations.
4.12. DNase Activity Test
4.13. DNase 1, PR-39, PMAP-23, PMAP-37 ELISA
4.14. Cell Culture
- Cell Culture Model of the Porcine Blood–CSF Barrier (Figure 5A): After three days’ incubation, transepithelial electrical resistance (TEER) was measured with a Millicell® ERS-2 (Millipore, Billerica, MA, USA) voltmeter to control the barrier density. Some filters were infected with S. suis 10 for one hour in the blood compartment. Before adding the freshly isolated porcine neutrophils, medium in the blood compartment was changed in all filters. Transmigration of neutrophils was performed for 4 h at 37 °C and 5% CO2. Serial dilutions of transmigrated bacteria were plated on blood agar (Columbia Agar with 7% Sheep Blood; Thermo Scientific™ PB5008A, Waltham, MA, USA) to determine the CFU/mL and 250 µL medium of the CSF compartment was fixed with a final concentration of 4% PFA (Paraformaldehyde 16% Science Services E15710, Munich, Germany) for flow cytometric analysis. TEER was measured again. The experiment was repeated three times on independent days.
- Cell Culture Model of the Porcine Blood–CSF Barrier Mimicking Physiological Conditions (Figure 6): Filters were transferred in new wells with 990 µL pooled CSF of healthy piglets (euthanasia of these pigs was approved and registered by the local Animal Welfare Officer in accordance with the German Animal Welfare Law under number TiHo-T-2019-14, last update 07/2019). To attract neutrophils, 50 ng interleukin 8 (IL8) (Recombinant Porcine IL-8/CXCL8, R&D Systems 535-IN-025, Minneapolis, MN, USA) was added to all wells. To half of the wells, 20 U DNase 1 (Serva 18535.02) was added. CSF of half of the wells was infected with 1–2 × 102 CFU S. suis 10. Thus ultimately yielded: non-infected wells, non-infected wells with DNase 1, infected wells and infected wells with DNase 1. In the upper compartment, 400 µL heparinized whole pig blood (animal permit registered at the Lower Saxonian State Office for Consumer Protection and Food Safety (Niedersächsisches Landesamt für Verbraucherschutz und Lebensmittelsicherheit) under no. 33.9-42502-05-18A302, last update 07/2018) was added and incubated at 37 °C and 5% CO2 for 4 h. After incubation, serial dilutions of CSF were plated on blood agar plates (Columbia Agar with 7% Sheep Blood; Thermo Scientific™ PB5008A, Waltham, MA, USA) to determine the CFU/mL, and 250 µL CSF was fixed with a final concentration of 4% PFA (Paraformaldehyde 16% Science Services E15710, Munich, Germany) for flow cytometric analysis. TEER was measured again. The experiment was repeated five times on independent days. CSF was collected from healthy euthanized pigs. The euthanasia of these pigs was approved and registered by the local Animal Welfare Officer in accordance with the German Animal Welfare Law under number TiHo-T-2019-14.
4.15. Flow Cytometry
4.16. Neutrophil Isolation
4.17. Neutrophil Killing Assay
4.18. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hersi, K.; Gonzalez, F.J.; Kondamudi, N.P. Meningitis. In StatPearls [Internet]; [Updated 2020 May 23]; StatPearls Publishing: Treasure Island, FL, USA, January 2020. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459360/ (accessed on 25 July 2020).
