Molecular and Structural Effects of Percutaneous Interventions in Chronic Achilles Tendinopathy
Abstract
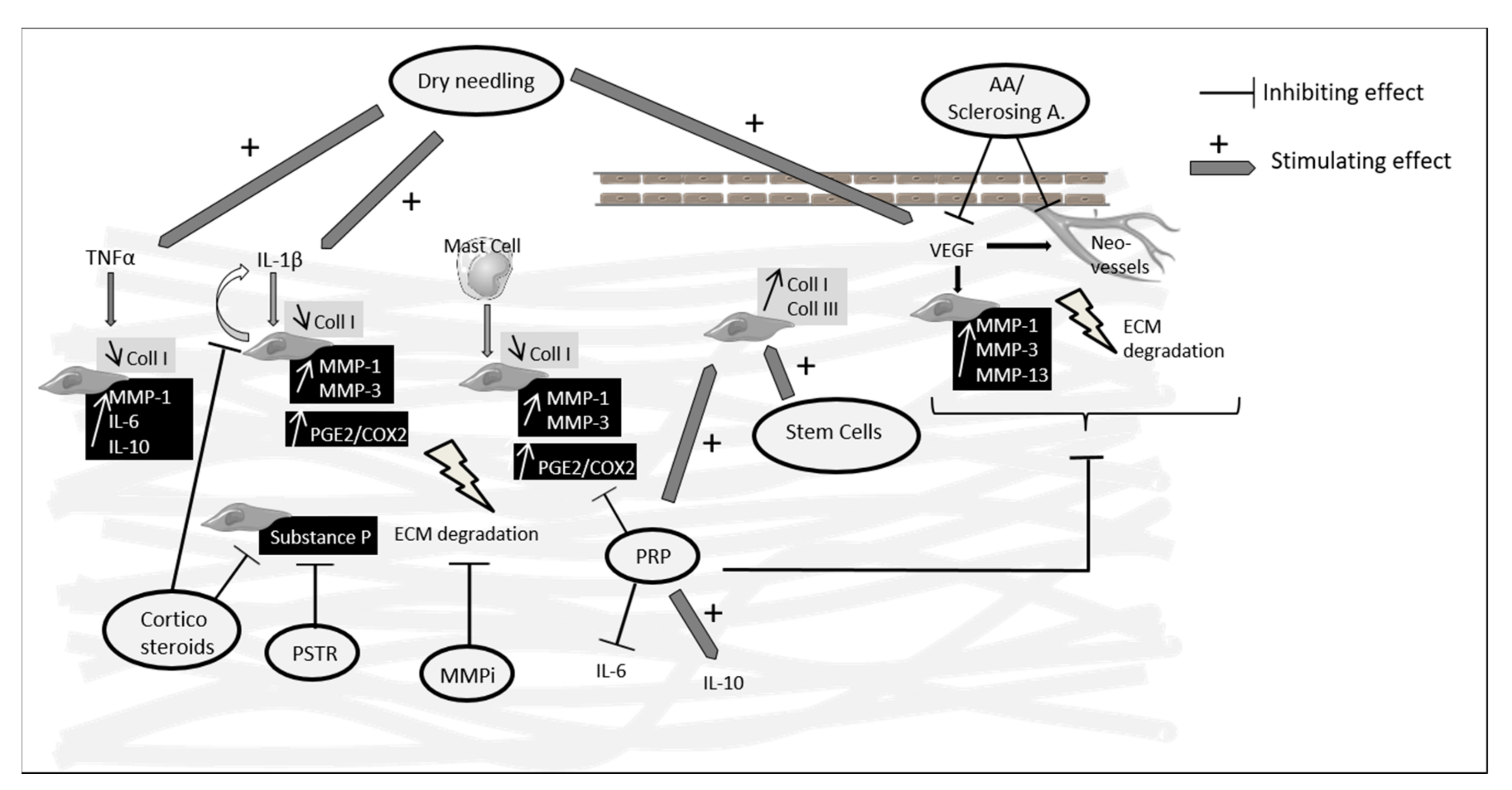
:1. Introduction
2. Ultrasound-Guided Injections
2.1. Platelet-Rich Plasma
2.2. Corticosteroids
2.3. Stem Cells and Autologous Tenocyte Injections
2.4. Sclerosing and Anti-Angiogenic Agents (AA)
2.5. MMP Inhibitors (Aprotinin)
3. Ultrasound-Guided Procedures Without Injection
3.1. Percutaneous Soft Tissue Release (PRST)
3.2. Percutaneous Tenotomy/Dry Needling
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AA | Anti-angiogenic agents |
| AT | Achilles tendinopathy |
| AT-MSCs | Adipose tissue-derived mesenchymal stem cells |
| CS | Corticosteroids |
| Coll | Collagen |
| COX | Cyclooxygenase |
| ECM | Extracellular matrix |
| HGF | Hepatocyte growth factor |
| IL | Interleukin |
| Lr-PRP | Leucocyte-rich PRP |
| Lp-PRP | Leucocyte-poor PRP |
| MIF | Macrophage migration inhibitory factor |
| MMPs | Matrix metalloproteinases |
| MSC | Mesenchymal stem cells |
| PGE2 | Prostaglandine E2 |
| PRP | Platelet-rich plasma |
| PRST | Percutaneous soft tissue release |
| TEM | Transmission electron microscopy |
| TGF-β | Transforming growth factor β |
| TIMPs | Tissue inhibitors of metalloproteinase |
| TLC | Tenocyte-like cells |
| TNF | Tumor necrosing factor |
| TSCs | Tendon stem cells |
| SP | Substance P |
| VAS | Visual analogic scale |
| VEGF | Vascular endothelial growth factor |
| VISA | Victorian Institute of Sports Assessment self-administered |
References
- De Jonge, S.; van den Berg, C.; de Vos, R.J.; van der Heide, H.J.L.; Weir, A.; Verhaar, J.A.N.; Bierma-Zeinstra, S.M.A.; Tol, J.L. Incidence of midportion Achilles tendinopathy in the general population. Br. J. Sports Med. 2011, 45, 1026–1028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yelin, E.; Weinstein, S.; King, T. The burden of musculoskeletal diseases in the United States. Semin. Arthritis Rheum. 2016, 46, 259–260. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-Y.; Hua, Y.-H. Achilles Tendinopathy: Current Concepts about the Basic Science and Clinical Treatments. BioMed Res. Int. 2016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riley, G. Tendinopathy—From basic science to treatment. Nat. Clin. Pract. Rheumatol. 2008, 4, 82–89. [Google Scholar] [CrossRef] [Green Version]
- Riley, G.P. Gene expression and matrix turnover in overused and damaged tendons. Scand. J. Med. Sci. Sports 2005, 15, 241–251. [Google Scholar] [CrossRef] [Green Version]
- Jones, G.C.; Corps, A.N.; Pennington, C.J.; Clark, I.M.; Edwards, D.R.; Bradley, M.M.; Hazleman, B.L.; Riley, G.P. Expression profiling of metalloproteinases and tissue inhibitors of metalloproteinases in normal and degenerate human achilles tendon. Arthritis Rheum. 2006, 54, 832–842. [Google Scholar] [CrossRef] [Green Version]
- Chisari, E.; Rehak, L.; Khan, W.S.; Maffulli, N. Tendon healing in presence of chronic low-level inflammation: A systematic review. Br. Med. Bull. 2019, 132, 97–116. [Google Scholar] [CrossRef]
- John, T.; Lodka, D.; Kohl, B.; Ertel, W.; Jammrath, J.; Conrad, C.; Stoll, C.; Busch, C.; Schulze-Tanzil, G. Effect of pro-inflammatory and immunoregulatory cytokines on human tenocytes. J. Orthop. Res. 2010, 28, 1071–1077. [Google Scholar] [CrossRef]
- Millar, N.L.; Wei, A.Q.; Molloy, T.J.; Bonar, F.; Murrell, G.A.C. Cytokines and apoptosis in supraspinatus tendinopathy. J. Bone Joint Surg. Br. 2009, 91, 417–424. [Google Scholar] [CrossRef] [Green Version]
- Dean, B.J.F.; Gettings, P.; Dakin, S.G.; Carr, A.J. Are inflammatory cells increased in painful human tendinopathy? A systematic review. Br. J. Sports Med. 2016, 50, 216–220. [Google Scholar] [CrossRef]
- Pingel, J.; Wienecke, J.; Kongsgaard, M.; Behzad, H.; Abraham, T.; Langberg, H.; Scott, A. Increased mast cell numbers in a calcaneal tendon overuse model. Scand. J. Med. Sci. Sports 2013, 23, e353–e360. [Google Scholar] [CrossRef] [PubMed]
