Unveiling the Actual Functions of Awns in Grasses: From Yield Potential to Quality Traits
Abstract
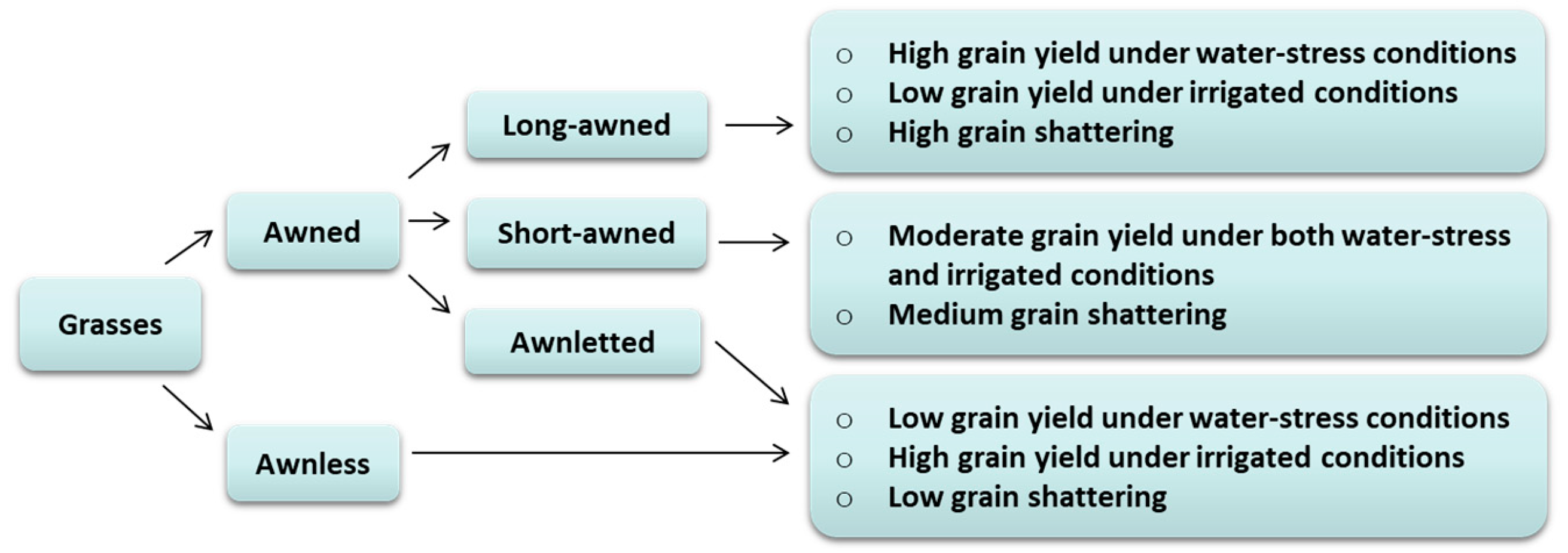
:1. Introduction
2. Impact of Awns on Grain Yield and Plant Biomass
2.1. Awns and Their Contribution to Canopy Temperature
2.2. The Photosynthetic Role of Awns and Their Possible Contribution to Grain Filling
2.3. Impact of Awns on Grain Yield and Components
2.4. Role of Awns on Biomass Yield
2.5. The Impact of Awns on Grain Shattering
3. Impact of Awns on Grain and Forage Quality Traits
3.1. Grain Quality Traits as Affected by Awns
3.2. Impact of Awns on Forage Quality Traits
4. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NILs | Near-isogenic Lines |
| PEPCase | Phosphoenolpyruvate Carboxylase |
References
- Hua, L.; Wang, D.R.; Tan, L.; Fu, Y.; Liu, F.; Xiao, L.; Zhu, Z.; Fu, Q.; Sun, X.; Gu, P.; et al. LABA1, a domestication gene associated with long, barbed awns in wild rice. Plant Cell 2015, 27, 1875–1888. [Google Scholar] [CrossRef] [Green Version]
- Yoshioka, M.; Iehisa, J.C.; Ohno, R.; Kimura, T.; Enoki, H.; Nishimura, S.; Nasuda, S.; Takumi, S. Three dominant awnless genes in common wheat: Fine mapping, interaction and contribution to diversity in awn shape and length. PLoS ONE 2017, 12, e0176148. [Google Scholar] [CrossRef] [Green Version]
- Huang, D.; Zheng, Q.; Melchkart, T.; Bekkaoui, Y.; Konkin, D.J.F.; Kagale, S.; Martucci, M.; You, F.M.; Clarke, M.; Adamski, N.M.; et al. Dominant inhibition of awn development by a putative zinc-finger transcriptional repressor expressed at the B1 locus in wheat. New Phytol. 2020, 225, 340–355. [Google Scholar] [CrossRef] [Green Version]
- Gu, X.-Y.; Kianian, S.F.; Foley, M.E. Phenotypic selection for dormancy introduced a set of adaptive haplotypes from weedy into cultivated rice. Genetics 2005, 171, 695–704. [Google Scholar] [CrossRef] [Green Version]
- Blum, A. Photosynthesis and transpiration in leaves and ears of wheat and barley varieties. J. Exp. Bot. 1985, 36, 432–440. [Google Scholar] [CrossRef]
- Maydup, M.L.; Antonietta, M.; Graciano, C.; Guiamet, J.J.; Tambussi, E.A. The contribution of the awns of bread wheat (Triticum aestivum L.) to grain filling: Responses to water deficit and the effects of awns on ear temperature and hydraulic conductance. Field Crops Res. 2014, 167, 102–111. [Google Scholar] [CrossRef]
- Sanchez-Bragado, R.; Vicente, R.; Molero, G.; Serret, M.D.; Maydup, M.L.; Araus, J.L. New avenues for increasing yield and stability in C3 cereals: Exploring ear photosynthesis. Curr. Opin. Plant Biol. 2020, 56, 223–234. [Google Scholar] [CrossRef]
- Motzo, R.; Giunta, F. Awnedness affects grain yield and kernel weight in near-isogenic lines of durum wheat. Aust. J. Agric. Res. 2002, 53, 1285–1293. [Google Scholar] [CrossRef]
- Peleg, Z.; Saranga, Y.; Fahima, T.; Aharoni, A.; Elbaum, R. Genetic control over silica deposition in wheat awns. Physiol. Plant. 2010, 140, 10–20. [Google Scholar] [CrossRef]
