Amorphization of Thiamine Chloride Hydrochloride: Effects of Physical State and Polymer Type on the Chemical Stability of Thiamine in Solid Dispersions
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Physical State on Thiamine Degradation
2.2. Relationship of T−Tg with Thiamine Stability
2.3. Effect of Polymer Type on Thiamine Degradation in Amorphous Solid Dispersions
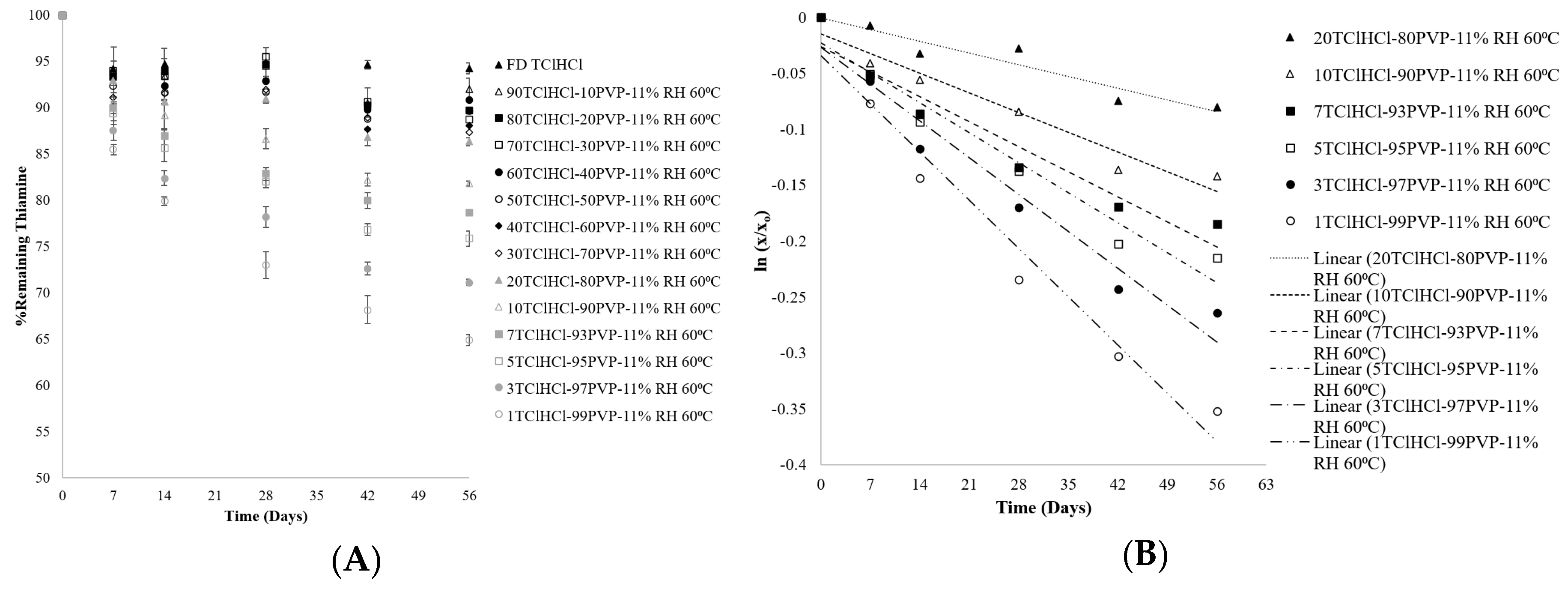
2.4. Chemical Stability of Thiamine Related to Polymer Proportion in a Solid Dispersion
3. Materials and Methods
3.1. Materials
3.2. Preparations of TClHCl Solid Dispersions via Lyophilization
3.3. Storage Treatments
3.4. Powder X-Ray Diffraction (PXRD)
3.5. Chemical Stability Determination with HPLC
3.6. Water Content and Moisture Sorption
3.7. Determination of Glass Transition Temperature (Tg) by Differential Scanning Calorimetry (DSC)
3.8. Reaction Kinetics Calculations
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| DSC | Differential scanning calorimetry |
| FD | Freeze dried |
| FTIR | Fourier transform infrared spectroscopy |
| HBA | Hydrogen bond acceptor |
| HBD | Hydrogen bond donor |
| HPLC | High-performance liquid chromatography |
| PEC | Pectin |
| PM | Physical mixture |
| PVP | Poly(vinylpyrrolidone) |
| PXRD | Powder x-ray diffraction |
| RH | Relative humidity |
| SD | Solid dispersion |
| TClHCl | Thiamine chloride hydrochloride |
| TFA | Trifluoroacetic acid |
| Tg | Glass transition temperature |
References
- Nath, A.; Shope, T.; Koch, T. Thiamine (vitamin B1) after weight loss bariatric surgery A2. In Metabolism and Pathophysiology of Bariatric Surgery; Rajendram, R., Martin, C.R., Eds.; Academic Press: Boston, MA, USA, 2017; pp. 479–489. [Google Scholar] [CrossRef]
- U.S Food & Drug Administration. FDA Vitamins and Minerals Chart. Available online: https://www.accessdata.fda.gov/scripts/interactivenutritionfactslabel/assets/InteractiveNFL_Vitamins&MineralsChart_March2020.pdf (accessed on 8 July 2020).
