Tumour-Infiltrating Inflammatory Cells in Early Breast Cancer: An Underrated Prognostic and Predictive Factor?
Abstract
:1. Introduction
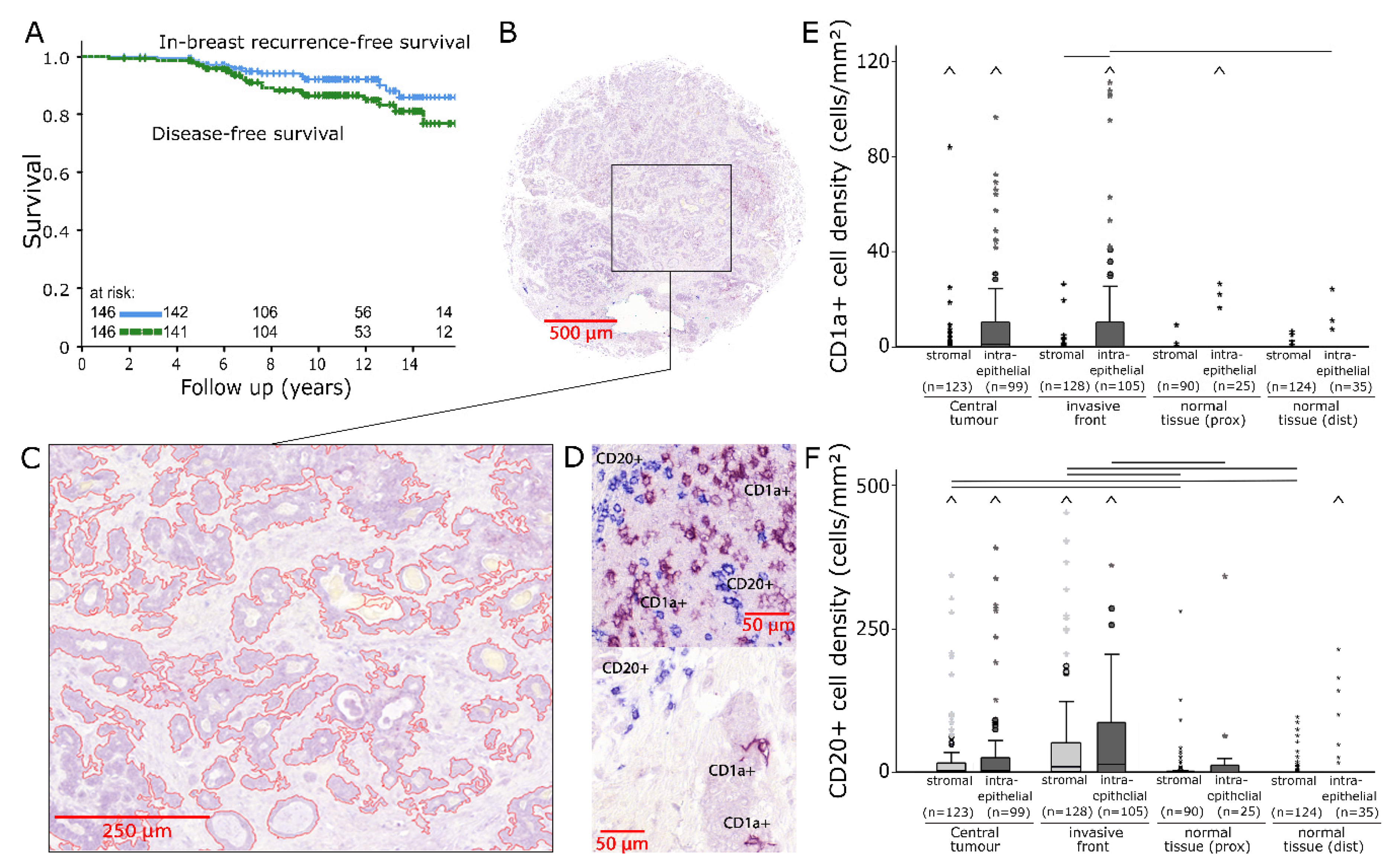
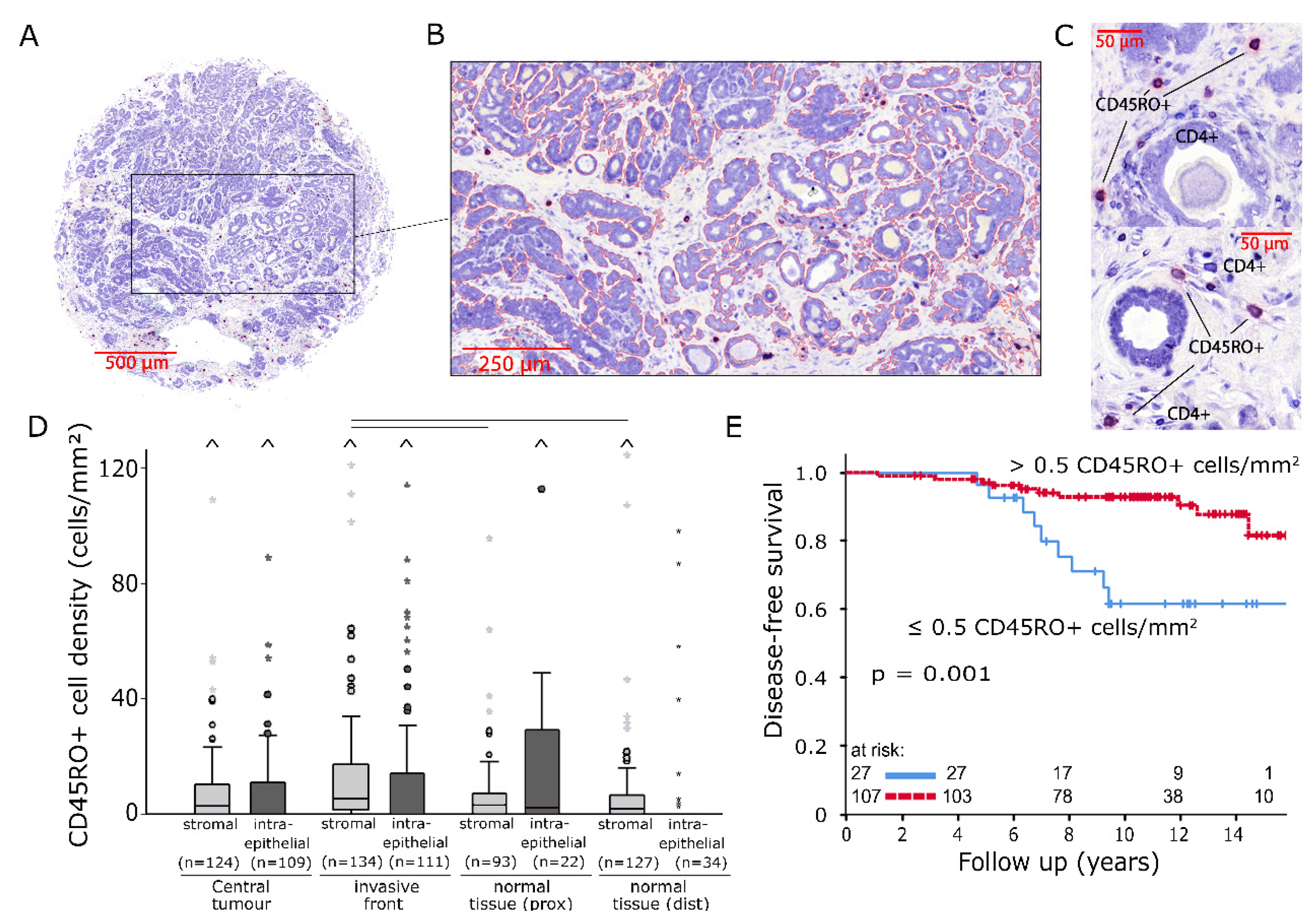
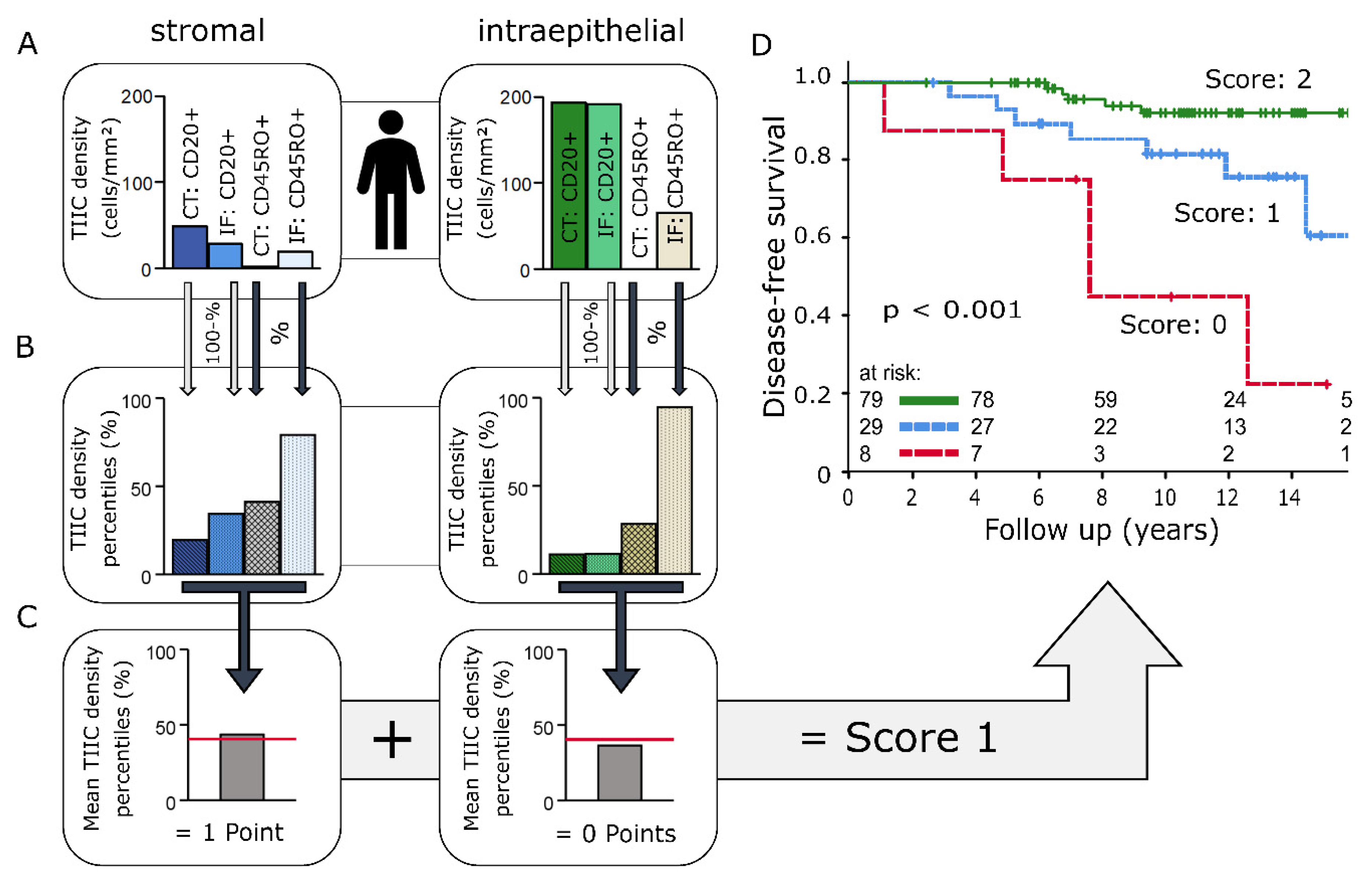
2. Results
2.1. CD1a/CD20 Immunostaining
2.2. CD45RO/CD4 Immunostaining
2.3. Immunoscore
3. Discussion
3.1. CD1a+ Dendritic Cells
3.2. CD45RO+ Memory T Cells
3.3. CD20+ B Cells
3.4. CD4+ T-Helper Cells
3.5. Immunoscore
4. Materials and Methods
4.1. Breast Cancer Patients and Clinical Data
4.2. TMA Construction and Immunohistochemistry
4.3. Quantification of Inflammatory Cells
4.4. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| TIIC | Tumour-infiltrating inflammatory cell |
| APBI | Accelerated partial breast irradiation |
| TMA | Tissue microarray |
| TAM | Tumour-associated macrophage |
| CT | Central tumour |
| IF | Invasive front |
| prox | Normal tissue in tumour proximity |
| dist | Normal tissue sampled distant from the tumour |
| HR+/- | Hormone receptor positive/negative |
| BC | Breast cancer |
| RT | Radiotherapy |
| ER | Oestrogen receptor |
| PR | Progesterone receptor |
| TNBC | Triple-negative breast cancer |
| DFS | Disease-free survival |
References
- Vonderheide, R.H.; Domchek, S.M.; Clark, A.S. Immunotherapy for Breast Cancer: What Are We Missing? Clin. Cancer Res. 2017, 23, 2640–2646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nieder, C. Combination immunotherapy for patients with brain metastases of malignant melanoma: De-escalation only under controlled conditions with frequent imaging. Strahlenther Onkol. 2019, 195, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Angell, H.K.; Bruni, D.; Barrett, J.C.; Herbst, R.; Galon, J. The Immunoscore: Colon Cancer and Beyond. Clin. Cancer Res. 2020, 26, 332–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salgado, R.; Denkert, C.; Demaria, S.; Sirtaine, N.; Klauschen, F.; Pruneri, G.; Wienert, S.; Van den Eynden, G.; Baehner, F.L.; Penault-Llorca, F.; et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: Recommendations by an International TILs Working Group 2014. Ann. Oncol. 2015, 26, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Stanton, S.E.; Adams, S.; Disis, M.L. Variation in the Incidence and Magnitude of Tumor-Infiltrating Lymphocytes in Breast Cancer Subtypes: A Systematic Review. JAMA Oncol. 2016, 2, 1354–1360. [Google Scholar] [CrossRef]
- Mao, Y.; Qu, Q.; Chen, X.; Huang, O.; Wu, J.; Shen, K. The Prognostic Value of Tumor-Infiltrating Lymphocytes in Breast Cancer: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0152500. [Google Scholar] [CrossRef]
- Ibrahim, E.M.; Al-Foheidi, M.E.; Al-Mansour, M.M.; Kazkaz, G.A. The prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancer: A meta-analysis. Breast Cancer Res. Treat. 2014, 148, 467–476. [Google Scholar] [CrossRef]
