Surface Modification by Nano-Structures Reduces Viable Bacterial Biofilm in Aerobic and Anaerobic Environments
Abstract
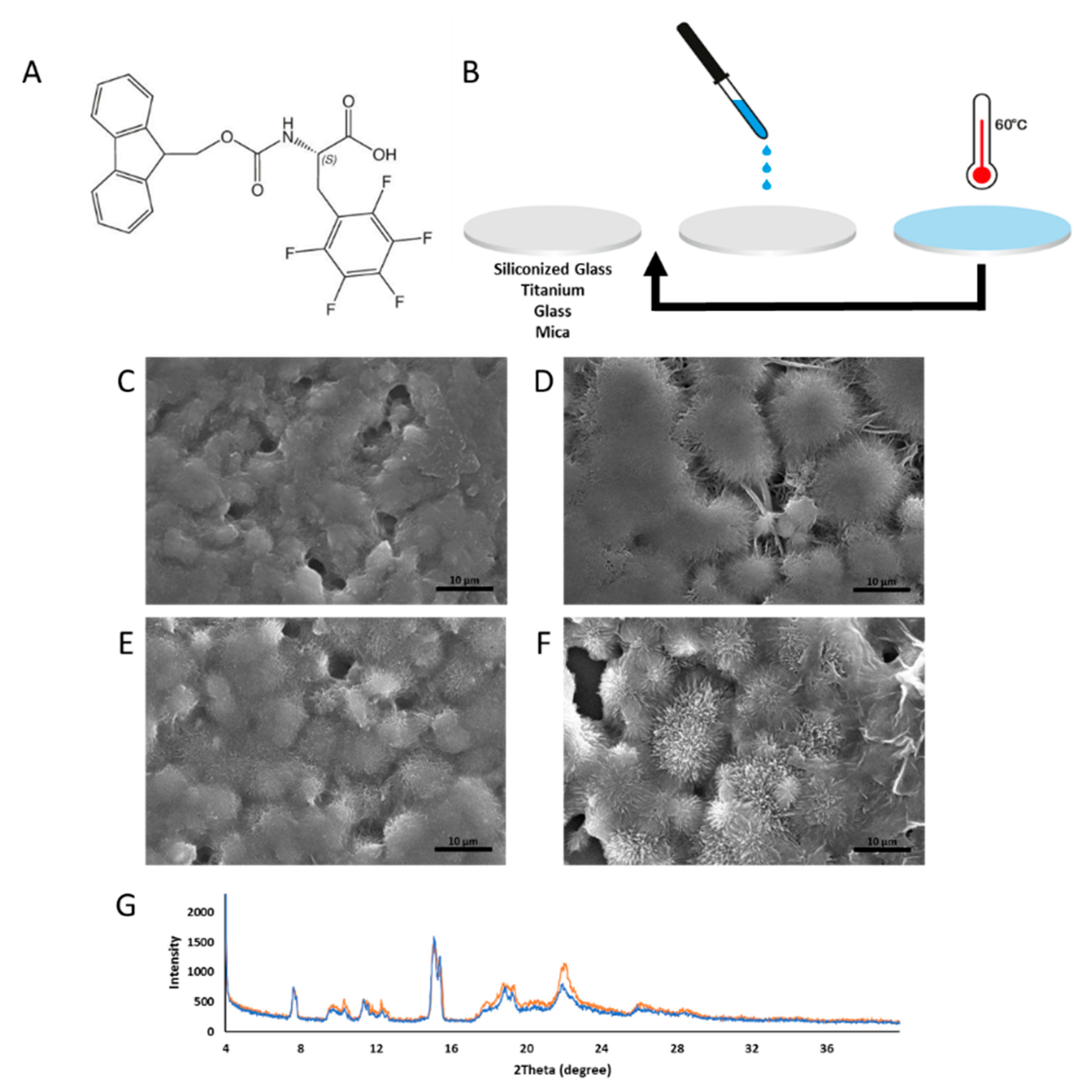
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Substrates
3.2. Coating Preparation Using the Drop-Cast Method
3.3. Scanning Electron Microscopy (SEM)
3.4. Contact Angle Measurements
3.5. X-ray Diffraction (XRD)
3.6. Bacterial Strains and Growth Conditions
3.7. Evaluation of Antibacterial Properties Using Luminescence Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xu, Y.; Dhaouadi, Y.; Stoodley, P.; Ren, D. Sensing the unreachable: Challenges and opportunities in biofilm detection. Curr. Opin. Biotechnol. 2019, 64, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Palestro, C.J. Molecular imaging of infection: The first 50 years. Semin. Nucl. Med. 2020, 50, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Douterelo, I.; Husband, S.; Loza, V.; Boxall, J. Dynamics of biofilm regrowth in drinking water distribution systems. Appl. Environ. Microbiol. 2016, 82, 4155–4168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flemming, H.C. Biofouling in water systems—Cases, causes and countermeasures. Appl. Microbiol. Biotechnol. 2002, 59, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, A.; Nir, S.; Reches, M.; Shemesh, M. Preventing biofilm formation by dairy-associated bacteria using peptide-coated surfaces. Front Microbiol. 2019, 10, 1405. [Google Scholar] [CrossRef] [Green Version]
- Elmoslemany, A.M.; Keefe, G.P.; Dohoo, I.R.; Jayarao, B.M. Risk factors for bacteriological quality of bulk tank milk in Prince Edward Island dairy herds. Part 2: Bacteria count-specific risk factors. J. Dairy Sci. 2009, 92, 2644–2652. [Google Scholar] [CrossRef] [Green Version]
- Chambers, L.D.; Stokes, K.R.; Walsh, F.C.; Wood, R.J. Modern approaches to marine antifouling coatings. Surf. Coat. Technol. 2006, 201, 3642–3652. [Google Scholar] [CrossRef] [Green Version]
- Asuri, P.; Karajanagi, S.S.; Kane, R.S.; Dordick, J.S. Polymer-nanotube-enzyme composites as active antifouling films. Small 2007, 3, 50–53. [Google Scholar] [CrossRef]
- Banerjee, I.; Pangule, R.C.; Kane, R.S. Antifouling coatings: Recent developments in the design of surfaces that prevent fouling by proteins, bacteria, and marine organisms. Adv. Mater. 2011, 23, 690–718. [Google Scholar] [CrossRef]
- Lejars, M.; Margaillan, A.; Bressy, C. Fouling release coatings: A nontoxic alternative to biocidal antifouling coatings. Chem. Rev. 2012, 112, 4347–4390. [Google Scholar] [CrossRef]
- Roy, R.; Tiwari, M.; Donelli, G.; Tiwari, V. Strategies for combating bacterial biofilms: A focus on anti-biofilm agents and their mechanisms of action. Virulence 2018, 9, 522–554. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Yu, K.; Lo, J.C.Y.; Takeuchi, L.E.; Hadjesfandiari, N.; Yazdani-Ahmadabadi, H.; Brooks, D.E.; Lange, D.; Kizhakkedathu, J.N. Polymer-nanoparticle interaction as a design principle in the development of a durable ultrathin universal binary antibiofilm coating with long-term activity. ACS Nano 2018, 12, 11881–11891. [Google Scholar] [CrossRef] [PubMed]