- McCandless, D. Thiamine Deficiency and Associated Clinical Disorders; Humana Press: New York, NY, USA, 2010; pp. 1–192. [Google Scholar]
- Ball, G. Vitamins in Foods: Analysis, Bioavailability, and Stability; CRC/Taylor & Francis: Boca Raton, FL, USA, 2006. [Google Scholar]
- Shepherd, S.; Gibson, P. Nutritional inadequacies of the gluten-free diet in both recently-diagnosed and long-term patients with coeliac disease. J. Hum. Nutr. Diet. 2013, 26, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Prinzo, Z.W. Thiamine deficiency and its prevention and control in major emergencies. In Micronutrient Series; World Health Organization (WHO), Department of Nutrition for Health and Development, Office of the United Nations High Commissioner for Refugees (UNHCR): Geneva, Switzerland, 1999. [Google Scholar]
- Dwivedi, B.K.; Arnold, R.G. Chemistry of thiamine degradation on food products and model systems. Review. J. Agric. Food. Chem. 1973, 21, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Lucille, E.; Michael, E.; Teresa Bridget, F. Examining strategies to facilitate vitamin B1 biofortification of plants by genetic engineering. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef] [Green Version]
- Connors, K.A. Chemical Stability of Pharmaceuticals: A Handbook for Pharmacists, 2nd ed.; Wiley: New York, NY, USA, 1986. [Google Scholar]
- Feliciotti, E.; Esselen, W. Thermal destruction rates of thiamine in pureed meats and vegetables. Food Technol. 1957, 11, 77–84. [Google Scholar]
- Buttery, R.G.; Haddon, W.F.; Seifert, R.M.; Turnbaugh, J.G. Thiamin odor and bis(2-methyl-3-furyl) disulfide. J. Agric. Food. Chem. 1984, 32, 674–676. [Google Scholar] [CrossRef]
- Voelker, A.L.; Miller, J.; Running, C.A.; Taylor, L.S.; Mauer, L.J. Chemical stability and reaction kinetics of two thiamine salts (thiamine mononitrate and thiamine chloride hydrochloride) in solution. Food Res. Int. 2018, 443–456. [Google Scholar] [CrossRef] [Green Version]
- Yen, C.-C.; Liang, Y.-K.; Cheng, C.-P.; Hsu, M.-C.; Wu, Y.-T. Oral bioavailability enhancement and anti-fatigue assessment of the andrographolide loaded solid dispersion. Int. J. Mol. Sci. 2020, 21, 2506. [Google Scholar] [CrossRef] [Green Version]
- Hancock, B.C.; Zografi, G. Characteristics and significance of the amorphous state in pharmaceutical systems. J. Pharm. Sci. 1997, 86, 1–12. [Google Scholar] [CrossRef]
- Arioglu-Tuncil, S.; Bhardwaj, V.; Taylor, L.S.; Mauer, L.J. Amorphization of thiamine chloride hydrochloride: A study of the crystallization inhibitor properties of different polymers in thiamine chloride hydrochloride amorphous solid dispersions. Food Res. Int. 2017, 99, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Roos, Y.H.; Drusch, S. Reaction kinetics. In Phase Transitions in Foods, 2nd ed.; Academic Press: San Diego, CA, USA, 2016; pp. 275–313. [Google Scholar] [CrossRef]
- Labuza, T.; Kamman, J. Comparison of stability of thiamin salts at high temperature and water activity. J. Food Sci. 1982, 47, 664–665. [Google Scholar] [CrossRef]
- Pachapurkar, D.; Bell, L.N. Kinetics of thiamin degradation in solutions under ambient storage conditions. J. Food Sci. 2006, 70. [Google Scholar] [CrossRef]
