The Evolution of Duplicated Genes of the Cpi-17/Phi-1 (ppp1r14) Family of Protein Phosphatase 1 Inhibitors in Teleosts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification of the Zebrafish ppp1r14 Gene Family
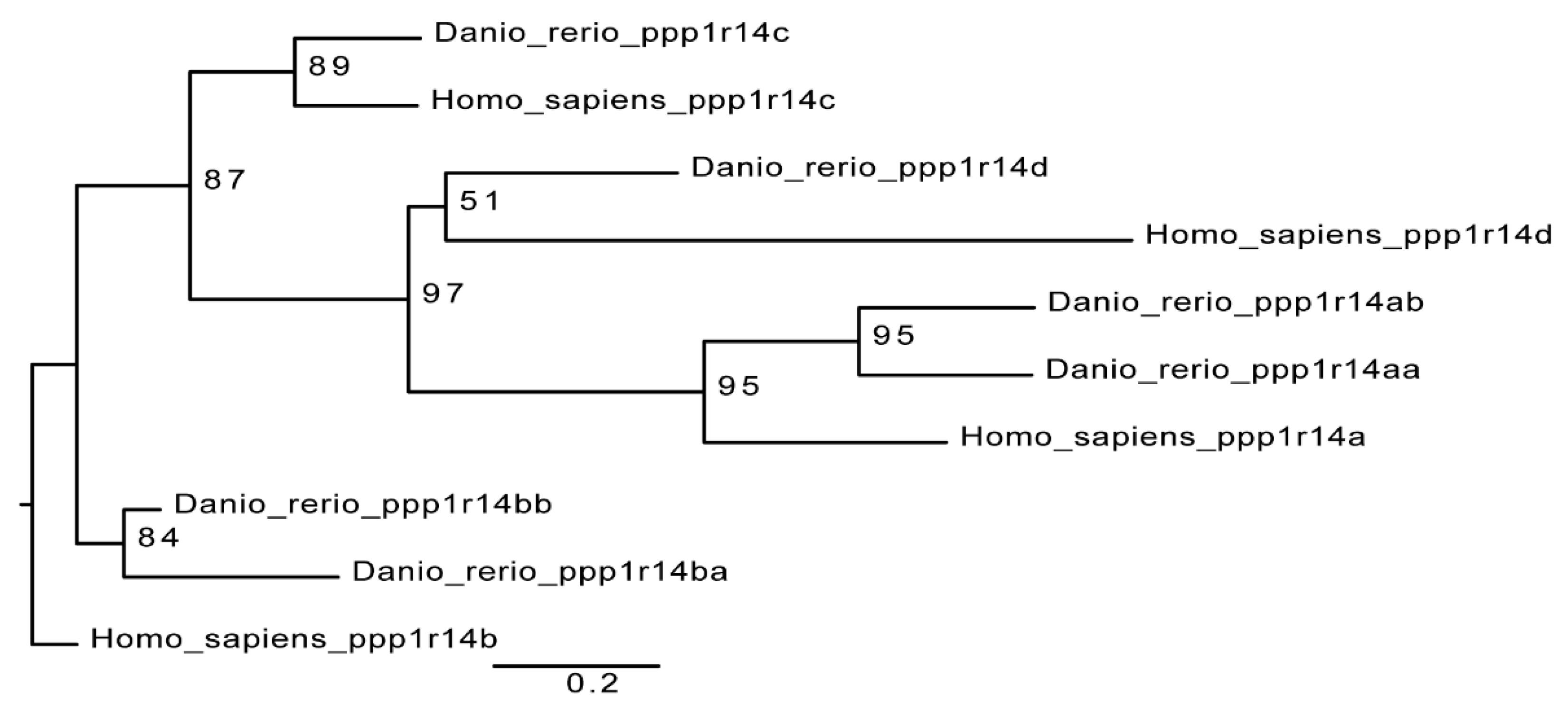
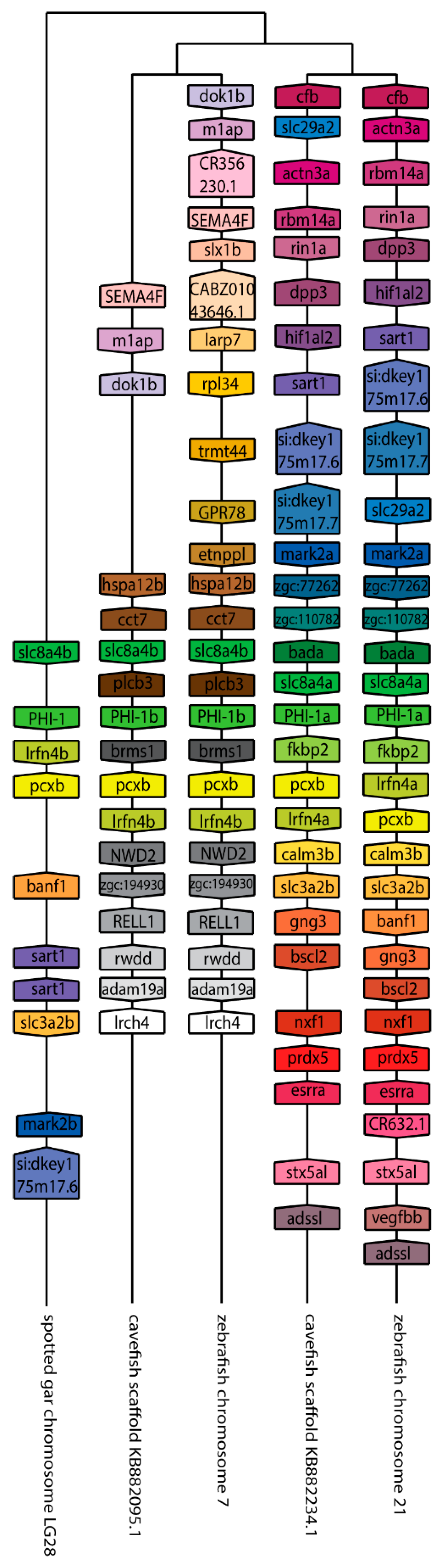
2.2. Synteny and Phylogenetic Analysis of Duplicated ppp1r14 Genes
2.3. Expression Patterns of Cpi-17 and Phi-1 Paralogs During Early Zebrafish Development
2.4. Inhibition of the Myosin Phosphatase by Cpi-17 Family Members
2.5. The Highly Conserved PHIN Domain of Cpi-17 is Responsible for High Affinity Myosin Phosphatase Inhibition
2.6. Expression of Cpi-17 Paralogs, But Not Phi-1 Paralogs, Rescue Knockdown of ppp1r14ab in Zebrafish Embryos
3. Materials and Methods
3.1. Animal Care
3.2. Cell Culture
3.3. Cloning ppp1r14 Family Members and Other Plasmids
3.4. Morpholinos and Zebrafish Injection
3.5. In Situ Hybridization
3.6. Quantitative Reverse Transcriptase PCR
3.7. Antibodies and Western Blots
3.8. Stress Fiber Assays
3.9. Phosphatase Assays
3.10. Homology and Phylogenetic Analysis
3.11. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PP1 | Protein phosphatase 1 |
| Mlc2 | Myosin light chain 2 |
| Mypt1 | Myosin phosphatase targeting subunit 1 |
| Cpi-17 | C-kinase-activated PP1 inhibitor of 17kDa—ppp1r14a |
| Phi-1 | PP1 holoenzyme inhibitor—ppp1r14b |
| KEPI | Kinase-C-enhanced PP1 inhibitor |
| GBPI | Gastro-intestinal and brain specific PP1 inhibitor |
| PHIN | PP1 holoenzyme inhibitory |
| CE | Convergent extension |
| qPCR | Quantitative polymerase chain reaction |
| MO | Morpholino antisense oligonucleotide |
| PKC | Protein kinase C |
References
- Ravi, V.; Venkatesh, B. The Divergent Genomes of Teleosts. Annu. Rev. Anim. Biosci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hoegg, S.; Brinkmann, H.; Taylor, J.S.; Meyer, A. Phylogenetic timing of the fish-specific genome duplication correlates with the diversification of teleost fish. J. Mol. Evol. 2004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amores, A.; Catchen, J.; Ferrara, A.; Fontenot, Q.; Postlethwait, J.H. Genome evolution and meiotic maps by massively parallel DNA sequencing: Spotted gar, an outgroup for the teleost genome duplication. Genetics 2011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, A.; Schartl, M. Gene and genome duplications in vertebrates: The one-to-four (-to-eight in fish) rule and the evolution of novel gene functions. Curr. Opin. Cell Biol. 1999, 11, 699–704. [Google Scholar] [CrossRef] [Green Version]