- Loi, S.; Michiels, S.; Salgado, R.; Sirtaine, N.; Jose, V.; Fumagalli, D.; Kellokumpu-Lehtinen, P.L.; Bono, P.; Kataja, V.; Desmedt, C.; et al. Tumor infiltrating lymphocytes are prognostic in triple negative breast cancer and predictive for trastuzumab benefit in early breast cancer: Results from the FinHER trial. Ann. Oncol. 2014, 25, 1544–1550. [Google Scholar] [CrossRef]
- Dieci, M.V.; Mathieu, M.C.; Guarneri, V.; Conte, P.; Delaloge, S.; Andre, F.; Goubar, A. Prognostic and predictive value of tumor-infiltrating lymphocytes in two phase III randomized adjuvant breast cancer trials. Ann. Oncol. 2015, 26, 1698–1704. [Google Scholar] [CrossRef]
- Iqbal, J.; Ginsburg, O.; Rochon, P.A.; Sun, P.; Narod, S.A. Differences in breast cancer stage at diagnosis and cancer-specific survival by race and ethnicity in the United States. JAMA 2015, 313, 165–173. [Google Scholar] [CrossRef] [Green Version]
- Abo-Madyan, Y.; Welzel, G.; Sperk, E.; Neumaier, C.; Keller, A.; Clausen, S.; Schneider, F.; Ehmann, M.; Sutterlin, M.; Wenz, F. Single-center long-term results from the randomized phase-3 TARGIT-A trial comparing intraoperative and whole-breast radiation therapy for early breast cancer. Strahlenther Onkol. 2019, 195, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Solinas, C.; Carbognin, L.; De Silva, P.; Criscitiello, C.; Lambertini, M. Tumor-infiltrating lymphocytes in breast cancer according to tumor subtype: Current state of the art. Breast 2017, 35, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Denkert, C.; von Minckwitz, G.; Darb-Esfahani, S.; Lederer, B.; Heppner, B.I.; Weber, K.E.; Budczies, J.; Huober, J.; Klauschen, F.; Furlanetto, J.; et al. Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: A pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol. 2018, 19, 40–50. [Google Scholar] [CrossRef]
- Dieci, M.V.; Griguolo, G.; Miglietta, F.; Guarneri, V. The immune system and hormone-receptor positive breast cancer: Is it really a dead end? Cancer Treat. Rev. 2016, 46, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Schnellhardt, S.; Erber, R.; Buttner-Herold, M.; Rosahl, M.C.; Ott, O.J.; Strnad, V.; Beckmann, M.W.; King, L.; Hartmann, A.; Fietkau, R.; et al. Accelerated Partial Breast Irradiation: Macrophage Polarisation Shift Classification Identifies High-Risk Tumours in Early Hormone Receptor-Positive Breast Cancer. Cancers 2020, 12, 446. [Google Scholar] [CrossRef] [Green Version]
- Cancer Genome Atlas, N. Comprehensive molecular portraits of human breast tumours. Nature 2012, 490, 61–70. [Google Scholar] [CrossRef] [Green Version]
- Luen, S.; Virassamy, B.; Savas, P.; Salgado, R.; Loi, S. The genomic landscape of breast cancer and its interaction with host immunity. Breast 2016, 29, 241–250. [Google Scholar] [CrossRef]
- Zhang, Q.; Qin, J.; Zhong, L.; Gong, L.; Zhang, B.; Zhang, Y.; Gao, W.Q. CCL5-Mediated Th2 Immune Polarization Promotes Metastasis in Luminal Breast Cancer. Cancer Res. 2015, 75, 4312–4321. [Google Scholar] [CrossRef] [Green Version]
