Monitoring of the Surface Charge Density Changes of Human Glioblastoma Cell Membranes upon Cinnamic and Ferulic Acids Treatment
Abstract
1. Introduction
2. Results
2.1. The Influence of CA and FA on Cell Viability
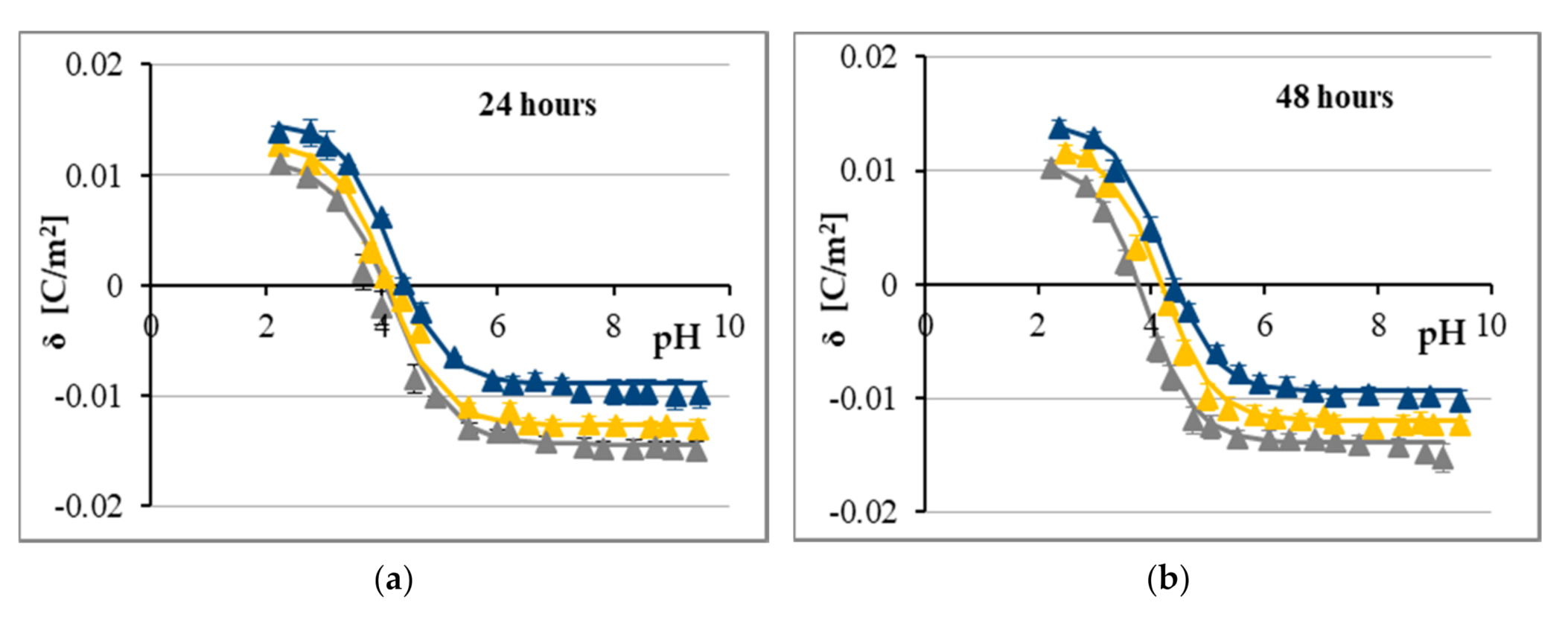
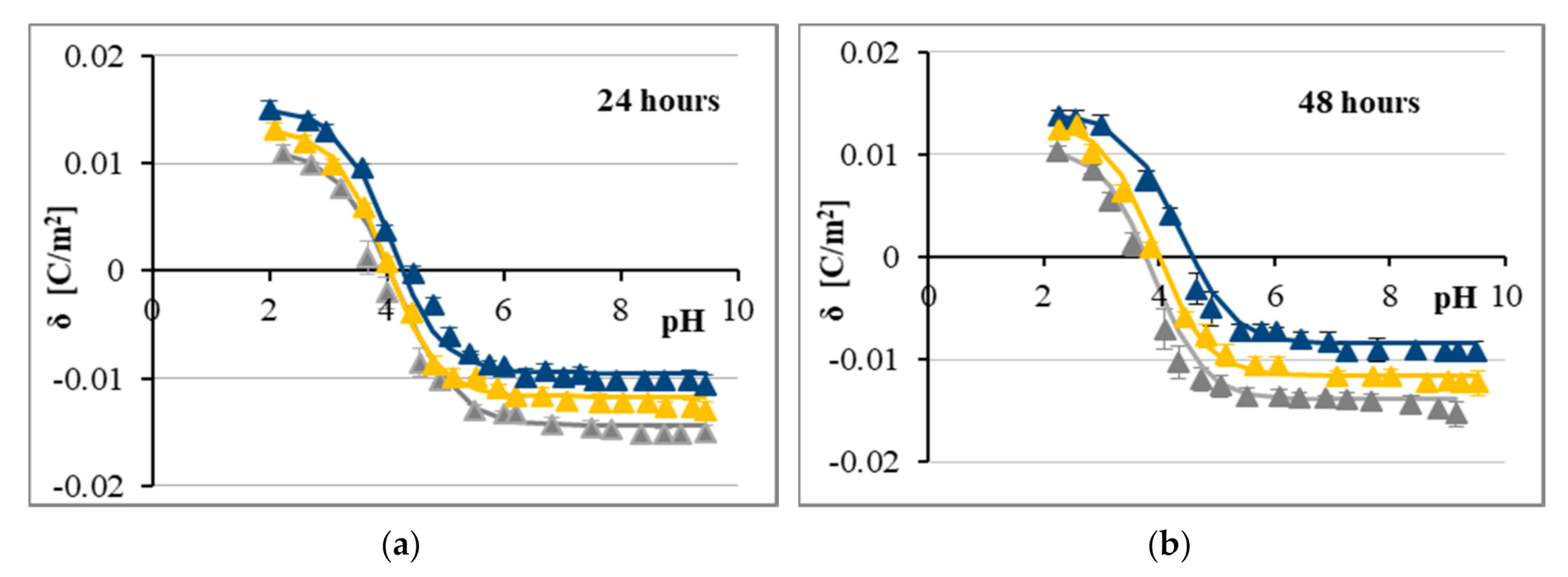
2.2. The Influence of CA and FA on Surface Charge Densities of Cell Membranes
3. Discussion
4. Materials and Methods
4.1. Reagents
Cell Culture and MTT Assay
4.2. Methods
4.2.1. Microelectrophoretic Mobility Measurements
4.2.2. Experimental Surface Charge Density Determination
4.2.3. Theoretical Surface Charge Density Determination
- -
- the equation describing surface charge density of the cell membrane is
- -
- the equations obtained by simplifications of Equation (3) to a linear form at low H+ and high H+ ion concentrations arein whichσ is the surface charge density;aH+, aNa+, aOH−, aCl—are the volume concentrations of solution ions;CTA is the total surface concentration of the membrane acidic groups;CTB is the total surface concentration of the membrane basic groups;KAH, KANa, KBOH, KBC are associations constants;F is the Faraday constant.
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CA | Cinnamic acid |
| FA | Ferulic acid |
| GBM | Glioblastoma multiforme |
| BBB | Blood–brain barrier |
| ELS | Electrophoretic light scattering |
| σ | Surface charge density |
| CTA | Concentration of functional acidic groups |
| CTB | Concentration of functional basic groups |
| KAH | Association constant with hydrogen ions |
| KBOH | Association constant with hydroxyl ions |
| MWT | Molecular weight |
| clogP | Calculated n-octanol/water partition coefficient |
| HBA | Hydrogen bond acceptor groups |
| HBD | Hydrogen bond donor groups |
| RB | Rotable bonds |
References
- Matejczyk, M.; Świsłocka, R.; Golonko, A.; Lewandowski, W.; Hawrylik, E. Cytotoxic, genotoxic and antimicrobial activity of caffeic and rosmarinic acids and their lithium, sodium and potassium salts as potential anticancer compounds. Adv. Med. Sci. 2018, 63, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.; Bravo, J.; Summavielle, T.; Remião, F.; Pérez, C.; Gil, C.; Martínez, A.; Borges, F. Biology-oriented development of novel lipophilic antioxidants with neuroprotective activity. RSC Adv. 2015, 5, 15800–15811. [Google Scholar] [CrossRef][Green Version]