- Meyer, A.; Van De Peer, Y. From 2R to 3R: Evidence for a fish-specific genome duplication (FSGD). BioEssays 2005, 27, 937–945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van De Peer, Y.; Maere, S.; Meyer, A. The evolutionary significance of ancient genome duplications. Nat. Rev. Genet. 2009, 10, 725–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berthelot, C.; Brunet, F.; Chalopin, D.; Juanchich, A.; Bernard, M.; Noël, B.; Bento, P.; Da Silva, C.; Labadie, K.; Alberti, A.; et al. The rainbow trout genome provides novel insights into evolution after whole-genome duplication in vertebrates. Nat. Commun. 2014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macqueen, D.J.; Johnston, I.A. A well-constrained estimate for the timing of the salmonid whole genome duplication reveals major decoupling from species diversification. Proc. R. Soc. B Biol. Sci. 2014. [Google Scholar] [CrossRef] [Green Version]
- Ohno, S. Evolution by Gene Duplication; Springer: Berlin/Heidelberg, Germany, 1970. [Google Scholar]
- Force, A.; Lynch, M.; Pickett, F.B.; Amores, A.; Yan, Y.L.; Postlethwait, J. Preservation of duplicate genes by complementary, degenerative mutations. Genetics 1999, 151, 1531–1545. [Google Scholar]
- Force, A.; Force, A.; Lynch, M.; Lynch, M.; Postlethwait, J.; Postlethwait, J. Preservation of duplicate genes by subfunctionalization. Am. Zool. 1999. [Google Scholar]
- Eto, M. Regulation of cellular protein phosphatase-1 (PP1) by phosphorylation of the CPI-17 family, C-kinase-activated PP1 Inhibitors. J. Biol. Chem. 2009, 284, 35273–35277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Virshup, D.M.; Shenolikar, S. From Promiscuity to Precision: Protein Phosphatases Get a Makeover. Mol. Cell 2009, 33, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Ceulemans, H.; Bollen, M. Functional Diversity of Protein Phosphatase-1, a Cellular Economizer and Reset Button. Physiol. Rev. 2004, 84, 1–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bollen, M.; Peti, W.; Ragusa, M.J.; Beullens, M. The extended PP1 toolkit: Designed to create specificity. Trends Biochem. Sci. 2010, 35, 450–458. [Google Scholar] [CrossRef] [Green Version]
- Bertolotti, A. The split protein phosphatase system. Biochem. J. 2018, 475, 3707–3723. [Google Scholar] [CrossRef] [Green Version]
- Eto, M.; Elliott, E.; Prickett, T.D.; Brautigan, D.L. Inhibitor-2 regulates protein phosphatase-1 complexed with NimA-related kinase to induce centrosome separation. J. Biol. Chem. 2002. [Google Scholar] [CrossRef] [Green Version]
- Terry-Lorenzo, R.T.; Elliot, E.; Weiser, D.C.; Prickett, T.D.; Brautigan, D.L.; Shenolikar, S. Neurabins recruit protein phosphatase-1 and inhibitor-2 to the actin cytoskeleton. J. Biol. Chem. 2002, 277. [Google Scholar] [CrossRef] [Green Version]
- Connor, J.H.; Weiser, D.C.; Li, S.; Hallenbeck, J.M.; Shenolikar, S. Growth arrest and DNA damage-inducible protein GADD34 assembles a novel signaling complex containing protein phosphatase 1 and inhibitor 1. Mol. Cell. Biol. 2001, 21. [Google Scholar] [CrossRef] [Green Version]
