Differentiation Induction of Human Stem Cells for Corneal Epithelial Regeneration
Abstract
:1. Introduction
2. Corneal Epithelial Cell
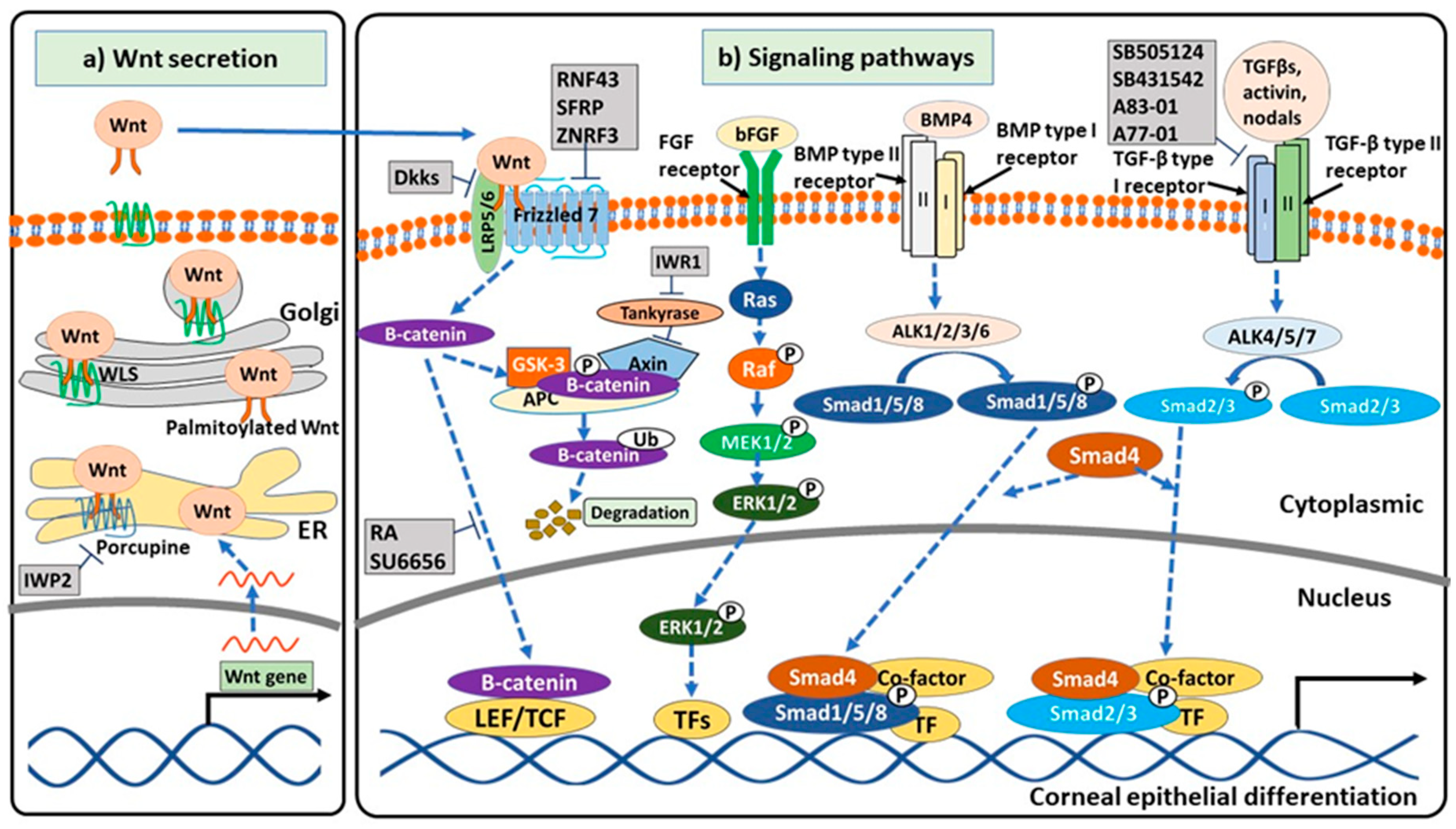
3. Signaling Pathways Related to Corneal Epithelial Differentiation
4. Corneal Epithelial Differentiation of Human MSCs
5. Corneal Epithelial Differentiation of Human PSCs
6. Future Perspective
Author Contributions
Funding
Conflicts of Interest
References
- Palanker, D.; Blumenkranz, M.S. Chapter 39—Retinal Laser Therapy: Biophysical Basis and Applications. In Retina, 5th ed.; Ryan, S.J., Sadda, S.R., Hinton, D.R., Schachat, A.P., Sadda, S.R., Wilkinson, C.P., Wiedemann, P., Schachat, A.P., Eds.; W.B. Saunders: London, UK, 2013; pp. 746–760. [Google Scholar] [CrossRef]
- Chen, L.W.; Chen, Y.-M.; Lu, C.-J.; Chen, M.-Y.; Lin, S.-Y.; Hu, F.-R.; Chen, W.-L. Effect of air-lifting on the stemness, junctional protein formation, and cytokeratin expression of in vitro cultivated limbal epithelial cell sheets. Taiwan J. Ophthalmol. 2017, 7, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Endo, K.-I.; Cooper, L.J.; Fullwood, N.J.; Tanifuji, N.; Tsuzuki, M.; Koizumi, N.; Inatomi, T.; Sano, Y.; Kinoshita, S. The Successful Culture and Autologous Transplantation of Rabbit Oral Mucosal Epithelial Cells on Amniotic Membrane. Investig. Ophthalmol. Vis. Sci. 2003, 44, 106–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, R.J.; Li, L.M.; Chen, J.K. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N. Eng. J. Med. 2000, 343, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Mathews, S.; Prasad, T.; Venugopal, B.; Palakkan, A.A.; Anil Kumar, P.R.; Kumary, T.V. Standardizing transdifferentiation of rabbit bone marrow mesenchymal stem cells to corneal lineage by simulating corneo-limbal cues. J. Stem Cell Res. Med. 2017, 2, 1–10. [Google Scholar] [CrossRef]
- Samoila, O.; Gocan, D. Clinical Outcomes from Cultivated Allogenic Stem Cells vs. Oral Mucosa Epithelial Transplants in Total Bilateral Stem Cells Deficiency. Front. Med. 2020, 7. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Xu, Y.; Xiao, Z.; Yang, W.; Zhang, C.; Song, E.; Du, Y.; Li, L. Reconstruction of chemically burned rat corneal surface by bone marrow-derived human mesenchymal stem cells. Stem Cells 2006, 24, 315–321. [Google Scholar] [CrossRef]
- Bandeira, F.; Goh, T.-W.; Setiawan, M.; Yam, G.H.-F.; Mehta, J.S. Cellular therapy of corneal epithelial defect by adipose mesenchymal stem cell-derived epithelial progenitors. Stem Cell Res. Ther. 2020, 11, 14. [Google Scholar] [CrossRef] [Green Version]
- Da Mata Martins, T.M.; da Silva Cunha, P.; Rodrigues, M.A.; de Carvalho, J.L.; de Souza, J.E.; de Carvalho Oliveira, J.A.; Gomes, D.A.; de Goes, A.M. Epithelial basement membrane of human decellularized cornea as a suitable substrate for differentiation of embryonic stem cells into corneal epithelial-like cells. Mater. Sci. Eng. C 2020, 116. [Google Scholar] [CrossRef]
- Zhang, P.; Ma, X.-y.; Huang, D.-t.; Yang, X.-y. The capacity of goat epidermal adult stem cells to reconstruct the damaged ocular surface of total LSCD and activate corneal genetic programs. J. Mol. Histol. 2020, 51, 277–286. [Google Scholar] [CrossRef]
- He, J.; Ou, S.; Ren, J.; Sun, H.; He, X.; Zhao, Z.; Wu, H.; Qu, Y.; Liu, T.; Jeyalatha, V.; et al. Tissue engineered corneal epithelium derived from clinical-grade human embryonic stem cells. Ocul. Surf. 2020, 18, 672–680. [Google Scholar] [CrossRef]
- Le-Bel, G.; Guérin, L.-P.; Carrier, P.; Mouriaux, F.; Germain, L.; Guérin, S.L.; Bazin, R. Grafting of an autologous tissue-engineered human corneal epithelium to a patient with limbal stem cell deficiency (LSCD). Am. J. Ophthalmol. Case Rep. 2019, 15, 100532. [Google Scholar] [CrossRef] [PubMed]
- Marquez-Curtis, L.A.; Janowska-Wieczorek, A.; McGann, L.E.; Elliott, J.A.W. Mesenchymal stromal cells derived from various tissues: Biological, clinical and cryopreservation aspects. Cryobiology 2015, 71, 181–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elahi, K.C.; Klein, G.; Avci-Adali, M.; Sievert, K.D.; MacNeil, S.; Aicher, W.K. Human Mesenchymal Stromal Cells from Different Sources Diverge in Their Expression of Cell Surface Proteins and Display Distinct Differentiation Patterns. Stem Cells Int. 2016, 2016, 5646384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Payushina, O.V.; Domaratskaya, E.I.; Starostin, V.I. Mesenchymal stem cells: Sources, phenotype, and differentiation potential. Biol. Bull. 2006, 33, 2–18. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef] [Green Version]
- Prescott, C. The business of exploiting induced pluripotent stem cells. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2011, 366, 2323–2328. [Google Scholar] [CrossRef] [Green Version]
- D’Antonio, M.; Woodruff, G.; Nathanson, J.L.; D’Antonio-Chronowska, A.; Arias, A.; Matsui, H.; Williams, R.; Herrera, C.; Reyna, S.M.; Yeo, G.W.; et al. High-Throughput and Cost-Effective Characterization of Induced Pluripotent Stem Cells. Stem Cell Rep. 2017, 8, 1101–1111. [Google Scholar] [CrossRef]
- Katikireddy, K.R.; Dana, R.; Jurkunas, U.V. Differentiation Potential of Limbal Fibroblasts and Bone Marrow Mesenchymal Stem Cells to Corneal Epithelial Cells. Stem Cells 2014, 32, 717–729. [Google Scholar] [CrossRef]