- Maity, S.; Nir, S.; Zada, T.; Reches, M. Self-assembly of a tripeptide into a functional coating that resists fouling. Chem. Commun. 2014, 50, 11154–11157. [Google Scholar] [CrossRef] [PubMed]
- Sakala, G.P.; Reches, M. Peptide-based approaches to fight biofouling. Adv. Mater. Interfaces 2018, 5, 1800073. [Google Scholar] [CrossRef]
- Garcia, A.M.; Lavendomme, R.; Kralj, S.; Kurbasic, M.; Bellotto, O.; Cringoli, M.C.; Semeraro, S.; Bandiera, A.; De Zorzi, R.; Marchesan, S. Self-assembly of an amino acid derivative into an antimicrobial hydrogel biomaterial. Chemistry 2020, 26, 1880–1886. [Google Scholar] [CrossRef]
- Ye, Z.; Aparicio, C. Modulation of supramolecular self-assembly of an antimicrobial designer peptide by single amino acid substitution: Implications on peptide activity. Nanoscale Adv. 2019, 1, 4679–4682. [Google Scholar] [CrossRef] [Green Version]
- Youssef, H.; DeWolf, C.E. Interfacial self-assembly of antimicrobial peptide GL13K into non-fibril crystalline beta-sheets. Langmuir 2020, 36, 660–665. [Google Scholar] [CrossRef]
- Ye, Z.; Zhu, X.; Acosta, S.; Kumar, D.; Sang, T.; Aparicio, C. Self-assembly dynamics and antimicrobial activity of all l- and d-amino acid enantiomers of a designer peptide. Nanoscale 2018, 11, 266–275. [Google Scholar] [CrossRef]
- Porter, S.L.; Coulter, S.M.; Pentlavalli, S.; Thompson, T.P.; Laverty, G. Self-assembling diphenylalanine peptide nanotubes selectively eradicate bacterial biofilm infection. Acta Biomater. 2018, 77, 96–105. [Google Scholar] [CrossRef] [Green Version]
- Schnaider, L.; Brahmachari, S.; Schmidt, N.W.; Mensa, B.; Shaham-Niv, S.; Bychenko, D.; Adler-Abramovich, L.; Shimon, L.J.W.; Kolusheva, S.; DeGrado, W.F.; et al. Self-assembling dipeptide antibacterial nanostructures with membrane disrupting activity. Nat. Commun. 2017, 8, 1365. [Google Scholar] [CrossRef]
- Nir, S.; Zanuy, D.; Zada, T.; Agazani, O.; Aleman, C.; Shalev, D.E.; Reches, M. Tailoring the self-assembly of a tripeptide for the formation of antimicrobial surfaces. Nanoscale 2019, 11, 8752–8759. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Silva, S.M.; Lei, W.; Quigley, A.; Kapsa, R.M.I.; Moulton, S.E.; Greene, G.W. Adhesion and self-assembly of lubricin (PRG4) brush layers on different substrate surfaces. Langmuir 2019, 35, 15834–15848. [Google Scholar] [CrossRef] [PubMed]
- Schnaider, L.; Toprakcioglu, Z.; Ezra, A.; Liu, X.; Bychenko, D.; Levin, A.; Gazit, E.; Knowles, T.P.J. Biocompatible hybrid organic/inorganic microhydrogels promote bacterial adherence and eradication in vitro and in vivo. Nano Lett. 2020, 20, 1590–1597. [Google Scholar] [CrossRef]
