Antimicrobial Chitosan Conjugates: Current Synthetic Strategies and Potential Applications
Abstract
1. Introduction
2. Methodologies for Covalent Bioactive Substances
2.1. Free Radical-Induced Conjugation
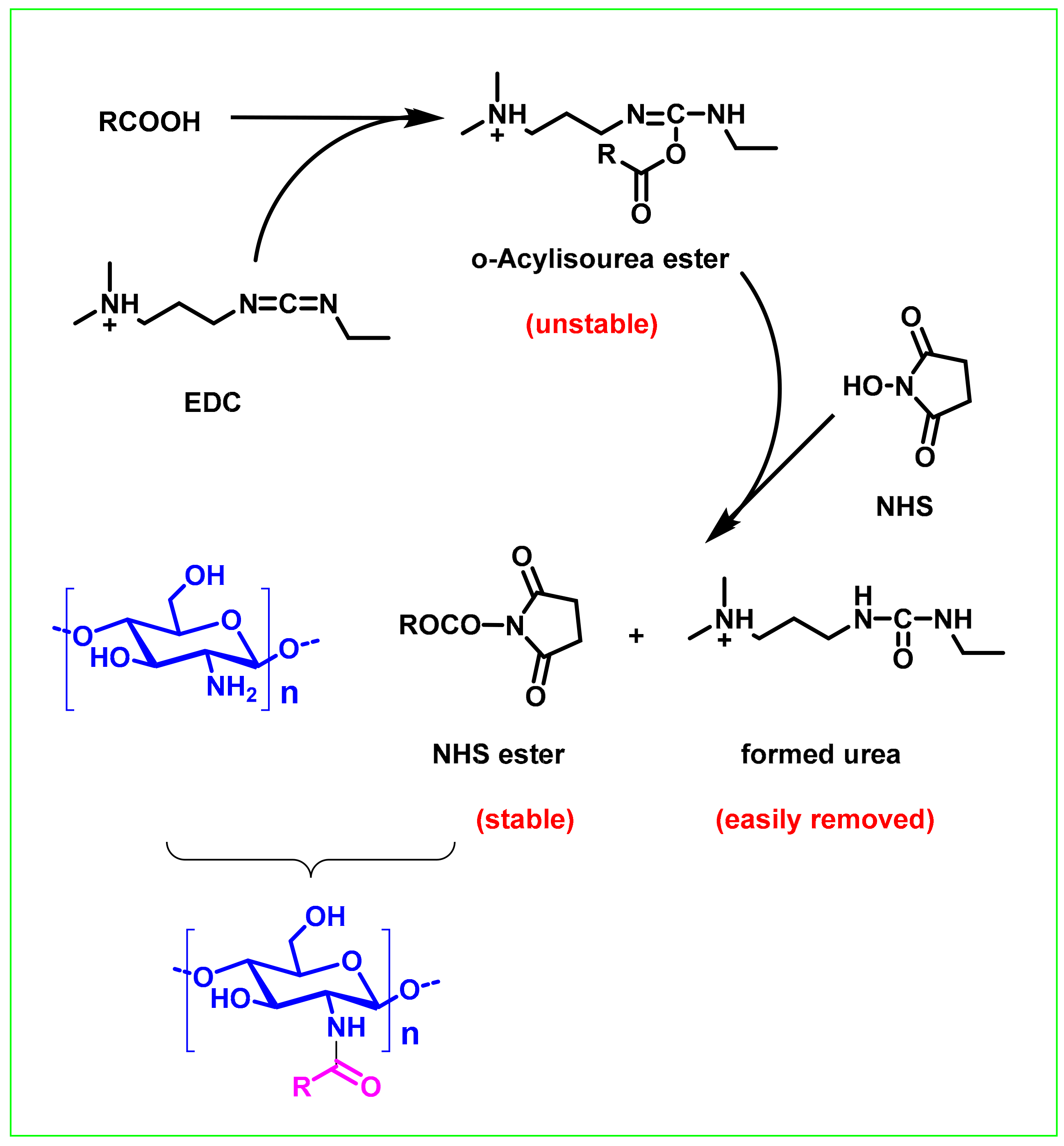
2.2. Carbodiimide Chemistry
2.3. Coupling by Forming a Schiff Base
2.4. Functional Group Conversion Strategy
2.5. Enzyme-Assisted Coupling Reaction
2.6. Other Methods
3. Physiochemical Properties, Antimicrobial Activities, and Potential Applications
3.1. Physiochemical Properties and Antimicrobial Activities
3.2. Potential Applications
4. Challenge and Limitations
4.1. How Can Chitosan Conjugates Be Properly Designdesigned and Synthesized to Ensure Their Effectiveness?
4.2. How Can the Structure-Activity Relationship and Mechanism of Action of the Chitosan Conjugates Be Clarified?
5. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Muxika, A.; Etxabide, A.; Uranga, J.; Guerrero, P.; De La Caba, K. Chitosan as a bioactive polymer: Processing, properties and applications. Int. J. Boil. Macromol. 2017, 105, 1358–1368. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef]
- Halim, A.S.; Periayah, M.H.; Saad, A.Z.M. Chitosan: A promising marine polysaccharide for biomedical research. Pharmacogn. Rev. 2016, 10, 39–42. [Google Scholar] [CrossRef]
- Pillai, C.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678. [Google Scholar] [CrossRef]
- Cheung, R.C.F.; Ng, T.B.; Wong, J.H.; Chan, W.Y. Chitosan: An Update on Potential Biomedical and Pharmaceutical Applications. Mar. Drugs 2015, 13, 5156–5186. [Google Scholar] [CrossRef]
- Kumar, M.N.V.R.; Muzzarelli, R.A.A.; Muzzarelli, C.; Sashiwa, H.; Domb, A.J. Chitosan Chemistry and Pharmaceutical Perspectives. Chem. Rev. 2004, 104, 6017–6084. [Google Scholar] [CrossRef]
- Lunkov, A.P.; Ilyina, A.V.; Varlamov, V.P. Antioxidant, Antimicrobial, and Fungicidal Properties of Chitosan Based Films (Review). Appl. Biochem. Microbiol. 2018, 54, 449–458. [Google Scholar] [CrossRef]
- Sahariah, P.; Másson, M. Antimicrobial Chitosan and Chitosan Derivatives: A Review of the Structure–Activity Relationship. Biomacromolecules 2017, 18, 3846–3868. [Google Scholar] [CrossRef]
- Rabea, E.I.; Badawy, M.E.T.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as antimicrobial agent: Applications and mode of action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar] [CrossRef]
- Sigroha, S.; Khatkar, A. Chitosan—A Naturally Derived Antioxidant Polymer with Diverse Applications. Curr. Org. Chem. 2017, 21, 333–341. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef]
- Ngo, D.-H.; Kim, S.-K. Antioxidant Effects of Chitin, Chitosan, and Their Derivatives. Adv. Food Nutr. Res. 2014, 73, 15–31. [Google Scholar]
- Yuan, G.; Chen, X.; Li, D. Chitosan films and coatings containing essential oils: The antioxidant and antimicrobial activity, and application in food systems. Food Res. Int. 2016, 89, 117–128. [Google Scholar] [CrossRef]
- Zargar, V.; Asghari, M.; Dashti, A. A Review on Chitin and Chitosan Polymers: Structure, Chemistry, Solubility, Derivatives, and Applications. ChemBioEng Rev. 2015, 2, 204–226. [Google Scholar] [CrossRef]
- El Knidri, H.; Belaabed, R.; Addaou, A.; Laajeb, A.; Lahsini, A. Extraction, chemical modification and characterization of chitin and chitosan. Int. J. Boil. Macromol. 2018, 120, 1181–1189. [Google Scholar] [CrossRef]
- Islam, S.-U.; Butola, B. Recent advances in chitosan polysaccharide and its derivatives in antimicrobial modification of textile materials. Int. J. Boil. Macromol. 2019, 121, 905–912. [Google Scholar] [CrossRef]
- Sajid, M.A.; Shahzad, S.A.; Hussain, F.; Skene, W.G.; Khan, Z.A.; Yar, M. Synthetic modifications of chitin and chitosan as multipurpose biopolymers: A review. Synth. Commun. 2018, 48, 1893–1908. [Google Scholar] [CrossRef]
- Mourya, V.; Inamdar, N.N. Chitosan-modifications and applications: Opportunities galore. React. Funct. Polym. 2008, 68, 1013–1051. [Google Scholar] [CrossRef]
- Shinde, U.A.; Joshi, P.N.; Jain, D.D.; Singh, K. Preparation and Evaluation of N-Trimethyl Chitosan Nanoparticles of Flurbiprofen for Ocular Delivery. Curr. Eye Res. 2019, 44, 575–582. [Google Scholar] [CrossRef]
- Rekha, M.R.; Sharma, C.P. Simultaneous Effect of Thiolation and Carboxylation of Chitosan Particles Towards Mucoadhesive Oral Insulin Delivery Applications: An In Vitro and In Vivo Evaluation. J. Biomed. Nanotechnol. 2015, 11, 165–176. [Google Scholar] [CrossRef]