- Behzad, H.; Sharma, A.; Mousavizadeh, R.; Lu, A.; Scott, A. Mast cells exert pro-inflammatory effects of relevance to the pathophyisology of tendinopathy. Arthritis Res. Ther. 2013, 15, R184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warden, S.J. Animal models for the study of tendinopathy. Br. J. Sports Med. 2007, 41, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Soslowsky, L.J.; Thomopoulos, S.; Tun, S.; Flanagan, C.L.; Keefer, C.C.; Mastaw, J.; Carpenter, J.E. Neer Award 1999. Overuse activity injures the supraspinatus tendon in an animal model: A histologic and biomechanical study. J. Shoulder Elbow Surg. 2000, 9, 79–84. [Google Scholar] [CrossRef]
- Huang, T.-F.; Perry, S.M.; Soslowsky, L.J. The effect of overuse activity on Achilles tendon in an animal model: A biomechanical study. Ann. Biomed. Eng. 2004, 32, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Glazebrook, M.A.; Wright, J.R.; Langman, M.; Stanish, W.D.; Lee, J.M. Histological analysis of achilles tendons in an overuse rat model. J. Orthop. Res. 2008, 26, 840–846. [Google Scholar] [CrossRef]
- Lui, P.P.Y.; Maffulli, N.; Rolf, C.; Smith, R.K.W. What are the validated animal models for tendinopathy? Scand. J. Med. Sci. Sports 2011, 21, 3–17. [Google Scholar] [CrossRef]
- Malliaras, P.; Barton, C.J.; Reeves, N.D.; Langberg, H. Achilles and patellar tendinopathy loading programmes: A systematic review comparing clinical outcomes and identifying potential mechanisms for effectiveness. Sports Med. 2013, 43, 267–286. [Google Scholar] [CrossRef]
- Roche, A.J.; Calder, J.D.F. Achilles tendinopathy: A review of the current concepts of treatment. Bone Jt. J. 2013, 95, 1299–1307. [Google Scholar] [CrossRef]
- Roos, E.M.; Engström, M.; Lagerquist, A.; Söderberg, B. Clinical improvement after 6 weeks of eccentric exercise in patients with mid-portion Achilles tendinopathy — a randomized trial with 1-year follow-up. Scand. J. Med. Sci. Sports 2004, 14, 286–295. [Google Scholar] [CrossRef] [Green Version]
- Ohberg, L.; Lorentzon, R.; Alfredson, H. Eccentric training in patients with chronic Achilles tendinosis: Normalised tendon structure and decreased thickness at follow up. Br. J. Sports Med. 2004, 38, 8–11, discussion 11. [Google Scholar] [CrossRef] [Green Version]
- Ohberg, L.; Alfredson, H. Effects on neovascularisation behind the good results with eccentric training in chronic mid-portion Achilles tendinosis? Knee Surg. Sports Traumatol. Arthrosc. 2004, 12, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Sayana, M.K.; Maffulli, N. Eccentric calf muscle training in non-athletic patients with Achilles tendinopathy. J. Sci. Med. Sport 2007, 10, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Maffulli, N.; Walley, G.; Sayana, M.K.; Longo, U.G.; Denaro, V. Eccentric calf muscle training in athletic patients with Achilles tendinopathy. Disabil. Rehabil. 2008, 30, 1677–1684. [Google Scholar] [CrossRef] [PubMed]
- Rhim, H.C.; Kim, M.S.; Choi, S.; Tenforde, A.S. Comparative Efficacy and Tolerability of Nonsurgical Therapies for the Treatment of Midportion Achilles Tendinopathy: A Systematic Review with Network Meta-analysis. Orthop. J. Sports Med. 2020, 8. [Google Scholar] [CrossRef]
- Stania, M.; Juras, G.; Chmielewska, D.; Polak, A.; Kucio, C.; Król, P. Extracorporeal Shock Wave Therapy for Achilles Tendinopathy. BioMed Res. Int. 2019. [Google Scholar] [CrossRef]
- Wang, X.; Qiu, Y.; Triffitt, J.; Carr, A.; Xia, Z.; Sabokbar, A. Proliferation and differentiation of human tenocytes in response to platelet rich plasma: An in vitro and in vivo study. J. Orthop. Res. 2012, 30, 982–990. [Google Scholar] [CrossRef]
- Tallon, C.; Coleman, B.D.; Khan, K.M.; Maffulli, N. Outcome of surgery for chronic Achilles tendinopathy. A critical review. Am. J. Sports Med. 2001, 29, 315–320. [Google Scholar] [CrossRef]