- Song, J.; Yuan, C.; Jiao, T.; Xing, R.; Yang, M.; Adams, D.J.; Yan, X. Multifunctional antimicrobial biometallohydrogels based on amino acid coordinated self-assembly. Small 2020, 16, e1907309. [Google Scholar] [CrossRef] [PubMed]
- Karsenti, E. Self-organization in cell biology: A brief history. Nat. Rev. Mol. Cell Biol. 2008, 9, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Whitesides, G.M.; Grzybowski, B. Self-assembly at all scales. Science 2002, 295, 2418–2421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, Q.; Zhu, H.; Zhang, C.; Zhang, F.; Zhang, B.; Kaplan, D.L. Silk self-assembly mechanisms and control from thermodynamics to kinetics. Biomacromolecules 2012, 13, 826–832. [Google Scholar] [CrossRef] [Green Version]
- Helbing, C.; Deckert-Gaudig, T.; Firkowska-Boden, I.; Wei, G.; Deckert, V.; Jandt, K.D. Protein Handshake on the nanoscale: How albumin and hemoglobin self-assemble into nanohybrid fibers. ACS Nano 2018, 12, 1211–1219. [Google Scholar] [CrossRef]
- Gazit, E. A possible role for pi-stacking in the self-assembly of amyloid fibrils. FASEB J. 2002, 16, 77–83. [Google Scholar] [CrossRef]
- Tao, K.; Levin, A.; Adler-Abramovich, L.; Gazit, E. Fmoc-modified amino acids and short peptides: Simple bio-inspired building blocks for the fabrication of functional materials. Chem. Soc. Rev. 2016, 45, 3935–3953. [Google Scholar] [CrossRef]
- Lee, S.; Trinh, T.H.T.; Yoo, M.; Shin, J.; Lee, H.; Kim, J.; Hwang, E.; Lim, Y.B.; Ryou, C. Self-assembling peptides and their application in the treatment of diseases. Int. J. Mol. Sci. 2019, 20, 5850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lakshmanan, A.; Zhang, S.; Hauser, C.A. Short self-assembling peptides as building blocks for modern nanodevices. Trends Biotechnol. 2012, 30, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Amit, M.; Yuran, S.; Gazit, E.; Reches, M.; Ashkenasy, N. Tailor-made functional peptide self-assembling nanostructures. Adv. Mater. 2018, 30, e1707083. [Google Scholar] [CrossRef] [PubMed]
- Kasotakis, E.; Mitraki, A. Designed self-assembling peptides as templates for the synthesis of metal nanoparticles. Methods Mol. Biol. 2013, 996, 195–202. [Google Scholar] [PubMed]
- Wei, G.; Su, Z.; Reynolds, N.P.; Arosio, P.; Hamley, I.W.; Gazit, E.; Mezzenga, R. Self-assembling peptide and protein amyloids: From structure to tailored function in nanotechnology. Chem. Soc. Rev. 2017, 46, 4661–4708. [Google Scholar] [CrossRef] [PubMed]
