Modulation of (Homo)Glutathione Metabolism and H2O2 Accumulation during Soybean Cyst Nematode Infections in Susceptible and Resistant Soybean Cultivars
Abstract
1. Introduction
2. Results
2.1. Root Penetration
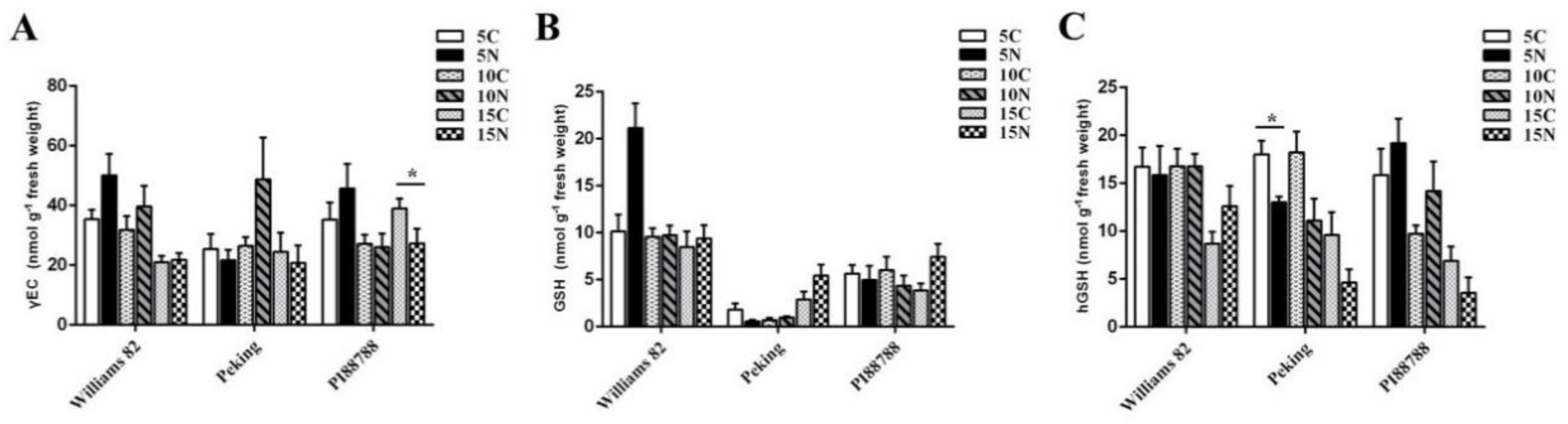
2.2. (h) GSH Metabolism Was Modified in Nematode-Induced Soybean Roots
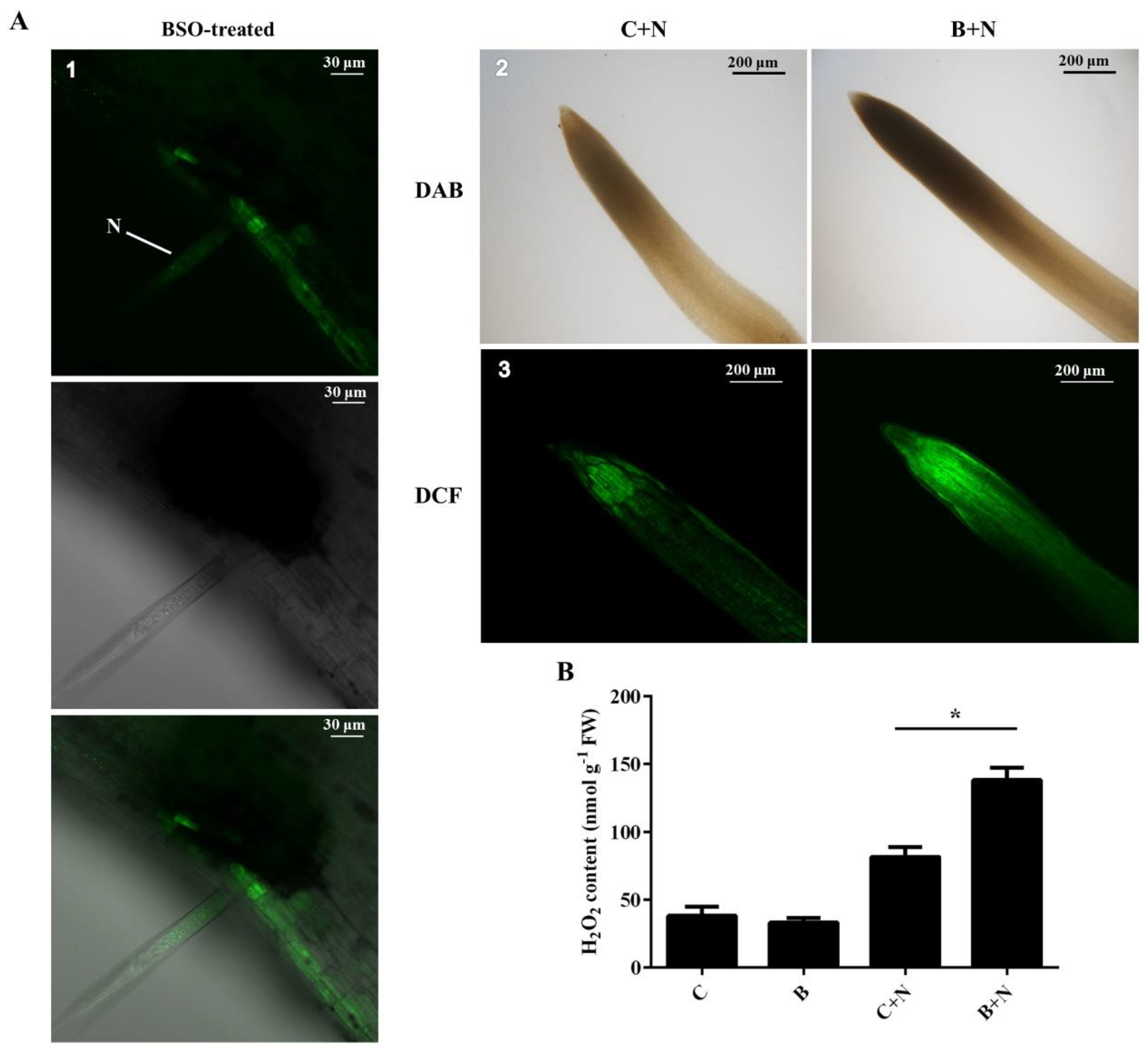
2.3. Quantitative Detection of ROS Production
2.4. (h)GSH Deficiency Impairs Nematode Development and Reproduction
2.5. Association between (h)GSH Deficiency and H2O2 Accumulation in Response to SCN
3. Discussion
4. Materials and Methods
4.1. Plant Material, Growth Condition, and Treatments
4.2. Nematode Inoculum
4.3. Root Penetration Studies
4.4. Total RNA Extraction and Gene Expression Analysis
4.5. γ-Glu-Cys and (h)GSH Determination
4.6. H2O2 Quantification and Cytochemical Detection
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| γ-Glu-Cys | γ-Glutamylcysteine |
| GSH | Reduced glutathione |
| GSSG | Oxidized glutathione |
| hGSH | homoGlutathione |
| γ-ECS | γ-Glutamylcysteine synthetase |
| GSHS | Glutathione synthetase |
| hGSHS | homoGlutathione synthetase |
| ROS | Reactive oxygen species |
| BSO | L-buthionine sulfoximine |
| H2O2 | Hydrogen peroxide |
| O2− | Superoxide |
| HR | Hypersensitive reaction |
| RKN | Root-knot nematode |
| SCN | Soybean cyst nematode |
| J2 | Second-stage juvenile |
| J3 | Third-stage juvenile |
| J4 | Fourth-stage juvenile |
| dpi | Days post-inoculation |
| NaOCl | Sodium hypochlorite |
| SDW | Sterile distilled water |
| HPLC | High-performance liquid chromatography |
| qRT-PCR | Quantitative reverse transcription-PCR |
| CLSM | Confocal laser-scanning microscope |
| SA | Salicylic acid |
| JA | Jasmonic acid |
| ET | Ethylene |
References
- Mathur, S. Soybean the wonder legume. Beverage Food World 2004, 31, 61–62. [Google Scholar]
- Niblack, T.L.; Lambert, K.N.; Tylka, G.L. A model plant pathogen from the kingdom Animalia: Heterodera glycines, the soybean cyst nematode. Annu. Rev. Phytopathol. 2006, 44, 283–303. [Google Scholar] [CrossRef]
- Abad, P.; Gouzy, J.; Aury, J.M.; Castagnone-Sereno, P.; Danchin, E.G.; Deleury, E.; Perfus-Barbeoch, L.; Anthouard, V.; Artiguenave, F.; Blok, V.C.; et al. Genome sequence of the metazoan plant-parasitic nematode Meloidogyne incognita. Nat. Biotechnol. 2008, 26, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Cook, R. Genetic resistance to nematodes: Where is it useful? Australas. Plant. Pathol. 2004, 33, 139–150. [Google Scholar] [CrossRef]
- Hartwig, E.E.; AR-SEA, U. Breeding Productive Soybean Cultivars Resistant to the Soybean Cyst Nematode. Plant. Dis. 1981, 65, 303. [Google Scholar] [CrossRef]
