Adipose-Derived Mesenchymal Stem Cells do not Affect the Invasion and Migration Potential of Oral Squamous Carcinoma Cells
Abstract
1. Introduction
2. Results
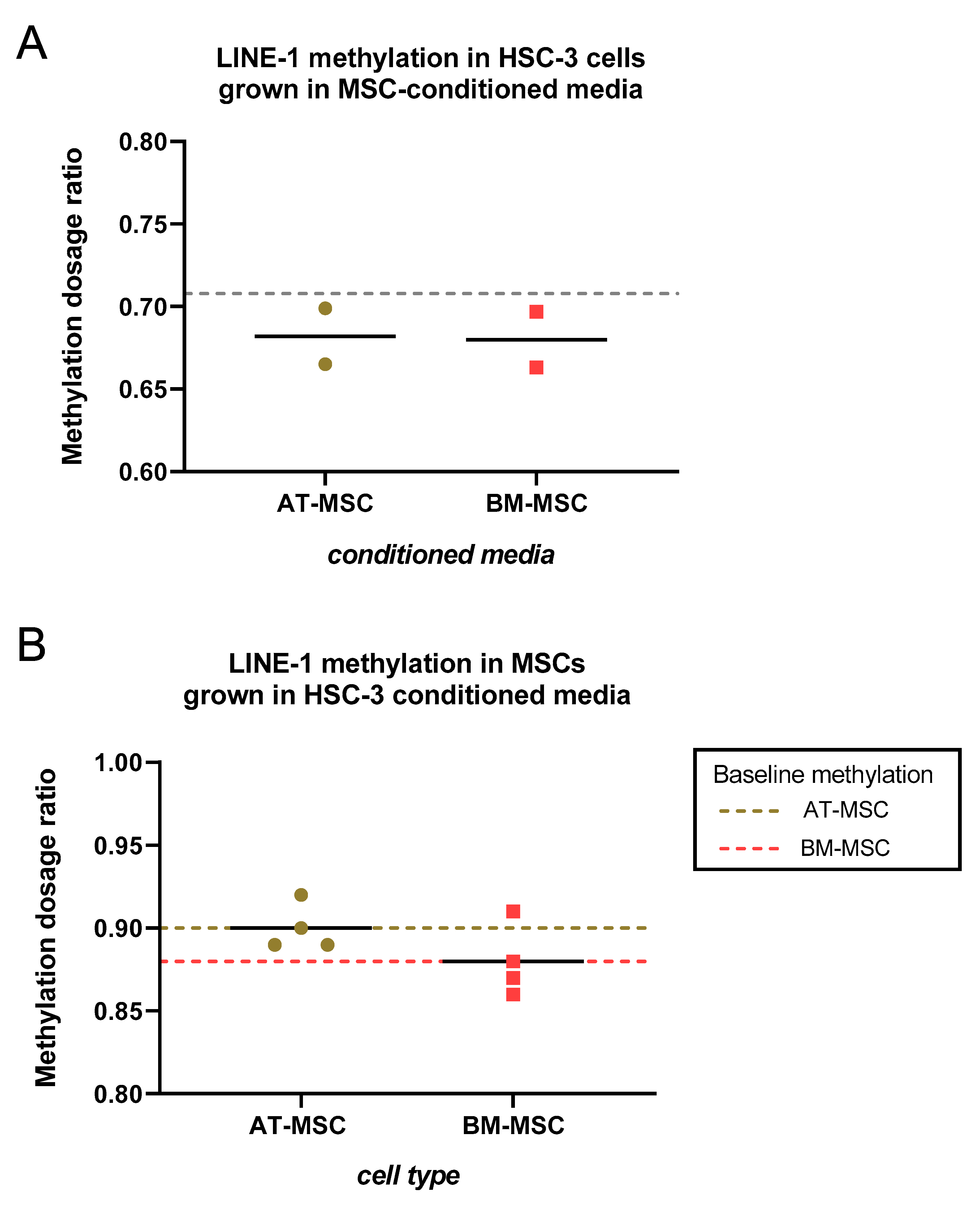
2.1. HSC-3- and MSCs-Conditioned Media had no effect on LINE-1 Methylation
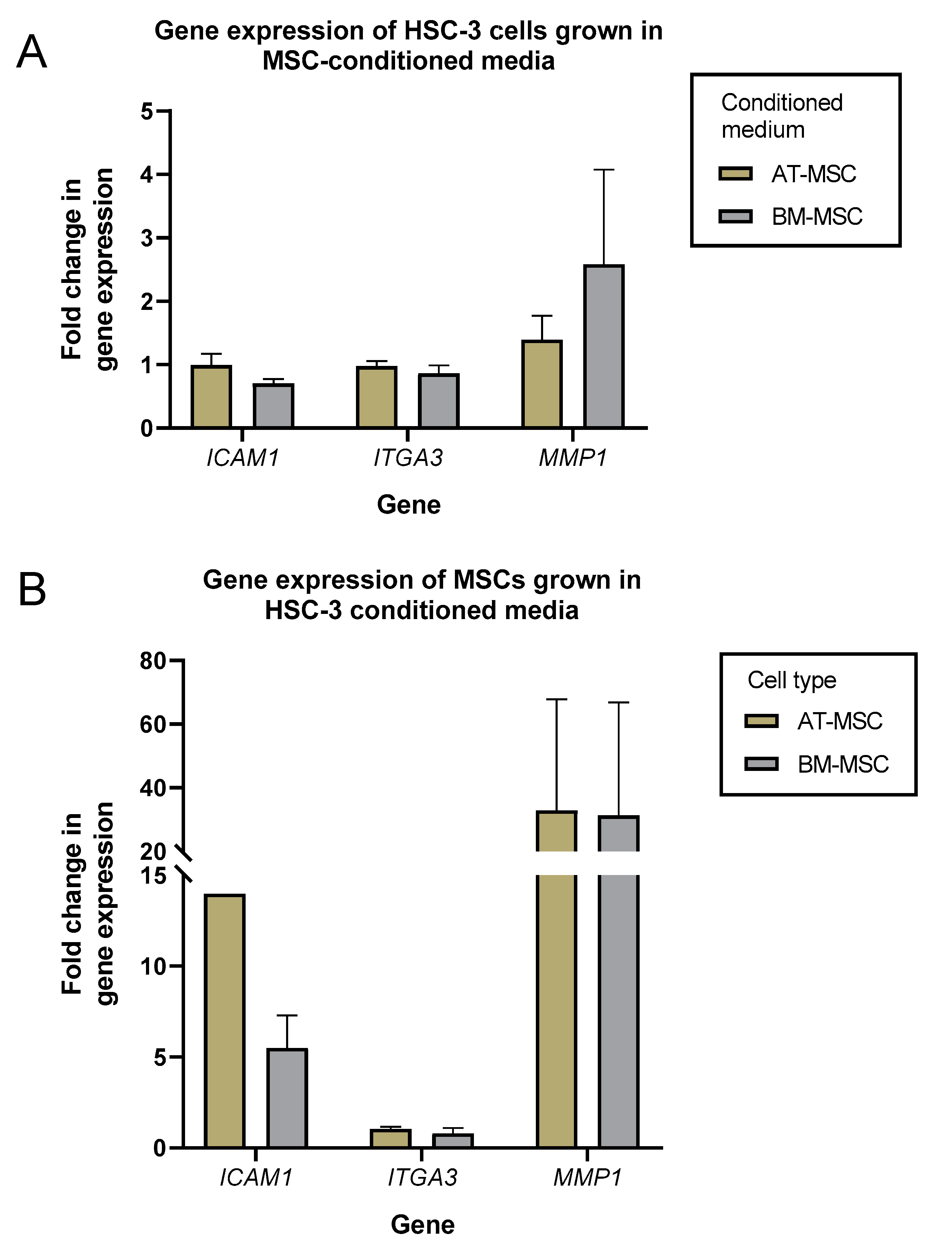
2.2. Microenvironment Remodeling genes Expression of HSC-3 and MSCs cells were Mainly Unaffected when Cultured in MSCs- or HSC-3-Conditioned Media
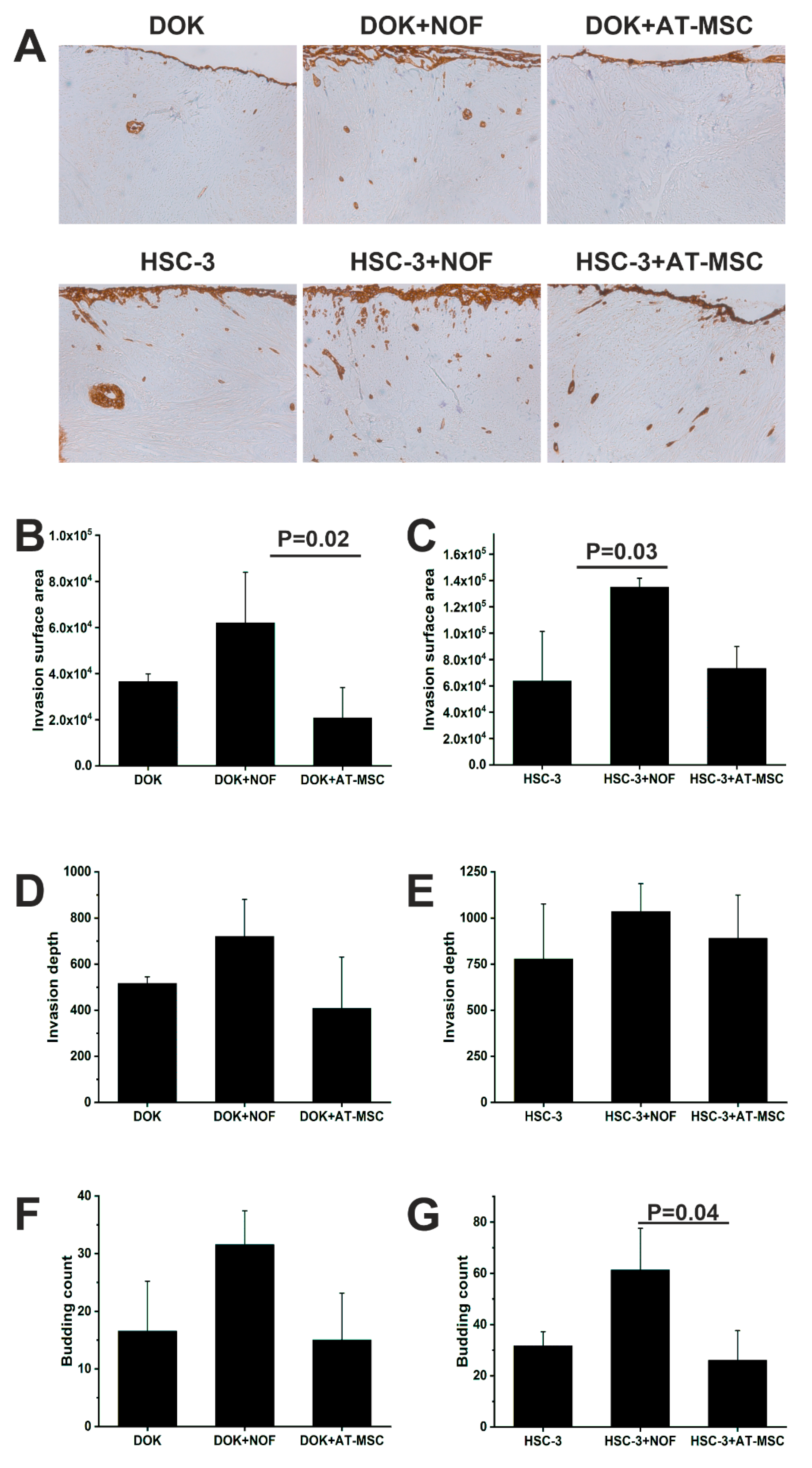
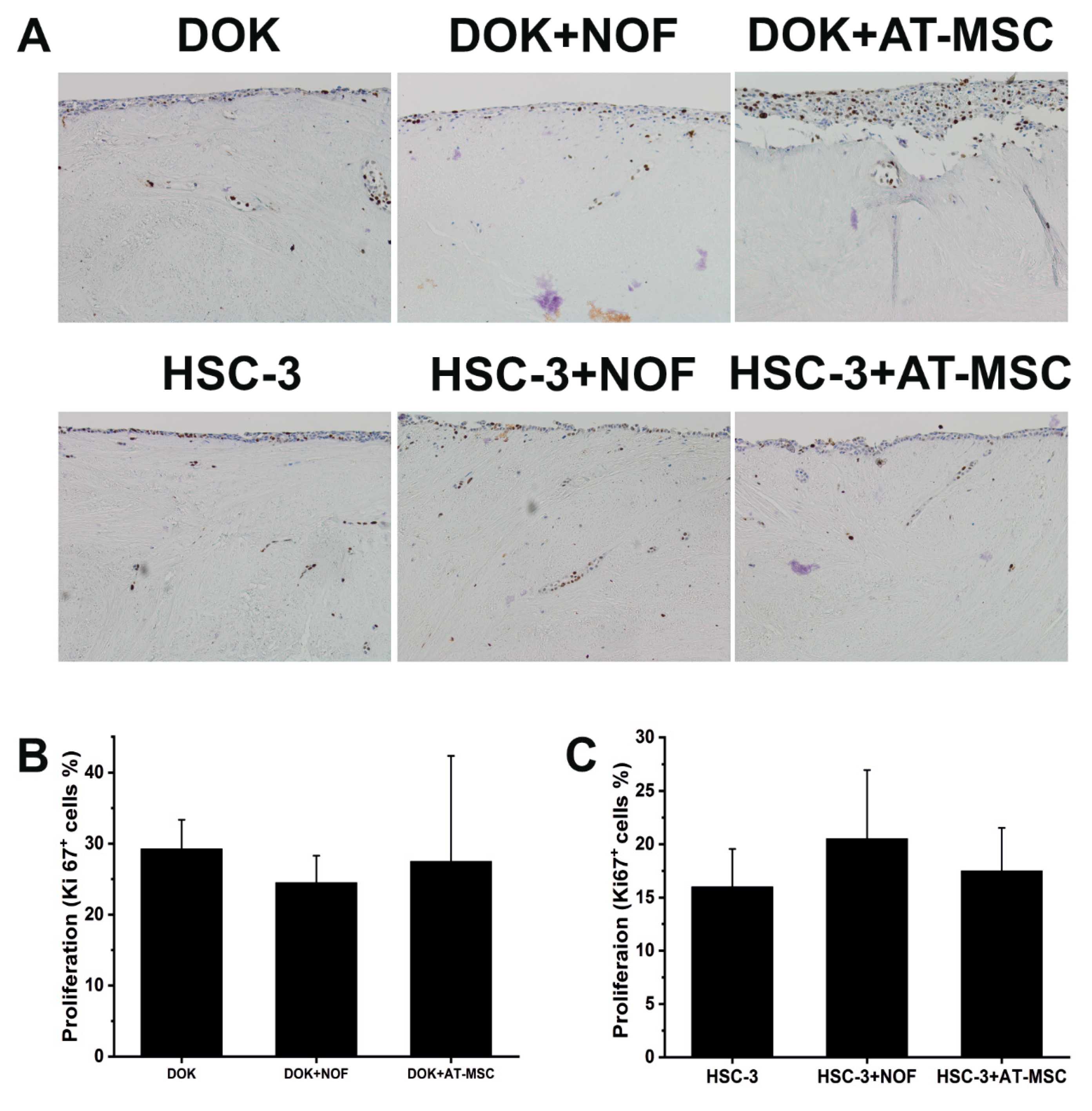
2.3. AT-MSCs had no Effect on the Invasion and the Proliferation Abilities of HSC-3 or DOK Cells
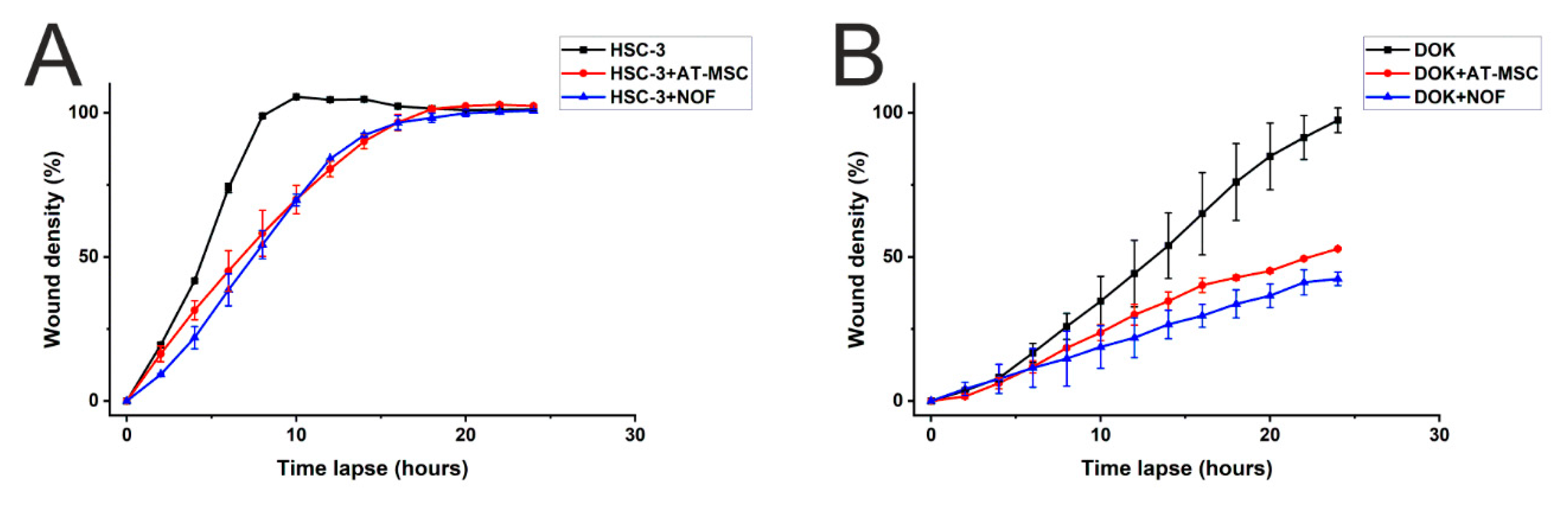
2.4. AT-MSCs and NOF did not Increase the Migration of HSC-3/DOK Cells
3. Discussion
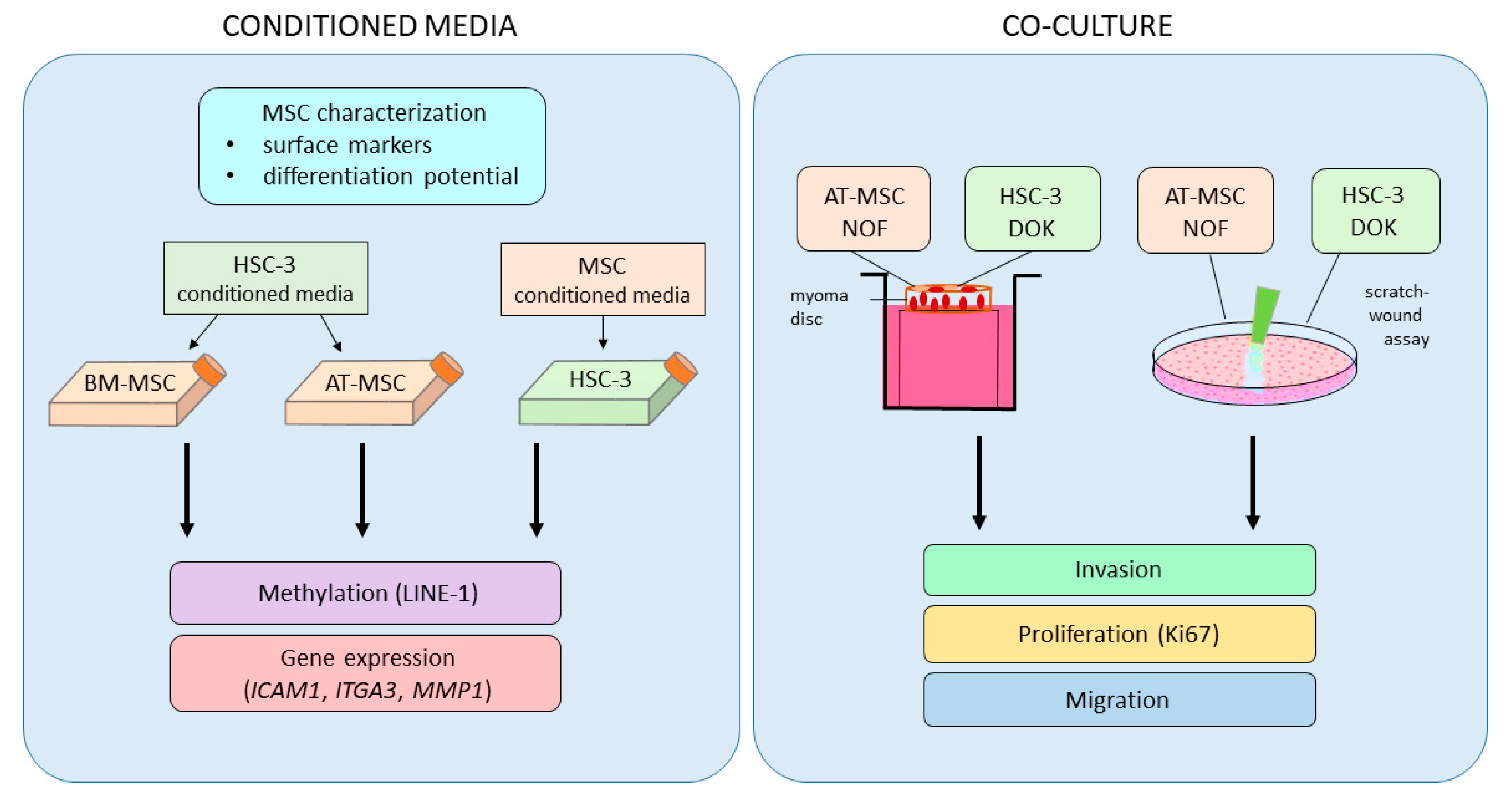
4. Materials and Methods
4.1. Cell Cultures
4.2. MSCs Characterization
4.3. Culture of MSCs and HSC-3 Cells in Conditioned Media
4.4. Gene Expression Analysis with qRT-PCR
4.5. LINE-1 Methylation Analysis
4.6. Organotypic Myoma Discs
4.7. Immunohistochemical Staining of the Myoma Discs
4.8. Scratch-Wound Cell Migration Assay
4.9. Microscopic and Histomorphometric Analysis
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| AT-MSC | Adipose tissue-derived mesenchymal stem cell |
| BM-MSC | Bone marrow-derived mesenchymal stem cell |
| DOK | Dysplastic oral keratinocyte |
| EV | Extracellular vesicle |
| HSC-3 | Human tongue squamous carcinoma |
| ICAM1 | Intercellular adhesion molecule-1 |
| ITGA3 | Integrin alpha-3 |
| LINE-1 | Long interspersed nuclear element-1 |
| MMP | Matrix metallopeptidase |
| MSC | Mesenchymal stem cell |
| MS-MLPA | Multiplex ligation-dependent probe amplification |
| NOF | Normal oral fibroblast |
| OTSCC | Oral tongue squamous cell carcinoma |
References