- Svensson, S.; Abrahamsson, A.; Rodriguez, G.V.; Olsson, A.K.; Jensen, L.; Cao, Y.; Dabrosin, C. CCL2 and CCL5 Are Novel Therapeutic Targets for Estrogen-Dependent Breast Cancer. Clin. Cancer Res. 2015, 21, 3794–3805. [Google Scholar] [CrossRef] [Green Version]
- Joffroy, C.M.; Buck, M.B.; Stope, M.B.; Popp, S.L.; Pfizenmaier, K.; Knabbe, C. Antiestrogens induce transforming growth factor beta-mediated immunosuppression in breast cancer. Cancer Res. 2010, 70, 1314–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strnad, V.; Hildebrandt, G.; Potter, R.; Hammer, J.; Hindemith, M.; Resch, A.; Spiegl, K.; Lotter, M.; Uter, W.; Bani, M.; et al. Accelerated partial breast irradiation: 5-year results of the German-Austrian multicenter phase II trial using interstitial multicatheter brachytherapy alone after breast-conserving surgery. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 17–24. [Google Scholar] [CrossRef]
- Ott, O.J.; Hildebrandt, G.; Potter, R.; Hammer, J.; Lotter, M.; Resch, A.; Sauer, R.; Strnad, V. Accelerated partial breast irradiation with multi-catheter brachytherapy: Local control, side effects and cosmetic outcome for 274 patients. Results of the German-Austrian multi-centre trial. Radiother. Oncol. 2007, 82, 281–286. [Google Scholar] [CrossRef]
- Coventry, B.; Heinzel, S. CD1a in human cancers: A new role for an old molecule. Trends Immunol. 2004, 25, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Treilleux, I.; Blay, J.Y.; Bendriss-Vermare, N.; Ray-Coquard, I.; Bachelot, T.; Guastalla, J.P.; Bremond, A.; Goddard, S.; Pin, J.J.; Barthelemy-Dubois, C.; et al. Dendritic cell infiltration and prognosis of early stage breast cancer. Clin. Cancer Res. 2004, 10, 7466–7474. [Google Scholar] [CrossRef] [Green Version]
- Coventry, B.J.; Morton, J. CD1a-positive infiltrating-dendritic cell density and 5-year survival from human breast cancer. Br. J. Cancer 2003, 89, 533–538. [Google Scholar] [CrossRef] [Green Version]
- Bell, D.; Chomarat, P.; Broyles, D.; Netto, G.; Harb, G.M.; Lebecque, S.; Valladeau, J.; Davoust, J.; Palucka, K.A.; Banchereau, J. In breast carcinoma tissue, immature dendritic cells reside within the tumor, whereas mature dendritic cells are located in peritumoral areas. J. Exp. Med. 1999, 190, 1417–1426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwamoto, M.; Shinohara, H.; Miyamoto, A.; Okuzawa, M.; Mabuchi, H.; Nohara, T.; Gon, G.; Toyoda, M.; Tanigawa, N. Prognostic value of tumor-infiltrating dendritic cells expressing CD83 in human breast carcinomas. Int. J. Cancer 2003, 104, 92–97. [Google Scholar] [CrossRef]
- Michie, C.A.; McLean, A.; Alcock, C.; Beverley, P.C. Lifespan of human lymphocyte subsets defined by CD45 isoforms. Nature 1992, 360, 264–265. [Google Scholar] [CrossRef]