- Concibido, V.C.; Diers, B.W.; Arelli, P.R. A decade of QTL mapping for cyst nematode resistance in soybean. Crop. Sci. 2004, 44, 1121–1131. [Google Scholar] [CrossRef]
- Klink, V.P.; Hosseini, P.; Matsye, P.D.; Alkharouf, N.W.; Matthews, B.F. Syncytium gene expression in Glycine max [PI 88788] roots undergoing a resistant reaction to the parasitic nematode Heterodera glycines. Plant. Physiol. Biochem. 2010, 48, 176–193. [Google Scholar] [CrossRef]
- Endo, B. Histological responses of resistant and susceptible soybean varieties and backcross progeny to entry and development of Heterodera glycines. Phytopathology 1965, 55, 375–381. [Google Scholar]
- Riggs, R.; Kim, K.; Gipson, I. Ultrastructural changes in Peking soybeans infected with Heterodera glycines. Phytopathology 1973, 63, 76–84. [Google Scholar] [CrossRef]
- Acedo, J.R.; Dropkin, V.H.; Luedders, V.D. Nematode Population Attrition and Histopathology of Heterodera glycines-Soybean Associations. J. Nematol. 1984, 16, 48–56. [Google Scholar]
- Kim, Y.H.; Riggs, R.D.; Kim, K.S. Structural Changes Associated with Resistance of Soybean to Heterodera glycines. J. Nematol. 1987, 19, 177–187. [Google Scholar]
- Klink, V.P.; Hosseini, P.; MacDonald, M.H.; Alkharouf, N.W.; Matthews, B.F. Population-specific gene expression in the plant pathogenic nematode Heterodera glycines exists prior to infection and during the onset of a resistant or susceptible reaction in the roots of the Glycine max genotype Peking. BMC Genom. 2009, 10, 111. [Google Scholar] [CrossRef] [PubMed]
- Strohm, M.; Jouanin, L.; Kunert, K.J.; Pruvost, C.; Polle, A.; Foyer, C.H.; Rennenberg, H. Regulation of glutathione synthesis in leaves of transgenic poplar (Populus tremula x P. alba) overexpressing glutathione synthetase. Plant J. 1995, 7, 141–145. [Google Scholar] [CrossRef]
- Noctor, G.; De Paepe, R.; Foyer, C.H. Mitochondrial redox biology and homeostasis in plants. Trends Plant Sci. 2007, 12, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Kopriva, S.; Rennenberg, H. Control of sulphate assimilation and glutathione synthesis: Interaction with N and C metabolism. J. Exp. Bot. 2004, 55, 1831–1842. [Google Scholar] [CrossRef]
- Melillo, M.T.; Leonetti, P.; Bongiovanni, M.; Castagnone-Sereno, P.; Bleve-Zacheo, T. Modulation of reactive oxygen species activities and H2O2 accumulation during compatible and incompatible tomato–root-knot nematode interactions. New Phytol. 2006, 170, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.M.; Murata, Y.; Benning, G.; Thomine, S.; Klusener, B.; Allen, G.J.; Grill, E.; Schroeder, J.I. Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 2000, 406, 731–734. [Google Scholar] [CrossRef]