- Simkin, A.J.; Faralli, M.; Ramamoorthy, S.; Lawson, T. Photosynthesis in non-foliar tissues: Implications for yield. Plant J. 2020, 101, 1001–1015. [Google Scholar] [CrossRef]
- Li, Y.; Li, H.; Li, Y.; Zhang, S. Improving water-use efficiency by decreasing stomatal conductance and transpiration rate to maintain higher ear photosynthetic rate in drought-resistant wheat. Crop J. 2017, 5, 231–239. [Google Scholar] [CrossRef]
- Ferguson, H.; Eslick, R.; Aase, J. Canopy temperatures of barley as influenced by morphological characteristics 1. Agron. J. 1973, 65, 425–428. [Google Scholar] [CrossRef]
- Deng, X.; Zhen, S.; Liu, D.; Liu, Y.; Li, M.; Liu, N.; Yan, Y. Integrated proteome analyses of wheat glume and awn reveal central drought response proteins under water deficit conditions. J. Plant Physiol. 2019, 232, 270–283. [Google Scholar] [CrossRef]
- Bort, J.; Brown, R.H.; Araus, J.L. Refixation of respiratory CO2 in the ears of C3 cereals. J. Exp. Bot. 1996, 47, 1567–1575. [Google Scholar] [CrossRef] [Green Version]
- Furuta, T.; Komeda, N.; Asano, K.; Uehara, K.; Gamuyao, R.; Angeles-Shim, R.B.; Nagai, K.; Doi, K.; Wang, D.R.; Yasui, H. Convergent loss of awn in two cultivated rice species Oryza sativa and Oryza glaberrima is caused by mutations in different loci. G3 Genes Genomes Genet. 2015, 5, 2267–2274. [Google Scholar]
- Li, X.; Wang, H.; Li, H.; Zhang, L.; Teng, N.; Lin, Q.; Wang, J.; Kuang, T.; Li, Z.; Li, B. Awns play a dominant role in carbohydrate production during the grain-filling stages in wheat (Triticum aestivum). Physiol. Plant. 2006, 127, 701–709. [Google Scholar] [CrossRef]
- Merah, O.; Evon, P.; Monneveux, P. Participation of green organs to grain filling in Triticum turgidum var. durum grown under mediterranean conditions. Int. J. Mol. Sci. 2017, 19, 56. [Google Scholar] [CrossRef] [Green Version]
- Yuo, T.; Yamashita, Y.; Kanamori, H.; Matsumoto, T.; Lundqvist, U.; Sato, K.; Ichii, M.; Jobling, S.A.; Taketa, S. A SHORT INTERNODES (SHI) family transcription factor gene regulates awn elongation and pistil morphology in barley. J. Exp. Bot. 2012, 63, 5223–5232. [Google Scholar] [CrossRef] [Green Version]
- Jin, J.; Hua, L.; Zhu, Z.; Tan, L.; Zhao, X.; Zhang, W.; Liu, F.; Fu, Y.; Cai, H.; Sun, X. GAD1 encodes a secreted peptide that regulates grain number, grain length, and awn development in rice domestication. Plant Cell 2016, 28, 2453–2463. [Google Scholar] [CrossRef] [Green Version]
- Abebe, T.; Wise, R.P.; Skadsen, R.W. Comparative Transcriptional Profiling Established the Awn as the Major Photosynthetic Organ of the Barley Spike While the Lemma and the Palea Primarily Protect the Seed. Plant Genome 2009, 2, 247–259. [Google Scholar] [CrossRef]
- Evans, L.T.; Bingham, J.; Jackson, P.; Sutherland, J. Effect of awns and drought on the supply of photosynthate and its distribution within wheat ears. Ann. Appl. Biol. 1972, 70, 67–76. [Google Scholar] [CrossRef]