- Strioga, M.; Viswanathan, S.; Darinskas, A.; Slaby, O.; Michalek, J. Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells Dev. 2012, 21, 2724–2752. [Google Scholar] [CrossRef]
- Ridge, S.M.; Sullivan, F.J.; Glynn, S.A. Mesenchymal stem cells: Key players in cancer progression. Mol. Cancer 2017, 16, 31. [Google Scholar] [CrossRef]
- Wang, M.; Yuan, Q.; Xie, L. Mesenchymal stem cell-based immunomodulation: Properties and clinical application. Stem Cells Int. 2018, 2018, 3057624. [Google Scholar] [CrossRef]
- Gomez-Salazar, M.; Gonzalez-Galofre, Z.N.; Casamitjana, J.; Crisan, M.; James, A.W.; Péault, B. Five decades later, are mesenchymal stem cells still relevant? Front. Bioeng. Biotechnol. 2020, 8, 148. [Google Scholar] [CrossRef]
- Kern, S.; Eichler, H.; Stoeve, J.; Klüter, H.; Bieback, K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 2006, 24, 1294–1301. [Google Scholar] [CrossRef]
- Zhou, X.; Li, T.; Chen, Y.; Zhang, N.; Wang, P.; Liang, Y.; Long, M.; Liu, H.; Mao, J.; Liu, Q.; et al. Mesenchymal stem cell-derived extracellular vesicles promote the in vitro proliferation and migration of breast cancer cells through the activation of the ERK pathway. Int. J. Oncol. 2019, 54, 1843–1852. [Google Scholar] [CrossRef] [PubMed]
- Dmitrieva, R.I.; Minullina, I.R.; Bilibina, A.A.; Tarasova, O.V.; Anisimov, S.V.; Zaritskey, A.Y. Bone marrow- and subcutaneous adipose tissue-derived mesenchymal stem cells: Differences and similarities. Cell Cycle 2012, 11, 377–383. [Google Scholar] [CrossRef]
- Nishikawa, G.; Kawada, K.; Nakagawa, J.; Toda, K.; Ogawa, R.; Inamoto, S.; Mizuno, R.; Itatani, Y.; Sakai, Y. Bone marrow-derived mesenchymal stem cells promote colorectal cancer progression via CCR5. Cell Death Dis. 2019, 10, 264. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.; You, M.; Zhang, J.; Gao, G.; Han, R.; Luo, W.; Liu, T.; Zuo, J.; Wang, F. Adipose-derived mesenchymal stem cells enhance ovarian cancer growth and metastasis by increasing thymosin beta 4X-linked expression. Stem Cells Int. 2019, 2019, 9037197. [Google Scholar] [CrossRef] [PubMed]