- El-Seedi, H.R.; El-Said, A.M.A.; Khalifa, S.A.M.; Göransson, U.; Bohlin, L.; Borg-Karlson, A.K.; Verpoorte, R. Biosynthesis, natural sources, dietary intake, pharmacokinetic properties, and biological activities of hydroxycinnamic acids. J. Agric. Food Chem. 2012, 60, 10877–10895. [Google Scholar] [CrossRef]
- Carrera, E.; Vall Ould El Kebir, M.; Jacquemond, C.; Luro, F.; Lozano, Y.; Gaydou, E.M. Free and bound cinnamic acid derivatives in Corsica sweet blond oranges. Nat. Prod. Commun. 2010, 5, 435–550. [Google Scholar] [CrossRef]
- Rosazza, J.P.; Huang, Z.; Dostal, L.; Volm, T.; Rousseau, B. Review: Biocatalytic transformations of ferulic acid: An abundant aromatic natural product. J. Ind. Microbiol. 1995, 15, 457–471. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Kaji, K.; Matsuo, M. Protection of linoleic acid hydroperoxide-induced cytotoxicity by phenolic antioxidants. Free Radic. Biol. Med. 1994, 16, 405–409. [Google Scholar] [CrossRef]
- Lohner, K. Effects of small organic molecules on phospholidpid phase transitions. Chem. Phys. Lipids 1991, 57, 341–362. [Google Scholar] [CrossRef]
- Castelli, F.; Uccella, N.; Trombetta, D.; Saija, A. Differences between coumaric and cinnamic acids in membrane permeation as evidenced by time-dependent calorimetry. J. Agric. Food Chem. 1999, 47, 991–995. [Google Scholar] [CrossRef]
- Cumming, H.; Rücker, C. Octanol-water partition coefficient measurement by a simple 1 H NMR method. ACS Omega 2017, 2, 6244–6249. [Google Scholar] [CrossRef]
- Giménez, B.G.; Santos, M.S.; Ferrarini, M.; Dos Santos Fernandes, J.P. Evaluation of blockbuster drugs under the rule-of-five. Pharmazie 2010, 65, 148–152. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Kuroda, Y.; Watanabe, Y.; Shibukawa, A.; Nakagawa, T. Role of phospholipids in drug-LDL bindings as studied by high-performance frontal analysis/capillary electrophoresis. J. Pharm. Biomed. Anal. 2003, 30, 1869–1877. [Google Scholar] [CrossRef]
- Smith, M.C.; Crist, R.M.; Clogston, J.D.; McNeil, S.E. Zeta potential: A case study of cationic, anionic, and neutral liposomes. Anal. Bioanal. Chem. 2017, 409, 5779–5787. [Google Scholar] [CrossRef]
- Kusaczuk, M.; Krętowski, R.; Naumowicz, M.; Stypułkowska, A.; Cechowska-Pasko, M. Silica nanoparticle-induced oxidative stress and mitochondrial damage is followed by activation of intrinsic apoptosis pathway in glioblastoma cells. Int. J. Nanomed. 2018, 13, 2279–2294. [Google Scholar] [CrossRef] [PubMed]
- Krętowski, R.; Kusaczuk, M.; Naumowicz, M.; Kotyńska, J.; Szynaka, B.; Cechowska-Pasko, M. The effects of silica nanoparticles on apoptosis and autophagy of glioblastoma cell lines. Nanomaterials 2017, 7, 230. [Google Scholar] [CrossRef]