- Bell, L.; White, K. Thiamin stability in solids as affected by the glass transition. J. Food Sci. 2000, 65, 498–501. [Google Scholar] [CrossRef]
- Arabshahi, A.; Lund, D. Thiamin stability in simulated intermediate moisture food. J. Food Sci. 1988, 53, 199–203. [Google Scholar] [CrossRef]
- Dennison, D.; Kirk, J.; Bach, J.; Kokoczka, P.; Heldman, D. Storage stability of thiamin and riboflavin in a dehydrated food system. J. Food Process. Preserv. 1977, 1, 43–54. [Google Scholar] [CrossRef]
- Slade, L.; Levine, H. Beyond water activity: Recent advances based on an alternative approach to the assessment of food quality and safety. Crit. Rev. Food Sci. Nutr. 1991, 30, 115–360. [Google Scholar] [CrossRef]
- Lee, D.-J.; Lee, S.; Kim, I.W. Effects of humidity and surfaces on the melt crystallization of ibuprofen. Int. J. Mol. Sci. 2012, 13, 10296–10304. [Google Scholar] [CrossRef] [Green Version]
- Bell, L.; Hageman, M. Differentiating between the effects of water activity and glass transition dependent mobility on a solid state chemical reaction: Aspartame degradation. J. Agric. Food. Chem. 1994, 42, 2398–2401. [Google Scholar] [CrossRef]
- Lai, M.C.; Hageman, M.J.; Schowen, R.L.; Borchardt, R.T.; Topp, E.M. Chemical stability of peptides in polymers. 1. Effect of water on peptide deamidation in poly(vinyl alcohol) and poly(vinyl pyrrolidone) matrixes. J. Pharm. Sci. 1999, 88, 1073–1080. [Google Scholar] [CrossRef]
- Luthra, S.A.; Shalaev, E.Y.; Medek, A.; Hong, J.; Pikal, M.J. Chemical stability of amorphous materials: Specific and general media effects in the role of water in the degradation of freeze-dried zoniporide. J. Pharm. Sci. 2012, 101, 3110–3123. [Google Scholar] [CrossRef]
- Sanchez, J.O.; Ismail, Y.; Christina, B.; Mauer, L.J. Degradation of L-ascorbic acid in the amorphous solid state. J. Food Sci. 2018, 83, 670–681. [Google Scholar] [CrossRef]
- Murase, N.; Franks, F. Salt precipitation during the freeze-concentration of phosphate buffer solutions. Biophys. Chem. 1989, 34, 293. [Google Scholar] [CrossRef]
- Ismail, Y.; Mauer, L.J. Phase transitions of ascorbic acid and sodium ascorbate in a polymer matrix and effects on vitamin degradation. J. Food Process Eng. 2019, 43, e13073. [Google Scholar] [CrossRef]
- Waterman, K.C.; Gerst, P.; Dai, Z. A generalized relation for solid-state drug stability as a function of excipient dilution: Temperature-independent behavior. J. Pharm. Sci. 2012, 101, 4170–4177. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Hong, P.; Alden, B.; Boissel, C.; Swanson, D.; Chambers, E.; Diehl, D.; Iraneta, P.; Mazzeo, J.; Wyndham, K.; et al. Improvements in reversed-phase HPLC columns designed for polar compound retention. In Proceedings of the HPLC, San Francisco, CA, USA, 17–23 June 2006. [Google Scholar]
- Mauer, L.J.; Bradley, R.L. Moisture and total solids analysis. In Food Analysis; Nielsen, S.S., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 257–286. [Google Scholar] [CrossRef]
- Windheuser, J.J.; Higuchi, T. Kinetics of thiamine hydrolysis. J. Pharm. Sci. 1962, 51, 354–364. [Google Scholar] [CrossRef]





| Sample | kobs (day−1) | R2 | t90 (days) |