- Bethke, P.C.; Jones, R.L. Cell death of barley aleurone protoplasts is mediated by reactive oxygen species. Plant J. 2001, 25, 19–29. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Borden, S.; Higgins, V.J. Hydrogen peroxide plays a critical role in the defence response of tomato to Cladosporium fulvum. Physiol. Mol. Plant Pathol. 2002, 61, 227–236. [Google Scholar] [CrossRef]
- Mellersh, D.G.; Foulds, I.V.; Higgins, V.J.; Heath, M.C. H2O2 plays different roles in determining penetration failure in three diverse plant-fungal interactions. Plant J. 2002, 29, 257–268. [Google Scholar] [CrossRef]
- Melillo, M.T.; Leonetti, P.; Leone, A.; Veronico, P.; Bleve-Zacheo, T. ROS and NO production in compatible and incompatible tomato-Meloidogyne incognita interactions. Eur. J. Plant Pathol. 2011, 130, 489–502. [Google Scholar] [CrossRef]
- Vos, C.; Schouteden, N.; Van Tuinen, D.; Chatagnier, O.; Elsen, A.; De Waele, D.; Panis, B.; Gianinazzi-Pearson, V. Mycorrhiza-induced resistance against the root–knot nematode Meloidogyne incognita involves priming of defense gene responses in tomato. Soil Biol. Biochem. 2013, 60, 45–54. [Google Scholar] [CrossRef]
- Zhou, J.; Xu, X.-C.; Cao, J.-J.; Yin, L.-L.; Xia, X.-J.; Shi, K.; Zhou, Y.-H.; Yu, J.-Q. Heat Shock Factor HsfA1a Is Essential for R Gene-Mediated Nematode Resistance and Triggers H2O2 Production1. Plant Physiol. 2018, 176, 2456–2471. [Google Scholar] [CrossRef] [PubMed]
- Lamb, C.; Dixon, R.A. The Oxidative Burst in Plant Disease Resistance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 251–275. [Google Scholar] [CrossRef] [PubMed]
- De Gara, L.; de Pinto, M.C.; Tommasi, F. The antioxidant systems vis-à-vis reactive oxygen species during plant–pathogen interaction. Plant Physiol. Biochem. 2003, 41, 863–870. [Google Scholar] [CrossRef]
- Huysmans, M.; Coll, N.S.; Nowack, M.K. Dying two deaths—Programmed cell death regulation in development and disease. Curr. Opin. Plant Biol. 2017, 35, 37–44. [Google Scholar] [CrossRef]
- Zacheo, G.; Bleve-Zacheo, T. Involvement of superoxide dismutases and superoxide radicals in the susceptibility and resistance of tomato plants to Meloidogyne incognita attack. Physiol. Mol. Plant Pathol. 1988, 32, 313–322. [Google Scholar] [CrossRef]
- Zacheo, G.; Orlando, C.; Bleve-Zacheo, T. Characterization of anionic peroxidases in tomato isolines infected by Meloidogyne incognita. J. Nematol. 1993, 25, 249. [Google Scholar]
- Noctor, G.; Gomez, L.; Vanacker, H.; Foyer, C.H. Interactions between biosynthesis, compartmentation and transport in the control of glutathione homeostasis and signalling. J. Exp. Bot. 2002, 53, 1283–1304. [Google Scholar] [CrossRef]