- Kruszewski, M.; Kusaczuk, M.; Kotyńska, J.; Gál, M.; Krętowski, R.; Cechowska-Pasko, M.; Naumowicz, M. The effect of quercetin on the electrical properties of model lipid membranes and human glioblastoma cells. Bioelectrochemistry 2018, 124, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Naumowicz, M.; Kusaczuk, M.; Kruszewski, M.A.; Gál, M.; Krętowski, R.; Cechowska-Pasko, M.; Kotyńska, J. The modulating effect of lipid bilayer/p-coumaric acid interactions on electrical properties of model lipid membranes and human glioblastoma cells. Bioorg. Chem. 2019, 92, 103242. [Google Scholar] [CrossRef] [PubMed]
- Kruszewski, M.A.; Kotyńska, J.; Kusaczuk, M.; Gál, M.; Naumowicz, M. The modulating effect of p-coumaric acid on the surface charge density of human glioblastoma cell membranes. Int. J. Mol. Sci. 2019, 20, 5286. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Hudgins, W.R.; Shack, S.; Yin, M.Q.; Samid, D. Cinnamic acid: A natural product with potential use in cancer intervention. Int. J. Cancer 1995, 62, 345–350. [Google Scholar] [CrossRef]
- Niero, E.L.D.O.; MacHado-Santelli, G.M. Cinnamic acid induces apoptotic cell death and cytoskeleton disruption in human melanoma cells. J. Exp. Clin. Cancer Res. 2013, 32, 31. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Shang, B.; Li, Y.; Zhen, Y. Inhibition of histone deacetylases by trans-cinnamic acid and its antitumor effect against colon cancer xenografts in athymic mice. Mol. Med. Rep. 2016, 13, 4159–4166. [Google Scholar] [CrossRef] [PubMed]
- Ekmekcioglu, C.; Feyertag, J.; Marktl, W. Cinnamic acid inhibits proliferation and modulates brush border membrane enzyme activities in Caco-2 cells. Cancer Lett. 1998, 128, 137–144. [Google Scholar] [CrossRef]
- Rosa, L.S.; Silva, N.J.A.; Soares, N.C.P.; Monteiro, M.C.; Teodoro, A.J. Anticancer properties of phenolic acids in colon cancer—A review. J. Nutr. Food Sci. 2016, 6, 1000468. [Google Scholar] [CrossRef]
- Qi, G.; Chen, J.; Shi, C.; Wang, Y.; Mi, S.; Shao, W.; Yu, X.; Ma, Y.; Ling, J.; Huang, J. Cinnamic acid (CINN) induces apoptosis and oroliferation in human nasopharyngeal carcinoma cells. Cell. Physiol. Biochem. 2016, 40, 589–596. [Google Scholar] [CrossRef]
- Eroğlu, C.; Seçme, M.; Bağcı, G.; Dodurga, Y. Assessment of the anticancer mechanism of ferulic acid via cell cycle and apoptotic pathways in human prostate cancer cell lines. Tumor Biol. 2015, 36, 9437–9446. [Google Scholar] [CrossRef]