- Eto, M.; Kitazawa, T.; Brautigan, D.L. Phosphoprotein inhibitor CPI-17 specificity depends on allosteric regulation of protein phosphathase-1 by regulatory subunits. Proc. Natl. Acad. Sci. USA 2004. [Google Scholar] [CrossRef] [Green Version]
- Filter, J.J.; Williams, B.C.; Eto, M.; Shalloway, D.; Goldberg, M.L. Unfair competition governs the interaction of pCPI-17 with myosin phosphatase (PP1-MYPT1). Elife 2017. [Google Scholar] [CrossRef] [Green Version]
- Ito, M.; Nakano, T.; Erdodi, F.; Hartshorne, D.J. Myosin phosphatase: Structure, regulation and function. Mol. Cell. Biochem. 2004, 259, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Grassie, M.E.; Moffat, L.D.; Walsh, M.P.; MacDonald, J.A. The myosin phosphatase targeting protein (MYPT) family: A regulated mechanism for achieving substrate specificity of the catalytic subunit of protein phosphatase type 1δ. Arch. Biochem. Biophys. 2011, 510, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Kiss, A.; Erdődi, F.; Lontay, B. Myosin phosphatase: Unexpected functions of a long-known enzyme. Biochim. Biophys. Acta-Mol. Cell Res. 2019, 1866, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Hirano, K.; Derkach, D.N.; Hirano, M.; Nishimura, J.; Kanaide, H. Protein kinase network in the regulation of phosphorylation and dephosphorylation of smooth muscle myosin light chain. Mol. Cell. Biochem. 2003, 248, 105–114. [Google Scholar] [CrossRef]
- Landsverk, M.L.; Epstein, H.F. Genetic analysis of myosin II assembly and organization in model organisms. Cell. Mol. Life Sci. 2005, 62, 2270–2282. [Google Scholar] [CrossRef]
- Eto, M.; Ohmori, T.; Suzuki, M.; Furuya, K.; Morita, F. A novel protein phosphatase-1 inhibitory protein potentiated by protein kinase C. isolation from porcine aorta media and characterization. J. Biochem. 1995. [Google Scholar] [CrossRef]
- Deng, J.T.; Sutherland, C.; Brautigan, D.L.; Eto, M.; Walsh, M.P. Phosphorylation of the myosin phosphatase inhibitors, CPI-17 and PHI-1, by integrin-linked kinase. Biochem. J. 2002. [Google Scholar] [CrossRef] [Green Version]
- Kitazawa, T.; Eto, M.; Woodsome, T.P.; Brautigan, D.L. Agonists trigger G protein-mediated activation of the CPI-17 inhibitor phosphoprotein of myosin light chain phosphatase to enhance vascular smooth muscle contractility. J. Biol. Chem. 2000. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Mahavadi, S.; Sriwai, W.; Hu, W.; Murthy, K.S. Gi-coupled receptors mediate phosphorylation of CPI-17 and MLC20 via preferential activation of the PI3K/ILK pathway. Biochem. J. 2006. [Google Scholar] [CrossRef] [Green Version]