- Olugbemi, L.; Austin, R.; Bingham, J. Effects of awns on the photosynthesis and yield of wheat, Triticum aestivum. Ann. Appl. Biol. 1976, 84, 241–250. [Google Scholar] [CrossRef]
- Li, X.-F.; Bin, D.; Hong-Gang, W. Awn anatomy of common wheat (Triticum aestivum L.) and its relatives. Caryologia 2010, 63, 391–397. [Google Scholar] [CrossRef] [Green Version]
- Kaiser, E.; Correa Galvis, V.; Armbruster, U. Efficient photosynthesis in dynamic light environments: A chloroplast’s perspective. Biochem. J. 2019, 476, 2725–2741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caley, C.; Duffus, C.; Jeffcoat, B. Photosynthesis in the pericarp of developing wheat grains. J. Exp. Bot. 1990, 41, 303–307. [Google Scholar] [CrossRef]
- Slocombe, S.; Whitelam, G.; Cockburn, W. Investigation of phosphoeno/pyruvate carboxylase (PEPCase) in Mesembryanthemum crystallinum L. in C3 and CAM photosynthetic states. Plant Cell Environ. 1993, 16, 403–411. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, Y.; Xia, H.; Fan, S.; Song, J.; Lv, X.; Kong, L. Photosynthetic characteristics of non-foliar organs in main C3 cereals. Physiol. Plant. 2019, 166, 226–239. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Li, D.; Wang, S.; Zhang, S. Photosynthetic response of tetraploid and hexaploid wheat to water stress. Photosynthetica 2017, 55, 454–466. [Google Scholar] [CrossRef]
- Merah, O.; Monneveux, P. Contribution of Different Organs to Grain Filling in Durum Wheat under Mediterranean Conditions I. Contribution of Post-Anthesis Photosynthesis and Remobilization. J. Agron. Crop Sci. 2015, 201, 344–352. [Google Scholar] [CrossRef] [Green Version]
- Sanchez-Bragado, R.; Kim, J.W.; Rivera-Amado, C.; Molero, G.; Araus, J.L.; Savin, R.; Slafer, G.A. Are awns truly relevant for wheat yields? A study of performance of awned/awnless isogenic lines and their response to source–sink manipulations. Field Crops Res. 2020, 254, 107827. [Google Scholar] [CrossRef]
- Brinton, J.; Uauy, C. A reductionist approach to dissecting grain weight and yield in wheat. J. Integr. Plant Biol. 2019, 61, 337–358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huo, X.; Wu, S.; Zhu, Z.; Liu, F.; Fu, Y.; Cai, H.; Sun, X.; Gu, P.; Xie, D.; Tan, L.; et al. NOG1 increases grain production in rice. Nat. Commun. 2017, 8, 1497. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Hou, L.; Zhu, Y.; Zhang, Q.; Wang, H.; Xia, F.; Chen, L.; Mao, P.; Hannaway, D.B. Siberian wildrye seed yield limited by assimilate source. Field Crops Res. 2018, 218, 18–23. [Google Scholar] [CrossRef]
- Duwayri, M. Effect of flag leaf and awn removal on grain yield and yield components of wheat grown under dryland conditions. Field Crops Res. 1984, 8, 307–313. [Google Scholar] [CrossRef]