- Ferguson, L.R.; Zhu, S.T.; Harris, P.J. Antioxidant and antigenotoxic effects of plant cell wall hydroxycinnamic acids in cultured HT-29 cells. Mol. Nutr. Food Res. 2005, 49, 585–593. [Google Scholar] [CrossRef]
- Bouzaiene, N.N.; Soumaya, K.J.; Kovacic, H.; Chekir-Ghedira, L.; Ghedira, K.; Luis, J. The effects of caffeic, coumaric and ferulic acids on proliferation, superoxide production, adhesion and migration of human tumor cells in vitro. Eur. J. Pharmacol. 2015, 766, 99–105. [Google Scholar] [CrossRef]
- Fahrioğlu, U.; Dodurga, Y.; Elmas, L.; Seçme, M. Ferulic acid decreases cell viability and colony formation while inhibiting migration of MIA PaCa-2 human pancreatic cancer cells in vitro. Gene 2016, 576, 476–482. [Google Scholar] [CrossRef]
- Wang, T.; Gong, X.; Jiang, R.; Li, H.; Du, W.; Kuang, G. Ferulic acid inhibits proliferation and promotes apoptosis via blockage of PI3K/Akt pathway in osteosarcoma cell. Am. J. Transl. Res. 2016, 8, 968–980. [Google Scholar]
- López-Valero, I.; Saiz-Ladera, C.; Torres, S.; Hernández-Tiedra, S.; García-Taboada, E.; Rodríguez-Fornés, F.; Barba, M.; Dávila, D.; Salvador-Tormo, N.; Guzmán, M.; et al. Targeting Glioma Initiating Cells with A combined therapy of cannabinoids and temozolomide. Biochem. Pharmacol. 2018, 157, 266–274. [Google Scholar] [CrossRef]
- Colella, B.; Colardo, M.; Iannone, G.; Contadini, C.; Saiz-Ladera, C.; Fuoco, C.; Barilà, D.; Velasco, G.; Segatto, M.; Di Bartolomeo, S. Mtor inhibition leads to src-mediated egfr internalisation and degradation in glioma cells. Cancers 2020, 12, 2266. [Google Scholar] [CrossRef] [PubMed]
- Kusaczuk, M.; Krętowski, R.; Bartoszewicz, M.; Cechowska-Pasko, M. Phenylbutyrate—A pan-HDAC inhibitor—Suppresses proliferation of glioblastoma LN-229 cell line. Tumor Biol. 2016, 37, 931–942. [Google Scholar] [CrossRef]
- Han, M.; Li, J.-F.; Tan, Q.; Sun, Y.-Y.; Wang, Y.-Y. Limitations of the use of MTT assay for screening in drug discovery. J. Chin. Pharm. Sci. 2010, 19, 195–200. [Google Scholar] [CrossRef]
- Albatany, M.; Meakin, S.; Bartha, R. The monocarboxylate transporter inhibitor quercetin induces intracellular acidification in a mouse model of glioblastoma multiforme: In-vivo detection using magnetic resonance imaging. Investig. New Drugs 2019, 37, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Velasco, G.; Carracedo, A.; Blázquez, C.; Lorente, M.; Aguado, T.; Haro, A.; Sánchez, C.; Galve-Roperh, I.; Guzmán, M. Cannabinoids and gliomas. Mol. Neurobiol. 2007, 36, 60–67. [Google Scholar] [CrossRef]