- Popescu, V.; Muresan, A.; Popescu, G.; Balan, M.; Dobromir, M.; Balan-Porcarasu, M. Ethyl chitosan synthesis and quantification of the effects acquired after grafting it on a cotton fabric, using ANOVA statistical analysis. Carbohydr. Polym. 2016, 138, 94–105. [Google Scholar] [CrossRef]
- Mati-Baouche, N.; Delattre, C.; De Baynast, H.; Grédiac, M.; Mathias, J.-D.; Ursu, A.V.; Desbrières, J.; Michaud, P. Alkyl-Chitosan-Based Adhesive: Water Resistance Improvement. Molecules 2019, 24, 1987. [Google Scholar] [CrossRef]
- Mu, Y.; Wu, G.; Su, C.; Dong, Y.; Zhang, K.; Li, J.; Sun, X.; Li, Y.; Chen, X.; Feng, C. pH-sensitive amphiphilic chitosan-quercetin conjugate for intracellular delivery of doxorubicin enhancement. Carbohydr. Polym. 2019, 223, 115072. [Google Scholar] [CrossRef]
- Ding, J.; Xu, Z.; Qi, B.; Cui, S.; Wang, T.; Jiang, L.; Zhang, Y.; Sui, X. Fabrication and characterization of soybean oil bodies encapsulated in maltodextrin and chitosan-EGCG conjugates: An in vitro digestibility study. Food Hydrocoll. 2019, 94, 519–527. [Google Scholar] [CrossRef]
- Hu, Q.; Luo, Y. Polyphenol-chitosan conjugates: Synthesis, characterization, and applications. Carbohydr. Polym. 2016, 151, 624–639. [Google Scholar] [CrossRef]
- Ryu, J.H.; Hong, S.; Lee, H. Bio-inspired adhesive catechol-conjugated chitosan for biomedical applications: A mini review. Acta Biomater. 2015, 27, 101–115. [Google Scholar] [CrossRef]
- Greco, F.; Vicent, M.J. Combination therapy: Opportunities and challenges for polymer-drug conjugates as anticancer nanomedicines. Adv. Drug Deliv. Rev. 2009, 61, 1203–1213. [Google Scholar] [CrossRef]
- Kopeček, J. Polymer-drug conjugates: Origins, progress to date and future directions. Adv. Drug Deliv. Rev. 2013, 65, 49–59. [Google Scholar] [CrossRef]
- Xu, H.; Ma, H.; Yang, P.; Zhang, X.; Wu, X.; Yin, W.; Wang, H.; Xu, N. Targeted polymer-drug conjugates: Current progress and future perspective. Colloids Surf. B Biointerfaces 2015, 136, 729–734. [Google Scholar] [CrossRef]
- Haag, R.; Kratz, F. Polymer Therapeutics: Concepts and Applications. Angew. Chem. Int. Ed. 2006, 45, 1198–1215. [Google Scholar] [CrossRef]
- Ilyasoğlu, H.; Guo, Z. Water soluble chitosan-caffeic acid conjugates as a dual functional polymeric surfactant. Food Biosci. 2019, 29, 118–125. [Google Scholar] [CrossRef]
- Wang, D.; Mao, L.; Dai, L.; Yuan, F.; Gao, Y. Characterization of chitosan-ferulic acid conjugates and their application in the design of β-carotene bilayer emulsions with propylene glycol alginate. Food Hydrocoll. 2018, 80, 281–291. [Google Scholar] [CrossRef]
- Jing, Y.; Diao, Y.; Yu, X. Free radical-mediated conjugation of chitosan with tannic acid: Characterization and antioxidant capacity. React. Funct. Polym. 2019, 135, 16–22. [Google Scholar] [CrossRef]
- Cho, Y.-S.; Lee, D.-S.; Kim, Y.-M.; Ahn, C.-B.; Kim, D.-H.; Jung, W.-K.; Je, J.-Y. Protection of hepatic cell damage and antimicrobial evaluation of chitosan-catechin conjugate. J. Korean Soc. Appl. Boil. Chem. 2013, 56, 701–707. [Google Scholar] [CrossRef]
- Saranya, T.; Rajan, V.; Biswas, R.; Jayakumar, R.; Sathianarayanan, S. Synthesis, characterisation and biomedical applications of curcumin conjugated chitosan microspheres. Int. J. Boil. Macromol. 2018, 110, 227–233. [Google Scholar] [CrossRef]
- Bhat, I.A.; Nazir, M.I.; Ahmad, I.; Pathakota, G.-B.; Chanu, T.; Goswami, M.; Sundaray, J.; Sharma, R. Fabrication and characterization of chitosan conjugated eurycomanone nanoparticles: In vivo evaluation of the biodistribution and toxicity in fish. Int. J. Boil. Macromol. 2018, 112, 1093–1103. [Google Scholar] [CrossRef]
- Mu, H.; Liu, Q.; Niu, H.; Sun, Y.; Duan, J. Gold nanoparticles make chitosan–streptomycin conjugates effective towards Gram-negative bacterial biofilm. RSC Adv. 2016, 6, 8714–8721. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, Y.; He, S.; Zhu, Y.; Ao, M.; Li, J.; Cao, Y. Synthesis and characterization of gibberellin–chitosan conjugate for controlled-release applications. Int. J. Boil. Macromol. 2013, 57, 213–217. [Google Scholar] [CrossRef]
- Fernandes, M.M.; Francesko, A.; Torrent-Burgués, J.; Tzanov, T. Effect of thiol-functionalisation on chitosan antibacterial activity: Interaction with a bacterial membrane model. React. Funct. Polym. 2013, 73, 1384–1390. [Google Scholar] [CrossRef]