- Ntakirutimana, F.; Xiao, B.; Xie, W.; Zhang, J.; Zhang, Z.; Wang, N.; Yan, J. Potential Effects of Awn Length Variation on Seed Yield and Components, Seed Dispersal and Germination Performance in Siberian Wildrye (Elymus sibiricus L.). Plants 2019, 8, 561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaudhary, C.; Sharma, N.; Khurana, P. Decoding the wheat awn transcriptome and overexpressing TaRca1β in rice for heat stress tolerance. Plant Mol Biol 2020, 1–14. [Google Scholar] [CrossRef]
- Liu, K.; Xu, H.; Liu, G.; Guan, P.; Zhou, X.; Peng, H.; Yao, Y.; Ni, Z.; Sun, Q.; Du, J. QTL mapping of flag leaf-related traits in wheat (Triticum aestivum L.). Theor. Appl. Genet. 2018, 131, 839–849. [Google Scholar] [CrossRef] [Green Version]
- Wu, Q.; Chen, Y.; Fu, L.; Zhou, S.; Chen, J.; Zhao, X.; Zhang, D.; Ouyang, S.; Wang, Z.; Li, D.; et al. QTL mapping of flag leaf traits in common wheat using an integrated high-density SSR and SNP genetic linkage map. Euphytica 2016, 208, 337–351. [Google Scholar] [CrossRef]
- Chhabra, A.K.; Sethi, S.K. Contribution and association of awns and flag-leaf with yield and its components in durum wheat. Cereal Res. Commun. 1989, 17, 265–271. [Google Scholar]
- Khaliq, I.; Irshad, A.; Ahsan, M. Awns and Flag Leaf Contribution Towards Grain Yield in Spring Wheat (Triticum aestivum L.). Cereal Res. Commun. 2008, 36, 65–76. [Google Scholar] [CrossRef]
- Paluska, M.M. Effect of Flag Leaf and Awn Removal on Seed Weight of Arivat Barley. J. Ariz.-Nev. Acad. Sci. 1981, 16, 22–23. [Google Scholar]
- Chen, G.D.; Li, H.B.; Zheng, Z.; Wei, Y.M.; Zheng, Y.L.; McIntyre, C.L.; Zhou, M.X.; Liu, C.J. Characterization of a QTL affecting spike morphology on the long arm of chromosome 3H in barley (Hordeum vulgare L.) based on near isogenic lines and a NIL-derived population. Theor. Appl. Genet. 2012, 125, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Grundbacher, F. The physiological function of the cereal awn. Bot. Rev. 1963, 29, 366–381. [Google Scholar] [CrossRef]
- Teich, A.H. Interaction of awns and environment on grain yield in winter wheat (Triticum aestivum L.). Cereal Res. Commun. 1982, 10, 11–15. [Google Scholar]
- Rebetzke, G.; Bonnett, D.; Reynolds, M. Awns reduce grain number to increase grain size and harvestable yield in irrigated and rainfed spring wheat. J. Exp. Bot. 2016, 67, 2573–2586. [Google Scholar] [CrossRef]
- Olugbemi, L.; Bingham, J.; Austin, R. Ear and flag leaf photosynthesis of awned and awnless Triticum species. Ann. Appl. Biol. 1976, 84, 231–240. [Google Scholar] [CrossRef]
- Wright, T.I.C. Capturing Photosynthetic Traits from the Progenitors of Wheat; University of Cambridge: Cambridge, UK, 2019. [Google Scholar]
- Jin, F.-X.; Kim, D.-M.; Ju, H.-G.; Ahn, S.-N. Mapping quantitative trait loci for awnness and yield component traits in isogenic lines derived from an Oryza sativa/O. rufipogon Cross. J. Crop Sci. Biotechnol. 2009, 12, 9–16. [Google Scholar] [CrossRef]