- Molloy, T.; Wang, Y.; Murrell, G.A.C. The Roles of Growth Factors in Tendon and Ligament Healing. Sports Med. 2003, 33, 381–394. [Google Scholar] [CrossRef]
- Chen, L.; Dong, S.-W.; Tao, X.; Liu, J.-P.; Tang, K.-L.; Xu, J.-Z. Autologous platelet-rich clot releasate stimulates proliferation and inhibits differentiation of adult rat tendon stem cells towards nontenocyte lineages. J. Int. Med. Res. 2012, 40, 1399–1409. [Google Scholar] [CrossRef]
- De Mos, M.; van der Windt, A.E.; Jahr, H.; van Schie, H.T.M.; Weinans, H.; Verhaar, J.A.N.; van Osch, G.J.V.M. Can platelet-rich plasma enhance tendon repair? A cell culture study. Am. J. Sports Med. 2008, 36, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Klatte-Schulz, F.; Schmidt, T.; Uckert, M.; Scheffler, S.; Kalus, U.; Rojewski, M.; Schrezenmeier, H.; Pruss, A.; Wildemann, B. Comparative Analysis of Different Platelet Lysates and Platelet Rich Preparations to Stimulate Tendon Cell Biology: An In Vitro Study. Int. J. Mol. Sci. 2018, 19, 212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andia, I.; Rubio-Azpeitia, E.; Maffulli, N. Platelet-rich plasma modulates the secretion of inflammatory/angiogenic proteins by inflamed tenocytes. Clin. Orthop. 2015, 473, 1624–1634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Middleton, K.K.; Fu, F.H.; Im, H.-J.; Wang, J.H.-C. HGF mediates the anti-inflammatory effects of PRP on injured tendons. PLoS ONE 2013, 8, e67303. [Google Scholar] [CrossRef] [Green Version]
- Dallaudière, B.; Lempicki, M.; Pesquer, L.; Louedec, L.; Preux, P.M.; Meyer, P.; Hummel, V.; Larbi, A.; Deschamps, L.; Journe, C.; et al. Efficacy of intra-tendinous injection of platelet-rich plasma in treating tendinosis: Comprehensive assessment of a rat model. Eur. Radiol. 2013, 23, 2830–2837. [Google Scholar] [CrossRef]
- Li, S.; Wu, Y.; Jiang, G.; Tian, X.; Hong, J.; Chen, S.; Yan, R.; Feng, G.; Cheng, Z. Intratendon delivery of leukocyte-rich platelet-rich plasma at early stage promotes tendon repair in a rabbit Achilles tendinopathy model. J. Tissue Eng. Regen. Med. 2020, 14, 452–463. [Google Scholar] [CrossRef]
- Jiang, G.; Wu, Y.; Meng, J.; Wu, F.; Li, S.; Lin, M.; Gao, X.; Hong, J.; Chen, W.; Yan, S.; et al. Comparison of Leukocyte-Rich Platelet-Rich Plasma and Leukocyte-Poor Platelet-Rich Plasma on Achilles Tendinopathy at an Early Stage in a Rabbit Model. Am. J. Sports Med. 2020, 48, 1189–1199. [Google Scholar] [CrossRef]
- Yan, R.; Gu, Y.; Ran, J.; Hu, Y.; Zheng, Z.; Zeng, M.; Heng, B.C.; Chen, X.; Yin, Z.; Chen, W.; et al. Intratendon Delivery of Leukocyte-Poor Platelet-Rich Plasma Improves Healing Compared With Leukocyte-Rich Platelet-Rich Plasma in a Rabbit Achilles Tendinopathy Model. Am. J. Sports Med. 2017, 45, 1909–1920. [Google Scholar] [CrossRef]
- Chen, L.; Liu, J.-P.; Tang, K.-L.; Wang, Q.; Wang, G.-D.; Cai, X.-H.; Liu, X.-M. Tendon derived stem cells promote platelet-rich plasma healing in collagenase-induced rat achilles tendinopathy. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2014, 34, 2153–2168. [Google Scholar] [CrossRef]
- Dallaudiere, B.; Louedec, L.; Lenet, M.P.J.; Pesquer, L.; Blaise, E.; Perozziello, A.; Michel, J.B.; Moinard, M.; Meyer, P.; Serfaty, J.M. The molecular systemic and local effects of intra-tendinous injection of Platelet Rich Plasma in tendinosis: Preliminary results on a rat model with ELISA method. Muscles Ligaments Tendons J. 2015, 5, 99–105. [Google Scholar] [CrossRef]