- Sarkar, I.; Pati, S.; Dutta, A.; Basak, U.; Sa, G. T-memory cells against cancer: Remembering the enemy. Cell Immunol. 2019, 338, 27–31. [Google Scholar] [CrossRef]
- Pages, F.; Berger, A.; Camus, M.; Sanchez-Cabo, F.; Costes, A.; Molidor, R.; Mlecnik, B.; Kirilovsky, A.; Nilsson, M.; Damotte, D.; et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N. Engl. J. Med. 2005, 353, 2654–2666. [Google Scholar] [CrossRef]
- Gao, Q.; Zhou, J.; Wang, X.Y.; Qiu, S.J.; Song, K.; Huang, X.W.; Sun, J.; Shi, Y.H.; Li, B.Z.; Xiao, Y.S.; et al. Infiltrating memory/senescent T cell ratio predicts extrahepatic metastasis of hepatocellular carcinoma. Ann. Surg. Oncol. 2012, 19, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.E.; Chae, S.W.; Lee, Y.J.; Kim, M.A.; Lee, H.S.; Lee, B.L.; Kim, W.H. Prognostic implications of type and density of tumour-infiltrating lymphocytes in gastric cancer. Br. J. Cancer 2008, 99, 1704–1711. [Google Scholar] [CrossRef] [Green Version]
- Hu, G.; Wang, S. Tumor-infiltrating CD45RO(+) Memory T Lymphocytes Predict Favorable Clinical Outcome in Solid Tumors. Sci. Rep. 2017, 7, 10376. [Google Scholar] [CrossRef] [Green Version]
- Yajima, R.; Yajima, T.; Fujii, T.; Yanagita, Y.; Fujisawa, T.; Miyamoto, T.; Tsutsumi, S.; Iijima, M.; Kuwano, H. Tumor-infiltrating CD45RO(+) memory cells are associated with a favorable prognosis breast cancer. Breast Cancer 2016, 23, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Ahmadvand, S.; Faghih, Z.; Montazer, M.; Safaei, A.; Mokhtari, M.; Jafari, P.; Talei, A.R.; Tahmasebi, S.; Ghaderi, A. Importance of CD45RO+ tumor-infiltrating lymphocytes in post-operative survival of breast cancer patients. Cell Oncol. (Dordr) 2019, 42, 343–356. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, Y.; Wang, Z.; Zhang, T.; Wu, P.; Huang, J. Yin-yang effect of tumor infiltrating B cells in breast cancer: From mechanism to immunotherapy. Cancer Lett. 2017, 393, 1–7. [Google Scholar] [CrossRef]
- Shen, M.; Wang, J.; Ren, X. New Insights into Tumor-Infiltrating B Lymphocytes in Breast Cancer: Clinical Impacts and Regulatory Mechanisms. Front. Immunol. 2018, 9, 470. [Google Scholar] [CrossRef] [Green Version]
- Mahmoud, S.M.; Lee, A.H.; Paish, E.C.; Macmillan, R.D.; Ellis, I.O.; Green, A.R. The prognostic significance of B lymphocytes in invasive carcinoma of the breast. Breast Cancer Res. Treat. 2012, 132, 545–553. [Google Scholar] [CrossRef]
- Garaud, S.; Buisseret, L.; Solinas, C.; Gu-Trantien, C.; de Wind, A.; Van den Eynden, G.; Naveaux, C.; Lodewyckx, J.N.; Boisson, A.; Duvillier, H.; et al. Tumor infiltrating B-cells signal functional humoral immune responses in breast cancer. JCI Insight 2019, 5. [Google Scholar] [CrossRef] [Green Version]
- Guo, F.F.; Cui, J.W. The Role of Tumor-Infiltrating B Cells in Tumor Immunity. J. Oncol. 2019, 2019, 2592419. [Google Scholar] [CrossRef]