|---|---|---|---|
| 1TClHCl:99PVP | 0.0062 | 0.96 | 17 |
| 3TClHCl:97PVP | 0.0047 | 0.95 | 22 |
| 5TClHCl:95PVP | 0.0038 | 0.95 | 28 |
| 7TClHCl:93PVP | 0.0032 | 0.93 | 33 |
| 10TClHCl:90PVP | 0.0025 | 0.95 | 42 |
| 20TClHCl:80PVP | 0.0015 | 0.92 | 70 |
| Sample | Onset Tg (°C) | % Moisture Content (wb) |
|---|---|---|
| 1TClHCl:99 PVP SD | 61 ± 2 A | 2.1 ± 0.5 a |
| 5TClHCl:95 PVP SD | 61 ± 2 A | 1.8 ± 0.1 a |
| 20TClHCl:80 PVP SD | 54 ± 7 BA | 1.3 ± 0.7 a |
| 30TClHCl:70 PVP SD | 55 ± 4 BA | 1.39 ± 0.07 a |
| 40TClHCl:60 PVP SD | 42 ± 5 B | 0.8 ± 0.2 a |
| Sample | Storage Condition | Onset Tg (°C) |
|---|---|---|
| 5TClHCl: 95PVP SD | 11% RH and 30 °C | 53.6 ± 0.1 A |
| 5TClHCl:95PVP SD | 11% RH and 60 °C | 47 ± 1 B |
| Pre-Lyophilized Solution Composition (100 mg Solids/10 mL Water) | pH (at 25 °C) |
|---|---|
| 1TClHCl:99PVP | 3.94 ± 0.01 B |
| 3TClHCl:97PVP | 3.905 ± 0.007 B |
| 5TClHCl:95PVP | 3.800 ± 0.004 BC |
| 7TClHCl:93PVP | 3.73 ± 0.06 C |
| 10TClHCl:90PVP | 3.71 ± 0.01 CD |
| 20TClHCl:80PVP | 3.54 ± 0 E |
| 30TClHCl:70PVP | 3.47 ± 0.03 EFG |
| 40TClHCl:60PVP | 3.44 ± 0.02 EFGH |
| 50TClHCl:50PVP | 3.38 ± 0.01 FGHI |
| 60TClHCl:40PVP | 3.345 ± 0.007 GHIJ |
| 70TClHCl:30PVP | 3.315 ± 0.007 HIJ |
| 80TClHCl:20PVP | 3.27 ± 0.01 IJ |
| 90TClHCl:10PVP | 3.25 ± 0.02 IJ |
| 100TClHCl | 3.23 ± 0.02 J |
| 100PVP | 4.1 ± 0.1 A |
| 5TClHCl:95PEC | 3.525 ± 0.007 EF |
| 100PEC | 3.58 ± 0 ED |
| 50TClHCl:50PEC | 3.31 ± 0 HIJ |
| % TClHCl in Formulation | % PVP in Formulation | % Pectin in Formulation |
|---|---|---|
| - | 100% | - |
| 1% | 99% | - |
| 3% | 97% | - |
| 5% | 95% | - |
| 7% | 93% | - |
| 10% | 90% | - |
| 20% | 80% | - |
| 30% | 70% | - |
| 40% | 60% | - |
| 50% | 50% | - |
| 60% | 40% | - |
| 70% | 30% | - |
| 80% | 20% | - |
| 90% | 10% | - |
| 100% | - | - |
| - | - | 100% |
| 5% | - | 95% |
| 50% | - | 50% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arioglu-Tuncil, S.; Voelker, A.L.; Taylor, L.S.; Mauer, L.J. Amorphization of Thiamine Chloride Hydrochloride: Effects of Physical State and Polymer Type on the Chemical Stability of Thiamine in Solid Dispersions. Int. J. Mol. Sci. 2020, 21, 5935. https://doi.org/10.3390/ijms21165935
Arioglu-Tuncil S, Voelker AL, Taylor LS, Mauer LJ. Amorphization of Thiamine Chloride Hydrochloride: Effects of Physical State and Polymer Type on the Chemical Stability of Thiamine in Solid Dispersions. International Journal of Molecular Sciences. 2020; 21(16):5935. https://doi.org/10.3390/ijms21165935
Chicago/Turabian StyleArioglu-Tuncil, Seda, Adrienne L. Voelker, Lynne S. Taylor, and Lisa J. Mauer. 2020. "Amorphization of Thiamine Chloride Hydrochloride: Effects of Physical State and Polymer Type on the Chemical Stability of Thiamine in Solid Dispersions" International Journal of Molecular Sciences 21, no. 16: 5935. https://doi.org/10.3390/ijms21165935
APA StyleArioglu-Tuncil, S., Voelker, A. L., Taylor, L. S., & Mauer, L. J. (2020). Amorphization of Thiamine Chloride Hydrochloride: Effects of Physical State and Polymer Type on the Chemical Stability of Thiamine in Solid Dispersions. International Journal of Molecular Sciences, 21(16), 5935. https://doi.org/10.3390/ijms21165935