- Eto, M.; Senba, S.; Morita, F.; Yazawa, M. Molecular cloning of a novel phosphorylation-dependent inhibitory protein of protein phosphatase-1 (CPI17) in smooth muscle: Its specific localization in smooth muscle. FEBS Lett. 1997. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.; Ruan, H.; Aw, M.Y.; Hussain, A.; Guo, L.; Gao, C.; Qian, F.; Leung, T.; Song, H.; Kimelman, D.; et al. Mypt1-mediated spatial positioning of Bmp2-producing cells is essential for liver organogenesis. Development 2008, 135, 3209–3218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bremer, J.; Granato, M. Myosin phosphatase Fine-tunes Zebrafish Motoneuron Position during Axonogenesis. PLoS Genet. 2016, 12. [Google Scholar] [CrossRef] [PubMed]
- Gutzman, J.H.; Sive, H. Epithelial relaxation mediated by the myosin phosphatase regulator Mypt1 is required for brain ventricle lumen expansion and hindbrain morphogenesis. Development 2010, 137, 795–804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, G.; Huang, Y.; Ding, H.; Luo, L.; Zhang, Y.; Huang, H.; Ruan, H. Mypt1 regulates Bmp signaling to promote embryonic exocrine pancreas growth in zebrafish. Genesis 2020. [Google Scholar] [CrossRef]
- Weiser, D.C.; Row, R.H.; Kimelman, D. Rho-regulated Myosin phosphatase establishes the level of protrusive activity required for cell movements during zebrafish gastrulation. Development 2009, 136. [Google Scholar] [CrossRef] [Green Version]
- Diz-Muñoz, A.; Krieg, M.; Bergert, M.; Ibarlucea-Benitez, I.; Muller, D.J.; Paluch, E.; Heisenberg, C.P. Control of directed cell migration in vivo by membrane-to-cortex attachment. PLoS Biol. 2010, 8. [Google Scholar] [CrossRef]
- Keller, R. Shaping the vertebrate body plan by polarized embryonic cell movements. Science (80-.). 2002, 298, 1950–1954. [Google Scholar] [CrossRef]
- Tada, M.; Heisenberg, C.-P. Convergent extension: Using collective cell migration and cell intercalation to shape embryos. Development 2012, 139, 3897–3904. [Google Scholar] [CrossRef] [Green Version]
- Williams, M.L.K.; Solnica-Krezel, L. Cellular and molecular mechanisms of convergence and extension in zebrafish. Curr. Top. Dev. Biol. 2020, 136, 377–407. [Google Scholar]
- Marlow, F.; Topczewski, J.; Sepich, D.; Solnica-Krezel, L. Zebrafish Rho kinase 2 acts downstream of Wnt11 to mediate cell polarity and effective convergence and extension movements. Curr. Biol. 2002, 12, 876–884. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.R.; Zhang, P.W.; Zhen, Q.; Walther, D.; Wang, X.B.; Uhl, G.R. KEPI, a PKC-dependent protein phosphatase 1 inhibitor regulated by morphine. J. Biol. Chem. 2002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Q.R.; Zhang, P.W.; Lin, Z.; Li, Q.F.; Woods, A.S.; Troncoso, J.; Uhl, G.R. GBPI, a novel gastrointestinal- and brain-specific PP1-inhibitory protein, is activated by PKC and inactivated by PKA. Biochem. J. 2004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woodsome, T.P.; Eto, M.; Everett, A.; Brautigan, D.L.; Kitazawa, T. Expression of CPI-17 and myosin phosphatase correlates with Ca2+ sensitivity of protein kinase C-induced contraction in rabbit smooth muscle. J. Physiol. 2001. [Google Scholar] [CrossRef] [PubMed]
