Interrelated Mechanism by Which the Methide Quinone Celastrol, Obtained from the Roots of Tripterygium wilfordii, Inhibits Main Protease 3CLpro of COVID-19 and Acts as Superoxide Radical Scavenger
Abstract
:1. Introduction
2. Results and Discussion
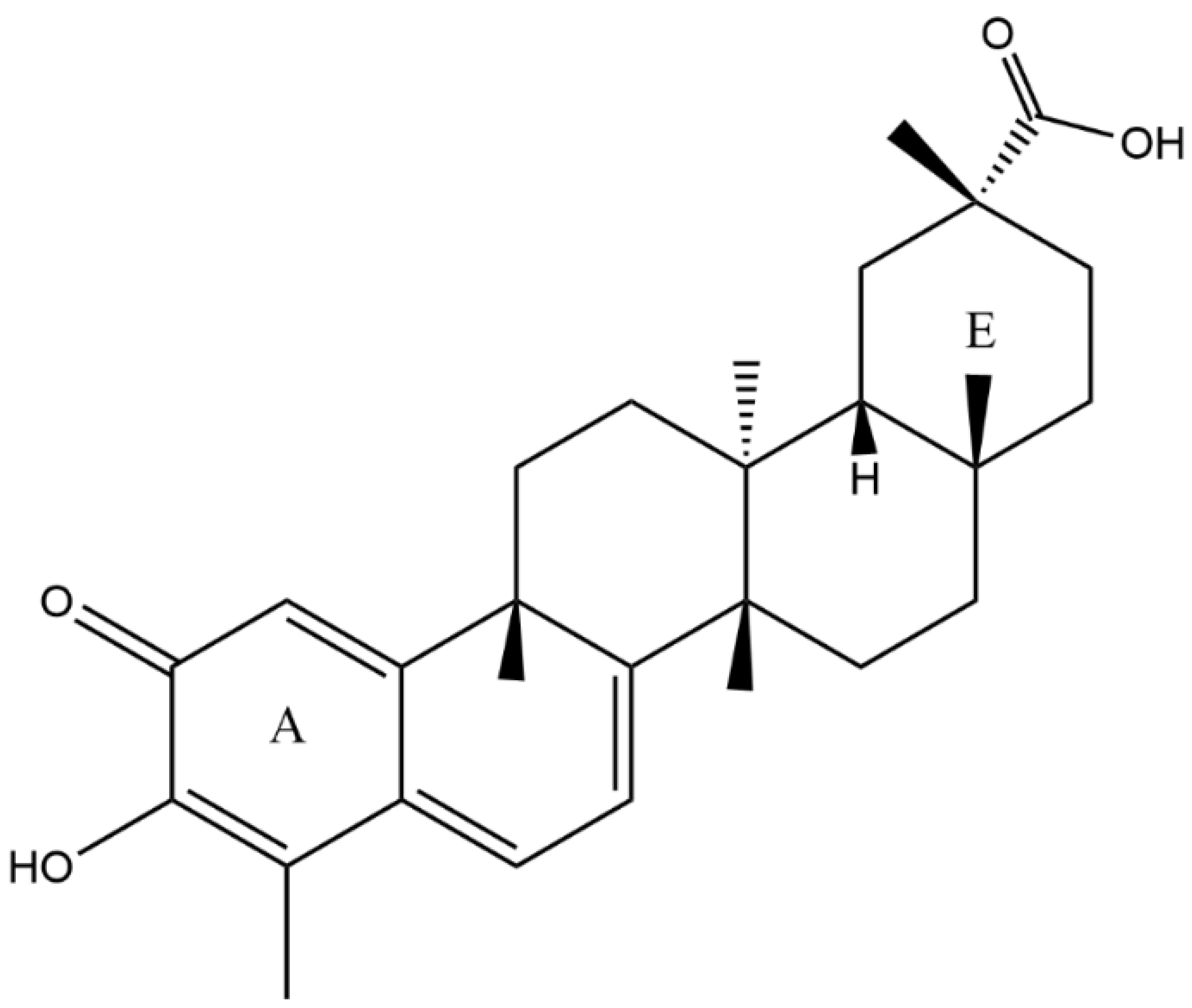
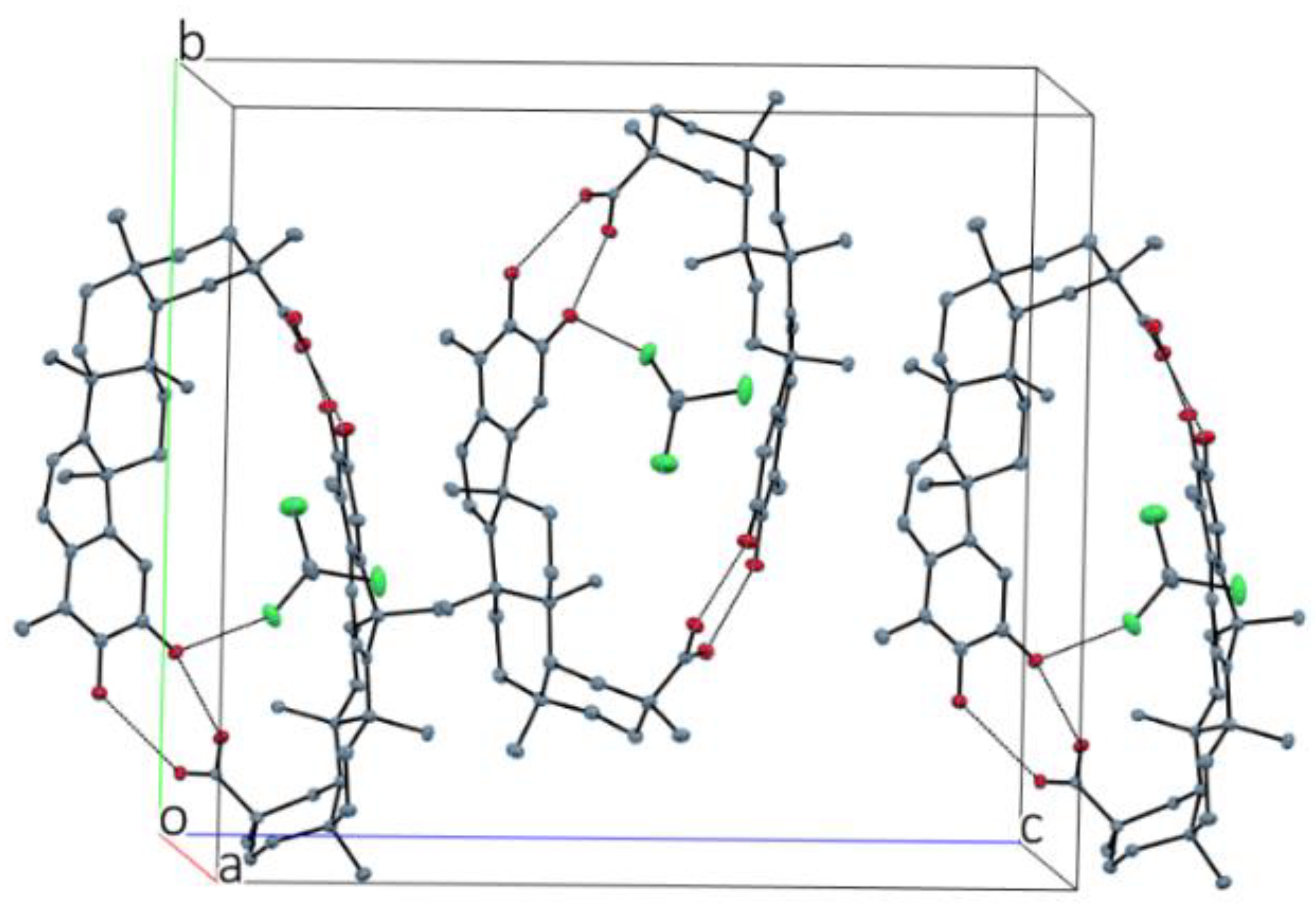