- Jain, V.; Langham, M.C.; Wehrli, F.W. MRI estimation of global brain oxygen consumption rate. J. Cereb. Blood Flow Metab. 2010, 30, 1598–1607. [Google Scholar] [CrossRef] [PubMed]
- Rajaraman, P.; Hutchinson, A.; Rothman, N.; Black, P.M.; Fine, H.A.; Loeffler, J.S.; Selker, R.G.; Shapiro, W.R.; Linet, M.S.; Inskip, P.D. Oxidative response gene polymorphisms and risk of adult brain tumors. Neuro. Oncol. 2008, 10, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Vidak, M.; Rozman, D.; Komel, R. Effects of flavonoids from food and dietary supplements on glial and glioblastoma multiforme cells. Molecules 2015, 20, 19406–19432. [Google Scholar] [CrossRef]
- Rossi, L.; Mazzitelli, S.; Arciello, M.; Capo, C.R.; Rotilio, G. Benefits from dietary polyphenols for brain aging and Alzheimer’s disease. Neurochem. Res. 2008, 33, 2390–2400. [Google Scholar] [CrossRef]
- Kelloff, G.J.; Crowell, J.A.; Hawk, E.T.; Steele, V.E.; Lubet, R.A.; Boone, C.W.; Covey, J.M.; Doody, L.A.; Omenn, G.S.; Greenwald, P.; et al. Strategy and planning for chemopreventive drug development: Clinical development plans II. J. Cell. Biochem. 1996, 63, 54–71. [Google Scholar] [CrossRef]
- Singh, M.; Arseneault, M.; Sanderson, T.; Murthy, V.; Ramassamy, C. Challenges for research on polyphenols from foods in Alzheimer’s disease: Bioavailability, metabolism, and cellular and molecular mechanisms. J. Agric. Food Chem. 2008, 56, 4855–4873. [Google Scholar] [CrossRef] [PubMed]
- Pajouhesh, H.; Lenz, G.R. Medicinal chemical properties of successful central nervous system drugs. NeuroRx 2005, 2, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Veber, D.F.; Johnson, S.R.; Cheng, H.Y.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular properties that influence the oral bioavailability of drug candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M.; Goulas, V.; Tsakona, S.; Manganaris, G.A.; Gekas, V. A knowledge base for the recovery of natural phenols with different solvents. Int. J. Food Prop. 2013, 16, 382–396. [Google Scholar] [CrossRef]
- Jablonsky, M.; Haz, A.; Burcova, Z.; Kreps, F.; Jablonsky, J. Pharmacokinetic properties of biomass-extracted substances isolated by green solvents. BioResources 2019, 14, 6294–6303. [Google Scholar] [CrossRef]
- Anantharaju, P.G.; Gowda, P.C.; Vimalambike, M.G.; Madhunapantula, S.V. An overview on the role of dietary phenolics for the treatment of cancers. Nutr. J. 2016, 15, 99. [Google Scholar] [CrossRef]