- Yuan, S.; Yin, J.; Jiang, W.; Liang, B.; Pehkonen, S.; Choong, C. Enhancing antibacterial activity of surface-grafted chitosan with immobilized lysozyme on bioinspired stainless steel substrates. Colloids Surf. B Biointerfaces 2013, 106, 11–21. [Google Scholar] [CrossRef]
- Jalvandi, J.; White, M.; Gao, Y.; Truong, Y.B.; Padhye, R.; Kyratzis, I.L. Polyvinyl alcohol composite nanofibres containing conjugated levofloxacin-chitosan for controlled drug release. Mater. Sci. Eng. C 2017, 73, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Pawar, V.; Dhanka, M.; Srivastava, R. Cefuroxime conjugated chitosan hydrogel for treatment of wound infections. Colloids Surf. B Biointerfaces 2019, 173, 776–787. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Kunduru, K.R.; Abtew, E.; Domb, A.J. Polysaccharide-Based Conjugates for Biomedical Applications. Bioconjugate Chem. 2015, 26, 1396–1412. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.A.; Abbas, K.; Jantan, I.; Bukhari, S.N.A. Polysaccharide-based materials in macromolecular prodrug design and development. Int. Mater. Rev. 2017, 62, 78–98. [Google Scholar] [CrossRef]
- Woo, J.-Y.; Je, J.-Y. Antioxidant and tyrosinase inhibitory activities of a novel chitosan–phloroglucinol conjugate. Int. J. Food Sci. Technol. 2013, 48, 1172–1178. [Google Scholar] [CrossRef]
- Cong, Y.; Geng, J.; Wang, H.; Su, J.; Arif, M.; Dong, Q.; Chi, Z.; Liu, C. Ureido-modified carboxymethyl chitosan-graft-stearic acid polymeric nano-micelles as a targeted delivering carrier of clarithromycin for Helicobacter pylori: Preparation and in vitro evaluation. Int. J. Boil. Macromol. 2019, 129, 686–692. [Google Scholar] [CrossRef]
- Kim, S.; Cui, Z.-K.; Koo, B.; Zheng, J.; Aghaloo, T.; Lee, M. Chitosan-Lysozyme Conjugates for Enzyme-Triggered Hydrogel Degradation in Tissue Engineering Applications. ACS Appl. Mater. Interfaces 2018, 10, 41138–41145. [Google Scholar] [CrossRef]
- Dumitriu, R.P.; Profire, L.; Nita, L.E.; Dragostin, O.M.; Ghetu, N.; Pieptu, D.; Vasile, C. Sulfadiazine-Chitosan Conjugates and Their Polyelectrolyte Complexes with Hyaluronate Destined to the Management of Burn Wounds. Materials 2015, 8, 317–338. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. Phytotherapy Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef]
- Francisco Javier, A.-M.; Enrique, B.-C.; Jose Antonio, E.; Juan Carlos, R.-D.; Vicente, M. Antimicrobial Capacity of Plant Polyphenols against Gram-positive Bacteria: A Comprehensive Review. Curr. Med. Chem. 2018, 25, 1–29. [Google Scholar]
- Sant’Anna, V.; Biondo, E.; Kolchinski, E.M.; da Silva, L.F.S.; Corrêa, A.P.F.; Bach, E.; Brandelli, A. Total Polyphenols, Antioxidant, Antimicrobial and Allelopathic Activities of Spend Coffee Ground Aqueous Extract. Waste Biomass Valoriz. 2017, 8, 439–442. [Google Scholar] [CrossRef]
- Trošt, K.; Vodopivec, B.M.; Lemut, M.S.; Jug, K.; Raspor, P.; Možina, S.S.; Klančnik, A. Polyphenol, antioxidant and antimicrobial potential of six different white and red wine grape processing leftovers. J. Sci. Food Agric. 2016, 96, 4809–4820. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhao, Y.; Padilla-Zakour, O.; Yang, G. Polyphenols, antioxidant and antimicrobial activities of leaf and bark extracts of Solidago canadensis L. Ind. Crop. Prod. 2015, 74, 803–809. [Google Scholar] [CrossRef]
- Lee, D.-S.; Woo, J.-Y.; Ahn, C.-B.; Je, J.-Y. Chitosan-hydroxycinnamic acid conjugates: Preparation, antioxidant and antimicrobial activity. Food Chem. 2014, 148, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Wang, T.; Zhou, M.; Xue, J.; Luo, Y. Formation of redispersible polyelectrolyte complex nanoparticles from gallic acid-chitosan conjugate and gum arabic. Int. J. Boil. Macromol. 2016, 92, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Dutta, P.K.; Kumar, H.; Kureel, A.K.; Rai, A.K. Improved antibacterial and antioxidant activities of gallic acid grafted chitin-glucan complex. J. Polym. Res. 2019, 26, 234. [Google Scholar] [CrossRef]
- Liu, J.; Lu, J.-F.; Kan, J.; Jin, C.-H. Synthesis of chitosan-gallic acid conjugate: Structure characterization and in vitro anti-diabetic potential. Int. J. Boil. Macromol. 2013, 62, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Wang, T.; Zhou, M.; Xue, J.; Luo, Y. In Vitro Antioxidant-Activity Evaluation of Gallic-Acid-Grafted Chitosan Conjugate Synthesized by Free-Radical-Induced Grafting Method. J. Agric. Food Chem. 2016, 64, 5893–5900. [Google Scholar] [CrossRef]