- Fedato, R.A.; Francisco, J.C.; Sliva, G.; de Noronha, L.; Olandoski, M.; Faria Neto, J.R.; Ferreira, P.E.; Simeoni, R.B.; Abdelwahid, E.; de Carvalho, K.A.T.; et al. Stem Cells and Platelet-Rich Plasma Enhance the Healing Process of Tendinitis in Mice. Stem Cells Int. 2019, 2019, 1497898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solchaga, L.A.; Bendele, A.; Shah, V.; Snel, L.B.; Kestler, H.K.; Dines, J.S.; Hee, C.K. Comparison of the effect of intra-tendon applications of recombinant human platelet-derived growth factor-BB, platelet-rich plasma, steroids in a rat achilles tendon collagenase model. J. Orthop. Res. 2014, 32, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-J.; Xu, S.-Z.; Gu, P.-C.; Du, J.-Y.; Cai, Y.-Z.; Zhang, C.; Lin, X.-J. Is Platelet-rich Plasma Injection Effective for Chronic Achilles Tendinopathy? A Meta-analysis. Clin. Orthop. 2018, 476, 1633–1641. [Google Scholar] [CrossRef] [PubMed]
- Madhi, M.I.; Yausep, O.E.; Khamdan, K.; Trigkilidas, D. The use of PRP in treatment of Achilles Tendinopathy: A systematic review of literature. Study design: Systematic review of literature. Ann. Med. Surg. 2020, 55, 320–326. [Google Scholar] [CrossRef]
- Coombes, B.K.; Bisset, L.; Vicenzino, B. Efficacy and safety of corticosteroid injections and other injections for management of tendinopathy: A systematic review of randomised controlled trials. Lancet 2010, 376, 1751–1767. [Google Scholar] [CrossRef] [Green Version]
- Wetke, E.; Johannsen, F.; Langberg, H. Achilles tendinopathy: A prospective study on the effect of active rehabilitation and steroid injections in a clinical setting. Scand. J. Med. Sci. Sports 2015, 25, e392–e399. [Google Scholar] [CrossRef]
- Metcalfe, D.; Achten, J.; Costa, M.L. Glucocorticoid injections in lesions of the achilles tendon. Foot Ankle Int. 2009, 30, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Mousavizadeh, R.; Backman, L.; McCormack, R.G.; Scott, A. Dexamethasone decreases substance P expression in human tendon cells: An in vitro study. Rheumatology 2015, 54, 318–323. [Google Scholar] [CrossRef] [Green Version]
- Gotoh, M.; Hamada, K.; Yamakawa, H.; Inoue, A.; Fukuda, H. Increased substance P in subacromial bursa and shoulder pain in rotator cuff diseases. J. Orthop. Res. 1998, 16, 618–621. [Google Scholar] [CrossRef]
- Tempfer, H.; Gehwolf, R.; Lehner, C.; Wagner, A.; Mtsariashvili, M.; Bauer, H.-C.; Resch, H.; Tauber, M. Effects of crystalline glucocorticoid triamcinolone acetonide on cultered human supraspinatus tendon cells. Acta Orthop. 2009, 80, 357–362. [Google Scholar] [CrossRef]
- Dinhane, K.G.I.; Godoy-Santos, A.L.; Fabro, A.T.; Moretto, M.R.; Deprá, I.; Yoshida, W.B. Short-term Changes After Corticosteroid Injections Into the Normal Tendons of Rabbits: A Controlled Randomized Study. Am. J. Sports Med. 2019, 47, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Muto, T.; Kokubu, T.; Mifune, Y.; Inui, A.; Harada, Y.; Yoshifumi, N.; Takase, F.; Kuroda, R.; Kurosaka, M. Temporary inductions of matrix metalloprotease-3 (MMP-3) expression and cell apoptosis are associated with tendon degeneration or rupture after corticosteroid injection. J. Orthop. Res. 2014, 32, 1297–1304. [Google Scholar] [CrossRef] [PubMed]
- Boesen, A.P.; Langberg, H.; Hansen, R.; Malliaras, P.; Boesen, M.I. High volume injection with and without corticosteroid in chronic midportion achilles tendinopathy. Scand. J. Med. Sci. Sports 2019, 29, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Boesen, A.P.; Hansen, R.; Boesen, M.I.; Malliaras, P.; Langberg, H. Effect of High-Volume Injection, Platelet-Rich Plasma, and Sham Treatment in Chronic Midportion Achilles Tendinopathy: A Randomized Double-Blinded Prospective Study. Am. J. Sports Med. 2017, 45, 2034–2043. [Google Scholar] [CrossRef]
- Ahmad, Z.; Wardale, J.; Brooks, R.; Henson, F.; Noorani, A.; Rushton, N. Exploring the Application of Stem Cells in Tendon Repair and Regeneration. Arthrosc. J. Arthrosc. Relat. Surg. 2012, 28, 1018–1029. [Google Scholar] [CrossRef]
- Ruzzini, L.; Longo, U.G.; Rizzello, G.; Denaro, V. Stem cells and tendinopathy: State of the art from the basic science to clinic application. Muscles Ligaments Tendons J. 2012, 2, 235–238. [Google Scholar]