- De Buhr, N.; Reuner, F.; Neumann, A.; Stump-Guthier, C.; Tenenbaum, T.; Schroten, H.; Ishikawa, H.; Müller, K.; Beineke, A.; Hennig-Pauka, I.; et al. Neutrophil extracellular trap formation in theStreptococcus suis-infected cerebrospinal fluid compartment. Cell. Microbiol. 2016, 19, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Goyette-Desjardins, G.; Auger, J.-P.; Xu, J.; Segura, M.; Gottschalk, M. Streptococcus suis, an important pig pathogen and emerging zoonotic agent-an update on the worldwide distribution based on serotyping and sequence typing. Emerg. Microbes Infect. 2014, 3, e45. [Google Scholar] [CrossRef]
- Tunkel, A.R.; Hartman, B.J.; Kaplan, S.L.; Kaufman, B.A.; Roos, K.L.; Scheld, W.M.; Whitley, R.J. Practice Guidelines for the Management of Bacterial Meningitis. Clin. Infect. Dis. 2004, 39, 1267–1284. [Google Scholar] [CrossRef]
- Brinkmann, V. Neutrophil Extracellular Traps Kill Bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Meng, W.; Paunel-Görgülü, A.; Flohé, S.; Witte, I.; Schädel-Höpfner, M.; Windolf, J.; Lögters, T.T. Deoxyribonuclease Is a Potential Counter Regulator of Aberrant Neutrophil Extracellular Traps Formation after Major Trauma. Mediat. Inflamm. 2012, 2012, 149560. [Google Scholar] [CrossRef] [Green Version]
- Jiménez-Alcázar, M.; Rangaswamy, C.; Panda, R.; Bitterling, J.; Simsek, Y.J.; Long, A.T.; Bilyy, R.; Krenn, V.; Renné, C.; Renné, T.; et al. Host DNases prevent vascular occlusion by neutrophil extracellular traps. Science 2017, 358, 1202–1206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hakkim, A.; Fürnrohr, B.G.; Amann, K.; Laube, B.; Abu Abed, U.; Brinkmann, V.; Herrmann, M.; Voll, R.E.; Zychlinsky, A. Impairment of neutrophil extracellular trap degradation is associated with lupus nephritis. Proc. Natl. Acad. Sci. USA 2010, 107, 9813–9818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Buhr, N.; Valentin-Weigand, P.; Neumann, A.; Von Köckritz-Blickwede, M.; Naim, H.Y.; Stehr, M.; Baums, C.G. Identification of a novel DNase of Streptococcus suis (EndAsuis) important for neutrophil extracellular trap degradation during exponential growth. Microbiology 2015, 161, 838–850. [Google Scholar] [CrossRef] [Green Version]
- De Buhr, N.; Neumann, A.; Jerjomiceva, N.; Von Köckritz-Blickwede, M.; Baums, C.G. Streptococcus suis DNase SsnA contributes to degradation of neutrophil extracellular traps (NETs) and evasion of NET-mediated antimicrobial activity. Microbiology 2014, 160, 385–395. [Google Scholar] [CrossRef] [Green Version]
- Mohanty, T.; Fisher, J.; Bakochi, A.; Neumann, A.; Cardoso, J.F.P.; Karlsson, C.A.Q.; Pavan, C.; Lundgaard, I.; Nilson, B.; Reinstrup, P.; et al. Neutrophil extracellular traps in the central nervous system hinder bacterial clearance during pneumococcal meningitis. Nat. Commun. 2019, 10, 1667. [Google Scholar] [CrossRef]
- De Buhr, N.; Bonilla, M.C.; Pfeiffer, J.; Akhdar, S.; Schwennen, C.; Kahl, B.C.; Waldmann, K.-H.; Valentin-Weigand, P.; Hennig-Pauka, I.; Von Köckritz-Blickwede, M. Degraded neutrophil extracellular traps promote the growth of Actinobacillus pleuropneumoniae. Cell Death Dis. 2019, 10, 657–716. [Google Scholar] [CrossRef] [PubMed]
- Baums, C.G.; Kaim, U.; Fulde, M.; Ramachandran, G.; Goethe, R.; Valentin-Weigand, P. Identification of a Novel Virulence Determinant with Serum Opacification Activity in Streptococcus suis. Infect. Immun. 2006, 74, 6154–6162. [Google Scholar] [CrossRef] [Green Version]