- Schafer, F.Q.; Buettner, G.R. Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radic. Biol. Med. 2001, 30, 1191–1212. [Google Scholar] [CrossRef]
- Cooper, C.E.; Patel, R.P.; Brookes, P.S.; Darley-Usmar, V.M. Nanotransducers in cellular redox signaling: Modification of thiols by reactive oxygen and nitrogen species. Trends Biochem. Sci. 2002, 27, 489–492. [Google Scholar] [CrossRef]
- Tausz, M.; Šircelj, H.; Grill, D. The glutathione system as a stress marker in plant ecophysiology: Is a stress-response concept valid? J. Exp. Bot. 2004, 55, 1955–1962. [Google Scholar] [CrossRef] [PubMed]
- Zechmann, B.; Koffler, B.E.; Russell, S.D. Glutathione synthesis is essential for pollen germination in vitro. BMC Plant Biol. 2011, 11, 54. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.C.; Ko, K.; Chang, W.L.; Kuo, W.C.; Chen, G.H.; Lin, T.P. Increased glutathione contributes to stress tolerance and global translational changes in Arabidopsis. Plant J. 2015, 83, 926–939. [Google Scholar] [CrossRef]
- Zhu, Y.L.; Pilon-Smits, E.A.; Tarun, A.S.; Weber, S.U.; Jouanin, L.; Terry, N. Cadmium tolerance and accumulation in Indian mustard is enhanced by overexpressing γ-glutamylcysteine synthetase. Plant Physiol. 1999, 121, 1169–1177. [Google Scholar] [CrossRef]
- Chen, J.H.; Jiang, H.W.; Hsieh, E.J.; Chen, H.Y.; Chien, C.T.; Hsieh, H.L.; Lin, T.P. Drought and salt stress tolerance of an Arabidopsis glutathione S-transferase U17 knockout mutant are attributed to the combined effect of glutathione and abscisic acid. Plant Physiol. 2012, 158, 340–351. [Google Scholar] [CrossRef]
- Dubreuil-Maurizi, C.; Poinssot, B. Role of glutathione in plant signaling under biotic stress. Plant Signal. Behav. 2012, 7, 210–212. [Google Scholar] [CrossRef]
- Baldacci-Cresp, F.; Chang, C.; Maucourt, M.; Deborde, C.; Hopkins, J.; Lecomte, P.; Bernillon, S.; Brouquisse, R.; Moing, A.; Abad, P.; et al. (Homo)glutathione deficiency impairs root-knot nematode development in Medicago truncatula. PLoS Pathog. 2012, 8, e1002471. [Google Scholar] [CrossRef]
- Datta, R.; Chattopadhyay, S. Glutathione as a crucial modulator of phytohormone signalling during pathogen defence in plants. Proc. Indian Natl. Sci. Acad. 2018, 84, 581–597. [Google Scholar] [CrossRef]
- Mukherjee, A.K.; Carp, M.-J.; Zuchman, R.; Ziv, T.; Horwitz, B.A.; Gepstein, S. Proteomics of the response of Arabidopsis thaliana to infection with Alternaria brassicicola. J. Proteom. 2010, 73, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Alscher, R.G. Biosynthesis and antioxidant function of glutathione in plants. Physiol. Plant. 1989, 77, 457–464. [Google Scholar] [CrossRef]