- Liu, J.; Pu, H.; Chen, C.; Liu, Y.; Bai, R.; Kan, J.; Jin, C. Reaction Mechanisms and Structural and Physicochemical Properties of Caffeic Acid Grafted Chitosan Synthesized in Ascorbic Acid and Hydroxyl Peroxide Redox System. J. Agric. Food Chem. 2018, 66, 279–289. [Google Scholar] [CrossRef]
- Marín, A.C.; Culcasi, M.; Cassien, M.; Stocker, P.; Thétiot-Laurent, S.; Robillard, B.; Chinnici, F.; Pietri, S. Chitosan as an antioxidant alternative to sulphites in oenology: EPR investigation of inhibitory mechanisms. Food Chem. 2019, 285, 67–76. [Google Scholar] [CrossRef]
- Saiki, S.; Nagasawa, N.; Hiroki, A.; Morishita, N.; Tamada, M.; Muroya, Y.; Kudo, H.; Katsumura, Y. ESR study on carboxymethyl chitosan radicals in an aqueous solution. Radiat. Phys. Chem. 2010, 79, 276–278. [Google Scholar] [CrossRef]
- Castagnino, E.; Ottaviani, M.F.; Cangiotti, M.; Morelli, M.; Casettari, L.; Muzzarelli, R.A. Radical scavenging activity of 5-methylpyrrolidinone chitosan and dibutyryl chitin. Carbohydr. Polym. 2008, 74, 640–647. [Google Scholar] [CrossRef]
- Jiao, W.; Shu, C.; Li, X.; Cao, J.; Fan, X.; Jiang, W. Preparation of a chitosan-chlorogenic acid conjugate and its application as edible coating in postharvest preservation of peach fruit. Postharvest Boil. Technol. 2019, 154, 129–136. [Google Scholar] [CrossRef]
- Woranuch, S.; Yoksan, R. Preparation, characterization and antioxidant property of water-soluble ferulic acid grafted chitosan. Carbohydr. Polym. 2013, 96, 495–502. [Google Scholar] [CrossRef]
- Netsomboon, K.; Suchaoin, W.; Laffleur, F.; Prüfert, F.; Bernkop-Schnürch, A. Multifunctional adhesive polymers: Preactivated thiolated chitosan-EDTA conjugates. Eur. J. Pharm. Biopharm. 2017, 111, 26–32. [Google Scholar] [CrossRef]
- Shrestha, A.; Hamblin, M.R.; Kishen, A. Characterization of a Conjugate between Rose Bengal and Chitosan for Targeted Antibiofilm and Tissue Stabilization Effects as a Potential Treatment of Infected Dentin. Antimicrob. Agents Chemother. 2012, 56, 4876–4884. [Google Scholar] [CrossRef]
- Xu, R.; Ma, S.; Wu, Y.; Lee, H.; Zhou, F.; Liu, W. Adaptive control in lubrication, adhesion, and hemostasis by Chitosan-Catechol-pNIPAM. Biomater. Sci. 2019, 7, 3599–3608. [Google Scholar] [CrossRef]
- Hozumi, K.; Nomizu, M. Mixed Peptide-Conjugated Chitosan Matrices as Multi-Receptor Targeted Cell-Adhesive Scaffolds. Int. J. Mol. Sci. 2018, 19, 2713. [Google Scholar] [CrossRef]
- Wang, Y.; Xie, M.; Ma, G.; Fang, Y.; Yang, W.; Ma, N.; Fang, D.; Hu, Q.; Pei, F. The antioxidant and antimicrobial activities of different phenolic acids grafted onto chitosan. Carbohydr. Polym. 2019, 225, 115238. [Google Scholar] [CrossRef]
- Quiñones, J.P.; Szopko, R.; Schmidt, C.; Covas, C.P. Novel drug delivery systems: Chitosan conjugates covalently attached to steroids with potential anticancer and agrochemical activity. Carbohydr. Polym. 2011, 84, 858–864. [Google Scholar] [CrossRef]
- Rui, L.; Xie, M.; Hu, B.; Zhou, L.; Saeeduddin, M.; Zeng, X. Enhanced solubility and antioxidant activity of chlorogenic acid-chitosan conjugates due to the conjugation of chitosan with chlorogenic acid. Carbohydr. Polym. 2017, 170, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Rui, L.; Xie, M.; Hu, B.; Zhou, L.; Yin, D.; Zeng, X. A comparative study on chitosan/gelatin composite films with conjugated or incorporated gallic acid. Carbohydr. Polym. 2017, 173, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Jing, Z.-W.; Jia, Y.-Y.; Wan, N.; Luo, M.; Huan, M.-L.; Kang, T.-B.; Zhou, S.-Y.; Zhang, B.-L. Design and evaluation of novel pH-sensitive ureido-conjugated chitosan/TPP nanoparticles targeted to Helicobacter pylori. Biomaterials 2016, 84, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Koh, J.; Kim, H.; Gupta, M.; Dutta, P. A new chitosan–thymine conjugate: Synthesis, characterization and biological activity. Int. J. Boil. Macromol. 2012, 50, 493–502. [Google Scholar] [CrossRef]
- Kafedjiiski, K.; Föger, F.; Werle, M.; Bernkop-Schnürch, A. Synthesis and in Vitro Evaluation of a Novel Chitosan–Glutathione Conjugate. Pharm. Res. 2005, 22, 1480–1488. [Google Scholar] [CrossRef]
- Antony, R.; Arun, T.; Manickam, S.T.D. A review on applications of chitosan-based Schiff bases. Int. J. Boil. Macromol. 2019, 129, 615–633. [Google Scholar] [CrossRef]