- Eto, M.; Bock, R.; Brautigan, D.L.; Linden, D.J. Cerebellar long-term synaptic depression requires PKC-mediated activation of CPI-17, a myosin/moesin phosphatase inhibitor. Neuron 2002. [Google Scholar] [CrossRef] [Green Version]
- Eto, M.; Karginov, A.; Brautigan, D.L. A novel phosphoprotein inhibitor of protein type-1 phosphatase holoenzymes. Biochemistry 1999. [Google Scholar] [CrossRef]
- Georgijevic, S.; Subramanian, Y.; Rollins, E.L.; Starovic-Subota, O.; Tang, A.C.Y.; Childs, S.J. Spatiotemporal expression of smooth muscle markers in developing zebrafish gut. Dev. Dyn. 2007. [Google Scholar] [CrossRef]
- Takada, N.; Appel, B. Identification of genes expressed by zebrafish oligodendrocytes using a differential microarray screen. Dev. Dyn. 2010. [Google Scholar] [CrossRef]
- Thisse, B.; Thisse, C. Fast Release Clones: A High Throughput Expression Analysis. ZFIN Direct Data Submiss. 2004. [Google Scholar]
- Choe, S.K.; Zhang, X.; Hirsch, N.; Straubhaar, J.; Sagerström, C.G. A screen for hoxb1-regulated genes identifies ppp1r14al as a regulator of the rhombomere 4 Fgf-signaling center. Dev. Biol. 2011. [Google Scholar] [CrossRef] [Green Version]
- Eto, M.; Kitazawa, T.; Matsuzawa, F.; Aikawa, S.-i.; Kirkbride, J.A.; Isozumi, N.; Nishimura, Y.; Brautigan, D.L.; Ohki, S.-y. Phosphorylation-Induced Conformational Switching of CPI-17 Produces a Potent Myosin Phosphatase Inhibitor. Structure 2007. [Google Scholar] [CrossRef] [Green Version]
- Hayashi, Y.; Senba, S.; Yazawa, M.; Brautigan, D.L.; Eto, M. Defining the Structural Determinants and a Potential Mechanism for Inhibition of Myosin Phosphatase by the Protein Kinase C-potentiated Inhibitor Protein of 17 kDa. J. Biol. Chem. 2001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muffato, M.; Louis, A.; Poisnel, C.E.; Crollius, H.R. Genomicus: A database and a browser to study gene synteny in modern and ancestral genomes. Bioinformatics 2010. [Google Scholar] [CrossRef] [PubMed]
- Tang, R.; Dodd, A.; Lai, D.; McNabb, W.C.; Love, D.R. Validation of zebrafish (Danio rerio) reference genes for quantitative real-time RT-PCR normalization. Acta Biochim. Biophys. Sin. (Shanghai) 2007, 39, 384–390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eto, M.; Wong, L.; Yazawa, M.; Brautigan, D.L. Inhibition of myosin/moesin phosphatase by expression of the phosphoinhibitor protein CPI-17 alters microfilament organization and retards cell spreading. Cell Motil. Cytoskeleton 2000. [Google Scholar] [CrossRef]
- Kitazawa, T.; Polzin, A.N.; Eto, M. CPI-17-deficient smooth muscle of chicken. J. Physiol. 2004. [Google Scholar] [CrossRef] [PubMed]