- Okesola, B.O.; Wu, Y.; Derkus, B.; Gani, S.; Wu, D.; Knani, D.; Smith, D.K.; Adams, D.J.; Mata, A. Supramolecular self-assembly to control structural and biological properties of multicomponent hydrogels. Chem. Mater. 2019, 31, 7883–7897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diaferia, C.; Ghosh, M.; Sibillano, T.; Gallo, E.; Stornaiuolo, M.; Giannini, C.; Morelli, G.; Adler-Abramovich, L.; Accardo, A. Fmoc-FF and hexapeptide-based multicomponent hydrogels as scaffold materials. Soft Matter. 2019, 15, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Seow, W.Y.; Kandasamy, K.; Purnamawati, K.; Sun, W.; Hauser, C.A.E. Thin peptide hydrogel membranes suitable as scaffolds for engineering layered biostructures. Acta Biomater. 2019, 88, 293–300. [Google Scholar] [CrossRef]
- Humenik, M.; Pawar, K.; Scheibel, T. Nanostructured, self-assembled spider silk materials for biomedical applications. Adv. Exp. Med. Biol. 2019, 1174, 187–221. [Google Scholar]
- Ghosh, M.; Halperin-Sternfeld, M.; Grigoriants, I.; Lee, J.; Nam, K.T.; Adler-Abramovich, L. Arginine-presenting peptide hydrogels decorated with hydroxyapatite as biomimetic scaffolds for bone regeneration. Biomacromolecules 2017, 18, 3541–3550. [Google Scholar] [CrossRef]
- Harper, M.M.; Connolly, M.L.; Goldie, L.; Irvine, E.J.; Shaw, J.E.; Jayawarna, V.; Richardson, S.M.; Dalby, M.J.; Lightbody, D.; Ulijn, R.V. Biogelx: Cell culture on self-assembling peptide gels. Methods Mol. Biol. 2018, 1777, 283–303. [Google Scholar] [PubMed]
- Jayawarna, V.; Smith, A.; Gough, J.E.; Ulijn, R.V. Three-dimensional cell culture of chondrocytes on modified di-phenylalanine scaffolds. Biochem. Soc. Trans. 2007, 35, 535–537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmes, T.C.; de Lacalle, S.; Su, X.; Liu, G.; Rich, A.; Zhang, S. Extensive neurite outgrowth and active synapse formation on self-assembling peptide scaffolds. Proc. Natl. Acad. Sci. USA 2000, 97, 6728–6733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, G.A.; Czeisler, C.; Niece, K.L.; Beniash, E.; Harrington, D.A.; Kessler, J.A.; Stupp, S.I. Selective differentiation of neural progenitor cells by high-epitope density nanofibers. Science 2004, 303, 1352–1355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roth-Konforti, M.E.; Comune, M.; Halperin-Sternfeld, M.; Grigoriants, I.; Shabat, D.; Adler-Abramovich, L. UV light-responsive peptide-based supramolecular hydrogel for controlled drug delivery. Macromol. Rapid Commun. 2018, 39, e1800588. [Google Scholar] [CrossRef]
- Matson, J.B.; Newcomb, C.J.; Bitton, R.; Stupp, S.I. Nanostructure-templated control of drug release from peptide amphiphile nanofiber gels. Soft Matter. 2012, 8, 3586–3595. [Google Scholar] [CrossRef] [Green Version]
- Tesauro, D.; Accardo, A.; Diaferia, C.; Milano, V.; Guillon, J.; Ronga, L.; Rossi, F. Peptide-based drug-delivery systems in biotechnological applications: Recent advances and perspectives. Molecules 2019, 24, 351. [Google Scholar] [CrossRef] [Green Version]
- Bucci, R.; Das, P.; Iannuzzi, F.; Feligioni, M.; Gandolfi, R.; Gelmi, M.L.; Reches, M.; Pellegrino, S. Self-assembly of an amphipathic alphaalphabeta-tripeptide into cationic spherical particles for intracellular delivery. Org. Biomol. Chem. 2017, 15, 6773–6779. [Google Scholar] [CrossRef]