- Olkhanud, P.B.; Damdinsuren, B.; Bodogai, M.; Gress, R.E.; Sen, R.; Wejksza, K.; Malchinkhuu, E.; Wersto, R.P.; Biragyn, A. Tumor-evoked regulatory B cells promote breast cancer metastasis by converting resting CD4(+) T cells to T-regulatory cells. Cancer Res. 2011, 71, 3505–3515. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Morgan, R.; Podack, E.R.; Rosenblatt, J. B cell regulation of anti-tumor immune response. Immunol. Res. 2013, 57, 115–124. [Google Scholar] [CrossRef]
- Gu, Y.; Liu, Y.; Fu, L.; Zhai, L.; Zhu, J.; Han, Y.; Jiang, Y.; Zhang, Y.; Zhang, P.; Jiang, Z.; et al. Tumor-educated B cells selectively promote breast cancer lymph node metastasis by HSPA4-targeting IgG. Nat. Med. 2019, 25, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gallastegui, N.; Rosenblatt, J.D. Regulatory B cells in anti-tumor immunity. Int. Immunol. 2015, 27, 521–530. [Google Scholar] [CrossRef]
- Wortel, C.M.; Heidt, S. Regulatory B cells: Phenotype, function and role in transplantation. Transpl. Immunol. 2017, 41, 1–9. [Google Scholar] [CrossRef]
- Caza, T.; Landas, S. Functional and Phenotypic Plasticity of CD4(+) T Cell Subsets. Biomed. Res. Int. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Ruffell, B.; DeNardo, D.G.; Affara, N.I.; Coussens, L.M. Lymphocytes in cancer development: Polarization towards pro-tumor immunity. Cytokine Growth Factor Rev. 2010, 21, 3–10. [Google Scholar] [CrossRef] [Green Version]
- Gu-Trantien, C.; Loi, S.; Garaud, S.; Equeter, C.; Libin, M.; de Wind, A.; Ravoet, M.; Le Buanec, H.; Sibille, C.; Manfouo-Foutsop, G.; et al. CD4(+) follicular helper T cell infiltration predicts breast cancer survival. J. Clin. Investig. 2013, 123, 2873–2892. [Google Scholar] [CrossRef]
- Liu, S.; Foulkes, W.D.; Leung, S.; Gao, D.; Lau, S.; Kos, Z.; Nielsen, T.O. Prognostic significance of FOXP3+ tumor-infiltrating lymphocytes in breast cancer depends on estrogen receptor and human epidermal growth factor receptor-2 expression status and concurrent cytotoxic T-cell infiltration. Breast Cancer Res. 2014, 16, 432. [Google Scholar] [CrossRef] [Green Version]
- Bates, G.J.; Fox, S.B.; Han, C.; Leek, R.D.; Garcia, J.F.; Harris, A.L.; Banham, A.H. Quantification of regulatory T cells enables the identification of high-risk breast cancer patients and those at risk of late relapse. J. Clin. Oncol. 2006, 24, 5373–5380. [Google Scholar] [CrossRef]
- Chung, Y.R.; Kim, H.J.; Jang, M.H.; Park, S.Y. Prognostic value of tumor infiltrating lymphocyte subsets in breast cancer depends on hormone receptor status. Breast Cancer Res. Treat. 2017, 161, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Weyer-Elberich, V.; Hengstler, J.G.; Heimes, A.S.; Almstedt, K.; Gerhold-Ay, A.; Lebrecht, A.; Battista, M.J.; Hasenburg, A.; Sahin, U.; et al. Prognostic impact of CD4-positive T cell subsets in early breast cancer: A study based on the FinHer trial patient population. Breast Cancer Res. 2018, 20, 15. [Google Scholar] [CrossRef]