- Nieto-Nicolau, N.; Martín-Antonio, B.; Müller-Sánchez, C.; Casaroli-Marano, R.P. In vitro potential of human mesenchymal stem cells for corneal epithelial regeneration. Regen. Med. 2020, 15, 1409–1426. [Google Scholar] [CrossRef]
- Rohaina, C.M.; Then, K.Y.; Ng, A.M.H.; Wan Abdul Halim, W.H.; Zahidin, A.Z.M.; Saim, A.; Idrus, R.B.H. Reconstruction of limbal stem cell deficient corneal surface with induced human bone marrow mesenchymal stem cells on amniotic membrane. Transl. Res. 2014, 163, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Garzón, I.; Martín-Piedra, M.A.; Alfonso-Rodríguez, C.; González-Andrades, M.; Carriel, V.; Martínez-Gómez, C.; Campos, A.; Alaminos, M. Generation of a Biomimetic Human Artificial Cornea Model Using Wharton’s Jelly Mesenchymal Stem Cells. Investig. Ophthalmol. Vis. Sci. 2014, 55, 4073–4083. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soleimanifar, F.; Mortazavi, Y.; Nadri, S.; Islami, M.; Vakilian, S. Coculture of conjunctiva derived mesenchymal stem cells (CJMSCs) and corneal epithelial cells to reconstruct the corneal epithelium. Biologicals 2018, 54, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Stewart, R.; Yung, S.; Kolli, S.; Armstrong, L.; Stojkovic, M.; Figueiredo, F.; Lako, M. Differentiation of human embryonic stem cells into corneal epithelial-like cells by in vitro replication of the corneal epithelial stem cell niche. Stem Cells 2007, 25, 1145–1155. [Google Scholar] [CrossRef]
- Ginis, I.; Luo, Y.; Miura, T.; Thies, S.; Brandenberger, R.; Gerecht-Nir, S.; Amit, M.; Hoke, A.; Carpenter, M.K.; Itskovitz-Eldor, J.; et al. Differences between human and mouse embryonic stem cells. Dev. Biol. 2004, 269, 360–380. [Google Scholar] [CrossRef] [Green Version]
- Hillmann, A.; Ahrberg, A.B.; Brehm, W.; Heller, S.; Josten, C.; Paebst, F.; Burk, J. Comparative Characterization of Human and Equine Mesenchymal Stromal Cells: A Basis for Translational Studies in the Equine Model. Cell Transpl. 2016, 25, 109–124. [Google Scholar] [CrossRef] [Green Version]
- Schnerch, A.; Cerdan, C.; Bhatia, M. Distinguishing between mouse and human pluripotent stem cell regulation: The best laid plans of mice and men. Stem Cells 2010, 28, 419–430. [Google Scholar] [CrossRef]
- Bottagisio, M.; Lovati, A.B.; Lopa, S.; Moretti, M. Osteogenic Differentiation of Human and Ovine Bone Marrow Stromal Cells in response to β-Glycerophosphate and Monosodium Phosphate. Cell. Reprogram. 2015, 17, 235–242. [Google Scholar] [CrossRef]
- Kalaszczynska, I.; Ruminski, S.; Platek, A.E.; Bissenik, I.; Zakrzewski, P.; Noszczyk, M.; Lewandowska-Szumiel, M. Substantial differences between human and ovine mesenchymal stem cells in response to osteogenic media: How to explain and how to manage? Biores. Open Access 2013, 2, 356–363. [Google Scholar] [CrossRef]
- Arnalich-Montiel, F.; Pastor, S.; Blazquez-Martinez, A.; Fernandez-Delgado, J.; Nistal, M.; Alio, J.L.; De Miguel, M.P. Adipose-Derived Stem Cells Are a Source for Cell Therapy of the Corneal Stroma. Stem Cells 2008, 26, 570–579. [Google Scholar] [CrossRef]
- Oliva, J.; Bardag-Gorce, F.; Niihara, Y. Clinical Trials of Limbal Stem Cell Deficiency Treated with Oral Mucosal Epithelial Cells. Int. J. Mol. Sci. 2020, 21, 411. [Google Scholar] [CrossRef] [Green Version]
- Movahedan, A.; Majdi, M.; Afsharkhamseh, N.; Sagha, H.M.; Saadat, N.S.; Shalileh, K.; Milani, B.Y.; Ying, H.; Djalilian, A.R. Notch Inhibition during Corneal Epithelial Wound Healing Promotes Migration. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7476–7483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sridhar, M.S. Anatomy of cornea and ocular surface. Indian J. Ophthalmol. 2018, 66, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Joseph, A.; Powell-Richards, A.O.R.; Shanmuganathan, V.A.; Dua, H.S. Epithelial cell characteristics of cultured human limbal explants. Br. J. Ophthalmol. 2004, 88, 393–398. [Google Scholar] [CrossRef] [Green Version]
- Mariappan, I.; Maddileti, S.; Savy, S.; Tiwari, S.; Gaddipati, S.; Fatima, A.; Sangwan, V.S.; Balasubramanian, D.; Vemuganti, G.K. In vitro culture and expansion of human limbal epithelial cells. Nat. Protoc. 2010, 5, 1470. [Google Scholar] [CrossRef] [PubMed]
- Reinstein Merjava, S.; Neuwirth, A.; Tanzerova, M.; Jirsova, K. The spectrum of cytokeratins expressed in the adult human cornea, limbus and perilimbal conjunctiva. Histol. Histopathol. 2011, 26, 323–331. [Google Scholar] [CrossRef]
- Wijnholds, J. “Basal Cell Migration” in Regeneration of the Corneal Wound-Bed. Stem Cell Rep. 2019, 12, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Mort, R.L.; Douvaras, P.; Morley, S.D.; Dorà, N.; Hill, R.E.; Collinson, J.M.; West, J.D. Stem cells and corneal epithelial maintenance: Insights from the mouse and other animal models. Results Probl. Cell Differ. 2012, 55, 357–394. [Google Scholar] [CrossRef] [Green Version]
- Beebe, D.C.; Masters, B.R. Cell lineage and the differentiation of corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 1996, 37, 1815–1825. [Google Scholar]
- Swamynathan, S.K. Ocular surface development and gene expression. J. Ophthalmol. 2013, 2013, 103947. [Google Scholar] [CrossRef] [Green Version]
- Yazdanpanah, G.; Jabbehdari, S.; Djalilian, A.R. Limbal and corneal epithelial homeostasis. Curr. Opin. Ophthalmol. 2017, 28, 348–354. [Google Scholar] [CrossRef]
