High-Throughput Cloning and Characterization of Emerging Adenovirus Types 70, 73, 74, and 75
Abstract
1. Introduction
2. Results
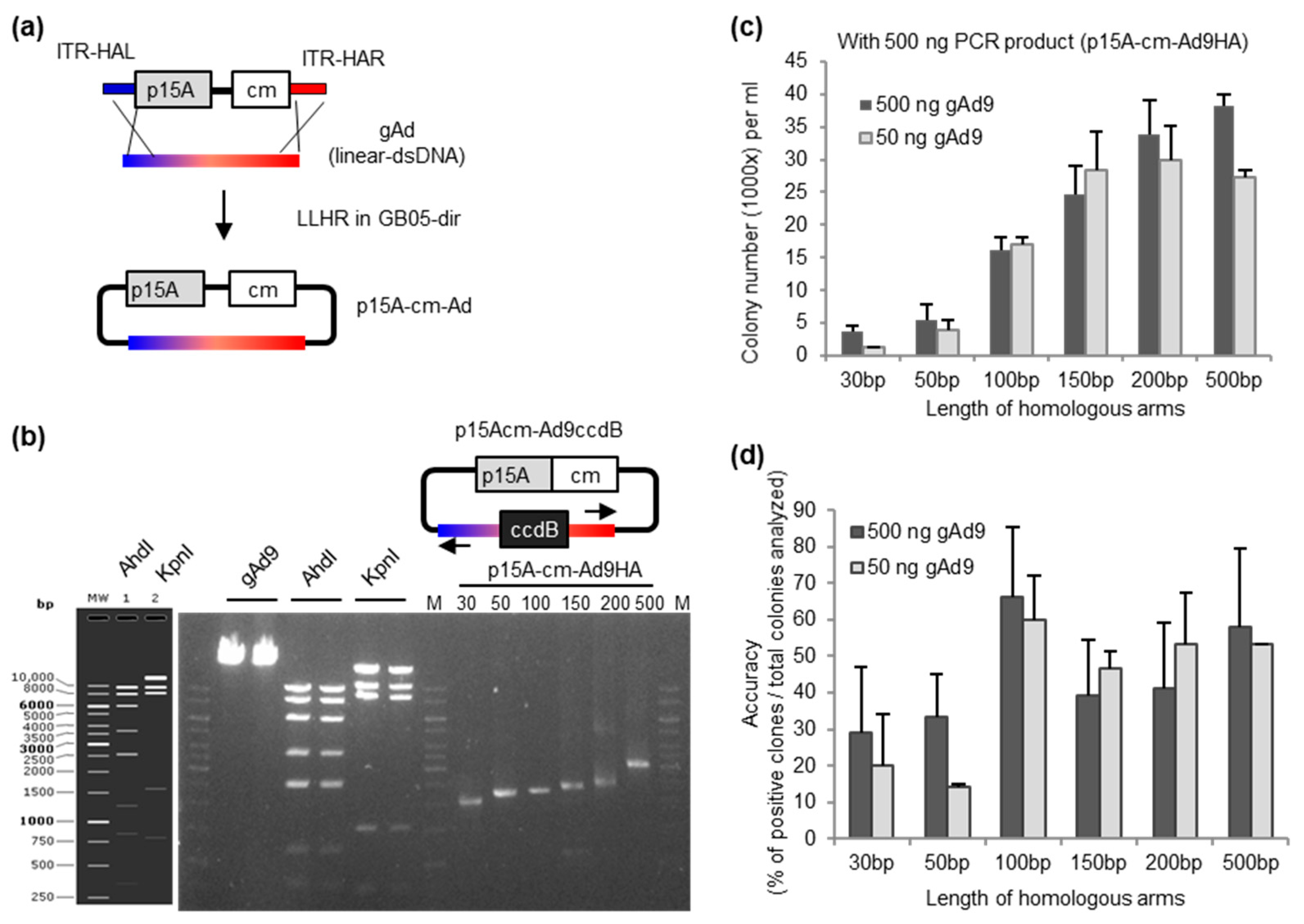
2.1. Role of Homology Arm-Length in the Efficiency of Adenoviral Genome Direct Cloning
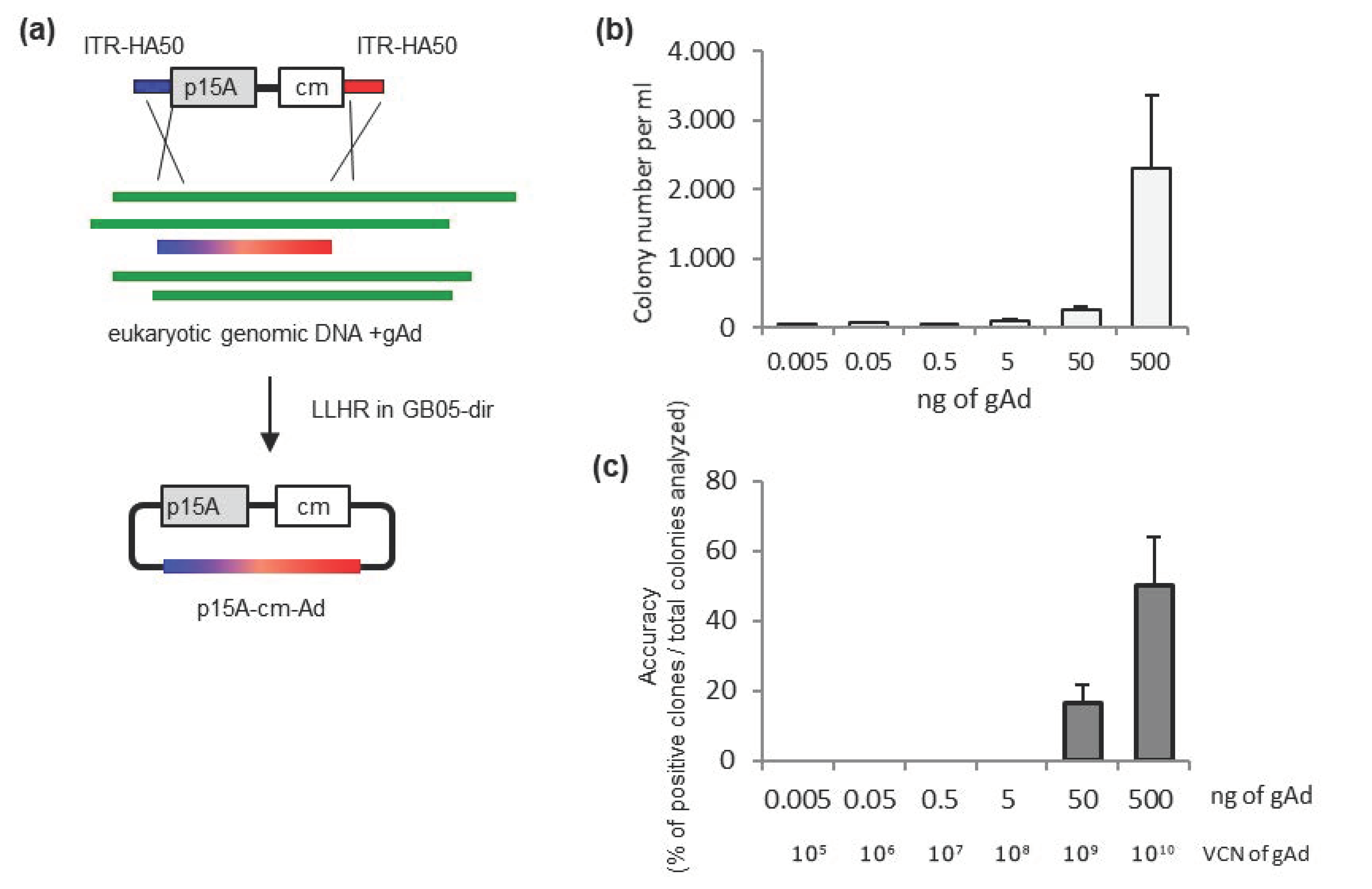
2.2. Sufficient Amount of Adenovirus Genome Is Required for Successful Cloning
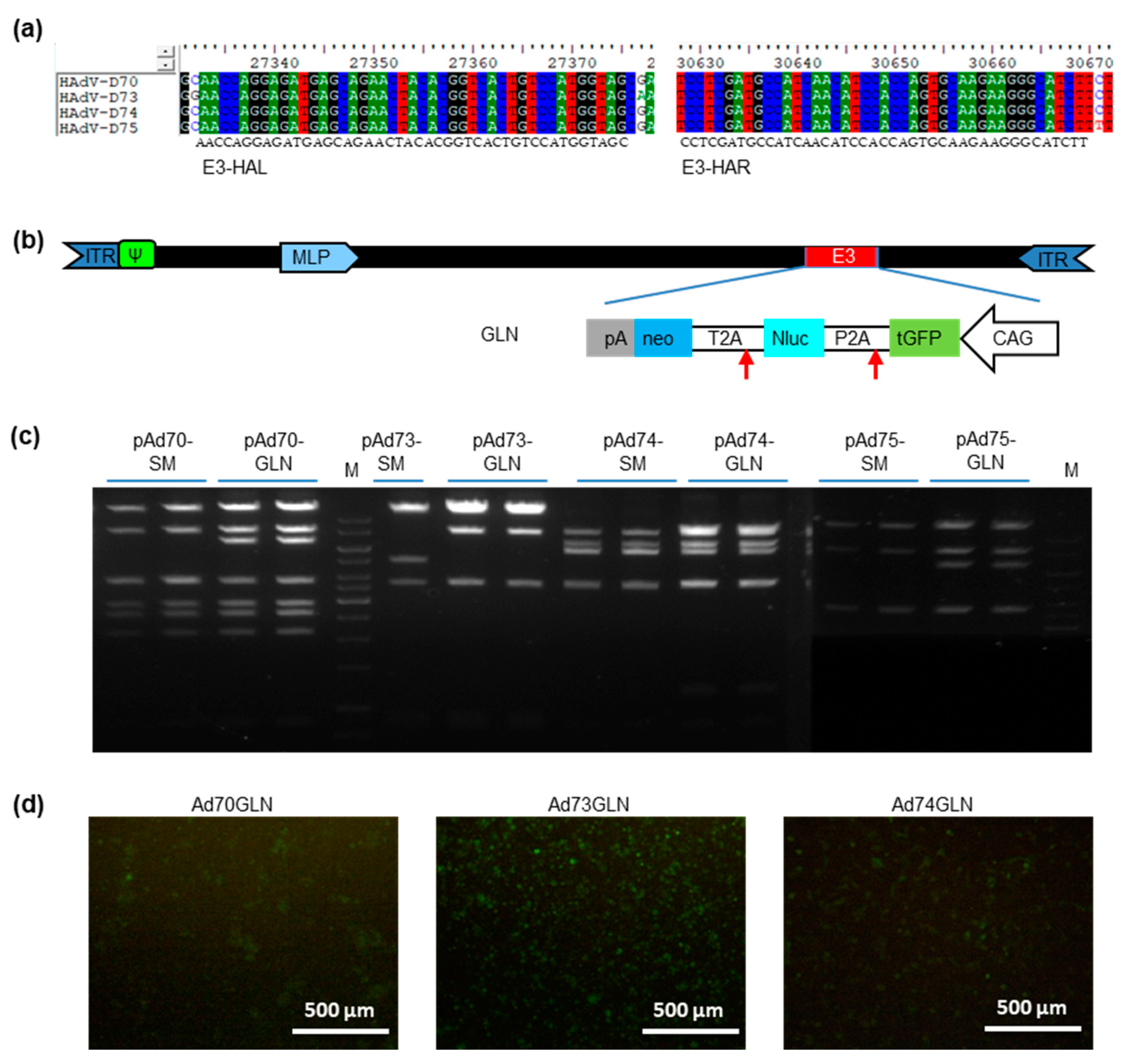
2.3. High-Throughput Cloning of Emerging Adenoviruses Types 70, 73, 74, and 75
2.4. High-Throughput Reporter Insertion
2.5. Screening of Adenovirus Tropisms
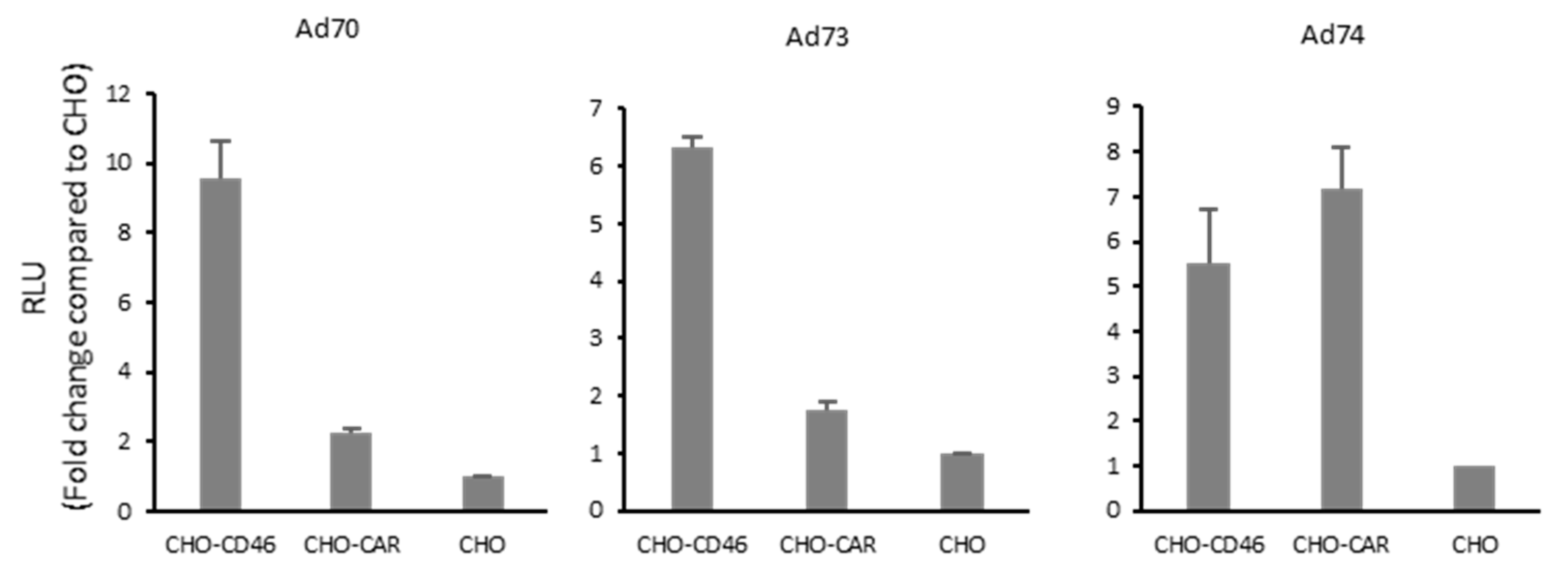
2.6. Screening of the Receptor Usage
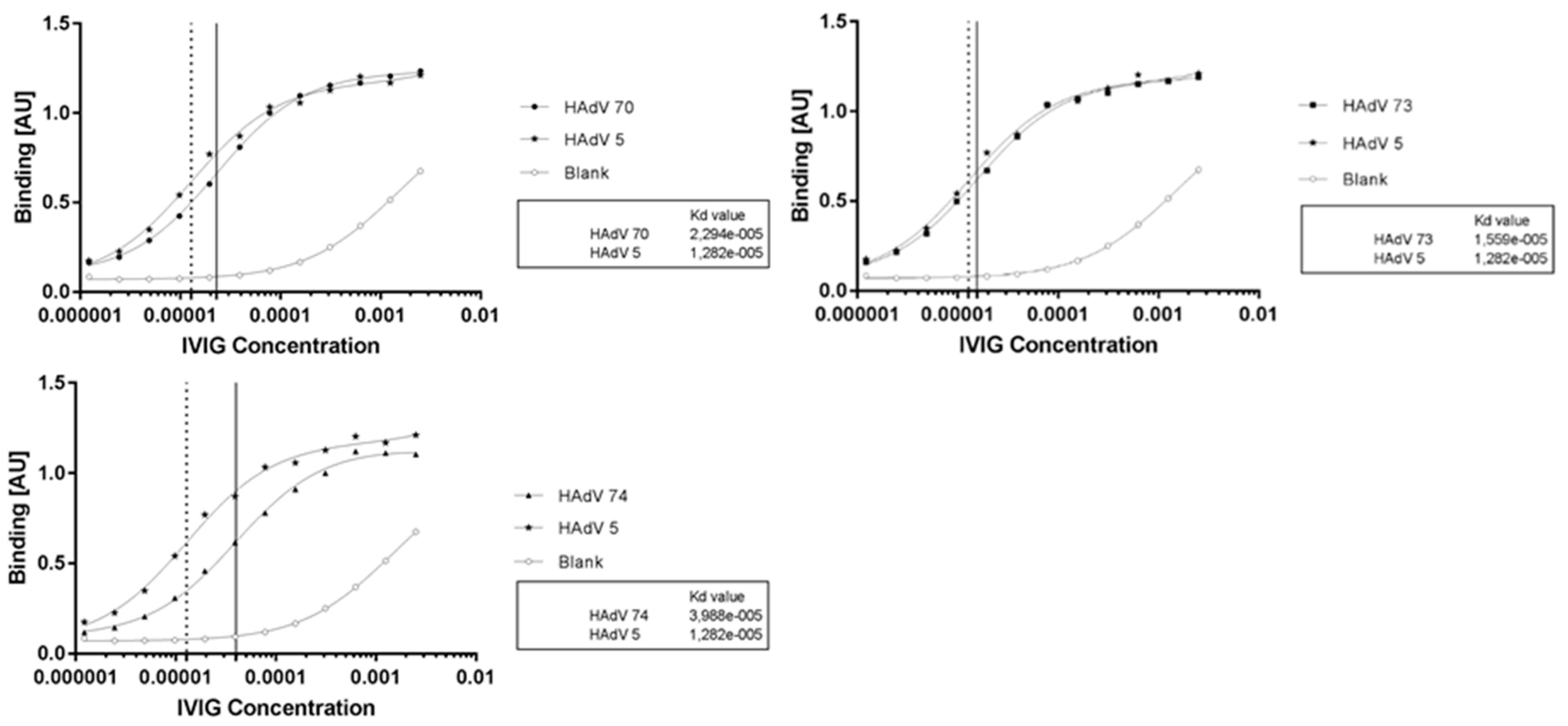
2.7. New Species D Human Adenoviruses with Lower or Comparable Prevalence of Anti-Adenoviral Antibodies
3. Discussion
4. Materials and Methods
4.1. Generation of Linear Cloning Vector p15A-cm-Ad9HA
4.2. Genomic DNA Isolation for Adenoviral Genome Cloning
4.3. Linear-Linear Homologous Recombination (LL-HR)
4.4. Colony-Polymerase Chain Reaction (PCR) Detection
4.5. Quantitative Real-Time PCR (qPCR) to Detect Virus Genome Copy Numbers (VCN)
4.6. Cell Cultures
4.7. New Species D Human Adenoviruses
4.8. Adenovirus Production
4.9. Evaluation of Adenovirus Transduction Efficiency via Luciferase Assay
4.10. ELISA to Measure Presence of Anti-Adenovirus Antibodies
4.11. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CAR | Coxsackievirus and adenovirus receptor |
| CD46 | Cluster of differentiation 46 |
| CsCl | Cesium chloride |
| CV | Cell/virus lysate |
| E3 | Early gene 3 |
| gAd | Human adenovirus genome |
| GFP | Green fluorescent protein |