- Hosseini, S.M.; Poustini, K.; Siddique, K.H.; Palta, J.A. Photosynthesis of barley awns does not play a significant role in grain yield under terminal drought. Crop Pasture Sci. 2012, 63, 489–499. [Google Scholar] [CrossRef]
- Teare, I.D.; Sij, J.W.; Waldren, R.P.; Goltz, S.M. Comparative data on the rate of photosynthesis, respiration, and transpiration of different organs in awned and awnless isogenic lines of wheat. Can. J. Plant Sci. 1972, 52, 965–971. [Google Scholar] [CrossRef]
- Knott, D. Effect of genes for photoperiodism, semidwarfism, and awns on agronomic characters in a wheat cross 1. Crop Sci. 1986, 26, 1158–1162. [Google Scholar] [CrossRef]
- Ntakirutimana, F.; Xie, W. Morphological and genetic mechanisms underlying awn development in monocotyledonous grasses. Genes 2019, 10, 573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mach, J. Domesticated versus wild rice? Bring it awn! Am. Soc. Plant Biol. 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeWitt, N.; Guedira, M.; Lauer, E.; Sarinelli, M.; Tyagi, P.; Fu, D.; Hao, Q.; Murphy, J.P.; Marshall, D.; Akhunova, A.; et al. Sequence-based mapping identifies a candidate transcription repressor underlying awn suppression at the B1 locus in wheat. New Phytol. 2020, 225, 326–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, J.; Liu, H.; Zhou, T.; Gu, B.; Huang, X.; Shangguan, Y.; Zhu, J.; Li, Y.; Zhao, Y.; Wang, Y. An-1 encodes a basic helix-loop-helix protein that regulates awn development, grain size, and grain number in rice. Plant Cell 2013, 25, 3360–3376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, B.; Zhou, T.; Luo, J.; Liu, H.; Wang, Y.; Shangguan, Y.; Zhu, J.; Li, Y.; Sang, T.; Wang, Z.; et al. An-2 Encodes a Cytokinin Synthesis Enzyme that Regulates Awn Length and Grain Production in Rice. Mol. Plant 2015, 8, 1635–1650. [Google Scholar] [CrossRef]
- Bessho-Uehara, K.; Wang, D.R.; Furuta, T.; Minami, A.; Nagai, K.; Gamuyao, R.; Asano, K.; Angeles-Shim, R.B.; Shimizu, Y.; Ayano, M.; et al. Loss of function at RAE2, a previously unidentified EPFL, is required for awnlessness in cultivated Asian rice. Proc. Natl. Acad. Sci. USA 2016, 113, 8969–8974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liller, C.B.; Walla, A.; Boer, M.P.; Hedley, P.; Macaulay, M.; Effgen, S.; von Korff, M.; van Esse, G.W.; Koornneef, M. Fine mapping of a major QTL for awn length in barley using a multiparent mapping population. Theor. Appl. Genet. 2017, 130, 269–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathan, J.; Bhattacharya, J.; Ranjan, A. Enhancing crop yield by optimizing plant developmental features. Development 2016, 143, 3283–3294. [Google Scholar] [CrossRef] [Green Version]
- Pampana, S.; Masoni, A.; Ercoli, L.; Mariotti, M.; Arduini, I. Effects of nitrogen splitting and source on durum wheat. Cereal Res. Commun. 2013, 41, 338–347. [Google Scholar] [CrossRef]