- Frendo, P.; Harrison, J.; Norman, C.; Jiménez, M.J.H.; Van de Sype, G.; Gilabert, A.; Puppo, A. Glutathione and homoglutathione play a critical role in the nodulation process of Medicago truncatula. Mol. Plant Microbe Interact. 2005, 18, 254–259. [Google Scholar] [CrossRef]
- Glazebrook, J.; Ausubel, F.M. Isolation of phytoalexin-deficient mutants of Arabidopsis thaliana and characterization of their interactions with bacterial pathogens. Proc. Natl. Acad. Sci. USA 1994, 91, 8955–8959. [Google Scholar] [CrossRef] [PubMed]
- Parisy, V.; Poinssot, B.; Owsianowski, L.; Buchala, A.; Glazebrook, J.; Mauch, F. Identification of PAD2 as a γ-glutamylcysteine synthetase highlights the importance of glutathione in disease resistance of Arabidopsis. Plant J. 2007, 49, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Becana, M.; Dalton, D.A.; Moran, J.F.; Iturbe-Ormaetxe, I.; Matamoros, M.A.; Rubio, M.C. Reactive oxygen species and antioxidants in legume nodules. Physiol. Plant. 2000, 109, 372–381. [Google Scholar] [CrossRef]
- Dalton, D.A.; Russell, S.A.; Hanus, F.; Pascoe, G.A.; Evans, H.J. Enzymatic reactions of ascorbate and glutathione that prevent peroxide damage in soybean root nodules. Proc. Natl. Acad. Sci. USA 1986, 83, 3811–3815. [Google Scholar] [CrossRef]
- Pucciariello, C.; Innocenti, G.; Van de Velde, W.; Lambert, A.; Hopkins, J.; Clement, M.; Ponchet, M.; Pauly, N.; Goormachtig, S.; Holsters, M.; et al. (Homo)glutathione Depletion Modulates Host Gene Expression during the Symbiotic Interaction between Medicago truncatula and Sinorhizobium meliloti. Plant Physiol. 2009, 151, 1186–1196. [Google Scholar] [CrossRef]
- Hernández, J.A.; Ferrer, M.A.; Jiménez, A.; Barceló, A.R.; Sevilla, F. Antioxidant systems and O2.−/H2O2 production in the apoplast of pea leaves. Its relation with salt-induced necrotic lesions in minor veins. Plant Physiol. 2001, 127, 817–831. [Google Scholar] [CrossRef]
- Pignocchi, C.; Foyer, C.H. Apoplastic ascorbate metabolism and its role in the regulation of cell signalling. Curr. Opin. Plant Biol. 2003, 6, 379–389. [Google Scholar] [CrossRef]
- Rouhier, N.; Lemaire, S.D.; Jacquot, J.P. The role of glutathione in photosynthetic organisms: Emerging functions for glutaredoxins and glutathionylation. Annu. Rev. Plant Biol. 2008, 59, 143–166. [Google Scholar] [CrossRef] [PubMed]
- Vernoux, T.; Wilson, R.C.; Seeley, K.A.; Reichheld, J.P.; Muroy, S.; Brown, S.; Maughan, S.C.; Cobbett, C.S.; Van Montagu, M.; Inze, D.; et al. The ROOT MERISTEMLESS1/CADMIUM SENSITIVE2 gene defines a glutathione-dependent pathway involved in initiation and maintenance of cell division during postembryonic root development. Plant Cell 2000, 12, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Reichheld, J.-P.; Khafif, M.; Riondet, C.; Droux, M.; Bonnard, G.; Meyer, Y. Inactivation of thioredoxin reductases reveals a complex interplay between thioredoxin and glutathione pathways in Arabidopsis development. Plant Cell 2007, 19, 1851–1865. [Google Scholar] [CrossRef] [PubMed]