- López-Paniagua, M.; Nieto-Miguel, T.; de la Mata, A.; Dziasko, M.; Galindo, S.; Rey, E.; Herreras, J.M.; Corrales, R.M.; Daniels, J.T.; Calonge, M. Comparison of functional limbal epithelial stem cell isolation methods. Exp. Eye Res. 2016, 146, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Kruse, F.E.; Tseng, S.C. Growth factors modulate clonal growth and differentiation of cultured rabbit limbal and corneal epithelium. Investig. Ophthalmol. Vis. Sci. 1993, 34, 1963–1976. [Google Scholar]
- Kruse, F.E. Stem cells and corneal epithelial regeneration. Eye 1994, 8, 170–183. [Google Scholar] [CrossRef]
- Moll, R.; Franke, W.W.; Schiller, D.L.; Geiger, B.; Krepler, R. The catalog of human cytokeratins: Patterns of expression in normal epithelia, tumors and cultured cells. Cell 1982, 31, 11–24. [Google Scholar] [CrossRef]
- Schermer, A.; Galvin, S.; Sun, T.T. Differentiation-related expression of a major 64K corneal keratin in vivo and in culture suggests limbal location of corneal epithelial stem cells. J. Cell Biol. 1986, 103, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Moll, R.; Divo, M.; Langbein, L. The human keratins: Biology and pathology. Histochem. Cell Biol. 2008, 129, 705–733. [Google Scholar] [CrossRef] [Green Version]
- Martínez García de la Torre, R.A.; Nieto-Nicolau, N.; Morales-Pastor, A.; Casaroli-Marano, R.P. Determination of the Culture Time Point to Induce Corneal Epithelial Differentiation in Induced Pluripotent Stem Cells. Transplant. Proc. 2017, 49, 2292–2295. [Google Scholar] [CrossRef]
- Harkin, D.G.; Foyn, L.; Bray, L.J.; Sutherland, A.J.; Li, F.J.; Cronin, B.G. Concise Reviews: Can Mesenchymal Stromal Cells Differentiate into Corneal Cells? A Systematic Review of Published Data. Stem Cells 2015, 33, 785–791. [Google Scholar] [CrossRef] [Green Version]
- Adhikary, G.; Crish, J.; Lass, J.; Eckert, R.L. Regulation of Involucrin Expression in Normal Human Corneal Epithelial Cells: A Role for Activator Protein One. Investig. Ophthalmol. Vis. Sci. 2004, 45, 1080–1087. [Google Scholar] [CrossRef] [Green Version]
- Adhikary, G.; Crish, J.F.; Gopalakrishnan, R.; Bone, F.; Eckert, R.L. Involucrin Expression in the Corneal Epithelium: An Essential Role for Sp1 Transcription Factors. Investig. Ophthalmol. Vis. Sci. 2005, 46, 3109–3120. [Google Scholar] [CrossRef] [PubMed]
- Khoh-Reiter, S.; Jessen, B.A. Evaluation of the cytotoxic effects of ophthalmic solutions containing benzalkonium chloride on corneal epithelium using an organotypic 3-D model. BMC Ophthalmol. 2009, 9, 5. [Google Scholar] [CrossRef] [Green Version]
- Guo, Z.H.; Zhang, W.; Jia, Y.Y.S.; Liu, Q.X.; Li, Z.F.; Lin, J.S. An Insight into the Difficulties in the Discovery of Specific Biomarkers of Limbal Stem Cells. Int. J. Mol. Sci. 2018, 19, 1982. [Google Scholar] [CrossRef] [Green Version]
- Yoon, J.J.; Ismail, S.; Sherwin, T. Limbal stem cells: Central concepts of corneal epithelial homeostasis. World J. Stem Cells 2014, 6, 391–403. [Google Scholar] [CrossRef] [PubMed]
- Arkell, R.M.; Tam, P.P.L. Initiating head development in mouse embryos: Integrating signalling and transcriptional activity. Open Biol. 2012, 2, 120030. [Google Scholar] [CrossRef] [Green Version]
- Gage, P.J.; Zacharias, A.L. Signaling “cross-talk” is integrated by transcription factors in the development of the anterior segment in the eye. Dev. Dyn. 2009, 238, 2149–2162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuhrmann, S. Wnt signaling in eye organogenesis. Organogenesis 2008, 4, 60–67. [Google Scholar] [CrossRef] [Green Version]
- Lian, X.; Selekman, J.; Bao, X.; Hsiao, C.; Zhu, K.; Palecek, S.P. A Small Molecule Inhibitor of Src Family Kinases Promotes Simple Epithelial Differentiation of Human Pluripotent Stem Cells. PLoS ONE 2013, 8, e60016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Yeh, L.-K.; Zhang, S.; Call, M.; Yuan, Y.; Yasunaga, M.; Kao, W.W.Y.; Liu, C.-Y. Wnt/β-catenin signaling modulates corneal epithelium stratification via inhibition of Bmp4 during mouse development. Development 2015, 142, 3383–3393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mikhailova, A.; Ilmarinen, T.; Uusitalo, H.; Skottman, H. Small-molecule induction promotes corneal epithelial cell differentiation from human induced pluripotent stem cells. Stem Cell Rep. 2014, 2, 219–231. [Google Scholar] [CrossRef] [Green Version]
- Glinka, A.; Wu, W.; Delius, H.; Monaghan, A.P.; Blumenstock, C.; Niehrs, C. Dickkopf-1 is a member of a new family of secreted proteins and functions in head induction. Nature 1998, 391, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Machon, O.; Kreslova, J.; Ruzickova, J.; Vacik, T.; Klimova, L.; Fujimura, N.; Lachova, J.; Kozmik, Z. Lens morphogenesis is dependent on Pax6-mediated inhibition of the canonical Wnt/beta-catenin signaling in the lens surface ectoderm. Genesis 2010, 48, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Gage, P.J.; Qian, M.; Wu, D.; Rosenberg, K.I. The canonical Wnt signaling antagonist DKK2 is an essential effector of PITX2 function during normal eye development. Dev. Biol. 2008, 317, 310–324. [Google Scholar] [CrossRef] [Green Version]