- Liotta, F.; Querci, V.; Mannelli, G.; Santarlasci, V.; Maggi, L.; Capone, M.; Rossi, M.C.; Mazzoni, A.; Cosmi, L.; Romagnani, S.; et al. Mesenchymal stem cells are enriched in head neck squamous cell carcinoma, correlates with tumour size and inhibit T-cell proliferation. Br. J. Med. 2015, 112, 745–754. [Google Scholar] [CrossRef]
- Lee, H.Y.; Hong, I.S. Double-edged sword of mesenchymal stem cells: Cancer-promoting versus therapeutic potential. Cancer Sci. 2017, 108, 1939–1946. [Google Scholar] [CrossRef]
- He, N.; Kong, Y.; Lei, X.; Liu, Y.; Wang, J.; Xu, C.; Wang, Y.; Du, L.; Ji, K.; Wang, Q.; et al. MSCs inhibit tumor progression and enhance radiosensitivity of breast cancer cells by down-regulating STAT3 signaling pathway. Cell Death Dis. 2018, 9, 1026. [Google Scholar] [CrossRef]
- Otsu, K.; Das, S.; Houser, S.D.; Quadri, S.K.; Bhattacharya, S.; Bhattacharya, J. Concentration-dependent inhibition of angiogenesis by mesenchymal stem cells. Blood 2009, 113, 4197–4205. [Google Scholar] [CrossRef]
- Rhee, K.J.; Lee, J.I.; Eom, Y.W. Mesenchymal stem cell-mediated effects of tumor support or suppression. Int. J. Mol. Sci. 2015, 16, 30015–30033. [Google Scholar] [CrossRef]
- Ankrum, J.A.; Ong, J.F.; Karp, J.M. Mesenchymal stem cells: Immune evasive, not immune privileged. Nat. Biotech. 2014, 32, 252–260. [Google Scholar] [CrossRef]
- Salo, S.; Bitu, C.; Merkku, K.; Nyberg, P.; Bello, I.O.; Vuoristo, J.; Sutinen, M.; Vähänikkilä, H.; Costea, D.E.; Kauppila, J.; et al. Human bone marrow mesenchymal stem cells induce collagen production and tongue cancer invasion. PLoS ONE. 2013, 8, e77692. [Google Scholar] [CrossRef]