- Yin, X.; Chen, J.; Yuan, W.; Lin, Q.; Ji, L.; Liu, F. Preparation and antibacterial activity of Schiff bases from O-carboxymethyl chitosan and para-substituted benzaldehydes. Polym. Bull. 2012, 68, 1215–1226. [Google Scholar] [CrossRef]
- Tamer, T.M.; Hassan, M.A.; Omer, A.M.; Baset, W.M.; Hassan, M.E.; El-Shafeey, M.E.; Eldin, M.S.M. Synthesis, characterization and antimicrobial evaluation of two aromatic chitosan Schiff base derivatives. Process. Biochem. 2016, 51, 1721–1730. [Google Scholar] [CrossRef]
- Liu, W.; Qin, Y.; Liu, S.; Xing, R.; Yu, H.; Chen, X.; Li, K.; Li, P. C-coordinated O-carboxymethyl chitosan metal complexes: Synthesis, characterization and antifungal efficacy. Int. J. Boil. Macromol. 2018, 106, 68–77. [Google Scholar] [CrossRef]
- Liu, W.; Qin, Y.; Liu, S.; Xing, R.; Yu, H.; Chen, X.; Li, K.; Li, P. Synthesis, characterization and antifungal efficacy of C-coordinated O-carboxymethyl chitosan Cu(II) complexes. Carbohydr. Polym. 2017, 160, 97–105. [Google Scholar] [CrossRef]
- Wei, L.; Tan, W.; Wang, G.; Li, Q.; Dong, F.; Guo, Z. The antioxidant and antifungal activity of chitosan derivatives bearing Schiff bases and quaternary ammonium salts. Carbohydr. Polym. 2019, 226, 115256. [Google Scholar] [CrossRef]
- Li, X.; Wu, P.; Cheng, S.; Lv, X. Synthesis and Assessment of Globotriose–Chitosan Conjugate, a Novel Inhibitor of Shiga Toxins Produced byEscherichia coli. J. Med. Chem. 2012, 55, 2702–2710. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Liu, J.; Li, R.; Jiao, S.; Feng, C.; Wang, Z.A.; Du, Y. Conjugation of Inulin Improves Anti-Biofilm Activity of Chitosan. Mar. Drugs 2018, 16, 151. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Han, B.; Zheng, Y.; Liu, L.; Li, C.; Sheng, S.; Zhang, J.; Wang, J.; Wu, F. The Quality Changes of Postharvest Mulberry Fruit Treated by Chitosan-g-Caffeic Acid during Cold Storage. J. Food Sci. 2016, 81, C881–C888. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Mu, H.; Zhang, W.; Cui, G.; Zhu, J.; Duan, J. Chitosan Coupling Makes Microbial Biofilms Susceptible to Antibiotics. Sci. Rep. 2013, 3, 3364. [Google Scholar] [CrossRef]
- Li, R.; Yuan, X.; Wei, J.; Zhang, X.; Cheng, G.; Wang, Z.A.; Du, Y. Synthesis and Evaluation of a Chitosan Oligosaccharide-Streptomycin Conjugate against Pseudomonas aeruginosa Biofilms. Mar. Drugs 2019, 17, 43. [Google Scholar] [CrossRef]
- Barbosa, M.; Vale, N.; Costa, F.M.; Martins, M.C.L.; Gomes, P. Tethering antimicrobial peptides onto chitosan: Optimization of azide-alkyne “click” reaction conditions. Carbohydr. Polym. 2017, 165, 384–393. [Google Scholar] [CrossRef]
- Kamoun, E.A. N-succinyl chitosan–dialdehyde starch hybrid hydrogels for biomedical applications. J. Adv. Res. 2016, 7, 69–77. [Google Scholar] [CrossRef]
- Petrin, T.H.C.; Fadel, V.; Martins, D.B.; Dias, S.A.; Cruz, A.; Sergio, L.M.; Arcisio-Miranda, M.; Castanho, M.A.R.B.; Cabrera, M.P.D.S. Synthesis and Characterization of Peptide-Chitosan Conjugates (PepChis) with Lipid Bilayer Affinity and Antibacterial Activity. Biomacromolecules 2019, 20, 2743–2753. [Google Scholar] [CrossRef]
- Zampano, G.; Bertoldo, M.; Ciardelli, F. Defined Chitosan-based networks by C-6-Azide–alkyne “click” reaction. React. Funct. Polym. 2010, 70, 272–281. [Google Scholar] [CrossRef]
- Hu, L.; Meng, X.; Xing, R.; Liu, S.; Chen, X.; Qin, Y.; Yu, H.; Li, P. Design, synthesis and antimicrobial activity of 6- N -substituted chitosan derivatives. Bioorganic Med. Chem. Lett. 2016, 26, 4548–4551. [Google Scholar] [CrossRef] [PubMed]
- Luan, F.; Li, Q.; Tan, W.; Wei, L.; Zhang, J.; Dong, F.; Gu, G.; Guo, Z. The evaluation of antioxidant and antifungal properties of 6-amino-6-deoxychitosan in vitro. Int. J. Boil. Macromol. 2018, 107, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Moussa, A.; Crépet, A.; Ladavière, C.; Trombotto, S. Reducing-end “clickable” functionalizations of chitosan oligomers for the synthesis of chitosan-based diblock copolymers. Carbohydr. Polym. 2019, 219, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Dutta, P.; Kumar, H.; Kureel, A.K.; Rai, A.K. Synthesis of chitin-glucan-aldehyde-quercetin conjugate and evaluation of anticancer and antioxidant activities. Carbohydr. Polym. 2018, 193, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, S.B.; Krolicka, M.; Broek, L.A.V.D.; Frissen, A.E.; Boeriu, C.G. Water-soluble chitosan derivatives and pH-responsive hydrogels by selective C-6 oxidation mediated by TEMPO-laccase redox system. Carbohydr. Polym. 2018, 186, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.; Costa, F.; Monteiro, C.; Duarte, F.; Martins, M.C.L.; Gomes, P. Antimicrobial coatings prepared from Dhvar-5-click-grafted chitosan powders. Acta Biomater. 2019, 84, 242–256. [Google Scholar] [CrossRef] [PubMed]