- Gennis, R.B. Biomembranes: Molecular Structure and Functions; Springer: New York, NY, USA, 1989; ISBN 0-387-96760-5. [Google Scholar]
- Heileman, K.; Daoud, J.; Tabrizian, M. Dielectric spectroscopy as a viable biosensing tool for cell and tissue characterization and analysis. Biosens. Bioelectron. 2013, 49, 348–359. [Google Scholar] [CrossRef]
- Beech, J.A. A theory of carcinogenesis based on an analysis of the effects of carcinogens. Med. Hypotheses 1987, 24, 265–286. [Google Scholar] [CrossRef]
- Camero, I.L.; Smith, N.K.R.; Pool, T.B.; Sparks, R.L. Intracellular concentration of sodium and other elements as related to mitogenesis and oncogenesis in vivo. Cancer Res. 1980, 40, 1493–1500. [Google Scholar]
- Seeger, P.G.; Wolz, S. Successful Biological Control of Cancer by Combat against the Causes; Neuwieder Verlagsgesellschaft: Neuweid, Germany, 1990. [Google Scholar]
- Erbil, K.M.; Sen, S.E.; Zincke, H.; Jones, J.D. Significance of serum protein and lipid-bound sialic acid as a marker for genitourinary malignancies. Cancer 1986, 57, 1389–1394. [Google Scholar] [CrossRef]
- Szachowicz-Petelska, B.; Dobrzynska, I.; Sulkowski, S.; Figaszewski, Z.A. Characterization of the cell membrane during cancer transformation. In Colorectal Cancer Biology-From Genes to Tumor; Ettarh, R., Ed.; InTech: Rijeka, Croatia, 2012; pp. 241–256. [Google Scholar]
- Spugnini, E.P.; Sonveaux, P.; Stock, C.; Perez-Sayans, M.; De Milito, A.; Avnet, S.; Garcìa, A.G.; Harguindey, S.; Fais, S. Proton channels and exchangers in cancer. Biochim. Biophys. Acta Biomembr. 2015, 1848, 2715–2726. [Google Scholar] [CrossRef] [PubMed]
- Dolowy, K. Bioelectrochemistry of cell surface. Prog. Surf. Sci. 1984, 15, 245–368. [Google Scholar] [CrossRef]
- Petelska, A.D.; Kotyńska, J.; Naumowicz, M.; Figaszewski, Z.A. Equilibria between cell membranes and electrolyte solution: Effect of fatal accidental hypothermia. J. Membr. Biol. 2016, 249, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Dobrzyńska, I.; Skrzydlewska, E.; Figaszewski, Z.A. Effects of novel dinuclear cisplatinum(II) complexes on the electrical properties of human molt-4 leukemia cells. Cell Biochem. Biophys. 2015, 71, 1517–1523. [Google Scholar] [CrossRef][Green Version]
- Dobrzyńska, I.; Skrzydlewska, E.; Figaszewski, Z.A. Effects of novel dinuclear cisplatinum(II) complexes on the electric properties of human breast cancer cells. J. Membr. Biol. 2014, 247, 167–173. [Google Scholar] [CrossRef][Green Version]