- Bellafiore, S.; Shen, Z.; Rosso, M.N.; Abad, P.; Shih, P.; Briggs, S.P. Direct identification of the Meloidogyne incognita secretome reveals proteins with host cell reprogramming potential. PLoS Pathog. 2008, 4, e1000192. [Google Scholar] [CrossRef] [PubMed]
- Dubreuil, G.; Magliano, M.; Deleury, E.; Abad, P.; Rosso, M.N. Transcriptome analysis of root-knot nematode functions induced in the early stages of parasitism. New Phytol. 2007, 176, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Klessig, D.F.; Durner, J.; Noad, R.; Navarre, D.A.; Wendehenne, D.; Kumar, D.; Zhou, J.M.; Shah, J.; Zhang, S.; Kachroo, P. Nitric oxide and salicylic acid signaling in plant defense. Proc. Natl. Acad. Sci. USA 2000, 97, 8849–8855. [Google Scholar] [CrossRef]
- Waetzig, G.; Sobczak, M.; Grundler, F. Localization of hydrogen peroxide during the defence response of Arabidopsis thaliana against the plant-parasitic nematode Heterodera glycines. Nematology 1999, 1, 681–686. [Google Scholar] [CrossRef]
- Delledonne, M.; Murgia, I.; Ederle, D.; Sbicego, P.F.; Biondani, A.; Polverari, A.; Lamb, C. Reactive oxygen intermediates modulate nitric oxide signaling in the plant hypersensitive disease-resistance response. Plant Physiol. Biochem. 2002, 40, 605–610. [Google Scholar] [CrossRef]
- Yoshida, S.; Tamaoki, M.; Ioki, M.; Ogawa, D.; Sato, Y.; Aono, M.; Kubo, A.; Saji, S.; Saji, H.; Satoh, S. Ethylene and salicylic acid control glutathione biosynthesis in ozone-exposed Arabidopsis thaliana. Physiol. Plant. 2009, 136, 284–298. [Google Scholar] [CrossRef]
- Mestre, T.C.; Garcia-Sanchez, F.; Rubio, F.; Martinez, V.; Rivero, R.M. Glutathione homeostasis as an important and novel factor controlling blossom-end rot development in calcium-deficient tomato fruits. J. Plant Physiol. 2012, 169, 1719–1727. [Google Scholar] [CrossRef]
- Yan, G.; Baidoo, R. Current research status of Heterodera glycines resistance and its implication on soybean breeding. Engineering 2018, 4, 534–541. [Google Scholar] [CrossRef]
- Dangl, J.L.; Jones, J.D. Plant pathogens and integrated defence responses to infection. Nature 2001, 411, 826. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.; Park, J.; Kim, Y.S.; Park, Y.; Kim, Y.H. Screening and histopathological characterization of Korean carrot lines for resistance to the root-knot nematode Meloidogyne incognita. Plant Pathol. J. 2014, 30, 75. [Google Scholar] [CrossRef] [PubMed]