- Gaballah, S.T.; El-Nazer, H.A.; Abdel-Monem, R.A.; El-Liethy, M.A.; Hemdan, B.A.; Rabie, S.T. Synthesis of novel chitosan-PVC conjugates encompassing Ag nanoparticles as antibacterial polymers for biomedical applications. Int. J. Boil. Macromol. 2019, 121, 707–717. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, B.; Javvaji, V.; Kim, E.; Lee, M.E.; Raghavan, S.R.; Wang, Q.; Payne, G.F. Tyrosinase-mediated grafting and crosslinking of natural phenols confers functional properties to chitosan. Biochem. Eng. J. 2014, 89, 21–27. [Google Scholar] [CrossRef]
- Sakai, S.; Khanmohammadi, M.; Khoshfetrat, A.B.; Taya, M.; Shinji, S. Horseradish peroxidase-catalyzed formation of hydrogels from chitosan and poly (vinyl alcohol) derivatives both possessing phenolic hydroxyl groups. Carbohydr. Polym. 2014, 111, 404–409. [Google Scholar] [CrossRef]
- Božič, M.; Gorgieva, S.; Kokol, V. Laccase-mediated functionalization of chitosan by caffeic and gallic acids for modulating antioxidant and antimicrobial properties. Carbohydr. Polym. 2012, 87, 2388–2398. [Google Scholar] [CrossRef]
- Aljawish, A.; Chevalot, I.; Jasniewski, J.; Paris, C.; Scher, J.; Muniglia, L. Laccase-catalysed oxidation of ferulic acid and ethyl ferulate in aqueous medium: A green procedure for the synthesis of new compounds. Food Chem. 2014, 145, 1046–1054. [Google Scholar] [CrossRef]
- Božič, M.; Gorgieva, S.; Kokol, V. Homogeneous and heterogeneous methods for laccase-mediated functionalization of chitosan by tannic acid and quercetin. Carbohydr. Polym. 2012, 89, 854–864. [Google Scholar] [CrossRef]
- Sampaio, S.; Taddei, P.; Monti, P.; Buchert, J.; Freddi, G. Enzymatic grafting of chitosan onto Bombyx mori silk fibroin: Kinetic and IR vibrational studies. J. Biotechnol. 2005, 116, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.; Littarru, G.; Muzzarelli, C.; Tosi, G. Selective reactivity of biochemically relevant quinones towards chitosans. Carbohydr. Polym. 2003, 53, 109–115. [Google Scholar] [CrossRef]
- Aljawish, A.; Chevalot, I.; Piffaut, B.; Rondeau-Mouro, C.; Girardin, M.; Jasniewski, J.; Scher, J.; Muniglia, L. Functionalization of chitosan by laccase-catalyzed oxidation of ferulic acid and ethyl ferulate under heterogeneous reaction conditions. Carbohydr. Polym. 2012, 87, 537–544. [Google Scholar] [CrossRef]
- Saito, H.; Sakakibara, Y.; Sakata, A.; Kurashige, R.; Murakami, D.; Kageshima, H.; Saito, A.; Miyazaki, Y. Antibacterial activity of lysozyme-chitosan oligosaccharide conjugates (LYZOX) against Pseudomonas aeruginosa, Acinetobacter baumannii and Methicillin-resistant Staphylococcus aureus. PLoS ONE 2019, 14, e0217504. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Liu, Y.; Yang, X.; Bentley, W.E.; Payne, G.F.; Shi, X.-W. Biomimetic Approach to Confer Redox Activity to Thin Chitosan Films. Adv. Funct. Mater. 2010, 20, 2683–2694. [Google Scholar] [CrossRef]
- Li, X.; Shi, X.; Wang, M.; Du, Y. Xylan chitosan conjugate—A potential food preservative. Food Chem. 2011, 126, 520–525. [Google Scholar] [CrossRef]
- Liang, C.; Yuan, F.; Liu, F.; Wang, Y.; Gao, Y. Structure and antimicrobial mechanism of ɛ-polylysine–chitosan conjugates through Maillard reaction. Int. J. Boil. Macromol. 2014, 70, 427–434. [Google Scholar] [CrossRef]
- Song, Y.E.; Babiker, E.; Usui, M.; Saito, A.; Kato, A. Emulsifying properties and bactericidal action of chitosan-lysozyme conjugates. Food Res. Int. 2002, 35, 459–466. [Google Scholar] [CrossRef]