| GLN | Turbo GFP, nanoLuciferase (Nluc), and neomycin resistance (neo) reporter cassette |
| HA | Homology arm |
| HAdV | Human adenovirus |
| IgG | Immunoglobulin G |
| IVIG | Intravenous immunoglobulin |
| LLHR | Linear-linear homologous recombination |
| ITR | Inverted terminal repeat |
| MLP | Major late prompter |
| PCR | Polymerase chain reaction |
| RLU | Relative light units |
| VCN | Virus genome copy number |
References
- Davison, A.J.; Benko, M.; Harrach, B. Genetic content and evolution of adenoviruses. J. Gen. Virol. 2003, 84, 2895–2908. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.M.; Singh, G.; Lee, J.Y.; Dehghan, S.; Rajaiya, J.; Liu, E.B.; Yousuf, M.A.; Betensky, R.A.; Jones, M.S.; Dyer, D.W.; et al. Molecular evolution of human adenoviruses. Sci. Rep. 2013, 3, 1812. [Google Scholar] [CrossRef] [PubMed]
- Nemerow, G.; Flint, J. Lessons learned from adenovirus (1970–2019). FEBS Lett. 2019, 593, 3395–3418. [Google Scholar] [CrossRef]
- Gu, J.; Su, Q.Q.; Zuo, T.T.; Chen, Y.B. Adenovirus diseases: A systematic review and meta-analysis of 228 case reports. Infection 2020. [Google Scholar] [CrossRef]
- Lion, T. Adenovirus infections in immunocompetent and immunocompromised patients. Clin. Microbiol. Rev. 2014, 27, 441–462. [Google Scholar] [CrossRef] [PubMed]
- Lion, T. Adenovirus persistence, reactivation, and clinical management. FEBS Lett. 2019, 593, 3571–3582. [Google Scholar] [CrossRef]
- Walsh, M.P.; Chintakuntlawar, A.; Robinson, C.M.; Madisch, I.; Harrach, B.; Hudson, N.R.; Schnurr, D.; Heim, A.; Chodosh, J.; Seto, D.; et al. Evidence of molecular evolution driven by recombination events influencing tropism in a novel human adenovirus that causes epidemic keratoconjunctivitis. PLoS ONE 2009, 4, e5635. [Google Scholar] [CrossRef]