- Ye, D.-Y.; Qi, Y.-H.; Cao, S.-F.; Wei, B.-Q.; Zhang, H.-S. Histopathology combined with transcriptome analyses reveals the mechanism of resistance to Meloidogyne incognita in Cucumis metuliferus. J. Plant Physiol. 2017, 212, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Abad, P.; Favery, B.; Rosso, M.N.; Castagnone-Sereno, P. Root-knot nematode parasitism and host response: Molecular basis of a sophisticated interaction. Mol. Plant Pathol. 2003, 4, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Rual, J.F.; Ceron, J.; Koreth, J.; Hao, T.; Nicot, A.S.; Hirozane-Kishikawa, T.; Vandenhaute, J.; Orkin, S.H.; Hill, D.E.; van den Heuvel, S.; et al. Toward improving Caenorhabditis elegans phenome mapping with an ORFeome-based RNAi library. Genome Res. 2004, 14, 2162–2168. [Google Scholar] [CrossRef]
- Kamath, R.S.; Fraser, A.G.; Dong, Y.; Poulin, G.; Durbin, R.; Gotta, M.; Kanapin, A.; Le Bot, N.; Moreno, S.; Sohrmann, M. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 2003, 421, 231. [Google Scholar] [CrossRef]
- Sonnichsen, B.; Koski, L.B.; Walsh, A.; Marschall, P.; Neumann, B.; Brehm, M.; Alleaume, A.M.; Artelt, J.; Bettencourt, P.; Cassin, E.; et al. Full-genome RNAi profiling of early embryogenesis in Caenorhabditis elegans. Nature 2005, 434, 462–469. [Google Scholar] [CrossRef]
- Klomsiri, C.; Karplus, P.A.; Poole, L.B. Cysteine-based redox switches in enzymes. Antioxid. Redox Signal. 2011, 14, 1065–1077. [Google Scholar] [CrossRef]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.M.; Fujita, M. Exogenous glutathione confers high temperature stress tolerance in mung bean (Vigna radiata L.) by modulating antioxidant defense and methylglyoxal detoxification system. Environ. Exp. Bot. 2015, 112, 44–54. [Google Scholar] [CrossRef]
- Cao, H.; Bowling, S.A.; Gordon, A.S.; Dong, X. Characterization of an Arabidopsis mutant that is nonresponsive to inducers of systemic acquired resistance. Plant Cell 1994, 6, 1583–1592. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.V.; Davis, K.R. Ozone-induced cell death occurs via two distinct mechanisms in Arabidopsis: The role of salicylic acid. Plant J. 1999, 17, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhou, L.; Liu, J.; Cao, Z.; Du, X.; Huang, F.; Pan, G.; Cheng, F. Involvement of CAT in the detoxification of HT-induced ROS burst in rice anther and its relation to pollen fertility. Plant Cell Rep. 2018, 37, 741–757. [Google Scholar] [CrossRef] [PubMed]
- Ghanta, S.; Bhattacharyya, D.; Chattopadhyay, S. Glutathione signaling acts through NPR1-dependent SA-mediated pathway to mitigate biotic stress. Plant Signal. Behav. 2011, 6, 607–609. [Google Scholar] [CrossRef] [PubMed]
- Douillet, P. Disinfection of rotifer cysts leading to bacteria-free populations. J. Exp. Mar. Biol. Ecol. 1998, 224, 183–192. [Google Scholar] [CrossRef]