- Zhao, X.; Xie, W.; Zhang, J.; Zhang, Z.; Wang, Y. Histological Characteristics, Cell Wall Hydrolytic Enzymes Activity and Candidate Genes Expression Associated with Seed Shattering of Elymus sibiricus Accessions. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Gill, B.S. Multiple genetic pathways for seed shattering in the grasses. Funct. Integr. Genom. 2006, 6, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.; Kim, S.-R.; Kim, Y.-H.; Kim, H.; Eun, M.-Y.; Jin, I.-D.; Cha, Y.-S.; Yun, D.-W.; Ahn, B.-O.; Lee, M.C. Inactivation of the CTD phosphatase-like gene OsCPL1 enhances the development of the abscission layer and seed shattering in rice. Plant J. 2010, 61, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Kandemir, N.; Kudrna, D.; Ullrich, S.; Kleinhofs, A. Molecular marker assisted genetic analysis of head shattering in six-rowed barley. Theor. Appl. Genet. 2000, 101, 203–210. [Google Scholar] [CrossRef]
- Thomson, M.; Tai, T.; McClung, A.; Lai, X.; Hinga, M.; Lobos, K.; Xu, Y.; Martinez, C.; McCouch, S.R. Mapping quantitative trait loci for yield, yield components and morphological traits in an advanced backcross population between Oryza rufipogon and the Oryza sativa cultivar Jefferson. Theor. Appl. Genet. 2003, 107, 479–493. [Google Scholar] [CrossRef]
- Zhou, Y.; Lu, D.; Li, C.; Luo, J.; Zhu, B.-F.; Zhu, J.; Shangguan, Y.; Wang, Z.; Sang, T.; Zhou, B. Genetic control of seed shattering in rice by the APETALA2 transcription factor SHATTERING ABORTION1. Plant Cell 2012, 24, 1034–1048. [Google Scholar] [CrossRef] [Green Version]
- Yoon, J.; Cho, L.H.; Kim, S.L.; Choi, H.; Koh, H.J.; An, G. The BEL 1-type homeobox gene SH 5 induces seed shattering by enhancing abscission-zone development and inhibiting lignin biosynthesis. Plant J. 2014, 79, 717–728. [Google Scholar] [CrossRef]
- Thurber, C.S.; Hepler, P.K.; Caicedo, A.L. Timing is everything: Early degradation of abscission layer is associated with increased seed shattering in US weedy rice. BMC Plant Biol. 2011, 11, 14. [Google Scholar] [CrossRef] [Green Version]
- Xie, W.; Zhang, J.; Zhao, X.; Zhang, Z.; Wang, Y. Transcriptome profiling of Elymus sibiricus, an important forage grass in Qinghai-Tibet plateau, reveals novel insights into candidate genes that potentially connected to seed shattering. BMC Plant Biol. 2017, 17, 78. [Google Scholar] [CrossRef]
- Domingos, S.; Fino, J.; Cardoso, V.; Sánchez, C.; Ramalho, J.C.; Larcher, R.; Paulo, O.S.; Oliveira, C.M.; Goulao, L.F. Shared and divergent pathways for flower abscission are triggered by gibberellic acid and carbon starvation in seedless Vitis vinifera L. BMC Plant Biol. 2016, 16, 38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magwa, R.A.; Zhao, H.; Yao, W.; Xie, W.; Yang, L.; Xing, Y.; Bai, X. Genomewide association analysis for awn length linked to the seed shattering gene qSH1 in rice. J. Genet. 2016, 95, 639–646. [Google Scholar] [CrossRef]