- Rodriguez, J.; Frigola, J.; Vendrell, E.; Risques, R.-A.; Fraga, M.F.; Morales, C.; Moreno, V.; Esteller, M.; Capellà, G.; Ribas, M.; et al. Chromosomal instability correlates with genome-wide DNA demethylation in human primary colorectal cancers. Cancer Res. 2006, 66, 8462–9468. [Google Scholar] [CrossRef]
- Feinberg, A.; Ohlsson, R.; Henikoff, S. The epigenetic progenitor origin of human cancer. Nat. Rev. Genet. 2006, 7, 21–33. [Google Scholar] [CrossRef]
- Figenschau, S.L.; Knutsen, E.; Urbarova, I.; Fenton, C.; Elston, B.; Perander, M.; Mortensen, E.S.; Fenton, K.A. ICAM1 expression is induced by proinflammatory cytokines and associated with TLS formation in aggressive breast cancer subtypes. Sci. Rep. 2018, 8, 11720. [Google Scholar] [CrossRef]
- Jiao, Y.; Li, Y.; Liu, S.; Chen, Q.; Liu, Y. ITGA3 serves as a diagnostic and prognostic biomarker for pancreatic cancer. Onco. Targets Ther. 2019, 12, 4141–4152. [Google Scholar] [CrossRef]
- Gobin, E.; Bagwell, K.; Wagner, J.; Mysona, D.; Sandirasegarane, S.; Smith, N.; Bai, S.; Sharma, A.; Schleifer, R.; She, J.-X. A pan-cancer perspective of matrix metalloproteases (MMP) gene expression profile and their diagnostic/prognostic potential. BMC Cancer 2019, 19, 581. [Google Scholar] [CrossRef]
- Caplan, A.I.; Dennis, J.E. Mesenchymal stem cells as trophic mediators. J. Cell. Biochem. 2006, 98, 1076–1084. [Google Scholar] [CrossRef]
- Rustad, K.C.; Gurtner, G.C. Mesenchymal stem cells home to sites of injury and inflammation. Adv. Wound Care (New Rochelle) 2012, 1, 147–152. [Google Scholar] [CrossRef]
- Kansy, B.A.; Dißmann, P.A.; Hemeda, H.; Bruderek, K.; Westerkamp, A.M.; Jagalski, V.; Schuler, P.; Kansy, K.; Lang, S.; Dumitru, C.A.; et al. The bidirectional tumor--mesenchymal stromal cell interaction promotes the progression of head and neck cancer. Stem Cell Res. Ther. 2014, 5, 95. [Google Scholar] [CrossRef]