- Kajan, G.L.; Kajon, A.E.; Pinto, A.C.; Bartha, D.; Arnberg, N. The complete genome sequence of human adenovirus 84, a highly recombinant new Human mastadenovirus D type with a unique fiber gene. Virus Res. 2017, 242, 79–84. [Google Scholar] [CrossRef]
- Rowe, W.P.; Huebner, R.J.; Gilmore, L.K.; Parrott, R.H.; Ward, T.G. Isolation of a cytopathogenic agent from human adenoids undergoing spontaneous degeneration in tissue culture. Proc. Soc. Exp. Biol. Med. 1953, 84, 570–573. [Google Scholar] [CrossRef]
- Crystal, R.G. Adenovirus: The first effective in vivo gene delivery vector. Hum. Gene Ther. 2014, 25, 3–11. [Google Scholar] [CrossRef]
- Zhang, C.; Zhou, D. Adenoviral vector-based strategies against infectious disease and cancer. Hum. Vaccin. Immunother. 2016, 12, 2064–2074. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.C.; Guan, X.H.; Li, Y.H.; Huang, J.Y.; Jiang, T.; Hou, L.H.; Li, J.X.; Yang, B.F.; Wang, L.; Wang, W.J.; et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2020, 396, 479–488. [Google Scholar] [CrossRef]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A.; et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet 2020. [Google Scholar] [CrossRef]
- Mast, T.C.; Kierstead, L.; Gupta, S.B.; Nikas, A.A.; Kallas, E.G.; Novitsky, V.; Mbewe, B.; Pitisuttithum, P.; Schechter, M.; Vardas, E.; et al. International epidemiology of human pre-existing adenovirus (Ad) type-5, type-6, type-26 and type-36 neutralizing antibodies: Correlates of high Ad5 titers and implications for potential HIV vaccine trials. Vaccine 2010, 28, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Abbink, P.; Lemckert, A.A.; Ewald, B.A.; Lynch, D.M.; Denholtz, M.; Smits, S.; Holterman, L.; Damen, I.; Vogels, R.; Thorner, A.R.; et al. Comparative seroprevalence and immunogenicity of six rare serotype recombinant adenovirus vaccine vectors from subgroups B and D. J. Virol. 2007, 81, 4654–4663. [Google Scholar] [CrossRef] [PubMed]
- Vogels, R.; Zuijdgeest, D.; van Rijnsoever, R.; Hartkoorn, E.; Damen, I.; de Bethune, M.P.; Kostense, S.; Penders, G.; Helmus, N.; Koudstaal, W.; et al. Replication-deficient human adenovirus type 35 vectors for gene transfer and vaccination: Efficient human cell infection and bypass of preexisting adenovirus immunity. J. Virol. 2003, 77, 8263–8271. [Google Scholar] [CrossRef]
- Alonso-Padilla, J.; Papp, T.; Kajan, G.L.; Benko, M.; Havenga, M.; Lemckert, A.; Harrach, B.; Baker, A.H. Development of Novel Adenoviral Vectors to Overcome Challenges Observed with HAdV-5-based Constructs. Mol. Ther. 2016, 24, 6–16. [Google Scholar] [CrossRef]
- Zhang, W.; Ehrhardt, A. Getting genetic access to natural adenovirus genomes to explore vector diversity. Virus Genes 2017, 53, 675–683. [Google Scholar] [CrossRef]
- Zou, X.H.; Bi, Z.X.; Guo, X.J.; Zhang, Z.; Zhao, Y.; Wang, M.; Zhu, Y.L.; Jie, H.Y.; Yu, Y.; Hung, T.; et al. ☆DNA assembly technique simplifies the construction of infectious clone of fowl adenovirus. J. Virol. Methods 2018, 257, 85–92. [Google Scholar] [CrossRef]
- Pan, H.; Yan, Y.; Zhang, J.; Zhao, S.; Feng, L.; Ou, J.; Cao, N.; Li, M.; Zhao, W.; Wan, C.; et al. Rapid Construction of a Replication-Competent Infectious Clone of Human Adenovirus Type 14 by Gibson Assembly. Viruses 2018, 10, 568. [Google Scholar] [CrossRef]