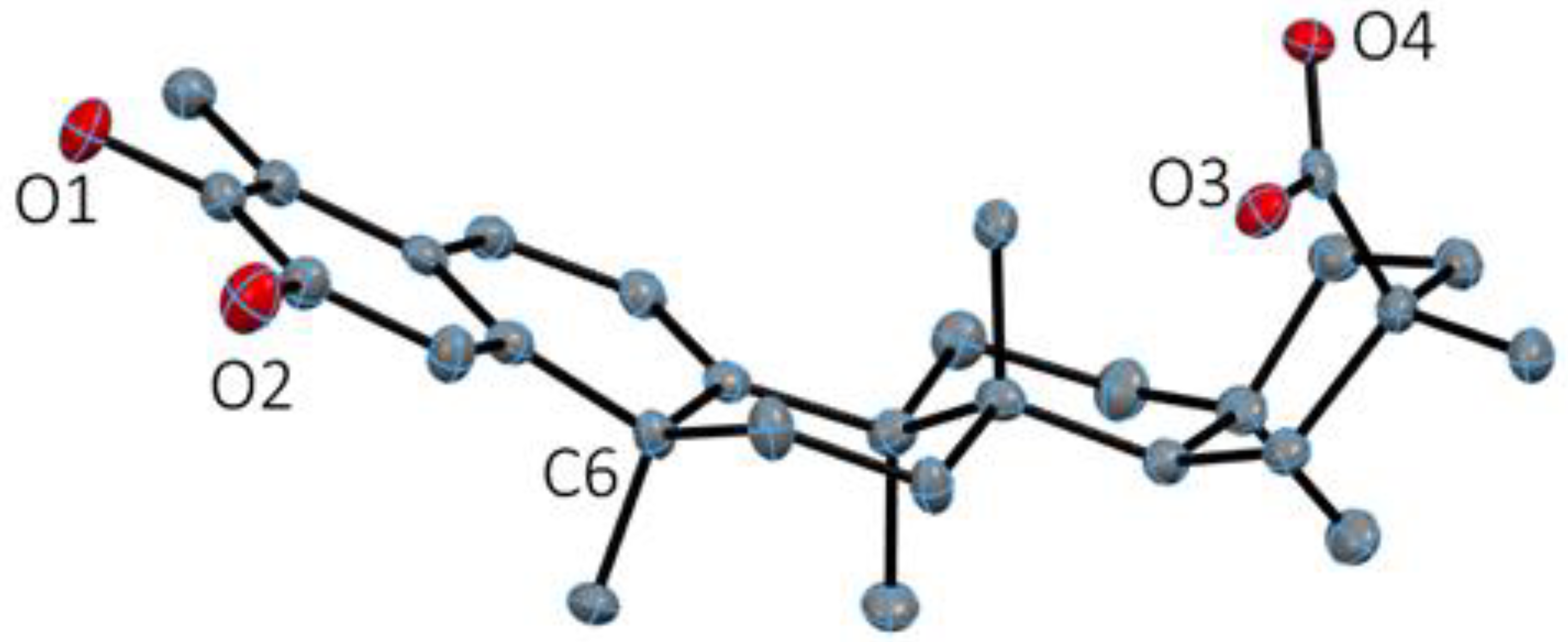
2.1. X-ray Diffraction