- Generali, D.; Bates, G.; Berruti, A.; Brizzi, M.P.; Campo, L.; Bonardi, S.; Bersiga, A.; Allevi, G.; Milani, M.; Aguggini, S.; et al. Immunomodulation of FOXP3+ regulatory T cells by the aromatase inhibitor letrozole in breast cancer patients. Clin. Cancer Res. 2009, 15, 1046–1051. [Google Scholar] [CrossRef] [Green Version]
- Ott, O.J.; Hildebrandt, G.; Potter, R.; Hammer, J.; Hindemith, M.; Resch, A.; Spiegl, K.; Lotter, M.; Uter, W.; Kortmann, R.D.; et al. Accelerated partial breast irradiation with interstitial implants: Risk factors associated with increased local recurrence. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 1458–1463. [Google Scholar] [CrossRef]
- Strnad, V.; Krug, D.; Sedlmayer, F.; Piroth, M.D.; Budach, W.; Baumann, R.; Feyer, P.; Duma, M.N.; Haase, W.; Harms, W.; et al. DEGRO practical guideline for partial-breast irradiation. Strahlenther Onkol. 2020, 196, 749–763. [Google Scholar] [CrossRef]
- Echarti, A.; Hecht, M.; Buttner-Herold, M.; Haderlein, M.; Hartmann, A.; Fietkau, R.; Distel, L. CD8+ and Regulatory T cells Differentiate Tumor Immune Phenotypes and Predict Survival in Locally Advanced Head and Neck Cancer. Cancers 2019, 11, 1398. [Google Scholar] [CrossRef] [Green Version]
- Goldhirsch, A.; Winer, E.P.; Coates, A.S.; Gelber, R.D.; Piccart-Gebhart, M.; Thurlimann, B.; Senn, H.J.; Panel, m. Personalizing the treatment of women with early breast cancer: Highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann. Oncol. 2013, 24, 2206–2223. [Google Scholar] [CrossRef]
- Fasching, P.A.; Gass, P.; Haberle, L.; Volz, B.; Hein, A.; Hack, C.C.; Lux, M.P.; Jud, S.M.; Hartmann, A.; Beckmann, M.W.; et al. Prognostic effect of Ki-67 in common clinical subgroups of patients with HER2-negative, hormone receptor-positive early breast cancer. Breast Cancer Res. Treat. 2019, 175, 617–625. [Google Scholar] [CrossRef]


| Variables | Groups | |||||||
|---|---|---|---|---|---|---|---|---|
| Age (years) | mean: 59.1; | <50: 33 (22.6%); | ≥50: 113 (77.4%) | |||||
| T category | pT1mic: 10 (6.8%) | pT1a: 10 (6.8%) | pT1b: 38 (26%) | pT1c: 82 (56.2%) | pT2: 6 (4.1%) | |||
| N category | N0: 143 (97.9%) | N1: 3 (2.1%) | ||||||
| Stage | UICC I: 134 (91.8%) | UICC II: 12 (8.2%) | ||||||
| Tumour size (mm) | <10: 40 (27.4%); | 10–20: 96 (65.8%); | >20: 10 (6.8%) | |||||
| Histological grading | G1: 38 (26%) | G2: 101 (69.2%) | G3: 4 (2.7%) | n.a. 3 (2.1%) | ||||
| Histological typing | lobular: 23 (15.8%) | no special type: 100 (68.5%) | other: 23 (15.8%) | |||||
| Ki67 | <20: 110 (75.3%) | ≥20: 32 (21.9%) | n.a. 4 (2.7%) | |||||
| Oestrogen receptor status | positive: 141 (96.6%) | negative: 1 (0.7%) | n.a. 4 (2.7%) | |||||
| Progesterone receptor status | positive: 132 (90.4%) | negative: 11 (7.5%) | n.a. 3 (2.1%) | |||||
| Her2 status | positive: 8 (5.5%) | negative: 133 (91.1%) | n.a. 5 (3.4%) | |||||
| Molecular subtype | Luminal A: 97 (66.4%) | Luminal B: 44 (30.1%) | n.a. 5 (3.4%) | |||||