- Forsdahl, S.; Kiselev, Y.; Hogseth, R.; Mjelle, J.E.; Mikkola, I. Pax6 Regulates the Expression of Dkk3 in Murine and Human Cell Lines, and Altered Responses to Wnt Signaling Are Shown in FlpIn-3T3 Cells Stably Expressing Either the Pax6 or the Pax6(5a) Isoform. PLoS ONE 2014, 9, e102559. [Google Scholar] [CrossRef] [Green Version]
- Tsukiyama, T.; Fukui, A.; Terai, S.; Fujioka, Y.; Shinada, K.; Takahashi, H.; Yamaguchi, T.P.; Ohba, Y.; Hatakeyama, S. Molecular Role of RNF43 in Canonical and Noncanonical Wnt Signaling. Mol. Cell. Biol. 2015, 35, 2007–2023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Madan, B.; Virshup, D.M. Targeting Wnts at the source--new mechanisms, new biomarkers, new drugs. Mol. Cancer Ther. 2015, 14, 1087–1094. [Google Scholar] [CrossRef] [Green Version]
- Gan, Q.; Lee, A.; Suzuki, R.; Yamagami, T.; Stokes, A.; Nguyen, B.C.; Pleasure, D.; Wang, J.; Chen, H.-W.; Zhou, C.J. Pax6 mediates ß-catenin signaling for self-renewal and neurogenesis by neocortical radial glial stem cells. Stem Cells 2014, 32, 45–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ueno, H.; Kurokawa, M.S.; Kayama, M.; Homma, R.; Kumagai, Y.; Masuda, C.; Takada, E.; Tsubota, K.; Ueno, S.; Suzuki, N. Experimental Transplantation of Corneal Epithelium-like Cells Induced by Pax6 Gene Transfection of Mouse Embryonic Stem Cells. Cornea 2007, 26, 1220–1227. [Google Scholar] [CrossRef]
- Sun, J.; Liu, W.-H.; Deng, F.-M.; Luo, Y.-H.; Wen, K.; Zhang, H.; Liu, H.-R.; Wu, J.; Su, B.-Y.; Liu, Y.-L. Differentiation of rat adipose-derived mesenchymal stem cells into corneal-like epithelial cells driven by PAX6. Exp. Ther. Med. 2018, 15, 1424–1432. [Google Scholar] [CrossRef]
- Sasamoto, Y.; Hayashi, R.; Park, S.-J.; Saito-Adachi, M.; Suzuki, Y.; Kawasaki, S.; Quantock, A.J.; Nakai, K.; Tsujikawa, M.; Nishida, K. PAX6 Isoforms, along with Reprogramming Factors, Differentially Regulate the Induction of Cornea-specific Genes. Sci. Rep. 2016, 6, 20807. [Google Scholar] [CrossRef] [Green Version]
- Heavner, W.; Pevny, L. Eye development and retinogenesis. Cold Spring Harb. Perspect. Biol. 2012, 4, a008391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, B.; Dodge, M.E.; Tang, W.; Lu, J.; Ma, Z.; Fan, C.-W.; Wei, S.; Hao, W.; Kilgore, J.; Williams, N.S.; et al. Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer. Nat. Chem. Biol. 2009, 5, 100–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.J.; Wolosin, J.M.; Chung, S.-H. Divergent effects of Wnt/β-catenin signaling modifiers on the preservation of human limbal epithelial progenitors according to culture condition. Sci. Rep. 2017, 7, 15241. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; He, X. Wnt/beta-catenin signaling: New (and old) players and new insights. Curr. Opin. Cell Biol. 2008, 20, 119–125. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Park, J.W.; Zheng, D.; Xu, R.-H. Universal Corneal Epithelial-Like Cells Derived from Human Embryonic Stem Cells for Cellularization of a Corneal Scaffold. Transl. Vis. Sci. Technol. 2018, 7, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamilton, P.W.; Sun, Y.; Henry, J.J. Lens regeneration from the cornea requires suppression of Wnt/β-catenin signaling. Exp. Eye Res. 2016, 145, 206–215. [Google Scholar] [CrossRef] [Green Version]
- Kahata, K.; Dadras, M.S.; Moustakas, A. TGF-beta Family Signaling in Epithelial Differentiation and Epithelial-Mesenchymal Transition. Cold Spring Harb. Perspect. Biol. 2018, 10. [Google Scholar] [CrossRef] [Green Version]
- Inman, G.J.; Nicolás, F.J.; Callahan, J.F.; Harling, J.D.; Gaster, L.M.; Reith, A.D.; Laping, N.J.; Hill, C.S. SB-431542 Is a Potent and Specific Inhibitor of Transforming Growth Factor-β Superfamily Type I Activin Receptor-Like Kinase (ALK) Receptors ALK4, ALK5, and ALK7. Mol. Pharmacol. 2002, 62, 65–74. [Google Scholar] [CrossRef]
- DaCosta Byfield, S.; Major, C.; Laping, N.J.; Roberts, A.B. SB-505124 is a selective inhibitor of transforming growth factor-beta type I receptors ALK4, ALK5, and ALK7. Mol. Pharm. 2004, 65, 744–752. [Google Scholar] [CrossRef]
- Tojo, M.; Hamashima, Y.; Hanyu, A.; Kajimoto, T.; Saitoh, M.; Miyazono, K.; Node, M.; Imamura, T. The ALK-5 inhibitor A-83-01 inhibits Smad signaling and epithelial-to-mesenchymal transition by transforming growth factor-β. Cancer Sci. 2005, 96, 791–800. [Google Scholar] [CrossRef]
- Wang, D.; Wang, E.; Liu, K.; Xia, C.-h.; Li, S.; Gong, X. Roles of TGFβ and FGF signals during growth and differentiation of mouse lens epithelial cell in vitro. Sci. Rep. 2017, 7, 7274. [Google Scholar] [CrossRef] [PubMed]
- Kamarudin, T.A.; Bojic, S.; Collin, J.; Yu, M.; Alharthi, S.; Buck, H.; Shortt, A.; Armstrong, L.; Figueiredo, F.C.; Lako, M. Differences in the Activity of Endogenous Bone Morphogenetic Protein Signaling Impact on the Ability of Induced Pluripotent Stem Cells to Differentiate to Corneal Epithelial-Like Cells. Stem Cells 2018, 36, 337–348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, H.M.; Cheng, J.C.; Fang, L.; Qiu, X.; Klausen, C.; Taylor, E.L.; Leung, P.C. Recombinant BMP4 and BMP7 downregulate pentraxin 3 in human granulosa cells. J. Clin. Endocrinol. Metab. 2015, 100, E365–E374. [Google Scholar] [CrossRef] [Green Version]