- Sirk, T.W.; Brown, E.F.; Sum, A.K.; Friedman, M. Molecular dynamics study on the biophysical interactions of seven green tea catechins with lipid bilayers of cell membranes. J. Agric. Food Chem. 2008, 56, 7750–7758. [Google Scholar] [CrossRef]
- Chen, M.; Meng, H.; Zhao, Y.; Chen, F.; Yu, S. Antioxidant and in vitro anticancer activities of phenolics isolated from sugar beet molasses. BMC Complement Altern. Med. 2015, 15, 313. [Google Scholar] [CrossRef]
- Habauzit, V.; Morand, C. Evidence for a protective effect of polyphenols-containing foods on cardiovascular health: An update for clinicians. Ther. Adv. Chronic Dis. 2012, 3, 87–106. [Google Scholar] [CrossRef]
- Carmichael, J.; Degraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a tetrazolium-based of semiautomated assay: Colorimetric assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–942. [Google Scholar]
- Alexander, A.E.; Johnson, P. Colloid Science; Clarendon Press: Oxford, UK, 1949. [Google Scholar]
- Barrow, G.M. Physical Chemistry; McGraw-Hill: New York, NY, USA, 1996. [Google Scholar]
- Dobrzynska, I.; Skrzydlewska, E.; Figaszewski, Z. Parameters characterizing acid-base equilibria between cell membrane and solution and their application to monitoring the effect of various factors on the membrane. Bioelectrochemistry 2006, 69, 142–147. [Google Scholar] [CrossRef]

| Phenolic Acid | Type of Cancers | Cell Lines Used | Reported IC50 1 [mmol/dm3] | Reference |
|---|---|---|---|---|
 Cinnamic acid (CA) Cinnamic acid (CA) | Glioblastoma | A172 | 4.50 ± 0.50 | [19] |
| U251 | 4.00 ± 1.00 | [19] | ||
| Melanoma | HT-144 | 2.40 | [20] | |
| MEL 1011 | 2.40 ± 1.00 | [19] | ||
| AS75(M) | 1.00 ± 0.20 | [19] | ||
| SKMEL28 | 2.50 ± 0.10 | [19] | ||
| Prostate | PC3(M) | 2.70 ± 0.50 | [19] | |
| Du145 | 4.00 ± 0.50 | [19] | ||
| LNCaP | 1.90 ± 1.00 | [19] | ||
| Colon | HT-29 | 1.00 | [21] | |
| Caco-2 | 4.00–5.00 | [22] | ||
| HCT 15 | 0.80 | [23] | ||
| Nasopharyngeal | CNE2 | −−− 2 | [24] | |
| Lung | A549 | 1.20 ± 0.10 | [19] | |
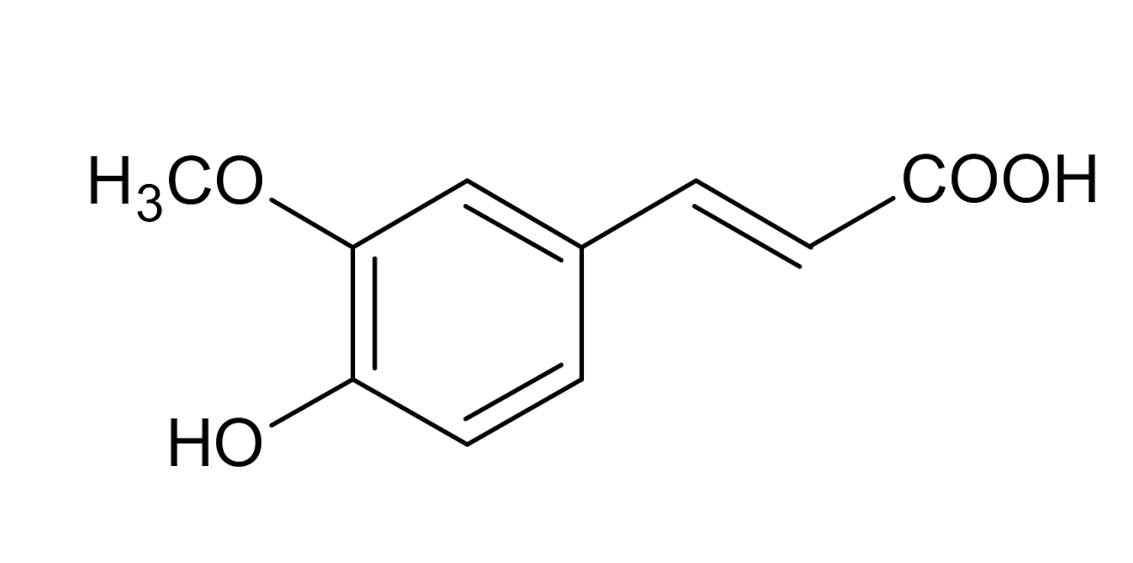 Ferulic acid (FA) Ferulic acid (FA) | Prostate | PC-3 | 0.30 | [25] |
| LNCaP | 0.50 | [25] | ||
| Colon | HT-29 | 1.21 ± 0.07 | [26] | |
| LOVO | 0.69 ± 0.10 | [26] | ||
| HT29-D4 | −−− 2 | [27] | ||
| HCT-8 | 1.12 ± 0.03 | [26] | ||
| Lung | A549 | −−− 2 | [27] | |
| Breast | EMT-6 | 0.87 ± 0.09 | [26] | |
| Pancreatic | MIA-Pa-Ca-2 | 0.50 | [28] | |
| Lymphoma | SW-620 | 0.98 ± 0.12 | [26] | |
| Osteosarcoma | 143B | 0.60 | [29] |
| Compound | System | Parameters | |||