- Zhang, W.; Fu, J.; Ehrhardt, A. Novel Vector Construction Based on Alternative Adenovirus Types via Homologous Recombination. Hum. Gene Ther. Methods 2018, 29, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Fu, J.; Liu, J.; Wang, H.; Schiwon, M.; Janz, S.; Schaffarczyk, L.; von der Goltz, L.; Ehrke-Schulz, E.; Dorner, J.; et al. An Engineered Virus Library as a Resource for the Spectrum-wide Exploration of Virus and Vector Diversity. Cell Rep. 2017, 19, 1698–1709. [Google Scholar] [CrossRef] [PubMed]
- Hage, E.; Gerd Liebert, U.; Bergs, S.; Ganzenmueller, T.; Heim, A. Human mastadenovirus type 70: A novel, multiple recombinant species D mastadenovirus isolated from diarrhoeal faeces of a haematopoietic stem cell transplantation recipient. J. Gen. Virol. 2015, 96, 2734–2742. [Google Scholar] [CrossRef] [PubMed]
- Hage, E.; Dhingra, A.; Liebert, U.G.; Bergs, S.; Ganzenmueller, T.; Heim, A. Three novel, multiple recombinant types of species of human mastadenovirus D (HAdV-D 73, 74 & 75) isolated from diarrhoeal faeces of immunocompromised patients. J. Gen. Virol. 2017, 98, 3037–3045. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Bian, X.; Hu, S.; Wang, H.; Huang, F.; Seibert, P.M.; Plaza, A.; Xia, L.; Muller, R.; Stewart, A.F.; et al. Full-length RecE enhances linear-linear homologous recombination and facilitates direct cloning for bioprospecting. Nat. Biotechnol. 2012, 30, 440–446. [Google Scholar] [CrossRef]
- Robinson, C.M.; Seto, D.; Jones, M.S.; Dyer, D.W.; Chodosh, J. Molecular evolution of human species D adenoviruses. Infect. Genet. Evol. 2011, 11, 1208–1217. [Google Scholar] [CrossRef]