- Seele, J.; Hillermann, L.-M.; Beineke, A.; Seitz, M.; Von Pawel-Rammingen, U.; Valentin-Weigand, P.; Baums, C.G. The immunoglobulin M-degrading enzyme of Streptococcus suis, Ide Ssuis, is a highly protective antigen against serotype 2. Vaccine 2015, 33, 2207–2212. [Google Scholar] [CrossRef] [PubMed]
- De Buhr, N.; Von Köckritz-Blickwede, M. How Neutrophil Extracellular Traps Become Visible. J. Immunol. Res. 2016, 2016, 4604713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosoda, H.; Nakamura, K.; Hu, Z.; Tamura, H.; Reich, J.; Kuwahara-Arai, K.; Iba, T.; Tabe, Y.; Nagaoaka, I. Antimicrobial cathelicidin peptide LL-37 induces NET formation and suppresses the inflammatory response in a mouse septic model. Mol. Med. Rep. 2017, 16, 5618–5626. [Google Scholar] [CrossRef] [PubMed]
- Neumann, A.; Völlger, L.; Berends, E.T.; Molhoek, E.M.; Stapels, D.A.; Midon, M.; Friães, A.; Pingoud, A.; Rooijakkers, S.H.; Gallo, R.L.; et al. Novel Role of the Antimicrobial Peptide LL-37 in the Protection of Neutrophil Extracellular Traps against Degradation by Bacterial Nucleases. J. Innate Immun. 2014, 6, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, M.; Segura, M. The pathogenesis of the meningitis caused by Streptococcus suis: The unresolved questions. Vet. Microbiol. 2000, 76, 259–272. [Google Scholar] [CrossRef]
- Beineke, A.; Bennecke, K.; Neis, C.; Schröder, C.; Waldmann, K.-H.; Baumgärtner, W.; Valentin-Weigand, P.; Baums, C. Comparative evaluation of virulence and pathology of Streptococcus suis serotypes 2 and 9 in experimentally infected growers. Vet. Microbiol. 2008, 128, 423–430. [Google Scholar] [CrossRef]
- Lun, S.; Perez-Casal, J.; Connor, W.; Willson, P.J. Role of suilysin in pathogenesis of Streptococcus suis capsular serotype 2. Microb. Pathog. 2003, 34, 27–37. [Google Scholar] [CrossRef]
- Williams, A.E.; Blakemore, W.F. Pathogenesis of Meningitis Caused by Streptococcus suis Type 2. J. Infect. Dis. 1990, 162, 474–481. [Google Scholar] [CrossRef]
- Madsen, L.W.; Svensmark, B.; Elvestad, K.; Aalbaek, B.; Jensen, H.E. Streptococcus suis Serotype 2 Infection in Pigs: New Diagnostic and Pathogenetic Aspects. J. Comp. Pathol. 2002, 126, 57–65. [Google Scholar] [CrossRef]
- Lauer, A.N.; März, M.; Meyer, S.; Meurer, M.; De Buhr, N.; Borkowski, J.; Weiß, C.; Schroten, H.; Schwerk, C. Optimized cultivation of porcine choroid plexus epithelial cells, a blood-cerebrospinal fluid barrier model, for studying granulocyte transmigration. Lab. Investig. 2019, 99, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.E.; Damman, M.; Van Der Velde, J.; Wagenaar, F.; Wisselink, H.J.; Stockhofe-Zurwieden, N.; Smits, M.A. Identification and Characterization of the cps Locus of Streptococcus suis Serotype 2: The Capsule Protects against Phagocytosis and Is an Important Virulence Factor. Infect. Immun. 1999, 67, 1750–1756. [Google Scholar] [CrossRef]
- Zhao, J.; Pan, S.; Lin, L.; Fu, L.; Yang, C.; Xu, Z.; Wei, Y.; Jin, M.; Zhang, A. Streptococcus suis serotype 2 strains can induce the formation of neutrophil extracellular traps and evade trapping. FEMS Microbiol. Lett. 2015, 362, fnv022. [Google Scholar] [CrossRef] [PubMed]
- Bickle, T.A.; Krüger, D.H. Biology of DNA restriction. Microbiol. Rev. 1993, 57, 434–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loll, B.; Gebhardt, M.; Wahle, E.; Meinhart, A. Crystal structure of the EndoG/EndoGI complex: Mechanism of EndoG inhibition. Nucleic Acids Res. 2009, 37, 7312–7320. [Google Scholar] [CrossRef] [Green Version]