- Machova Urdzikova, L.; Sedlacek, R.; Suchy, T.; Amemori, T.; Ruzicka, J.; Lesny, P.; Havlas, V.; Sykova, E.; Jendelova, P. Human multipotent mesenchymal stem cells improve healing after collagenase tendon injury in the rat. Biomed. Eng. Online 2014, 13. [Google Scholar] [CrossRef] [Green Version]
- Oshita, T.; Tobita, M.; Tajima, S.; Mizuno, H. Adipose-Derived Stem Cells Improve Collagenase-Induced Tendinopathy in a Rat Model. Am. J. Sports Med. 2016, 44, 1983–1989. [Google Scholar] [CrossRef]
- Chen, J.; Yu, Q.; Wu, B.; Lin, Z.; Pavlos, N.J.; Xu, J.; Ouyang, H.; Wang, A.; Zheng, M.H. Autologous tenocyte therapy for experimental Achilles tendinopathy in a rabbit model. Tissue Eng. 2011, 17, 2037–2048. [Google Scholar] [CrossRef]
- Wang, Y.; He, G.; Guo, Y.; Tang, H.; Shi, Y.; Bian, X.; Zhu, M.; Kang, X.; Zhou, M.; Lyu, J.; et al. Exosomes from tendon stem cells promote injury tendon healing through balancing synthesis and degradation of the tendon extracellular matrix. J. Cell. Mol. Med. 2019, 23, 5475–5485. [Google Scholar] [CrossRef] [Green Version]
- Goldberg, A.J.; Zaidi, R.; Brooking, D.; Kim, L.; Korda, M.; Masci, L.; Green, R.; O’Donnell, P.; Smith, R. Autologous Stem Cells in Achilles Tendinopathy (ASCAT): Protocol for a phase IIA, single-centre, proof-of-concept study. BMJ Open 2018, 8, e021600. [Google Scholar] [CrossRef]
- Clarke, A.W.; Alyas, F.; Morris, T.; Robertson, C.J.; Bell, J.; Connell, D.A. Skin-derived tenocyte-like cells for the treatment of patellar tendinopathy. Am. J. Sports Med. 2011, 39, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Kim, W.; Lim, C.; Chung, S.G. Treatment of Lateral Epicondylosis by Using Allogeneic Adipose-Derived Mesenchymal Stem Cells: A Pilot Study. Stem Cells 2015, 33, 2995–3005. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.; Ran, S. Regulation of tumor angiogenesis by the local environment. Front. Biosci. 2010, 15, 195–212. [Google Scholar] [CrossRef]
- Tempfer, H.; Traweger, A. Tendon Vasculature in Health and Disease. Front. Physiol. 2015, 6, 330. [Google Scholar] [CrossRef]
- Dallaudière, B.; Lempicki, M.; Pesquer, L.; Louedec, L.; Preux, P.M.; Meyer, P.; Hess, A.; Durieux, M.H.M.; Hummel, V.; Larbi, A.; et al. Acceleration of tendon healing using US guided intratendinous injection of bevacizumab: First pre-clinical study on a murine model. Eur. J. Radiol. 2013, 82, e823–e828. [Google Scholar] [CrossRef] [PubMed]
- Dallaudiere, B.; Zurlinden, O.; Perozziello, A.; Deschamps, L.; Larbi, A.; Louedec, L.; Pesquer, L.; Benayoun, Y.; Silvestre, A.; Serfaty, J.M. Combined intra-tendinous injection of Platelet Rich Plasma and bevacizumab accelerates and improves healing compared to Platelet Rich Plasma in tendinosis: Comprehensive assessment on a rat model. Muscles Ligaments Tendons J. 2014, 4, 351–356. [Google Scholar] [CrossRef]
- Alfredson, H. Ultrasound and Doppler-guided mini-surgery to treat midportion Achilles tendinosis: Results of a large material and a randomised study comparing two scraping techniques. Br. J. Sports Med. 2011, 45, 407–410. [Google Scholar] [CrossRef]
- Lind, B.; Öhberg, L.; Alfredson, H. Sclerosing polidocanol injections in mid-portion Achilles tendinosis: Remaining good clinical results and decreased tendon thickness at 2-year follow-up. Knee Surg. Sports Traumatol. Arthrosc. 2006, 14. [Google Scholar] [CrossRef]
- Ebbesen, B.H.; Mølgaard, C.M.; Olesen, J.L.; Gregersen, H.E.; Simonsen, O. No beneficial effect of Polidocanol treatment in Achilles tendinopathy: A randomised controlled trial. Knee Surg. Sports Traumatol. Arthrosc. 2018, 26, 2038–2044. [Google Scholar] [CrossRef]