- Hongisto, H.; Ilmarinen, T.; Vattulainen, M.; Mikhailova, A.; Skottman, H. Xeno- and feeder-free differentiation of human pluripotent stem cells to two distinct ocular epithelial cell types using simple modifications of one method. Stem Cell Res. Ther. 2017, 8, 291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hongisto, H.; Vattulainen, M.; Ilmarinen, T.; Mikhailova, A.; Skottman, H. Efficient and Scalable Directed Differentiation of Clinically Compatible Corneal Limbal Epithelial Stem Cells from Human Pluripotent Stem Cells. J. Vis. Exp. 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vattulainen, M.; Ilmarinen, T.; Koivusalo, L.; Viiri, K.; Hongisto, H.; Skottman, H. Modulation of Wnt/BMP pathways during corneal differentiation of hPSC maintains ABCG2-positive LSC population that demonstrates increased regenerative potential. Stem Cell Res. Ther. 2019, 10, 236. [Google Scholar] [CrossRef]
- Fitzsimmons, R.E.B.; Mazurek, M.S.; Soos, A.; Simmons, C.A. Mesenchymal Stromal/Stem Cells in Regenerative Medicine and Tissue Engineering. Stem Cells Int. 2018, 2018, 8031718. [Google Scholar] [CrossRef]
- Najar, M.; Fahmi, H. Of Mesenchymal Stem/Stromal Cells and Osteoarthritis: Time to Merge the Latest Breakthroughs. Stem Cell Rev. Rep. 2020, 16, 1016–1018. [Google Scholar] [CrossRef]
- Kim, H.J.; Park, J.-S. Usage of Human Mesenchymal Stem Cells in Cell-based Therapy: Advantages and Disadvantages. Dev. Reprod. 2017, 21, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Musiał-Wysocka, A.; Kot, M.; Majka, M. The Pros and Cons of Mesenchymal Stem Cell-Based Therapies. Cell Transplant. 2019, 28, 801–812. [Google Scholar] [CrossRef] [Green Version]
- Squillaro, T.; Peluso, G.; Galderisi, U. Clinical Trials With Mesenchymal Stem Cells: An Update. Cell Transplant. 2016, 25, 829–848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrzejewska, A.; Lukomska, B.; Janowski, M. Concise Review: Mesenchymal Stem Cells: From Roots to Boost. Stem Cells 2019, 37, 855–864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viswanathan, S.; Shi, Y.; Galipeau, J.; Krampera, M.; Leblanc, K.; Martin, I.; Nolta, J.; Phinney, D.G.; Sensebe, L. Mesenchymal stem versus stromal cells: International Society for Cell & Gene Therapy (ISCT®) Mesenchymal Stromal Cell committee position statement on nomenclature. Cytotherapy 2019, 21, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Miwa, H.; Era, T. Tracing the destiny of mesenchymal stem cells from embryo to adult bone marrow and white adipose tissue via Pdgfrα expression. Development 2018, 145, dev155879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeon, B.-G.; Jang, S.-J.; Park, J.-S.; Subbarao, R.B.; Jeong, G.-J.; Park, B.-W.; Rho, G.-J. Differentiation potential of mesenchymal stem cells isolated from human dental tissues into non-mesodermal lineage. Anim. Cells Syst. 2015, 19, 321–331. [Google Scholar] [CrossRef] [Green Version]
- Azandeh, S.; Mohammad Gharravi, A.; Orazizadeh, M.; Khodadi, A.; Hashemi Tabar, M. Improvement of mesenchymal stem cell differentiation into the endoderm lineage by four step sequential method in biocompatible biomaterial. Bioimpacts 2016, 6, 9–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jadalannagari, S.; Aljitawi, O.S. Ectodermal Differentiation of Wharton’s Jelly Mesenchymal Stem Cells for Tissue Engineering and Regenerative Medicine Applications. Tissue Eng. Part B Rev. 2014, 21, 314–322. [Google Scholar] [CrossRef]
- Jadalannagari, S.; Berry, A.M.; Hopkins, R.A.; Bhavsar, D.; Aljitawi, O.S. Potential mechanisms underlying ectodermal differentiation of Wharton’s jelly mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2016, 478, 831–837. [Google Scholar] [CrossRef]
- Sacchetti, M.; Rama, P.; Bruscolini, A.; Lambiase, A. Limbal Stem Cell Transplantation: Clinical Results, Limits, and Perspectives. Stem Cells Int. 2018, 2018, 8086269. [Google Scholar] [CrossRef] [Green Version]
- Guo, T.; Wang, W.; Zhang, J.; Chen, X.; Li, B.; Li, L. Experimental study on repairing damage of corneal surface by mesenchymal stem cells transplantation. Chin. J. Ophthalmol. 2006, 42, 246–250. [Google Scholar]
- Monteiro, B.G.; Serafim, R.C.; Melo, G.B.; Silva, M.C.P.; Lizier, N.F.; Maranduba, C.M.C.; Smith, R.L.; Kerkis, A.; Cerruti, H.; Gomes, J.A.P.; et al. Human immature dental pulp stem cells share key characteristic features with limbal stem cells. Cell Prolif. 2009, 42, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.Á.P.; Geraldes Monteiro, B.; Melo, G.B.; Smith, R.L.; Cavenaghi Pereira da Silva, M.; Lizier, N.F.; Kerkis, A.; Cerruti, H.; Kerkis, I. Corneal Reconstruction with Tissue-Engineered Cell Sheets Composed of Human Immature Dental Pulp Stem Cells. Investig. Ophthalmol. Vis. Sci. 2010, 51, 1408–1414. [Google Scholar] [CrossRef]
- Lin, K.-J.; Loi, M.-X.; Lien, G.-S.; Cheng, C.-F.; Pao, H.-Y.; Chang, Y.-C.; Ji, A.T.-Q.; Ho, J.H.-C. Topical administration of orbital fat-derived stem cells promotes corneal tissue regeneration. Stem Cell Res. Ther. 2013, 4, 72. [Google Scholar] [CrossRef] [Green Version]
- Zeppieri, M.; Salvetat, M.; Beltrami, A.; Cesselli, D.; Bergamin, N.; Russo, R.; Cavaliere, F.; Varano, G.; Alcalde, I.; Merayo-Lloves, J.; et al. Human Adipose-Derived Stem Cells for the Treatment of Chemically Burned Rat Cornea: Preliminary Results. Curr. Eye Res. 2013, 38. [Google Scholar] [CrossRef] [PubMed]