- Fontaine, M.C.; Perez-Casal, J.; Willson, P.J. Investigation of a Novel DNase of Streptococcus suis Serotype 2. Infect. Immun. 2004, 72, 774–781. [Google Scholar] [CrossRef] [Green Version]
- Berends, E.T.M.; Horswill, A.R.; Haste, N.M.; Monestier, M.; Nizet, V.; Von Köckritz-Blickwede, M. Nuclease expression by Staphylococcus aureus facilitates escape from neutrophil extracellular traps. J. Innate Immun. 2010, 2, 576–586. [Google Scholar] [CrossRef] [Green Version]
- Weiner, D.J.; Bucki, R.; Janmey, P.A. The Antimicrobial Activity of the Cathelicidin LL37 Is Inhibited by F-actin Bundles and Restored by Gelsolin. Am. J. Respir. Cell Mol. Biol. 2003, 28, 738–745. [Google Scholar] [CrossRef]
- Allen, C.; Thornton, P.; Denes, A.; McColl, B.W.; Pierozynski, A.; Monestier, M.; Pinteaux, E.; Rothwell, N.J.; Allan, S.M. Neutrophil cerebrovascular transmigration triggers rapid neurotoxicity through release of proteases associated with decondensed DNA. J. Immunol. 2012, 189, 381–392. [Google Scholar] [CrossRef]
- Lazzaretto, B.; Fadeel, B. Intra- and Extracellular Degradation of Neutrophil Extracellular Traps by Macrophages and Dendritic Cells. J. Immunol. 2019, 203, 2276–2290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwerk, C.; Adam, R.; Borkowski, J.; Schneider, H.; Klenk, M.; Zink, S.; Quednau, N.; Schmidt, N.; Stump, C.; Sagar, A.; et al. In vitro transcriptome analysis of porcine choroid plexus epithelial cells in response to Streptococcus suis: Release of pro-inflammatory cytokines and chemokines. Microbes Infect. 2011, 13, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Ostergaard, C.; Benfield, T.L.; Sellebjerg, F.; Kronborg, G.; Lohse, N.; Lundgren, J.D. Interleukin-8 in cerebrospinal fluid from patients with septic and aseptic meningitis. Eur. J. Clin. Microbiol. Infect. Dis. 1996, 15, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Prasad, R.; Kapoor, R.; Srivastava, R.K.; Mishra, O.P.; Singh, T.B. Cerebrospinal Fluid TNF-?, IL-6, and IL-8 in Children With Bacterial Meningitis. Pediatr. Neurol. 2014, 50, 60–65. [Google Scholar] [CrossRef]
- Sambrook, M.A. The relationship between cerebrospinal fluid and plasma electrolytes in patients with meningitis. J. Neurol. Sci. 1974, 23, 265–273. [Google Scholar] [CrossRef]
- Barichello, T.; Fagundes, G.D.; Generoso, J.S.; Moreira, A.P.; Costa, C.S.; Zanatta, J.R.; Simões, L.R.; Petronilho, F.; Dal-Pizzol, F.; Vilela, M.C.; et al. Brain–blood barrier breakdown and pro-inflammatory mediators in neonate rats submitted meningitis by Streptococcus pneumoniae. Brain Res. 2012, 1471, 162–168. [Google Scholar] [CrossRef] [Green Version]
- Gaipl, U.S.; Beyer, T.D.; Heyder, P.; Kuenkele, S.; Böttcher, A.; Voll, R.E.; Kalden, J.R.; Herrmann, M. Cooperation between C1q and DNase I in the clearance of necrotic cell-derived chromatin. Arthritis Rheum. 2004, 50, 640–649. [Google Scholar] [CrossRef]
- Mook-Kanamori, B.B.; Brouwer, M.C.; Geldhoff, M.; Van Der Ende, A.; Van De Beek, D. Cerebrospinal fluid complement activation in patients with pneumococcal and meningococcal meningitis. J. Infect. 2014, 68, 542–547. [Google Scholar] [CrossRef]
- Hitchcock, S.E. Actin Deoxyribonuclease I Interaction. J. Biol. Chem. 1980, 255, 5668–5673. [Google Scholar]
- Papayannopoulos, V.; Staab, D.; Zychlinsky, A. Neutrophil Elastase Enhances Sputum Solubilization in Cystic Fibrosis Patients Receiving DNase Therapy. PLoS ONE 2011, 6, e28526. [Google Scholar] [CrossRef]
- Berge, M.T.; Van Der Wiel, E.; Tiddens, H.A.; Merkus, P.J.; Hop, W.C.; De Jongste, J.C. DNase in stable cystic fibrosis infants: A pilot study. J. Cyst. Fibros. 2003, 2, 183–188. [Google Scholar] [CrossRef] [Green Version]