- Magra, M.; Maffulli, N. Matrix metalloproteases: A role in overuse tendinopathies. Br. J. Sports Med. 2005, 39, 789–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Buono, A.; Oliva, F.; Osti, L.; Maffulli, N. Metalloproteases and tendinopathy. Muscles Ligaments Tendons J. 2013, 3, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Orchard, J.; Massey, A.; Brown, R.; Cardon-Dunbar, A.; Hofmann, J. Successful Management of Tendinopathy With Injections of the MMP-inhibitor Aprotinin. Clin. Orthop. 2008, 466, 1625–1632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maffulli, N.; Spiezia, F.; Longo, U.G.; Denaro, V.; Maffulli, G.D. High volume image guided injections for the management of chronic tendinopathy of the main body of the Achilles tendon. Phys. Ther. Sport 2013, 14, 163–167. [Google Scholar] [CrossRef]
- Brown, R.; Orchard, J.; Kinchington, M.; Hooper, A.; Nalder, G. Aprotinin in the management of Achilles tendinopathy: A randomised controlled trial. Br. J. Sports Med. 2006, 40, 275–279. [Google Scholar] [CrossRef] [Green Version]
- Maffulli, N.; Longo, U.G.; Kadakia, A.; Spiezia, F. Achilles tendinopathy. Foot Ankle Surg. 2020, 26, 240–249. [Google Scholar] [CrossRef]
- Hsieh, Y.-L.; Lin, M.-T.; Hong, C.-Z.; Chen, H.-S. Percutaneous soft tissue release performed using a blunt cannula in rabbits with chronic collagenase-induced Achilles tendinopathy. Foot Ankle Surg. Off. J. Eur. Soc. Foot Ankle Surg. 2019, 25, 186–192. [Google Scholar] [CrossRef]
- Testa, V.; Capasso, G.; Benazzo, F.; Maffulli, N. Management of Achilles tendinopathy by ultrasound-guided percutaneous tenotomy. Med. Sci. Sports Exerc. 2002, 34, 573–580. [Google Scholar] [CrossRef]
- Maffulli, N.; Oliva, F.; Testa, V.; Capasso, G.; Del Buono, A. Multiple percutaneous longitudinal tenotomies for chronic Achilles tendinopathy in runners: A long-term study. Am. J. Sports Med. 2013, 41, 2151–2157. [Google Scholar] [CrossRef]
- Stoychev, V.; Finestone, A.S.; Kalichman, L. Dry Needling as a Treatment Modality for Tendinopathy: A Narrative Review. Curr. Rev. Musculoskelet. Med. 2020, 13, 133–140. [Google Scholar] [CrossRef]
- Hammerman, M.; Aspenberg, P.; Eliasson, P. Microtrauma stimulates rat Achilles tendon healing via an early gene expression pattern similar to mechanical loading. J. Appl. Physiol. 2014, 116, 54–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riggin, C.N.; Chen, M.; Gordon, J.A.; Schultz, S.M.; Soslowsky, L.J.; Khoury, V. Ultrasound-Guided Dry Needling of the Healthy Rat Supraspinatus Tendon Elicits Early Healing Without Causing Permanent Damage. J. Orthop. Res. 2019, 37, 2035–2042. [Google Scholar] [CrossRef] [PubMed]
- Housner, J.A.; Jacobson, J.A.; Misko, R. Sonographically guided percutaneous needle tenotomy for the treatment of chronic tendinosis. J. Ultrasound Med. 2009, 28, 1187–1192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeo, A.; Kendall, N.; Jayaraman, S. Ultrasound-guided dry needling with percutaneous paratenon decompression for chronic Achilles tendinopathy. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 2112–2118. [Google Scholar] [CrossRef] [PubMed]
- Abate, M.; Carlo, L.D.; Salini, V. Platelet rich plasma compared to dry needling in the treatment of non-insertional Achilles tendinopathy. Phys. Sportsmed. 2019, 47, 232–237. [Google Scholar] [CrossRef]
- Finnoff, J.T.; Fowler, S.P.; Lai, J.K.; Santrach, P.J.; Willis, E.A.; Sayeed, Y.A.; Smith, J. Treatment of Chronic Tendinopathy with Ultrasound-Guided Needle Tenotomy and Platelet-Rich Plasma Injection. PM&R 2011, 3, 900–911. [Google Scholar] [CrossRef]
| Author, Year | Type of Experiment | Main Results | ||||
|---|---|---|---|---|---|---|
| Animal Model | Time of Treatment | Time of Analysis | Histology | Gene and Protein | Biomechanical Testing | |
| Zhang, 2013 [31] | Wound of 1 mm Mouse | D0 | D0, D1, D3, D5 and D12 after I | - | - ↘ PGE2 production after injury - ↘ COX-1 and COX-2 expression | - |
| Dallaudière, 2013 [32] | Collagenase Rat | D3 | D6, D13, D18 and D25 after C | - Reduction of tendon thickening and fibrillary disorganization as well as neovascularization with PRP | - | - |