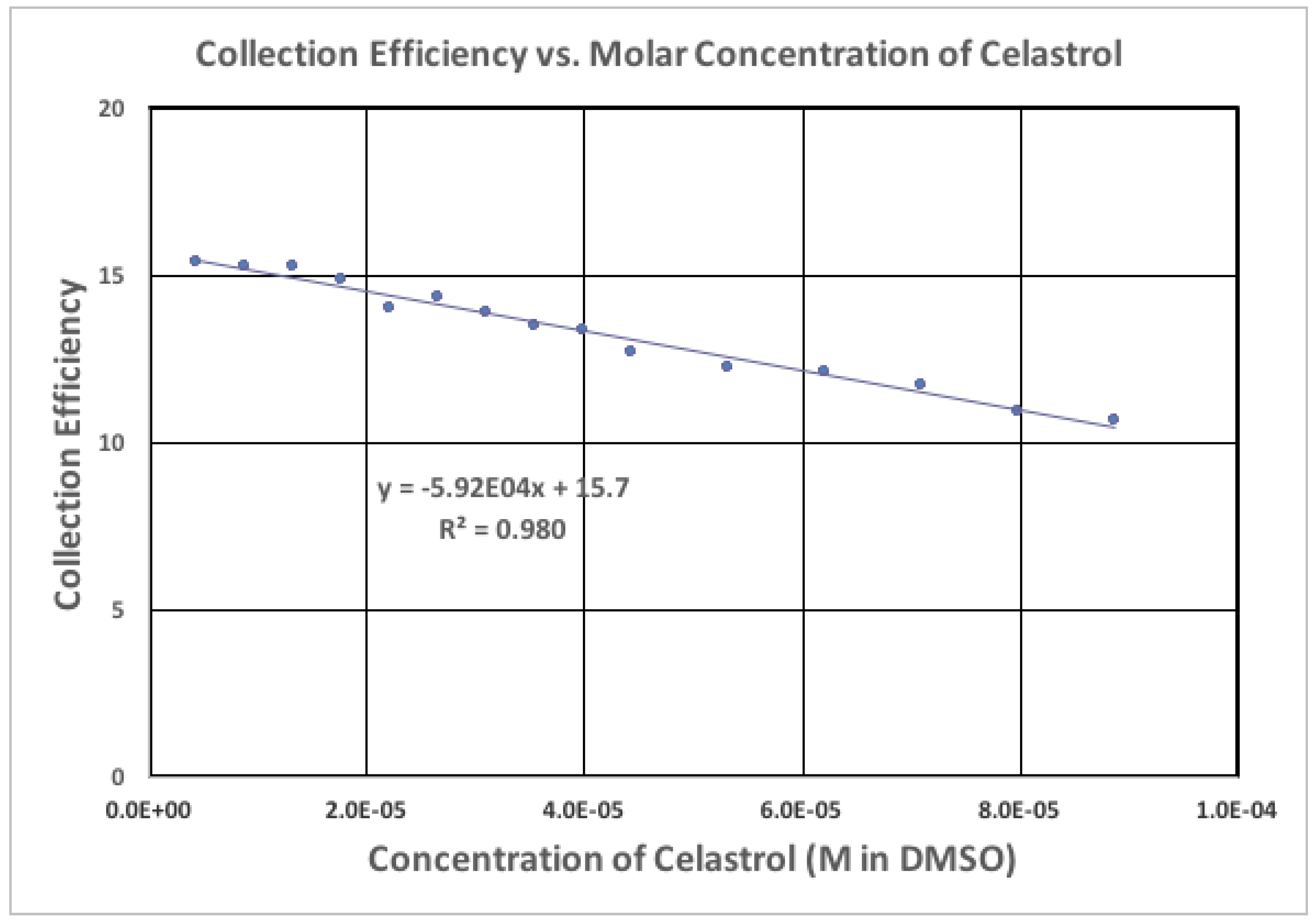
2.2. Electrochemistry

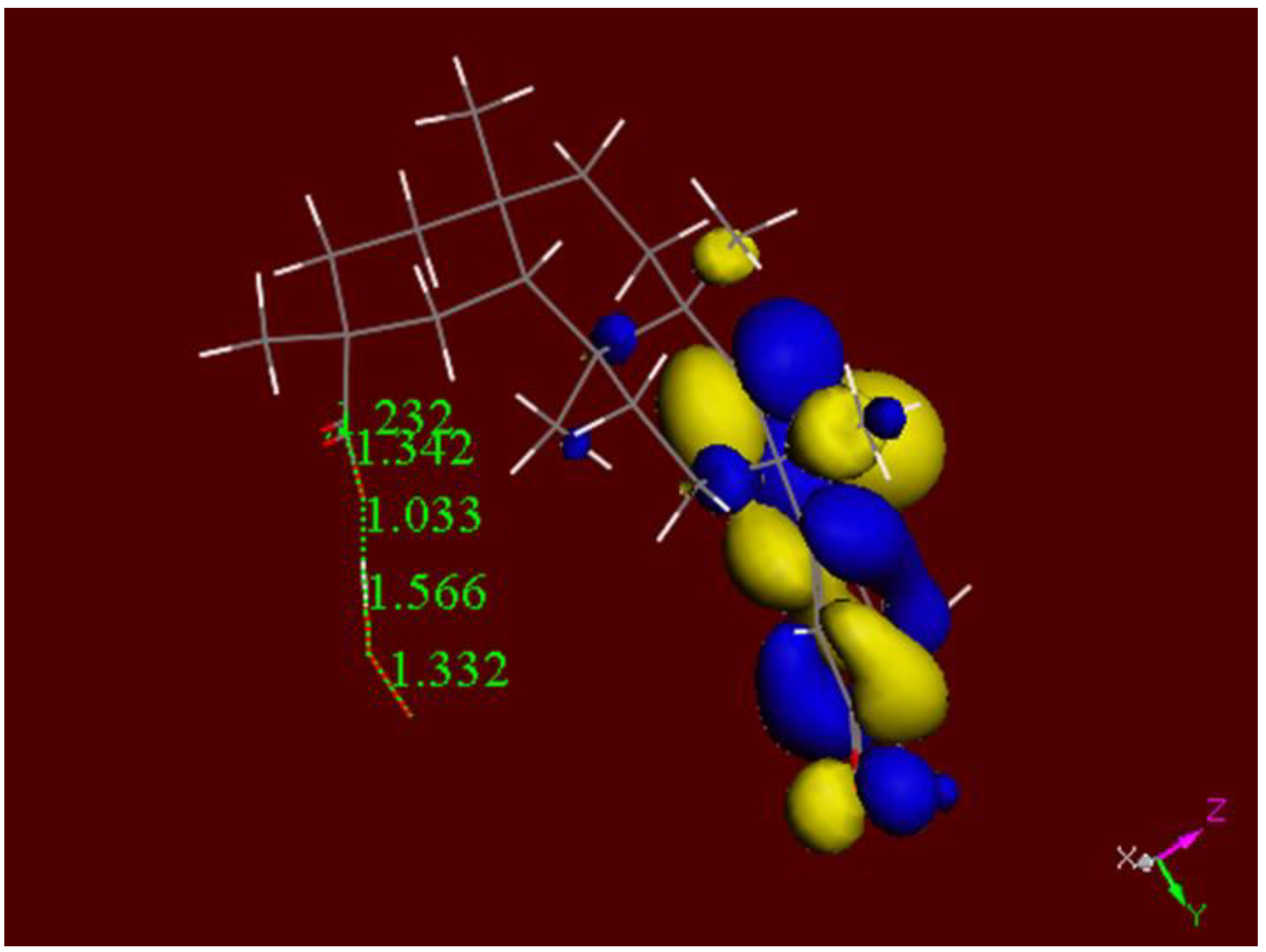