- Ischakov, R.; Adler-Abramovich, L.; Buzhansky, L.; Shekhter, T.; Gazit, E. Peptide-based hydrogel nanoparticles as effective drug delivery agents. Bioorg. Med. Chem. 2013, 21, 3517–3522. [Google Scholar] [CrossRef]
- Ni, M.; Zhuo, S.; Iliescu, C.; So, P.T.C.; Mehta, J.S.; Yu, H.; Hauser, C.A.E. Self-assembling amyloid-like peptides as exogenous second harmonic probes for bioimaging applications. J. Biophotonics 2019, 12, e201900065. [Google Scholar] [CrossRef]
- Diaferia, C.; Gianolio, E.; Accardo, A. Peptide-based building blocks as structural elements for supramolecular Gd-containing MRI contrast agents. J. Pept. Sci. 2019, 25, e3157. [Google Scholar] [CrossRef] [PubMed]
- Khadeja, L.; Grigoriants, I.; Halperin-Sternfeld, M.; Yona, A.; Adler-Abramovich, L. Sonochemical functionalization of cotton and non-woven fabrics with bio-inspired self-assembled nanostructures. Isr. J. Chem. 2019. [Google Scholar] [CrossRef]
- Gaw, S.L.; Sakala, G.; Nir, S.; Saha, A.; Xu, Z.J.; Lee, P.S.; Reches, M. Rational design of amphiphilic peptides and its effect on antifouling performance. Biomacromolecules 2018, 19, 3620–3627. [Google Scholar] [CrossRef] [PubMed]
- Ceylan, H.; Tekinay, A.B.; Guler, M.O. Selective adhesion and growth of vascular endothelial cells on bioactive peptide nanofiber functionalized stainless steel surface. Biomaterials 2011, 32, 8797–8805. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, H.; Zheng, W.; Zhang, J.; Zhao, Q.; Wang, S.; Yang, Z.; Kong, D. Highly stable surface modifications of poly(3-caprolactone) (PCL) films by molecular self-assembly to promote cells adhesion and proliferation. Chem. Commun. 2011, 47, 8901–8903. [Google Scholar] [CrossRef]
- Alves, P.; Nir, S.; Reches, M.; Mergulhão, F. The effects of fluid composition and shear conditions on bacterial adhesion to an antifouling peptide-coated surface. MRS Commun. 2018, 8, 938–946. [Google Scholar] [CrossRef]
- Chakraborty, P.; Gazit, E. Amino acid based self-assembled nanostructures: Complex structures from remarkably simple building blocks. ChemNanoMat 2018, 4, 730–740. [Google Scholar] [CrossRef]
- Ghadiri, M.R.; Granja, J.R.; Milligan, R.A.; McRee, D.E.; Khazanovich, N. Self-assembling organic nanotubes based on a cyclic peptide architecture. Nature 1993, 366, 324–327. [Google Scholar] [CrossRef]
- Hartgerink, J.D.; Granja, J.R.; Milligan, R.A.; Ghadiri, M.R. Self-assembling peptide nanotubes. J. Am. Chem. Soc. 1996, 118, 43–50. [Google Scholar] [CrossRef]
- Reches, M.; Gazit, E. Casting metal nanowires within discrete self-assembled peptide nanotubes. Science 2003, 300, 625–627. [Google Scholar] [CrossRef] [Green Version]
- Reches, M.; Gazit, E. Formation of closed-cage nanostructures by self-assembly of aromatic dipeptides. Nano Lett. 2004, 4, 581–585. [Google Scholar] [CrossRef]