- Maj, M.; Bajek, A.; Nalejska, E.; Porowinska, D.; Kloskowski, T.; Gackowska, L.; Drewa, T. Influence of mesenchymal stem cells conditioned media on proliferation of urinary tract cancer cell lines and their sensitivity to ciprofloxacin. J. Cell. Biochem. 2017, 118, 1361–1368. [Google Scholar] [CrossRef]
- Li, L.; Tian, H.; Yue, W.; Zhu, F.; Li, S.; Li, W. Human mesenchymal stem cells play a dual role on tumor cell growth in vitro and in vivo. J. Cell. Physiol. 2011, 226, 1860–1867. [Google Scholar] [CrossRef]
- Howard, G.; Eiges, R.; Gaudet, F.; Jaenisch, R.; Eden, A. Activation and transposition of endogenous retroviral elements in hypomethylation induced tumors in mice. Oncogene 2008, 27, 404–408. [Google Scholar] [CrossRef]
- Mannerström, B.; Kornilov, R.; Abu-Shahba, A.G.; Chowdhury, I.M.; Sinha, S.; Seppänen-Kaijansinkko, R.; Kaur, S. Epigenetic alterations in mesenchymal stem cells by osteosarcoma-derived extracellular vesicles. Epigenetics 2019, 14, 352–364. [Google Scholar] [CrossRef]
- Di, D.; Chen, L.; Wang, L.; Sun, P.; Liu, Y.; Xu, Z.; Ju, J. Downregulation of human intercellular adhesion molecule-1 attenuates the metastatic ability in human breast cancer cell lines. Oncol. Rep. 2016, 35, 1541–1548. [Google Scholar] [CrossRef]
- Hou, C.H.; Lin, F.L.; Tong, K.B.; Hou, S.M.; Liu, J.F. Transforming growth factor alpha promotes osteosarcoma metastasis by ICAM-1 and PI3K/Akt signaling pathway. Biochem. Pharmacol. 2014, 89, 453–463. [Google Scholar] [CrossRef]
- Ho, I.A.; Chan, K.Y.; Ng, W.H.; Guo, C.M.; Hui, K.M.; Cheang, P.; Lam, P.Y. Matrix metalloproteinase 1 is necessary for the migration of human bone marrow-derived mesenchymal stem cells toward human glioma. Stem Cells 2009, 27, 1366–1375. [Google Scholar] [CrossRef]