- Singh, G.; Robinson, C.M.; Dehghan, S.; Jones, M.S.; Dyer, D.W.; Seto, D.; Chodosh, J. Homologous recombination in E3 genes of human adenovirus species D. J. Virol. 2013, 87, 12481–12488. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, J.S.; Materne, E.C.; Rajala, R.; Ismail, A.M.; Seto, D.; Dyer, D.W.; Rajaiya, J.; Chodosh, J. Bacterial RecA Protein Promotes Adenoviral Recombination during In Vitro Infection. mSphere 2018, 3. [Google Scholar] [CrossRef]
- Ismail, A.M.; Lee, J.S.; Lee, J.Y.; Singh, G.; Dyer, D.W.; Seto, D.; Chodosh, J.; Rajaiya, J. Adenoviromics: Mining the Human Adenovirus Species D Genome. Front. Microbiol. 2018, 9, 2178. [Google Scholar] [CrossRef]
- Arnberg, N. Adenovirus receptors: Implications for tropism, treatment and targeting. Rev. Med. Virol. 2009, 19, 165–178. [Google Scholar] [CrossRef]
- Arnberg, N. Adenovirus receptors: Implications for targeting of viral vectors. Trends Pharmacol. Sci. 2012, 33, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Boehme, P.; Zhang, W.; Fu, J.; Yumul, R.; Mese, K.; Tsoukas, R.; Solanki, M.; Kaufmann, M.; Lu, R.; et al. Human adenovirus type 17 from species D transduces endothelial cells and human CD46 is involved in cell entry. Sci. Rep. 2018, 8, 13442. [Google Scholar] [CrossRef] [PubMed]
- Gaggar, A.; Shayakhmetov, D.M.; Lieber, A. CD46 is a cellular receptor for group B adenoviruses. Nat. Med. 2003, 9, 1408–1412. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, Z.Y.; Liu, Y.; Persson, J.; Beyer, I.; Moller, T.; Koyuncu, D.; Drescher, M.R.; Strauss, R.; Zhang, X.B.; et al. Desmoglein 2 is a receptor for adenovirus serotypes 3, 7, 11 and 14. Nat. Med. 2011, 17, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Arnberg, N.; Edlund, K.; Kidd, A.H.; Wadell, G. Adenovirus type 37 uses sialic acid as a cellular receptor. J. Virol. 2000, 74, 42–48. [Google Scholar] [CrossRef]