2.3. DFT Antioxidant Properties
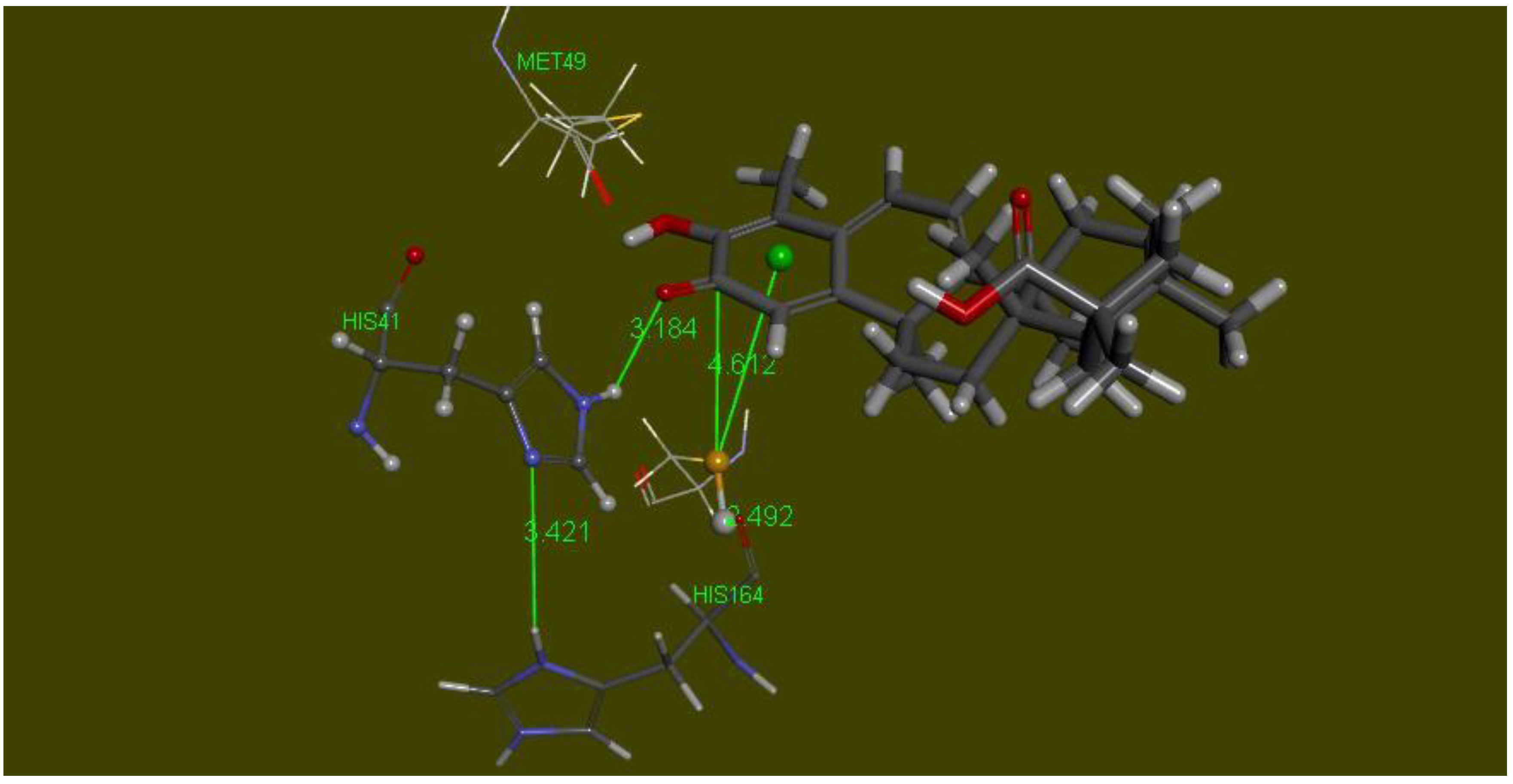