- Frederiksen, B.; Pressler, T.; Hansen, A.; Koch, C.; Høiby, N. Effect of aerosolized rhDNase (Pulmozyme®) on pulmonary colonization in patients with cystic fibrosis. Acta Paediatrica 2006, 95, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Yipp, B.G.; Petri, B.; Salina, D.; Jenne, C.N.; Scott, B.N.V.; Zbytnuik, L.D.; Pittman, K.; Asaduzzaman, M.; Wu, K.; Meijndert, H.C.; et al. Infection-induced NETosis is a dynamic process involving neutrophil multitasking in vivo. Nat. Med. 2012, 18, 1386–1393. [Google Scholar] [CrossRef] [Green Version]
- Boeltz, S.; Amini, P.; Anders, H.-J.; Andrade, F.; Bilyy, R.; Chatfield, S.; Cichon, I.; Clancy, D.M.; Desai, J.; Dumych, T.; et al. To NET or not to NET:current opinions and state of the science regarding the formation of neutrophil extracellular traps. Cell Death Differ. 2019, 26, 395–408. [Google Scholar] [CrossRef] [Green Version]
- Baums, C.G.; Kock, C.; Beineke, A.; Bennecke, K.; Goethe, R.; Schröder, C.; Waldmann, K.-H.; Valentin-Weigand, P. Streptococcus suis Bacterin and Subunit Vaccine Immunogenicities and Protective Efficacies against Serotypes 2 and 9. Clin. Vaccine Immunol. 2008, 16, 200–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vecht, U.; Wisselink, H.J.; Stockhofe-Zurwieden, N.; Smith, H.E. Characterization of virulence of the Sreptococcus suis serotype 2 reference strain Henrichsen S 735 in newborn gnotobiotic pigs. Vet. Microbiol. 1996, 51, 125–136. [Google Scholar] [CrossRef]
- Romagnoli, N.; Ventrella, D.; Giunti, M.; Dondi, F.; Sorrentino, N.C.; Fraldi, A.; Surace, E.M.; Lavitrano, M. Access to cerebrospinal fluid in piglets via the cisterna magna: Optimization and description of the technique. Lab. Anim. 2014, 48, 345–348. [Google Scholar] [CrossRef] [Green Version]
- Silva, L.; Baums, C.G.; Rehm, T.; Wisselink, H.J.; Goethe, R.; Valentinweigand, P. Virulence-associated gene profiling of Streptococcus suis isolates by PCR. Vet. Microbiol. 2006, 115, 117–127. [Google Scholar] [CrossRef]
- Schroten, M.; Hanisch, F.-G.; Quednau, N.; Stump, C.; Riebe, R.; Lenk, M.; Wolburg, H.; Tenenbaum, T.; Schwerk, C. A Novel Porcine In Vitro Model of the Blood-Cerebrospinal Fluid Barrier with Strong Barrier Function. PLoS ONE 2012, 7, e39835. [Google Scholar] [CrossRef] [Green Version]







© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meurer, M.; Öhlmann, S.; Bonilla, M.C.; Valentin-Weigand, P.; Beineke, A.; Hennig-Pauka, I.; Schwerk, C.; Schroten, H.; Baums, C.G.; Köckritz-Blickwede, M.v.; et al. Role of Bacterial and Host DNases on Host-Pathogen Interaction during Streptococcus suis Meningitis. Int. J. Mol. Sci. 2020, 21, 5289. https://doi.org/10.3390/ijms21155289
Meurer M, Öhlmann S, Bonilla MC, Valentin-Weigand P, Beineke A, Hennig-Pauka I, Schwerk C, Schroten H, Baums CG, Köckritz-Blickwede Mv, et al. Role of Bacterial and Host DNases on Host-Pathogen Interaction during Streptococcus suis Meningitis. International Journal of Molecular Sciences. 2020; 21(15):5289. https://doi.org/10.3390/ijms21155289
Chicago/Turabian StyleMeurer, Marita, Sophie Öhlmann, Marta C. Bonilla, Peter Valentin-Weigand, Andreas Beineke, Isabel Hennig-Pauka, Christian Schwerk, Horst Schroten, Christoph G. Baums, Maren von Köckritz-Blickwede, and et al. 2020. "Role of Bacterial and Host DNases on Host-Pathogen Interaction during Streptococcus suis Meningitis" International Journal of Molecular Sciences 21, no. 15: 5289. https://doi.org/10.3390/ijms21155289