- Zhong, P.; Liu, L.; Shen, A.; Chen, Z.; Hu, X.; Cai, Y.; Lin, J.; Wang, B.; Li, J.; Chen, Y.; et al. Five extracellular matrix-associated genes upregulated in oral tongue squamous cell carcinoma: An integrated bioinformatics analysis. Oncol. Lett. 2019, 18, 5959–5967. [Google Scholar] [CrossRef]
- Liu, X.; Yu, J.; Jiang, L.; Wang, A.; Shi, F.; Ye, H.; Zhou, X. MicroRNA-222 regulates cell invasion by targeting matrix metalloproteinase 1 (MMP1) and manganese superoxide dismutase 2 (SOD2) in tongue squamous cell carcinoma cell lines. Cancer Genom. Proteom. 2009, 6, 131–139. [Google Scholar]
- Chu, Y.; Tang, H.; Guo, Y.; Guo, J.; Huang, B.; Fang, F.; Cai, J.; Wang, Z. Adipose-derived mesenchymal stem cells promote cell proliferation and invasion of epithelial ovarian cancer. Exp. Cell Res. 2015, 337, 16–27. [Google Scholar] [CrossRef]
- Peltoniemi, H.H.; Salmi, A.; Miettinen, S.; Mannerström, B.; Saariniemi, K.; Mikkonen, R.; Kuokkanen, H.; Herold, C. Stem cell enrichment does not warrant a higher graft survival in lipofilling of the breast: A prospective comparative study. J. Plast. Reconstr. Aesthet. Surg. 2013, 66, 1494–1503. [Google Scholar] [CrossRef]
- Sobral, L.M.; Zecchin, K.G.; Nascimento de Aquino, S.; Lopes, M.A.; Graner, E.; Coletta, R.D. Isolation and characterization of myofibroblast cell lines from oral squamous cell carcinoma. Oncol. Rep. 2011, 25, 1013–1020. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society of Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Lindroos, B.; Boucher, S.; Chase, L.; Kuokkanen, H.; Huhtala, H.; Haataja, R.; Vemuri, M.; Suuronen, R.; Miettinen, S. Serum-free, xeno-free culture media maintain the proliferation rate and multipotentiality of adipose stem cells in vitro. Cytotherapy 2009, 11, 958–972. [Google Scholar] [CrossRef]