- Tamamis, P.; Adler-Abramovich, L.; Reches, M.; Marshall, K.; Sikorski, P.; Serpell, L.; Gazit, E.; Archontis, G. Self-assembly of phenylalanine oligopeptides: Insights from experiments and simulations. Biophys. J. 2009, 96, 5020–5029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, X.; Zhu, P.; Li, J. Self-assembly and application of diphenylalanine-based nanostructures. Chem. Soc. Rev. 2010, 39, 1877–1890. [Google Scholar] [CrossRef] [PubMed]
- Vegners, R.; Shestakova, I.; Kalvinsh, I.; Ezzell, R.M.; Janmey, P.A. Use of a gel-forming dipeptide derivative as a carrier for antigen presentation. J. Pept. Sci. 1995, 1, 371–378. [Google Scholar] [CrossRef]
- Mahler, A.; Reches, M.; Rechter, M.; Cohen, S.; Gazit, E. Rigid, self-assembled hydrogel composed of a modified aromatic dipeptide. Adv. Mater. 2006, 18, 1365–1370. [Google Scholar] [CrossRef]
- Smith, A.M.; Williams, R.J.; Tang, C.; Coppo, P.; Collins, R.F.; Turner, M.L.; Saiani, A.; Ulijn, R.V. Fmoc-diphenylalanine self assembles to a hydrogel via a novel architecture based on π–π interlocked β-sheets. Adv. Mater. 2008, 20, 37–41. [Google Scholar] [CrossRef]
- Adler-Abramovich, L.; Vaks, L.; Carny, O.; Trudler, D.; Magno, A.; Caflisch, A.; Frenkel, D.; Gazit, E. Phenylalanine assembly into toxic fibrils suggests amyloid etiology in phenylketonuria. Nat. Chem. Biol. 2012, 8, 701–706. [Google Scholar] [CrossRef]
- Ménard-Moyon, C.; Venkatesh, V.; Krishna, K.V.; Bonachera, F.; Verma, S.; Bianco, A. Self-assembly of tyrosine into controlled supramolecular nanostructures. Chem. Eur. J. 2015, 21, 11681–11686. [Google Scholar] [CrossRef]
- Shaham-Niv, S.; Rehak, P.; Vuković, L.; Adler-Abramovich, L.; Král, P.; Gazit, E. Formation of apoptosis-inducing amyloid fibrils by tryptophan. Isr. J. Chem. 2017, 57, 729–737. [Google Scholar] [CrossRef]
- Perween, S.; Chandanshive, B.; Kotamarthi, H.C.; Khushalani, D. Single amino acid based self-assembled structure. Soft Matter. 2013, 9, 10141–10145. [Google Scholar] [CrossRef]
- Das, T.; Häring, M.; Haldar, D.; Díaz, D.D. Phenylalanine and derivatives as versatile low-molecular-weight gelators: Design, structure and tailored function. Biomater. Sci. 2018, 6, 38–59. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Snigdha, K.; Singh, C.; Sinha, N.; Thakur, A.K. Understanding the self-assembly of Fmoc–phenylalanine to hydrogel formation. Soft Matter. 2015, 11, 5353–5364. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.M.; Anderson, S.B.; Senguen, F.T.; Youngman, R.E.; Nilsson, B.L. Self-assembly and hydrogelation promoted by F 5-phenylalanine. Soft Matter. 2010, 6, 475–479. [Google Scholar] [CrossRef]