- Galindo, S.; Herreras, J.M.; López-Paniagua, M.; Rey, E.; de la Mata, A.; Plata-Cordero, M.; Calonge, M.; Nieto-Miguel, T. Therapeutic Effect of Human Adipose Tissue-Derived Mesenchymal Stem Cells in Experimental Corneal Failure Due to Limbal Stem Cell Niche Damage. Stem Cells 2017, 35, 2160–2174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinach, P.; Pokorny, K. The corneal epithelium: Clinical relevance of cytokine-mediated responses to maintenance of corneal health. Arq. Bras. Oftalmol. 2008, 71, 80–86. [Google Scholar] [CrossRef] [Green Version]
- Kruse, F.E.; Tseng, S.C. Retinoic acid regulates clonal growth and differentiation of cultured limbal and peripheral corneal epithelium. Investig. Ophthalmol. Vis. Sci. 1994, 35, 2405–2420. [Google Scholar]
- Yu, F.-S.X.; Yin, J.; Xu, K.; Huang, J. Growth factors and corneal epithelial wound healing. Brain Res. Bull. 2010, 81, 229–235. [Google Scholar] [CrossRef] [Green Version]
- Xue, G.; Han, X.; Ma, X.; Wu, H.; Qin, Y.; Liu, J.; Hu, Y.; Hong, Y.; Hou, Y. Effect of Microenvironment on Differentiation of Human Umbilical Cord Mesenchymal Stem Cells into Hepatocytes In Vitro and In Vivo. BioMed Res. Int. 2016, 2016, 8916534. [Google Scholar] [CrossRef] [Green Version]
- Keung, A.J.; Kumar, S.; Schaffer, D.V. Presentation counts: Microenvironmental regulation of stem cells by biophysical and material cues. Annu. Rev. Cell Dev. Biol. 2010, 26, 533–556. [Google Scholar] [CrossRef]
- Wan, P.-X.; Wang, B.-W.; Wang, Z.-C. Importance of the stem cell microenvironment for ophthalmological cell-based therapy. World J. Stem Cells 2015, 7, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Steens, J.; Klein, D. Current Strategies to Generate Human Mesenchymal Stem Cells In Vitro. Stem Cells Int. 2018, 2018, 6726185. [Google Scholar] [CrossRef] [PubMed]
- Abdal Dayem, A.; Lee, S.B.; Kim, K.; Lim, K.M.; Jeon, T.-I.; Seok, J.; Cho, S.-G. Production of Mesenchymal Stem Cells Through Stem Cell Reprogramming. Int. J. Mol. Sci. 2019, 20, 1922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, T.-S.; Cai, L.; Ji, W.-Y.; Hui, Y.-N.; Wang, Y.-S.; Hu, D.; Zhu, J. Reconstruction of the corneal epithelium with induced marrow mesenchymal stem cells in rats. Mol. Vis. 2010, 16, 1304–1316. [Google Scholar]
- Raj, D.K.; Kumary, T.V.; Anil Kumar, P.R. Development of corneal epithelial cell sheet construct from trans-differentiated bone marrow mesenchymal stem cells. Adv. Tissue Eng. Regen. Med. 2018, 4, 127–135. [Google Scholar] [CrossRef]
- Venugopal, B.; Shenoy, S.J.; Mohan, S.; Anil Kumar, P.R.; Kumary, T.V. Bioengineered corneal epithelial cell sheet from mesenchymal stem cells-A functional alternative to limbal stem cells for ocular surface reconstruction. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 1033–1045. [Google Scholar] [CrossRef]
- Nieto-Miguel, T.; Galindo, S.; Reinoso, R.; Corell, A.; Martino, M.; Pérez-Simón, J.A.; Calonge, M. In vitro simulation of corneal epithelium microenvironment induces a corneal epithelial-like cell phenotype from human adipose tissue mesenchymal stem cells. Curr. Eye Res. 2013, 38, 933–944. [Google Scholar] [CrossRef]
- Sikora, B.; Skubis-Sikora, A.; Kimsa-Furdzik, M.; Ciszek, W.; Kostrzewski, M.; Stojko, J.; Mazurek, U.; Gola, J. Adipose-derived stem cells undergo differentiation after co-culture with porcine limbal epithelial stem cells. Stem Cell Res. 2019, 41, 101609. [Google Scholar] [CrossRef]
- Tsai, C.L.; Chuang, P.C.; Kuo, H.K.; Chen, Y.H.; Su, W.H.; Wu, P.C. Differentiation of Stem Cells From Human Exfoliated Deciduous Teeth Toward a Phenotype of Corneal Epithelium In Vitro. Cornea 2015, 34, 1471–1477. [Google Scholar] [CrossRef]
- Soleimanifar, F.; Mortazavi, Y.; Nadri, S.; Soleimani, M. Conjunctiva derived mesenchymal stem cell (CJMSCs) as a potential platform for differentiation into corneal epithelial cells on bioengineered electrospun scaffolds. J. Biomed. Mater. Res. Part A 2017, 105, 2703–2711. [Google Scholar] [CrossRef]
- Ding, Y.; Gan, Y.; Feng, J.B.; Qi, S.H.; Li, R.M.; Li, D.S. Efficient isolation inner cell mass from blastocysts by improved microsurgical technique. Cell Res. 2008, 18, S39. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Homma, R.; Yoshikawa, H.; Takeno, M.; Kurokawa, M.S.; Masuda, C.; Takada, E.; Tsubota, K.; Ueno, S.; Suzuki, N. Induction of Epithelial Progenitors In Vitro from Mouse Embryonic Stem Cells and Application for Reconstruction of Damaged Cornea in Mice. Investig. Ophthalmol. Vis. Sci. 2004, 45, 4320–4326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumagai, Y.; Kurokawa, M.S.; Ueno, H.; Kayama, M.; Tsubota, K.; Nakatsuji, N.; Nito, S.; Ueno, S.; Suzuki, N. Induction of Corneal Epithelium-Like Cells From Cynomorgus Monkey Embrionic Stem Cells and Their Experimental Transplantation to Damaged Cornea. Investig. Ophthalmol. Vis. Sci. 2007, 48, 432–438. [Google Scholar]
- Notara, M.; Hernandez, D.; Mason, C.; Daniels, J.T. Characterization of the phenotype and functionality of corneal epithelial cells derived from mouse embryonic stem cells. Regen. Med. 2012, 7, 167–178. [Google Scholar] [CrossRef]
- Hanson, C.; Hardarson, T.; Ellerström, C.; Nordberg, M.; Caisander, G.; Rao, M.; Hyllner, J.; Stenevi, U. Transplantation of human embryonic stem cells onto a partially wounded human cornea in vitro. Acta Ophthalmol. 2013, 91, 127–130. [Google Scholar] [CrossRef] [Green Version]