- Haas, M.; Schreiber, M.; Mascher, M. Domestication and crop evolution of wheat and barley: Genes, genomics, and future directions. J. Integr. Plant Biol. 2019, 61, 204–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKevith, B. Nutritional aspects of cereals. Nutr. Bull. 2004, 29, 111–142. [Google Scholar] [CrossRef]
- Sato, H.; Sakamoto, S.; Mitsuda, N.; Ohme-Takagi, M.; Takamizo, T. Improvement of cell wall digestibility in tall fescue by Oryza sativa SECONDARY WALL NAC DOMAIN PROTEIN2 chimeric repressor. Mol. Breed. 2018, 38, 36. [Google Scholar] [CrossRef]
- Glémin, S.; Bataillon, T. A comparative view of the evolution of grasses under domestication. New Phytol. 2009, 183, 273–290. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Hey, S. Do “ancient” wheat species differ from modern bread wheat in their contents of bioactive components? J. Cereal Sci. 2015, 65, 236–243. [Google Scholar] [CrossRef] [Green Version]
- Fasahat, P.; Muhammad, K.; Abdullah, A.; Ratnam, W. Proximate nutritional composition and antioxidant properties of ‘Oryza rufipogon’, a wild rice collected from Malaysia compared to cultivated rice, MR219. Aust. J. Crop Sci. 2012, 6, 1502. [Google Scholar]
- Ma, W.; Sutherland, M.W.; Kammholz, S.; Banks, P.; Brennan, P.; Bovill, W.; Daggard, G. Wheat flour protein content and water absorption analysis in a doubled haploid population. J. Cereal Sci. 2007, 45, 302–308. [Google Scholar] [CrossRef] [Green Version]
- Huang, D.-R.; Fan, Y.-Y.; Hu, B.-L.; Xiao, Y.-Q.; Chen, D.-Z.; Zhuang, J.-Y. Assessment and genetic analysis of heavy metal content in rice grain using an Oryza sativa × O. rufipogon backcross inbred line population. J. Sci. Food Agric. 2018, 98, 1339–1345. [Google Scholar] [CrossRef]
- Capstaff, N.M.; Miller, A.J. Improving the yield and nutritional quality of forage crops. Front. Plant Sci. 2018, 9, 535. [Google Scholar] [CrossRef] [Green Version]
- Moore, K.J.; Moser, L.E. Quantifying developmentalorphology of perennial grasses. Crop Sci. 1995, 35, 37–43. [Google Scholar] [CrossRef]
- Twidwell, E.; Johnson, K.; Cherney, J.; Volenec, J. Forage quality and digestion kinetics of switchgrass herbage and morphological components. Crop Sci. 1988, 28, 778–782. [Google Scholar] [CrossRef]
- Karn, J.; Berdahl, J.; Frank, A. Nutritive quality of four perennial grasses as affected by species, cultivar, maturity, and plant tissue. Agron. J. 2006, 98, 1400–1409. [Google Scholar] [CrossRef] [Green Version]
- Xue, Q.; Wang, L.; Newman, R.; Newman, C.; Graham, H. Influence of the hulless, waxy starch and short-awn genes on the composition of barleys. J. Cereal Sci. 1997, 26, 251–257. [Google Scholar] [CrossRef]
- Park, T.-I.; Han, O.-K.; Seo, J.-H.; Choi, J.-S.; Park, K.-H.; Kim, J.-G. New barley cultivars with improved morphological characteristics for whole crop forage in Korea. J. Korean Soc. Grassl. Forage Sci. 2008, 28, 193–202. [Google Scholar]