- Hofmann, T.; Bors, W.; Stettmaier, K. Studies on radical intermediates in the early stage of the nonenzymatic browning reaction of carbohydrates and amino acids. J. Agric. Food Chem. 1999, 47, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Berezin, A.S.; Skorik, Y.A. Chitosan-isoniazid conjugates: Synthesis, evaluation of tuberculostatic activity, biodegradability and toxicity. Carbohydr. Polym. 2015, 127, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Jing, Y.; Huang, J.; Yu, X. Preparation, characterization, and functional evaluation of proanthocyanidin-chitosan conjugate. Carbohydr. Polym. 2018, 194, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Chen, T.; Mei, N.; Li, B.; Xu, Z.; Wang, L.; Wang, X.; Tang, S. LED 209 conjugated chitosan as a selective antimicrobial and potential anti-adhesion material. Carbohydr. Polym. 2019, 206, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-S.; Je, J.-Y. Gallic Acid- Grafted-Chitosan Inhibits Foodborne Pathogens by a Membrane Damage Mechanism. J. Agric. Food Chem. 2013, 61, 6574–6579. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Yu, D.; Eom, S.-H.; Kim, S.-H.; Oh, J.; Jung, W.; Kim, Y.-M. Synergistic Antibacterial Effects of Chitosan-Caffeic Acid Conjugate against Antibiotic-Resistant Acne-Related Bacteria. Mar. Drugs 2017, 15, 167. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Hu, Y.; Xu, D.; Cai, K. Surface functionalization of titanium substrates with chitosan–lauric acid conjugate to enhance osteoblasts functions and inhibit bacteria adhesion. Colloids Surf. B Biointerfaces 2014, 119, 115–125. [Google Scholar] [CrossRef]
- Yang, C.; Zhou, Y.; Zheng, Y.; Li, C.; Sheng, S.; Wang, J.; Wu, F. Enzymatic modification of chitosan by cinnamic acids: Antibacterial activity against Ralstonia solanacearum. Int. J. Boil. Macromol. 2016, 87, 577–585. [Google Scholar] [CrossRef]
- Amato, A.; Migneco, L.M.; Martinelli, A.; Pietrelli, L.; Piozzi, A.; Francolini, I. Antimicrobial activity of catechol functionalized-chitosan versus Staphylococcus epidermidis. Carbohydr. Polym. 2018, 179, 273–281. [Google Scholar] [CrossRef]
- Xu, Y.; Fan, H.; Lu, C.; Gao, G.F.; Li, X. Synthesis of Galabiose-chitosan Conjugate as Potent Inhibitor of Streptococcus suis Adhesion. Biomacromolecules 2010, 11, 1701–1704. [Google Scholar] [CrossRef]
- Chatterjee, N.S.; Panda, S.K.; Navitha, M.; Asha, K.K.; Anandan, R.; Mathew, S. Vanillic acid and coumaric acid grafted chitosan derivatives: Improved grafting ratio and potential application in functional food. J. Food Sci. Technol. 2015, 52, 7153–7162. [Google Scholar] [CrossRef]
- Verlee, A.; Mincke, S.; Stevens, C.V. Recent developments in antibacterial and antifungal chitosan and its derivatives. Carbohydr. Polym. 2017, 164, 268–283. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, R.; Liu, W. Effects of Chitin and Its Derivative Chitosan on Postharvest Decay of Fruits: A Review. Int. J. Mol. Sci. 2011, 12, 917–934. [Google Scholar] [CrossRef] [PubMed]
- Raafat, D.; Sahl, H.-G. Chitosan and its antimicrobial potential—a critical literature survey. Microb. Biotechnol. 2009, 2, 186–201. [Google Scholar] [CrossRef] [PubMed]





| Number | Sample | Active Molecule | Linker | Bond Type | References |
|---|---|---|---|---|---|
| 1 | Chitosan-tannic acid: Conjugate | tannic acid | No | Amide/ester | [33] |
| 2 | Gallic acid-chitosan conjugate | gallic acid | No | Amide/ester | [55] |
| 3 | Chitosan–hydroxycinnamic acid conjugates | hydroxycinnamic acid | No | Amide/ester | [54] |
| 4 | Gibberellin–chitosan conjugate | gibberellin | No | Amide | [38] |
| 5 | Chitosan-caffeic acid conjugates | caffeic acid | No | Amide | [31] |
| 6 | Curcuminconjugated chitosan | curcumin | No | Imino | [35] |
| 7 | Chitosan-isoniazid conjugates | isoniazid | epichlorohydrin | Hydrazo/hydrazide | [112] |
| 8 | Chitosan–thymine conjugate | thymine | bromoacetic acid | Amide | [74] |
| 9 | chitosan-PVC conjugates | PVC | bromoacetyl bromide | Alkylamino | [97] |
| Sample | Microorganism | Antimicrobial Properties | References |
|---|---|---|---|
| Chitosan-Catechin Conjugate | Bacillus subtilis | MIC: 64 μg/mL (Conjugate); 128 μg/mL (chitosan) | [34] |
| Chitosan-Caffeic Acid Conjugate | Staphylococcus aureus | MIC: 8 μg/mL (Conjugate); 16 μg/m(chitosan) | [116] |
| Chitosan–hydroxycinnamic acid conjugates | Bacillus subtilis | MIC: 2 μg/mL (CFA (I)); 128 μg/mL(chitosan) | [54] |
| Chitosan thiolated conjugate | E. coli | MIC: 2.9 mg/mL (TCNAC); 4.1 mg/mL(chitosan) | [39] |
| Chitosan thiolated conjugate | Bacillus subtilis | MIC: 0.25 mg/mL (CA-g-CS); 2 mg/mL(chitosan) | [69] |
| chitosan–lauricacid conjugate | Staphylococcus aureus | Antibacterial rate: 95.6% (Ti-PDOP-Chi–2.5%LA) | [117] |
| Cinnamic acids conjugated chitosan | Ralstonia solanacearum−5 | IC50: 0.23 mg/mL (CTS-g-CA)); 0.56 mg/mL(chitosan) | [118] |