- Kaur, S.; Abu-Shahba, A.G.; Paananen, R.O.; Hongisto, H.; Hiidenmaa, H.; Skottman, H.; Seppänen-Kaijansinkko, R.; Mannerström, B. Small non-coding RNA landscape of extracellular vesicles from human stem cells. Sci. Rep. 2018, 8, 15503. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta C(t)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Pavicic, W.; Joensuu, E.I.; Nieminen, T.; Peltomäki, P. LINE-1 hypomethylation in familial and sporadic cancer. J. Mol. Med. 2012, 90, 827–835. [Google Scholar] [CrossRef]
- Sinha, S.; Mannerström, B.; Seppänen-Kaijansinkko, R.; Kaur, S. LINE-1 methylation analysis in msesnchymal stem cells treated with osteosarcoma-derived extracellular vesicles. J. Vis. Exp. 2020, 156, e60705. [Google Scholar] [CrossRef]
- Nurmenniemi, S.; Sinikumpu, T.; Alahuhta, I.; Salo, S.; Sutinen, M.; Santala, M.; Risteli, J.; Nyberg, P.; Salo, T. A novel organotypic model mimics the tumor microenvironment. Am. J. Pathol. 2009, 175, 1281–1291. [Google Scholar] [CrossRef]
- Åstrom, P.; Heljasvaara, R.; Nyberg, P.; Al-Samadi, A.; Salo, T. Human tumor tissue-based 3D in vitro invasion assays. Methods Mol. Biol. 2018, 1731, 213–221. [Google Scholar] [CrossRef]
- Salo, T.; Sutinen, M.; Hoque Apu, E.; Sundquist, E.; Cervigne, N.K.; De Oliveira, C.E.; Akram, S.U.; Ohlmeier, S.; Suomi, F.; Eklund, L.; et al. A novel human leiomyoma tissue derived matrix for cell culture studies. BMC Cancer 2015, 15, 981. [Google Scholar] [CrossRef]
- Dourado, M.R.; Korvala, J.; Åström, P.; De Oliveira, C.E.; Cervigne, N.K.; Mofatto, L.S.; Campanella Bastos, D.; Pereira Messetti, A.C.; Graner, E.; Paes Leme, A.F.; et al. Extracellular vesicles derived from cancer-associated fibroblasts induce the migration and invasion of oral squamous cell carcinoma. J. Extracell Vesicles 2019, 8, 1578525. [Google Scholar] [CrossRef]
| Marker | Clone | % Expression in Cells (Average ± Standard Deviation) | |
|---|---|---|---|
| AT-MSC (n = 5) | BM-MSC (n = 4) | ||