- Susaimanickam, P.J.; Maddileti, S.; Pulimamidi, V.K.; Boyinpally, S.R.; Naik, R.R.; Naik, M.N.; Reddy, G.B.; Sangwan, V.S.; Mariappan, I. Generating minicorneal organoids from human induced pluripotent stem cells. Development 2017, 144, 2338. [Google Scholar] [CrossRef] [Green Version]
- Foster, J.W.; Wahlin, K.; Adams, S.M.; Birk, D.E.; Zack, D.J.; Chakravarti, S. Cornea organoids from human induced pluripotent stem cells. Sci. Rep. 2017, 7, 41286. [Google Scholar] [CrossRef]
- Brzeszczynska, J.; Samuel, K.; Greenhough, S.; Ramaesh, K.; Dhillon, B.; Hay, D.C.; Ross, J.A. Differentiation and molecular profiling of human embryonic stem cell-derived corneal epithelial cells. Int. J. Mol. Med. 2014, 33, 1597–1606. [Google Scholar] [CrossRef] [Green Version]
- Cieslar-Pobuda, A.; Rafat, M.; Knoflach, V.; Skonieczna, M.; Hudecki, A.; Malecki, A.; Urasinska, E.; Ghavami, S.; Los, M.J. Human induced pluripotent stem cell differentiation and direct transdifferentiation into corneal epithelial-like cells. Oncotarget 2016, 7, 42314–42329. [Google Scholar] [CrossRef]
- Richards, M.; Fong, C.-Y.; Tan, S.; Chan, W.-K.; Bongso, A. An Efficient and Safe Xeno-Free Cryopreservation Method for the Storage of Human Embryonic Stem Cells. Stem Cells 2004, 22, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J.; Tilkins, M.L.; Eckert, R.; Boucher, S. Methods for culturing human embryonic stem cells in a xeno-free system. Methods Mol. Biol. 2013, 997, 115–126. [Google Scholar] [CrossRef]
- Tanjore, H.; Kalluri, R. The role of type IV collagen and basement membranes in cancer progression and metastasis. Am. J. Pathol. 2006, 168, 715–717. [Google Scholar] [CrossRef] [Green Version]
- Kalluri, R. Basement membranes: Structure, assembly and role in tumour angiogenesis. Nat. Rev. Cancer 2003, 3, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Cameron, J.D.; Skubitz, A.P.; Furcht, L.T. Type IV collagen and corneal epithelial adhesion and migration. Effects of type IV collagen fragments and synthetic peptides on rabbit corneal epithelial cell adhesion and migration in vitro. Investig. Ophthalmol. Vis. Sci. 1991, 32, 2766–2773. [Google Scholar] [PubMed]
- Hayashi, R.; Ishikawa, Y.; Ito, M.; Kageyama, T.; Takashiba, K.; Fujioka, T.; Tsujikawa, M.; Miyoshi, H.; Yamato, M.; Nakamura, Y.; et al. Generation of corneal epithelial cells from induced pluripotent stem cells derived from human dermal fibroblast and corneal limbal epithelium. PLoS ONE 2012, 7, e45435. [Google Scholar] [CrossRef] [Green Version]
- Saichanma, S.; Bunyaratvej, A.; Sila-Asna, M. In vitro transdifferentiation of corneal epithelial-like cells from human skin-derived precursor cells. Int. J. Ophthalmol. 2012, 5, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Man, R.C.; Yong, T.K.; Hwei, N.M.; Halim, W.H.W.A.; Zahidin, A.Z.M.; Ramli, R.; Saim, A.B.; Idrus, R.B.H. Corneal regeneration by induced human buccal mucosa cultivated on an amniotic membrane following alkaline injury. Mol. Vis. 2017, 23, 810–822. [Google Scholar]
- Gaddipati, S.; Muralidhar, R.; Sangwan, V.S.; Mariappan, I.; Vemuganti, G.K.; Balasubramanian, D. Oral epithelial cells transplanted on to corneal surface tend to adapt to the ocular phenotype. Indian J. Ophthalmol. 2014, 62, 644–648. [Google Scholar] [CrossRef]
- Dobrowolski, D.; Orzechowska-Wylegala, B.; Wowra, B.; Wroblewska-Czajka, E.; Grolik, M.; Szczubialka, K.; Nowakowska, M.; Puzzolo, D.; Wylegala, E.A.; Micali, A.; et al. Cultivated Oral Mucosa Epithelium in Ocular Surface Reconstruction in Aniridia Patients. Biomed. Res. Int. 2015, 2015, 281870. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.C.; Chen, H.L.; Lai, J.Y.; Chen, C.C.; Tsai, Y.J.; Kuo, M.T.; Chu, P.H.; Sun, C.C.; Chen, J.K.; Ma, D.H. Persistence of transplanted oral mucosal epithelial cells in human cornea. Investig. Ophthalmol. Vis. Sci. 2009, 50, 4660–4668. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reza, H.M.; Ng, B.-Y.; Gimeno, F.L.; Phan, T.T.; Ang, L.P.-K. Umbilical Cord Lining Stem Cells as a Novel and Promising Source for Ocular Surface Regeneration. Stem Cell Rev. Rep. 2011, 7, 935–947. [Google Scholar] [CrossRef]
- Ngan, N.D.; Chau, H.M.; Dong, P.N.; Cung, L.X.; Thuy, N.T.; Thang, P.T.; Thai, T.V.; Nga, V.T.; Bac, N.D. Tissue-Cultured Human Cord Lining Epithelial Cells in Treatment of Persistent Corneal Epithelial Defect. Open Access Maced. J. Med. Sci. 2019, 7, 4266–4271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Human MSCs | Signal Providing Cells | Culture Medium | Duration (Days) | Marker Expression at the Final Differentiation Stage | Reference |
|---|---|---|---|---|---|
| AT-MSCs | porcine limbal epithelial stem cells | DMEM FBS 10% | 14 | gene expression increase: CK3, CK12, p63 decrease: CD73, CD90, CD105, CK15, CK19, Cx43, ABCG2 | [119] |
| CJ-MSCs | human corneal epithelial cells | SHEM medium FBS 5% DMSO 0.5% EGF 5 ng/mL Insulin 5 μg/mL Transferrin 5 μg/mL Sodium selenite 5 ng/mL Hydrocortisone 0.5 μg/mL Cholera toxin A 30 ng/mL | 24 | gene expression increase: CK3, CK8, CK12, CK14, CK15, ABCG2, DSC1, DSG1, NP63-α, Nestin, Involucrin ICC positive: CK3 | [24] |
| EDT-MSCs | human corneal epithelial cells | DMEM/Ham F12 (1:1) FBS 5% DMSO 0.5% EGF 5 ng/mL | 21 | gene expression increase: CK3, CK19 ICC positive: CK3, CK19 | [120] |
| Human MSCs | Induction Medium | Duration (Days) | Marker Expression at the Final Differentiation Stage | Reference |