| Chitosan gallic acid conjugate | C. albicans | Reduction of fungi: 70%(GA); 61%(chitosan) | [100] |
| Sulfadiazine—Chitosan Conjugates | Listeria monocytogenes | Inhibition rate: 100% (PEC Lm-SDZ); 58%(chitosan) | [48] |
| Polylysine–chitosanconjugates | Beer yeast | Inhibition zone: 1.36 cm (conjugate) 0.96 cm (chitosan) | [109] |
| Hydrocaffeic acid conjugated chitosan | S. epidermidis | Enhanced antimicrobial activity compared to pure chitosan. | [119] |
| Sample | Potential Applications | Physiochemical Properties | Bio-Functions | Cytotoxicity | References |
|---|---|---|---|---|---|
| LED 209 conjugated chitosan | Antimicrobial and anti-adhesion material | Increasing solubility with increase in DS | Highly selective activity and anti-adhesion activity against MDR-E. coli | Minor cytotoxicity to mammalian cells. | [114] |
| Galabiose-chitosan Conjugate | Anti-adhesion agents | Showed good solubility in neutral water (1.0 mg/mL) | The highest inhibitory effect at the DP of 1839 (MIC:1.7 nM) | n.d | [120] |
| Gibberellin–chitosan | Fungicide release | Good water solubility and stability | n.d | n.d | [38] |
| Dhvar-5-chitosan conjugate | Antimicrobial surfaces | lower viscosity values | Displayed bactericidal effect. | No cytotoxic potential. | [96] |
| Curcumin conjugated chitosan | Anti-skin infection agent | n.d | Be effective against E. coli and S. Auerus | Cyto and hemo-compatible | [35] |
| Inulin–LCS Conjugate | Anti-biofilm reagent | Good water solubility | Showed similar biofilm eradication with florfenicol at 500 μg/mL | Low cellular toxicity to mammalian | [83] |
| Gallic acid-grafted-chitosan | Biomaterials in food packaging | An increase in water solubility; exhibited darker appearance and weaker transmittance | Significantly enhanced antibacterial ability | n.d | [72] |
| Gallic acid grafted chitin-glucan complex | Biomedical areas | n.d | Completely inhibited the growth of Bacillus subtilis and Escherichia coli | Non-hazardous and biocompatible | |
| Chitosan–hydroxycinnamic acid conjugates | Food and pharmaceutical industries | n.d | Exhibited better antimicrobial activity than chitosan | No cytotoxic activity | [56] |
| Chitosan–lysozyme conjugates | Ingredient with emulsifying properties | Greatly improved solubility and emulsion stability | Enhanced bactericidal action against Escherichia coil K-12, | n.d | [54] |
| lysozyme-chitosan oligosaccharide conjugates | Refractory infection drugs | n.d | Exhibited antibacterial activity and low drug resistance | Low hemolytic activity | [110] |
| Cefuroxime conjugated chitosan | Anti-chronic wound infection drugs | Decrease in the rate of swelling and degradation rate | Showed an efficient antibacterial activity over a longer period. | Have good blood compatibility | [106] |
| Chitosan-phenolic acid conjugates | Food preservatives | Improved water solubility | Showed broad spectrum antibacterial activity | n.d | [42] |
| Chitosan-isoniazid conjugates | Antituberculosis drugs | Enhanced solubility under physi-ological conditions | Comparable or slightly higher minimum inhibitory concentration for conjugates than for INH itself | Reduced biodegradability and decreased toxicity | [121] |
| Proanthocyanidin-chitosan conjugate | Food nutraceutical, and Biomedicine filed | Had lower crystallinity and thermal stability than chitosan | Showed bacterial strain-depended behavior in the antibacterial activity compared with chitosan | n.d | [112] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, Y.; Li, P. Antimicrobial Chitosan Conjugates: Current Synthetic Strategies and Potential Applications. Int. J. Mol. Sci. 2020, 21, 499. https://doi.org/10.3390/ijms21020499
Qin Y, Li P. Antimicrobial Chitosan Conjugates: Current Synthetic Strategies and Potential Applications. International Journal of Molecular Sciences. 2020; 21(2):499. https://doi.org/10.3390/ijms21020499
Chicago/Turabian StyleQin, Yukun, and Pengcheng Li. 2020. "Antimicrobial Chitosan Conjugates: Current Synthetic Strategies and Potential Applications" International Journal of Molecular Sciences 21, no. 2: 499. https://doi.org/10.3390/ijms21020499
APA StyleQin, Y., & Li, P. (2020). Antimicrobial Chitosan Conjugates: Current Synthetic Strategies and Potential Applications. International Journal of Molecular Sciences, 21(2), 499. https://doi.org/10.3390/ijms21020499