| Hormone therapy | Yes: 131 (89.7%) | No: 15 (10.3%) | ||||||
| Chemotherapy | Yes: 10 (6.8%) | No: 136 (93.2%) | ||||||
| CD20+ TIIC Densities Normal Tissue | CD20+ TIIC Densities | |||||
|---|---|---|---|---|---|---|
| Proximity | Distant | Central Tumour | Invasive Front | |||
| Correlation coefficient | CD4+ TIIC densities normal tissue (prox) | 0.139 | 0.080 | CD4+ TIIC densities central tumour | 0.521 | 0.193 |
| p | 0.192 | 0.459 | <0.001 | 0.038 | ||
| n | 90 | 89 | 122 | 116 | ||
| Correlation coefficient | CD4+ TIIC densities normal tissue (dist) | 0.126 | 0.081 | CD4+ TIIC densities invasive front | 0.179 | 0.397 |
| p | 0.248 | 0.373 | 0.049 | <0.001 | ||
| n | 86 | 123 | 121 | 128 | ||
| Stromal (n = 134) | Intraepithelial (n = 111) | ||||||
|---|---|---|---|---|---|---|---|
| N (total) | CD45RO+ Tiics Low | CD45RO+ TIICs High | p | CD45RO+ TIICs Low | CD45RO+ TIICs High | p | |
| Age (years) | 0.02 | 0.21 | |||||
| <50 | 33 | 1 (4%) | 26 (24%) | 16 (18%) | 6 (30%) | ||
| ≥50 | 113 | 25 (96%) | 82 (76%) | 75 (82%) | 14 (70%) | ||
| Tumour size (mm) | 0.43 | 0.49 | |||||
| <20 | 136 | 25 (96%) | 99 (92%) | 82 (90%) | 19 (95%) | ||
| ≥20 | 10 | 1 (4%) | 9 (8%) | 9 (10%) | 1 (5%) | ||
| Histological grading | 0.57 | 0.49 | |||||
| G1 | 38 | 6 (23%) | 30 (29%) | 24 (27%) | 7 (35%) | ||
| G2 + G3 | 105 | 20 (77%) | 75 (71%) | 64 (73%) | 13 (65%) | ||
| n.a. | 3 | ||||||
| Histological typing | 0.94 | 0.01 | |||||
| nonlobular | 123 | 22 (85%) | 92 (85%) | 83 (91%) | 14 (70%) | ||
| lobular | 23 | 4 (15%) | 16 (15%) | 8 (9%) | 6 (30%) | ||
| Ki67 | 0.38 | 0.43 | |||||
| <20 | 110 | 18 (72%) | 84 (80%) | 67 (77%) | 17 (85%) | ||
| ≥20 | 32 | 7 (28%) | 21 (20%) | 20 (23%) | 3 (15%) | ||
| n.a. | 4 | ||||||
| ER status | 0.62 | 0.63 | |||||
| neg | 1 | 0 (0%) | 1 (1%) | 1 (1%) | 0 (0%) | ||
| pos | 141 | 25 (100%) | 104 (99%) | 86 (99%) | 20 (100%) | ||
| n.a. | 4 | ||||||
| PR status | 0.13 | 0.47 | |||||
| neg | 11 | 4 (16%) | 7 (7%) | 9 (10%) | 1 (5%) | ||
| pos | 132 | 21 (84%) | 99 (93%) | 79 (90%) | 19 (95%) | ||
| n.a. | 3 | ||||||
| Her2 status | 0.67 | 0.94 | |||||
| neg | 133 | 23 (92%) | 99 (94%) | 83 (95%) | 19 (95%) | ||
| pos | 8 | 2 (8%) | 6 (6%) | 4 (5%) | 1 (5%) | ||
| n.a. | 5 | ||||||
| Subtype | 0.12 | 0.23 | |||||
| Luminal A | 97 | 14 (56%) | 75 (72%) | 57 (66%) | 16 (80%) | ||
| Luminal B | 44 | 11 (44%) | 29 (28%) | 29 (34%) | 4 (20%) | ||
| n.a. | 5 | ||||||
| Breast Cancer | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| Variable | Hazard Ratio | 95% C.I. | p | Hazard Ratio | 95% C.I. | p |
| Age (years) (<50 (n = 28) vs ≥50 (n = 118)) | 1.009 | 0.339–3.002 | 0.988 | --- | --- | --- |
| Stage (UICC I (n = 134) vs UICC II (n = 12 )) | 1.934 | 0.569–6.576 | 0.291 | --- | --- | --- |
| Tumour size (mm) (<20 (n = 136) vs ≥20 (n = 10)) | 1.379 | 0.321–5.927 | 0.666 | --- | --- | --- |