|---|---|---|---|---|---|
| CTA | CTB | KAH | KBOH | ||
| (10−6 mol/m2) | (10−6 mol/m2) | (m3/mol) | (107 m3/mol) | ||
| CA | LN-229 (24 h) | 4.18 ± 0.07 | 1.16 ± 0.07 | 194.30 ± 1.12 | 7.39 ± 0.09 |
| +1.0 mmol/dm3 | 3.39 ± 0.04 | 1.37 ± 0.07 | 208.10 ± 1.14 | 5.22 ± 0.08 | |
| +3.5 mmol/dm3 | 2.75 ± 0.10 | 1.56 ± 0.04 | 284.00 ± 1.09 | 3.85 ± 0.09 | |
| LN-229 (48 h) | 4.04 ± 0.05 | 1.11 ± 0.04 | 175.00 ± 1.15 | 9.24 ± 0.04 | |
| +1.0 mmol/dm3 | 3.51 ± 0.10 | 1.24 ± 0.03 | 308.20 ± 1.13 | 6.42 ± 0.17 | |
| +3.5 mmol/dm3 | 2.73 ± 0.06 | 1.45 ± 0.04 | 343.00 ± 1.22 | 4.30 ± 0.07 | |
| FA | LN-229 (24 h) | 4.18 ± 0.07 | 1.16 ± 0.07 | 194.30 ± 1.12 | 7.39 ± 0.09 |
| +1.0 mmol/dm3 | 3.67 ± 0.12 | 1.33 ± 0.05 | 270.40 ± 1.05 | 5.72 ± 0.10 | |
| +5.0 mmol/dm3 | 2.59 ± 0.08 | 1.51 ± 0.05 | 336.10 ± 1.08 | 4.56 ± 0.14 | |
| LN-229 (48 h) | 4.04 ± 0.05 | 1.11 ± 0.04 | 175.00 ± 1.15 | 9.24 ± 0.04 | |
| +1.0 mmol/dm3 | 3.37 ± 0.07 | 1.35 ± 0.03 | 208.00 ± 1.07 | 6.74 ± 0.15 | |
| +5.0 mmol/dm3 | 2.44 ± 0.04 | 1.44 ± 0.03 | 311.50 ± 1.18 | 3.04 ± 0.14 | |
| Compound | MWT1 | clogP2 | HBA 3 | HBD 4 | RB 5 |
|---|---|---|---|---|---|
| CA | 148.16 | 1.98 [44] | 2 [45] | 1 [45] | 2 [45] |
| FA | 194.18 | 1.42 [44] | 4 [45] | 2 [45] | 3 [2] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naumowicz, M.; Kusaczuk, M.; Zając, M.; Gál, M.; Kotyńska, J. Monitoring of the Surface Charge Density Changes of Human Glioblastoma Cell Membranes upon Cinnamic and Ferulic Acids Treatment. Int. J. Mol. Sci. 2020, 21, 6972. https://doi.org/10.3390/ijms21186972
Naumowicz M, Kusaczuk M, Zając M, Gál M, Kotyńska J. Monitoring of the Surface Charge Density Changes of Human Glioblastoma Cell Membranes upon Cinnamic and Ferulic Acids Treatment. International Journal of Molecular Sciences. 2020; 21(18):6972. https://doi.org/10.3390/ijms21186972
Chicago/Turabian StyleNaumowicz, Monika, Magdalena Kusaczuk, Marcin Zając, Miroslav Gál, and Joanna Kotyńska. 2020. "Monitoring of the Surface Charge Density Changes of Human Glioblastoma Cell Membranes upon Cinnamic and Ferulic Acids Treatment" International Journal of Molecular Sciences 21, no. 18: 6972. https://doi.org/10.3390/ijms21186972
APA StyleNaumowicz, M., Kusaczuk, M., Zając, M., Gál, M., & Kotyńska, J. (2020). Monitoring of the Surface Charge Density Changes of Human Glioblastoma Cell Membranes upon Cinnamic and Ferulic Acids Treatment. International Journal of Molecular Sciences, 21(18), 6972. https://doi.org/10.3390/ijms21186972