| CD14 | M5E2 | 1.7 ± 2.8 | 0.4 ± 0.3 |
| CD19 | HIB19 | 0.3 ± 0.4 | 0.3 ± 0.2 |
| CD34 | 581 | 31.9 ± 25.4 | 1.1 ± 1.4 |
| CD45RO | UCHL1 | 1.4 ± 2.3 | 0.5 ± 0.3 |
| CD54 | HA58 | 61.8 ± 22.8 | 12.4 ± 2.2 |
| CD73 | AD2 | 99.0 ± 1.8 | 99.5 ± 0.3 |
| CD90 | 5E10 | 99.3 ± 0.7 | 94.6 ± 5.1 |
| CD105 | 266 | 96.1 ± 7.1 | 98.2 ± 1.0 |
| HLA-DR | G46-6 | 1.7 ± 3.1 | 1.5 ± 0.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sinha, S.; Narjus-Sterba, M.; Tuomainen, K.; Kaur, S.; Seppänen-Kaijansinkko, R.; Salo, T.; Mannerström, B.; Al-Samadi, A. Adipose-Derived Mesenchymal Stem Cells do not Affect the Invasion and Migration Potential of Oral Squamous Carcinoma Cells. Int. J. Mol. Sci. 2020, 21, 6455. https://doi.org/10.3390/ijms21186455
Sinha S, Narjus-Sterba M, Tuomainen K, Kaur S, Seppänen-Kaijansinkko R, Salo T, Mannerström B, Al-Samadi A. Adipose-Derived Mesenchymal Stem Cells do not Affect the Invasion and Migration Potential of Oral Squamous Carcinoma Cells. International Journal of Molecular Sciences. 2020; 21(18):6455. https://doi.org/10.3390/ijms21186455
Chicago/Turabian StyleSinha, Snehadri, Matilda Narjus-Sterba, Katja Tuomainen, Sippy Kaur, Riitta Seppänen-Kaijansinkko, Tuula Salo, Bettina Mannerström, and Ahmed Al-Samadi. 2020. "Adipose-Derived Mesenchymal Stem Cells do not Affect the Invasion and Migration Potential of Oral Squamous Carcinoma Cells" International Journal of Molecular Sciences 21, no. 18: 6455. https://doi.org/10.3390/ijms21186455
APA StyleSinha, S., Narjus-Sterba, M., Tuomainen, K., Kaur, S., Seppänen-Kaijansinkko, R., Salo, T., Mannerström, B., & Al-Samadi, A. (2020). Adipose-Derived Mesenchymal Stem Cells do not Affect the Invasion and Migration Potential of Oral Squamous Carcinoma Cells. International Journal of Molecular Sciences, 21(18), 6455. https://doi.org/10.3390/ijms21186455