- Tountas, N.A.; Brautigan, D.L. Migration and retraction of endothelial and epithelial cells require PHI-1, a specific protein-phosphatase-1 inhibitor protein. J. Cell Sci. 2004. [Google Scholar] [CrossRef] [Green Version]
- Jayashankar, V.; Nguyen, M.J.; Carr, B.W.; Zheng, D.C.; Rosales, J.B.; Rosales, J.B.; Weiser, D.C. Protein phosphatase 1 β paralogs encode the zebrafish myosin phosphatase catalytic subunit. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [Green Version]
- Shimizu, N.; Ishitani, S.; Sato, A.; Shibuya, H.; Ishitani, T. Hipk2 and PP1c Cooperate to Maintain Dvl Protein Levels Required for Wnt Signal Transduction. Cell Rep. 2014. [Google Scholar] [CrossRef] [Green Version]
- Weiser, D.C.; Kimelman, D. Analysis of cell shape and polarity during zebrafish gastrulation. Methods Mol. Biol. 2012, 839, 53–68. [Google Scholar] [CrossRef] [Green Version]
- Nechiporuk, A.; Linbo, T.; Poss, K.D.; Raible, D.W. Specification of epibranchial placodes in zebrafish. Development 2007. [Google Scholar] [CrossRef] [Green Version]
- LaFlamme, A.; Young, K.E.; Lang, I.; Weiser, D.C. Alternative splicing of (ppp1r12a/mypt1) in zebrafish produces a novel myosin phosphatase targeting subunit. Gene 2018. [Google Scholar] [CrossRef] [PubMed]
- Carr, B.W.; Basepayne, T.L.; Chen, L.; Jayashankar, V.; Weiser, D.C. Characterization of the zebrafish homolog of zipper interacting protein kinase. Int. J. Mol. Sci. 2014, 15, 11597–11613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aguilar, H.N.; Tracey, C.N.; Tsang, S.C.F.; McGinnis, J.M.; Mitchell, B.F. Phos-tag-based analysis of myosin regulatory light chain phosphorylation in human uterine myocytes. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]








© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lang, I.; Virk, G.; Zheng, D.C.; Young, J.; Nguyen, M.J.; Amiri, R.; Fong, M.; Arata, A.; Chadaideh, K.S.; Walsh, S.; et al. The Evolution of Duplicated Genes of the Cpi-17/Phi-1 (ppp1r14) Family of Protein Phosphatase 1 Inhibitors in Teleosts. Int. J. Mol. Sci. 2020, 21, 5709. https://doi.org/10.3390/ijms21165709
Lang I, Virk G, Zheng DC, Young J, Nguyen MJ, Amiri R, Fong M, Arata A, Chadaideh KS, Walsh S, et al. The Evolution of Duplicated Genes of the Cpi-17/Phi-1 (ppp1r14) Family of Protein Phosphatase 1 Inhibitors in Teleosts. International Journal of Molecular Sciences. 2020; 21(16):5709. https://doi.org/10.3390/ijms21165709
Chicago/Turabian StyleLang, Irene, Guneet Virk, Dale C. Zheng, Jason Young, Michael J. Nguyen, Rojin Amiri, Michelle Fong, Alisa Arata, Katia S. Chadaideh, Susan Walsh, and et al. 2020. "The Evolution of Duplicated Genes of the Cpi-17/Phi-1 (ppp1r14) Family of Protein Phosphatase 1 Inhibitors in Teleosts" International Journal of Molecular Sciences 21, no. 16: 5709. https://doi.org/10.3390/ijms21165709