- Lenman, A.; Liaci, A.M.; Liu, Y.; Ardahl, C.; Rajan, A.; Nilsson, E.; Bradford, W.; Kaeshammer, L.; Jones, M.S.; Frangsmyr, L.; et al. Human adenovirus 52 uses sialic acid-containing glycoproteins and the coxsackie and adenovirus receptor for binding to target cells. PLoS Pathog. 2015, 11, e1004657. [Google Scholar] [CrossRef]
- Garnett, C.T.; Erdman, D.; Xu, W.; Gooding, L.R. Prevalence and quantitation of species C adenovirus DNA in human mucosal lymphocytes. J. Virol. 2002, 76, 10608–10616. [Google Scholar] [CrossRef]
- Raper, S.E.; Chirmule, N.; Lee, F.S.; Wivel, N.A.; Bagg, A.; Gao, G.P.; Wilson, J.M.; Batshaw, M.L. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer. Mol. Genet. Metab. 2003, 80, 148–158. [Google Scholar] [CrossRef]
- Jager, L.; Hausl, M.A.; Rauschhuber, C.; Wolf, N.M.; Kay, M.A.; Ehrhardt, A. A rapid protocol for construction and production of high-capacity adenoviral vectors. Nat. Protoc. 2009, 4, 547–564. [Google Scholar] [CrossRef]
- Kratzer, R.F.; Kreppel, F. Production, Purification, and Titration of First-Generation Adenovirus Vectors. Methods Mol. Biol. 2017, 1654, 377–388. [Google Scholar] [CrossRef]


| Virus | NCBI Access Number | Penton/Hexon/Fiber | Date/Place | Source (Isolated From) |
|---|---|---|---|---|
| Ad70 | KP641339 | P70H70F29 | 2014/Leipzig, Germany | Diarrheal feces of a hematopoietic stem cell transplantation recipient [23] |