| Li, 2020 [33] | Collagenase Rabbit | D7 or W4 | 6 weeks after C | - Significant reduction of histological lesions (PRP injected at D7) | - ↗ expression of Coll1 (D7) and ↗ expression of Coll3, MMP1 and MMP3 (W4) - ↗ level of IL-10 and ↘ level of IL-6 - No difference in IL-1β and TNFα concentrations | - No improvement in failure load and stiffness |
| Jiang, 2020 [34] | Collagenase Rabbit | D7 (Lp-PRP, Lr-PRP and saline) | 3 and 6 weeks after C | - Significant reduction in histological lesions in the 2 PRP groups compared with saline but better in Lr-PRP | - ↗ expression of Coll1 earlier in Lr-PRP group - ↗ then ↘ VEGF and VEGF-R expression (Lr-PRP group) - Transient ↗ IL-10 expression (Lr-PRP group) | - Failure load, stiffness, and tensile stress in the Lr-PRP group higher than those in the saline group |
| Yan, 2017 [35] | Collagenase Rat | W4 (Lp-PRP, Lr-PRP and saline) | 8 weeks after C | - Reduction of tendinopathic lesions compared to saline (Lp-PRP) | - ↗ expression of Coll1 compared with the saline group (Lp-PRP) - No impact on IL-1β and TNFα concentrations but ↘ of IL-6 level compared to saline (Lp-PRP) - ↘ MMP1 and MMP3 expression. No effect on MMP9 | - |
| Chen, 2014 [36] | Collagenase Rat | W4 | 8 or 12 weeks after C | - Significant improvement of histological parameters of tendon quality (fiber organization, nuclear rounding, and inflammation) | - ↗ expression of Coll1, scleraxis and Tenascin C | - Maximum load to failure and stiffness significantly superior to the control group by week 8 |
| Dallaudière, 2015 [37] | Collagenase Rat | D3 | D7, D13, D18 and D25 after C | - | No effect on local concentrations of IL-1α, IL-1β, IL-18, G-CSF, GM-CSF, M-CSF, MIP-1α, RANTES, TNFα | - |
| Fedato, 2019 [38] | Collagenase Rat | D5 | 4 weeks after PRP | - No significant effect on histological lesions compared to control group | - | - Better results for maximum deformation and elastic modulus. Ultimate tensile strength not improved. |
| Solchaga, 2014 [39] | Collagenase Rat | D7 | D14 and D28 after C | - No differences between groups in the extent and character of the repair | - | - No improvement in mechanical properties (maximum load, ultimate tensile stress, stiffness) |
| Author, Year | Design | Main Results | ||||
|---|---|---|---|---|---|---|
| Type | Cells/Model | Steroid | Histology | Gene and Protein | Biomechanical Testing | |
| Mousavizadeh, 2015 [45] | In vitro | Human tenocytes | Dexamethasone | - | - ↘ expression of IL-1α, IL-1β, and IL-6 - ↘ expression level of TAC1 (gene encoding for substance P (SP)) but no effect on the expression of NK1R, its receptor. Reduced secretion of SP - ↘ induction of SP by IL-1β and by mechanical loading | - |
| Tempfer, 2009 [47] | In vitro | Human tenocytes | Triamcinolone acetonide | - | - ↘ Expression and secretion of Coll1 - ↘ proliferation rate - ↘ expression of MMP2, MMP8, MMP9, and MMP13 but ↗ expression of TIMP1 - Increased expression of SOX9 | - |
| Solchaga, 2014 [39] | In vivo | Collagenase Rat | Intra-tendinous triamcinolone acetonide at D7 Analysis at D14 and D28 after C | - ↘ tendon thickness at insertion site and midsubstance - ↘ cell proliferation and ↘ inflammation relative to saline treatment | - | - No effect on mechanical properties compared to saline (maximum load, ultimate tensile stress, stiffness, ramping modulus) |
| Dinhane, 2019 [48] | In vivo | Normal Achilles tendons Rabbit | Intra-tendinous injection of betamethasone Analysis 48 h after I | - | - ↘ MMP2 expression compared with the control group - No difference in IL1 and IL6 levels in the tendon tissues | - No effect on mechanical resistance (maximum deformation, maximum force, energy at maximum force, elasticity modulus, and tension at maximum force) |
| Muto, 2014 [49] | In vivo | Normal Achilles tendons Rat | Triamcinolone acetonide (TCA) or prednisolone (PSL) around the tendon. Analysis at W1 and W3 after I | - Collagen fiber bundles irregularly aligned at W1 but at W3, these changes had ↘. - At W1, increased number of apoptotic cells in the surface layers of the tendons but no significant difference after 3 weeks | - at 1 week, ↗ MMP3 in the surface layers of tendons | - Reduction of maximum failure load 1 week after the injection with a return to normal after 3 weeks |