|---|---|---|---|---|
| CJ-MSCs | CnT-Prime 3D medium | 24 | gene expression increase: CK3, CK8, CK12, CK14, CK15, ABCG2, DSC1, DSG1, NP63-α, Nestin, Involucrin ICC positive: CK3 | [24] |
| CJ-MSCs | DMEM: Ham’s F-12 (3:1) FBS 5% EGF 10 ng/mL Insulin 5 μg/mL Hydrocortisone 0.5 μg/mL Triiodothyronine 2 nM Adenine 2 nM | 21 | gene expression increase: CK3, CK8, CK12, DSC1, DSG1 | [121] |
| BM-MSCs | Step 1 DMEM EGF 10 ng/mL BMP4 25 ng/mL All-trans retinoic acid 1 μM Step 2 DMEM: Ham’s F-12 (3:1) FBS 5% EGF 10 ng/mL Insulin 5 μg/mL Hydrocortisone 0.5 μg/mL Triiodothyronine 2 nM Adenine 2 nM | 4 (step 1) and 12 (step 2) | gene expression increase: CK3, CK8, CK12, DSC1, DSG1 decrease: Oct4, Sox2, Nanog, Rex1, p63, ABCG2 ICC positive: CK3, CK8, CK12, CK14, CK15, β-integrin, E-cadherin negative: α-SMA | [20] |
| BM-MSCs | DMEM FBS 2% L-glutamine 2 mM All-trans retinoic acid 1 μM | 7 | gene expression increase: CK3, CK12, CK19, E-cad, ITGB1, Wnt-2, Snail decrease: Vimentin Protein expression increase: CK12, CK19, ITGB1, N-cad decrease: CK3, Vimentin, Snail, α-sma | [21] |
| BM-MSCs | SHEM/Ham’s F12 (2:1) FBS 2% DMSO 0.5% L-Glutamine 2 mM EGF 10 ng/mL Insulin 5 μg/mL Hydrocortisone 0.4 μg/mL Triiodothyronine 2 nM Adenine 0.18 mM | 7 | gene expression increase: CK3, CK12, ITGB1 decrease: CK19, E-cad, Vimentin, Wnt-2, Snail Protein expression increase: CK3, CK12, ITGB1 decrease: CK19, Vimentin, Snail, α-sma, N-cad | [21] |
| Human Cell Line | Method | Step 1 | Step 2 | Step 3 | Duration (Days) | ICC Result | Flow Cytometry/Cytospin Result | Gene Expression Result | Reference |
|---|---|---|---|---|---|---|---|---|---|
| ESCs (H1, hES-NCL1) | CM of MMC-treated limbal fibroblasts | 3LG-DMEM/1F12 +hydrocortisone, insulin, adenine, EGF, tri-iodothyronine, cholera toxin | - | 21 | hES-NCL1: high positive with CK12, CK3/12, p63 on D7 | Flow cytometry p63 highest on D6 hES-NCL1: CK3/12~55% highest on D6 H1: CK3/12~55% highest on D9 | CK3 highest on D15 hES-NCL1: CK3+, CK12+ highest on D6 H1: CK3+++, CK12+++ highest on D9 | [25] | |
| iPSCs (L1B41, L1C51, 253G1, 201B7, L1B34) | Co-culture with MMC-treated PA6 | GMEM +KSR, 2-mercaptoethanol | 12–16 weeks | L1B41: CK12, CK3, PAX6, CK14 | L1B41 high responsive: CK3, CK12: high express from week 8–12 Other cell lines low or non-responsive | [137] | |||
| ESCs (H9, H3, H14) iPSCs (6-9-9, 19-9-11) | Defined medium | DMEM/F12 +β-mercaptoethanol, DMSO, SU6656 | DMEM/F12 +RA, BMP4, bFGF, β-mercaptoethanol | DKSFM | 19 | CK14, CK3, p63 | Flow cytometry CK3 5%, CK14 90.7% | [59] | |
| iPSCs | Defined medium | RegES medium +SB505124, IWP-2, bFGF | Cnt-30 | 44 | D20 CK12, CK3, CK15, Ki67, Pax6 | Cytospin on D44 p63 70%, CK3 30%, CK15 55%, CK12 70% | P63+++, CK15++, CK3+, and CK12+ | [61] | |
| ESCs (H9, RCM1) | CM from limbal fibroblasts | 3LG-DMEM/1F12 +hydrocortisone, insulin, adenine, EGF, triiodothyronine, cholera toxin | 21 | - | Flow cytometry H9: CK3 99% on D21, high rate from D4–21 RCM1: CK12 94% on D21, high rate from D16–21 | CK3, CK12: very low express, p63, CK19, ABCG2: high express on D16 and D21 | [130] | ||
| iPSCs | CM from limbal stromal cells | EpiLife medium +human corneal growth supplement | 21 | CK3, CK12, p63 express from D7–21 | CK3, CK12 high express on D14–21 | [131] | |||
| iPSCs (UTA.045111.WT) | Defined medium | XF-Ko-SR D1: blebbistatin D2: SB505124, bFGF D3–4: BMP4 | Cnt-30 | 21 | CK14, CK15, p63, PAX6 | Flow cytometry p63 71% | [85] | ||
| ESCs (H9) iPSCs (SBAd2, SBAd3) | Defined medium | mTeSR1 D1:Y27632 | DMEM/F12 +BMP4/RA/EGF/LDN193189 or BMP4+RA+EGF or SB505124+IWP2 w/wo BMP4 | CnT prime +10%FBS | 20 | CK12: not different on D20, CK3: lower express on D20 in SBAd3 group | BMP4+RA+EGF is the best SBAd2 &H9 responsive: CK3, CK12: high express SBAd3 non-responsive | [83] | |
| ESCs (H1, H9, CT3, Envy (GFP+)) | Defined medium | E6 +IWR1, A83-01, bFGF w/wo BMP4 | E6 | 75 | CK12, CK3 | Flow cytometry CK3 55.6%, CK12 28.2% | [76] | ||
| ESCs (Regea 08/017, Regea11/013) iPSCs (UTA.04607.WT) | Defined medium | XF-Ko-SR D1: blebbistatin D2: SB505124, bFGF D3–4: BMP4 | Cnt-30 | 24 | D11: ABCG2+++, ∆Np63++, LGR5+++ D24: ABCG2+, ∆Np63+++, LGR5+, CK14+++, CK15++ | Cytopsin: D11: ABCG2 62.4%, ∆Np63 23.2%, CD200 42.6% D24: ABCG2 1.8%, ∆Np63 54.3%, CK15 37%, CK14 56.2% | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theerakittayakorn, K.; Thi Nguyen, H.; Musika, J.; Kunkanjanawan, H.; Imsoonthornruksa, S.; Somredngan, S.; Ketudat-Cairns, M.; Parnpai, R. Differentiation Induction of Human Stem Cells for Corneal Epithelial Regeneration. Int. J. Mol. Sci. 2020, 21, 7834. https://doi.org/10.3390/ijms21217834
Theerakittayakorn K, Thi Nguyen H, Musika J, Kunkanjanawan H, Imsoonthornruksa S, Somredngan S, Ketudat-Cairns M, Parnpai R. Differentiation Induction of Human Stem Cells for Corneal Epithelial Regeneration. International Journal of Molecular Sciences. 2020; 21(21):7834. https://doi.org/10.3390/ijms21217834
Chicago/Turabian StyleTheerakittayakorn, Kasem, Hong Thi Nguyen, Jidapa Musika, Hataiwan Kunkanjanawan, Sumeth Imsoonthornruksa, Sirilak Somredngan, Mariena Ketudat-Cairns, and Rangsun Parnpai. 2020. "Differentiation Induction of Human Stem Cells for Corneal Epithelial Regeneration" International Journal of Molecular Sciences 21, no. 21: 7834. https://doi.org/10.3390/ijms21217834