| Ad73 | KY618676 | P67H45F27 | 2015/Leipzig, Germany | Diarrheal feces of a lymphoma patient treated with chemotherapy [24] |
| Ad74 | KY618677 | P70H74F51 | 2015/Leipzig, Germany | Diarrheal feces of a hematopoietic stem cell transplantation recipient [24] |
| Ad75 | KY618678 | P75H26F29 | 2015/Leipzig, Germany | The feces of an AIDS patient [24] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, W.; Mese, K.; Schellhorn, S.; Bahlmann, N.; Mach, N.; Bunz, O.; Dhingra, A.; Hage, E.; Lafon, M.-E.; Wodrich, H.; et al. High-Throughput Cloning and Characterization of Emerging Adenovirus Types 70, 73, 74, and 75. Int. J. Mol. Sci. 2020, 21, 6370. https://doi.org/10.3390/ijms21176370
Zhang W, Mese K, Schellhorn S, Bahlmann N, Mach N, Bunz O, Dhingra A, Hage E, Lafon M-E, Wodrich H, et al. High-Throughput Cloning and Characterization of Emerging Adenovirus Types 70, 73, 74, and 75. International Journal of Molecular Sciences. 2020; 21(17):6370. https://doi.org/10.3390/ijms21176370
Chicago/Turabian StyleZhang, Wenli, Kemal Mese, Sebastian Schellhorn, Nora Bahlmann, Nicolas Mach, Oskar Bunz, Akshay Dhingra, Elias Hage, Marie-Edith Lafon, Harald Wodrich, and et al. 2020. "High-Throughput Cloning and Characterization of Emerging Adenovirus Types 70, 73, 74, and 75" International Journal of Molecular Sciences 21, no. 17: 6370. https://doi.org/10.3390/ijms21176370
APA StyleZhang, W., Mese, K., Schellhorn, S., Bahlmann, N., Mach, N., Bunz, O., Dhingra, A., Hage, E., Lafon, M.-E., Wodrich, H., Heim, A., & Ehrhardt, A. (2020). High-Throughput Cloning and Characterization of Emerging Adenovirus Types 70, 73, 74, and 75. International Journal of Molecular Sciences, 21(17), 6370. https://doi.org/10.3390/ijms21176370