- Niblack, T.; Heinz, R.; Smith, G.; Donald, P. Distribution, density, and diversity of Heterodera glycines in Missouri. J. Nematol. 1993, 25, 880. [Google Scholar] [PubMed]
- Bybd, D.W., Jr.; Kirkpatrick, T.; Barker, K.R. An improved technique for clearing and staining plant tissues for detection of nematodes. J. Nematol. 1983, 15, 142–143. [Google Scholar] [PubMed]
- De Jesus Miranda, V.; Coelho, R.R.; Viana, A.A.B.; de Oliveira Neto, O.B.; Carneiro, R.M.D.G.; Rocha, T.L.; de Sa, M.F.G.; Fragoso, R.R. Validation of reference genes aiming accurate normalization of qPCR data in soybean upon nematode parasitism and insect attack. BMC Res. Notes 2013, 6, 196. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, J.; Li, X.; Xia, X.-J.; Zhou, Y.-H.; Shi, K.; Chen, Z.; Yu, J.-Q. H2O2 mediates the crosstalk of brassinosteroid and abscisic acid in tomato responses to heat and oxidative stresses. J. Exp. Bot. 2014, 65, 4371–4383. [Google Scholar] [CrossRef]
- Torres, M.A.; Dangl, J.L.; Jones, J.D. Arabidopsis gp91phox homologues AtrbohD and AtrbohF are required for accumulation of reactive oxygen intermediates in the plant defense response. Proc. Natl. Acad. Sci. USA 2002, 99, 517–522. [Google Scholar] [CrossRef] [PubMed]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Li, S.; Zhao, X.; Zhu, X.; Wang, Y.; Xuan, Y.; Liu, X.; Fan, H.; Chen, L.; Duan, Y. Modulation of (Homo)Glutathione Metabolism and H2O2 Accumulation during Soybean Cyst Nematode Infections in Susceptible and Resistant Soybean Cultivars. Int. J. Mol. Sci. 2020, 21, 388. https://doi.org/10.3390/ijms21020388
Chen X, Li S, Zhao X, Zhu X, Wang Y, Xuan Y, Liu X, Fan H, Chen L, Duan Y. Modulation of (Homo)Glutathione Metabolism and H2O2 Accumulation during Soybean Cyst Nematode Infections in Susceptible and Resistant Soybean Cultivars. International Journal of Molecular Sciences. 2020; 21(2):388. https://doi.org/10.3390/ijms21020388
Chicago/Turabian StyleChen, Xi, Shuang Li, Xuebing Zhao, Xiaofeng Zhu, Yuanyuan Wang, Yuanhu Xuan, Xiaoyu Liu, Haiyan Fan, Lijie Chen, and Yuxi Duan. 2020. "Modulation of (Homo)Glutathione Metabolism and H2O2 Accumulation during Soybean Cyst Nematode Infections in Susceptible and Resistant Soybean Cultivars" International Journal of Molecular Sciences 21, no. 2: 388. https://doi.org/10.3390/ijms21020388
APA StyleChen, X., Li, S., Zhao, X., Zhu, X., Wang, Y., Xuan, Y., Liu, X., Fan, H., Chen, L., & Duan, Y. (2020). Modulation of (Homo)Glutathione Metabolism and H2O2 Accumulation during Soybean Cyst Nematode Infections in Susceptible and Resistant Soybean Cultivars. International Journal of Molecular Sciences, 21(2), 388. https://doi.org/10.3390/ijms21020388