| Author, Year | Design | Main Results | ||||
|---|---|---|---|---|---|---|
| Animal Model | Treatment | Time of Analysis | Histology | Gene and Protein | Biomechanical Testing | |
| Machova Urdzikova, 2014 [52] | Collagenase Rat | Bone marrow-derived hMSC injected at D3 | W2, W4 and W6 after C | - Better organization of the collagen fibers and ↗ neovascularization in hMSC-treated rats - MSC always present at the site of injection 6 weeks later | - ↗ amounts of Coll1 and Coll3 - No difference in quantities of aggregan and versican in the ECM | - No difference between groups (testing of stiffness and load to failure) |
| Oshita, 2016 [53] | Collagenase Rat | Adipose-derived stem cells (ASCs) injected at D7 | W4 and W12 after ttt | - ↘ degree of tendon degeneration (↘ disrupted collagen fibers, ↘ cellularity, and less ground substance deposition between collagen fibers) | - ↘ Coll3/Coll1 ratio in the ASC group | - |
| Chen, 2011 [54] | Collagenase Rat | Autologous tenocytes injected at W4 | 4 and 8 weeks after ttt | - Histologic scores significantly better at W8 (fiber structure, rounding of the nuclei, inflammatory cells, and neovascularization) - Tenocytes incorporated into the ECM and distributed longitudinally and parallel to the fiber orientation in a typical spindle pattern | - At W8, ↗ synthesis of Coll1 compared to the control group | - Improvement of the ultimate failure load |
| Chen, 2014 [36] | Collagenase Rat | Tendon-derived stem cells injected at W4 | 8 or 12 weeks after C | - Significant improvement of histological parameters (fiber arrangement and structure, nuclear rounding, and inflammation) after combined injection of TDSC and PRP. - No effect of TDSC alone. | - No effect of TDSC on tenocyte-related gene expression (Coll1, Scleraxis, Tenascin C) and or non-tenocyte gene expression (Runx2, SOX9 and PPARγ) | - Maximum load to failure and stiffness significantly superior to the control group when TDSCs were injected with PRP but no effect of TDSC alone on mechanical parameters. |
| Fedato, 2019 [38] | Collagenase Rat | Bone marrow-derived stem cells injected at D5 | 4 weeks after ttt | - No significant improvement of histological lesions | - | - Significantly better results for elastic modulus in the stem cell group - Ultimate tensile strength and maximum deformation not improved. |
| Wang, 2019 [55] | Collagenase Rat | Tendon-derived stem cells injected at D7 | 5 weeks after collagenase | - Reduction of histological lesions | - | - ↗ Maximum loading and ultimate stress in the TDSC group compared with the control group |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Darrieutort-Laffite, C.; Soslowsky, L.J.; Le Goff, B. Molecular and Structural Effects of Percutaneous Interventions in Chronic Achilles Tendinopathy. Int. J. Mol. Sci. 2020, 21, 7000. https://doi.org/10.3390/ijms21197000
Darrieutort-Laffite C, Soslowsky LJ, Le Goff B. Molecular and Structural Effects of Percutaneous Interventions in Chronic Achilles Tendinopathy. International Journal of Molecular Sciences. 2020; 21(19):7000. https://doi.org/10.3390/ijms21197000
Chicago/Turabian StyleDarrieutort-Laffite, Christelle, Louis J. Soslowsky, and Benoit Le Goff. 2020. "Molecular and Structural Effects of Percutaneous Interventions in Chronic Achilles Tendinopathy" International Journal of Molecular Sciences 21, no. 19: 7000. https://doi.org/10.3390/ijms21197000
APA StyleDarrieutort-Laffite, C., Soslowsky, L. J., & Le Goff, B. (2020). Molecular and Structural Effects of Percutaneous Interventions in Chronic Achilles Tendinopathy. International Journal of Molecular Sciences, 21(19), 7000. https://doi.org/10.3390/ijms21197000