| Histological grading (G1 (n = 38) vs G2-3 (n = 105)) | 3.344 | 0.779–14.365 | 0.105 | 2.216 | 0.257–19.103 | 0.469 |
| Histological typing (nonlobular (n = 123) vs lobular (n = 23)) | 0.722 | 0.211–2.471 | 0.603 | --- | --- | --- |
| DCIS (no (n = 76) vs yes (n = 57)) | 0.798 | 0.312–2.04 | 0.637 | --- | --- | --- |
| Ki67 (<20 (n = 110) vs ≥20 (n = 32)) | 2.563 | 1.077–6.098 | 0.033 | 5.973 | 1.677–21.278 | 0.006 |
| Her2 status (negative (n = 133) vs positive (n = 8)) | 0.890 | 0.119–6.66 | 0.910 | --- | --- | --- |
| Luminal (A (n = 97) vs B (n = 44)) | 2.182 | 0.925–5.147 | 0.075 | 0.273 | 0.024–3.137 | 0.298 |
| Hormone therapy (no (n = 15) vs yes (n = 131)) | 1.003 | 0.234–4.309 | 0.996 | --- | --- | --- |
| Chemotherapy (no (n = 136) vs yes (n = 10 )) | 2.280 | 0.67–7.763 | 0.187 | --- | --- | --- |
| Stromal CD4+ TIICs (low (n = 59) vs high (n = 75)) | 2.712 | 0.899–8.185 | 0.077 | 1.250 | 0.154–10.156 | 0.834 |
| Intraepithelial CD4+ TIICs (low (n = 86) vs high (n = 25)) | 5.467 | 1.963–15.226 | 0.001 | 2.869 | 0.929–8.857 | 0.067 |
| Stromal CD45RO+ TIICs (low (n = 26) vs high (n = 108)) | 0.243 | 0.098–0.599 | 0.002 | 0.104 | 0.027–0.398 | 0.001 |
| Intraepithelial CD45RO+ TIICs (low (n = 91) vs high (n = 20)) | 0.854 | 0.191–3.816 | 0.836 | --- | --- | --- |
| Stromal CD1a+ TIICs (low (n = 106) vs high (n = 22)) | 0.545 | 0.124–2.398 | 0.422 | --- | --- | --- |
| Intraepithelial CD1a+ TIICs (low (n = 58) vs high (n = 47)) | 0.688 | 0.241 - 1.968 | 0.486 | --- | --- | --- |
| Stromal CD20+ TIICs (low (n = 76) vs high (n = 52)) | 2.535 | 0.935–6.872 | 0.067 | 11.601 | 2.485–54.152 | 0.002 |
| Intraepithelial CD20+ TIICs (low (n = 74) vs high (n = 31)) | 2.132 | 0.771–5.891 | 0.144 | 0.619 | 0.129–2.969 | 0.549 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schnellhardt, S.; Erber, R.; Büttner-Herold, M.; Rosahl, M.-C.; Ott, O.J.; Strnad, V.; Beckmann, M.W.; King, L.; Hartmann, A.; Fietkau, R.; et al. Tumour-Infiltrating Inflammatory Cells in Early Breast Cancer: An Underrated Prognostic and Predictive Factor? Int. J. Mol. Sci. 2020, 21, 8238. https://doi.org/10.3390/ijms21218238
Schnellhardt S, Erber R, Büttner-Herold M, Rosahl M-C, Ott OJ, Strnad V, Beckmann MW, King L, Hartmann A, Fietkau R, et al. Tumour-Infiltrating Inflammatory Cells in Early Breast Cancer: An Underrated Prognostic and Predictive Factor? International Journal of Molecular Sciences. 2020; 21(21):8238. https://doi.org/10.3390/ijms21218238
Chicago/Turabian StyleSchnellhardt, Sören, Ramona Erber, Maike Büttner-Herold, Marie-Charlotte Rosahl, Oliver J. Ott, Vratislav Strnad, Matthias W. Beckmann, Lillian King, Arndt Hartmann, Rainer Fietkau, and et al. 2020. "Tumour-Infiltrating Inflammatory Cells in Early Breast Cancer: An Underrated Prognostic and Predictive Factor?" International Journal of Molecular Sciences 21, no. 21: 8238. https://doi.org/10.3390/ijms21218238