- Schnaider, L.; Ghosh, M.; Bychenko, D.; Grigoriants, I.; Ya’ari, S.; Antsel, T.S.; Matalon, S.; Sarig, R.; Brosh, T.; Pilo, R.; et al. Enhanced nanoassembly-incorporated antibacterial composite materials. ACS Appl. Mater. Interfaces 2019, 11, 21334–21342. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Gerassi, D.; Arnon, Z.A.; Guterman, T.; Levin, A.; Ghosh, M.; Aviv, M.; Levy, D.; Knowles, T.P.J.; Shacham-Diamand, Y.; Adler-Abramovich, L. Phase transition and crystallization kinetics of a supramolecular system in a microfluidic platform. Chem. Mater. 2020. [Google Scholar] [CrossRef]
- Gahane, A.Y.; Ranjan, P.; Singh, V.; Sharma, R.K.; Sinha, N.; Sharma, M.; Chaudhry, R.; Thakur, A.K. Fmoc-phenylalanine displays antibacterial activity against gram-positive bacteria in gel and solution phases. Soft Matter. 2018, 14, 2234–2244. [Google Scholar] [CrossRef]
- Halperin-Sternfeld, M.; Ghosh, M.; Sevostianov, R.; Grigoriants, I.; Adler-Abramovich, L. Molecular co-assembly as a strategy for synergistic improvement of the mechanical properties of hydrogels. Chem. Commun. 2017, 53, 9586–9589. [Google Scholar] [CrossRef]
- Adler-Abramovich, L.; Gazit, E. Controlled patterning of peptide nanotubes and nanospheres using inkjet printing technology. J. Pept. Sci. 2008, 14, 217–223. [Google Scholar] [CrossRef]
- Dunavant, T.R.; Regan, J.D.; Glickman, G.N.; Solomon, E.S.; Honeyman, A.L. Comparative evaluation of endodontic irrigants against Enterococcus faecalis biofilms. J. Endod. 2006, 32, 527–531. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Zhang, C.F.; Chu, C.H.; Zhu, X.F. Prevalence of Enterococcus faecalis in saliva and filled root canals of teeth associated with apical periodontitis. Int. J. Oral Sci. 2012, 4, 19–23. [Google Scholar] [CrossRef] [Green Version]
- Hamada, S.; Slade, H.D. Biology, immunology, and cariogenicity of Streptococcus mutans. Microbiol. Rev. 1980, 44, 331–384. [Google Scholar] [CrossRef] [PubMed]
- Krzyściak, W.; Jurczak, A.; Kościelniak, D.; Bystrowska, B.; Skalniak, A. The virulence of Streptococcus mutans and the ability to form biofilms. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 499–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ya’ari, S.; Halperin-Sternfeld, M.; Rosin, B.; Adler-Abramovich, L. Surface Modification by Nano-Structures Reduces Viable Bacterial Biofilm in Aerobic and Anaerobic Environments. Int. J. Mol. Sci. 2020, 21, 7370. https://doi.org/10.3390/ijms21197370
Ya’ari S, Halperin-Sternfeld M, Rosin B, Adler-Abramovich L. Surface Modification by Nano-Structures Reduces Viable Bacterial Biofilm in Aerobic and Anaerobic Environments. International Journal of Molecular Sciences. 2020; 21(19):7370. https://doi.org/10.3390/ijms21197370
Chicago/Turabian StyleYa’ari, Sarah, Michal Halperin-Sternfeld, Boris Rosin, and Lihi Adler-Abramovich. 2020. "Surface Modification by Nano-Structures Reduces Viable Bacterial Biofilm in Aerobic and Anaerobic Environments" International Journal of Molecular Sciences 21, no. 19: 7370. https://doi.org/10.3390/ijms21197370