- Kim, J.; Choi, J.; Park, H.; Baek, S.; Kang, M.; Park, K.; Kwon, Y.; Heo, H.; Suh, S.; Nam, J. A new smooth awn barley cultivar, “Wooho” suitable for whole crop forage use. Korean J. Breed. Sci. 2007, 39, 238–239. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |

| Species | Growing Conditions | Genotypes Tested | Grains Per Inflorescence | Kernel Weight (mg) | Grain Yield Per Plant (g) | Biomass Yield (t ha−1) | References |
|---|---|---|---|---|---|---|---|
| Bread wheat (Triticum aestivum L.) | Dry land | Awned | 36.90 a | 31.20 c | 1.13 | nd | Teich [44] |
| Awnless | 37.60 a | 28.60 c | 1.03 | nd | |||
| Irrigated | Awned | 65.40 a | 31.40 c | 2.10 | nd | ||
| Awnless | 82.40 a | 30.60 c | 2.52 | nd | |||
| Bread wheat (Triticum aestivum L.) | Dry land | Awned | 42.60 | 48.20 | 7.36 d | nd | Duwayri [34] |
| De-awned | 36.10 | 45.80 | 6.19 d | nd | |||
| Bread wheat (Triticum aestivum L.) | Rainfed | Awned | Nd b | 34.13 | 305.00 e | nd | Knott [51] |
| Awnletted | Nd | 32.50 | 291.33 e | nd | |||
| Bread wheat (Triticum aestivum L.) | Rainfed | Awned | 61.95 | 40.96 c | 23.61 | nd | Khaliq et al. [40] |
| De-awned | 59.45 | 39.12 c | 21.30 | nd | |||
| Bread wheat (Triticum aestivum L.) | Rainfed | Awned | 23.30 | 39.00 | 2.84 d | 7.90 | Rebetzke et al. [45] |
| Awnletted | 24.30 | 37.10 | 2.74 d | 7.59 | |||
| Irrigated | Awned | 25.90 | 40.80 | 6.27 d | 16.50 | ||
| Awnletted | 27.40 | 38.90 | 6.32 d | 16.20 | |||
| Durum wheat (Triticum turgidum L. var.durum) | Irrigated | Awned | 45.54 | 54.60 | 238.00 f | nd | Chhabra and Seth [39] |
| De-awned | 45.13 | 47.50 | 205.90 f | nd | |||
| Durum wheat (Triticum turgidum L. var. durum) | Rainfed | Awned | 32.30 | 35.10 | 104.70 e | nd | Motzo and Giunta [8] |
| Rainfed | De-awned | 31.20 | 34.50 | 94.10 e | nd | ||
| Barley (Hordeum vulgare L.) | Rainfed | Awned | 42.40 | 46.90 c | Nd | nd | Paluska [41] |
| De-awned | 44.70 | 37.80 c | nd | nd | |||
| Barley (Hordeum vulgare L.) | Irrigated | Awned | 19.57 | 46.68 | 2.47 | 7.20 | Hosseini et al. [49] |
| Terminal drought | Awned | 17.78 | 39.03 | 1.74 | 5.67 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ntakirutimana, F.; Xie, W. Unveiling the Actual Functions of Awns in Grasses: From Yield Potential to Quality Traits. Int. J. Mol. Sci. 2020, 21, 7593. https://doi.org/10.3390/ijms21207593
Ntakirutimana F, Xie W. Unveiling the Actual Functions of Awns in Grasses: From Yield Potential to Quality Traits. International Journal of Molecular Sciences. 2020; 21(20):7593. https://doi.org/10.3390/ijms21207593
Chicago/Turabian StyleNtakirutimana, Fabrice, and Wengang Xie. 2020. "Unveiling the Actual Functions of Awns in Grasses: From Yield Potential to Quality Traits" International Journal of Molecular Sciences 21, no. 20: 7593. https://doi.org/10.3390/ijms21207593