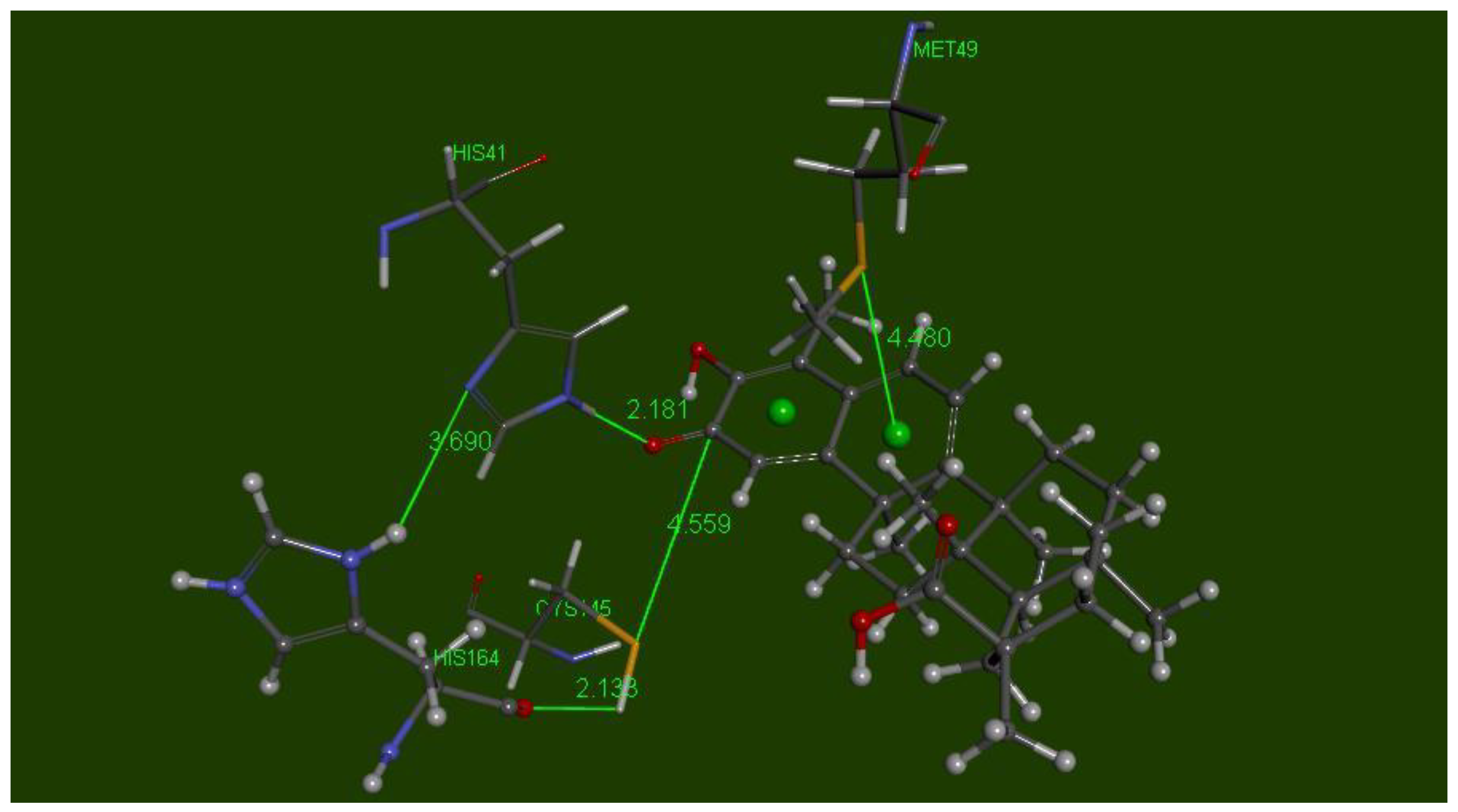
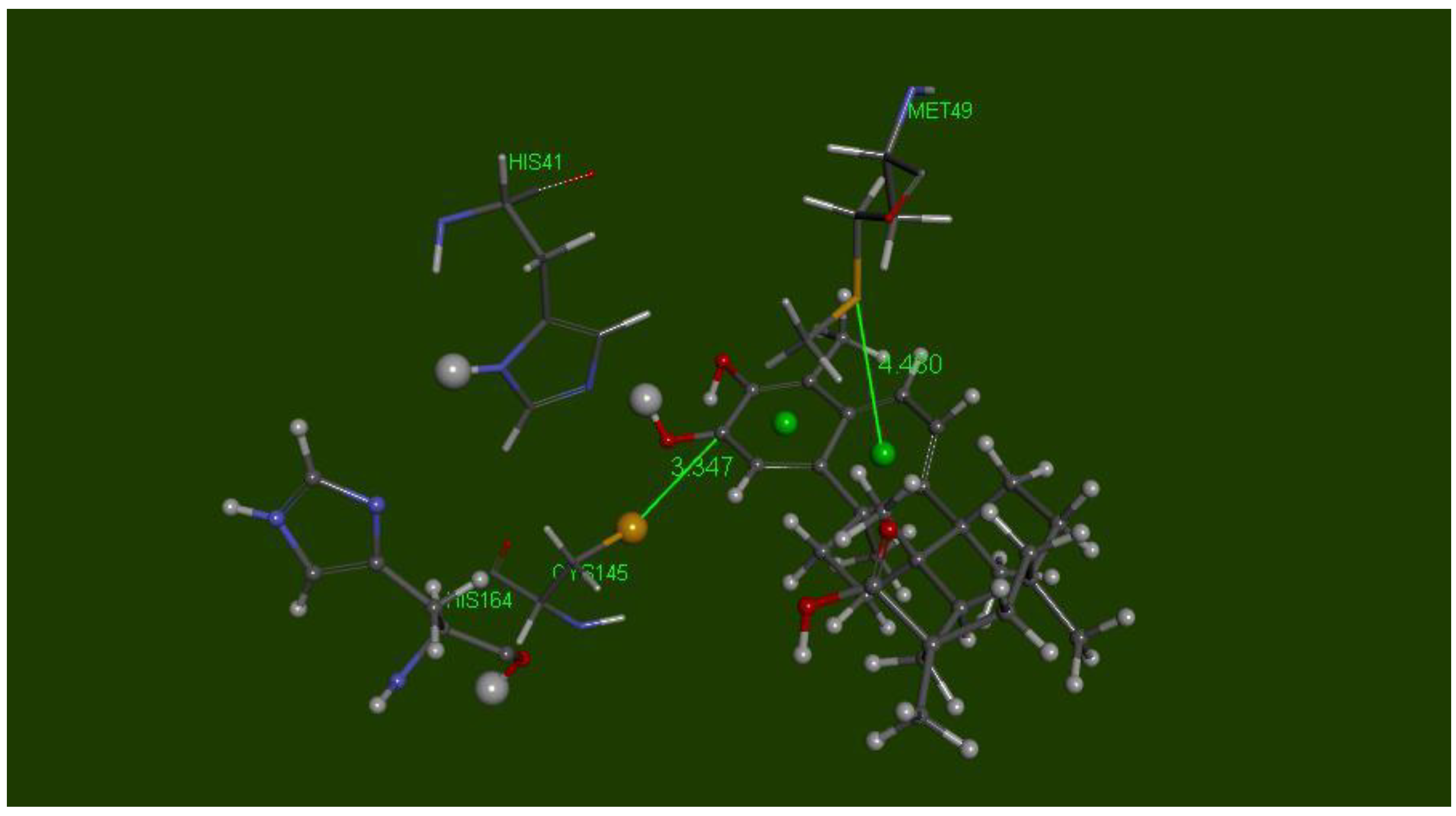
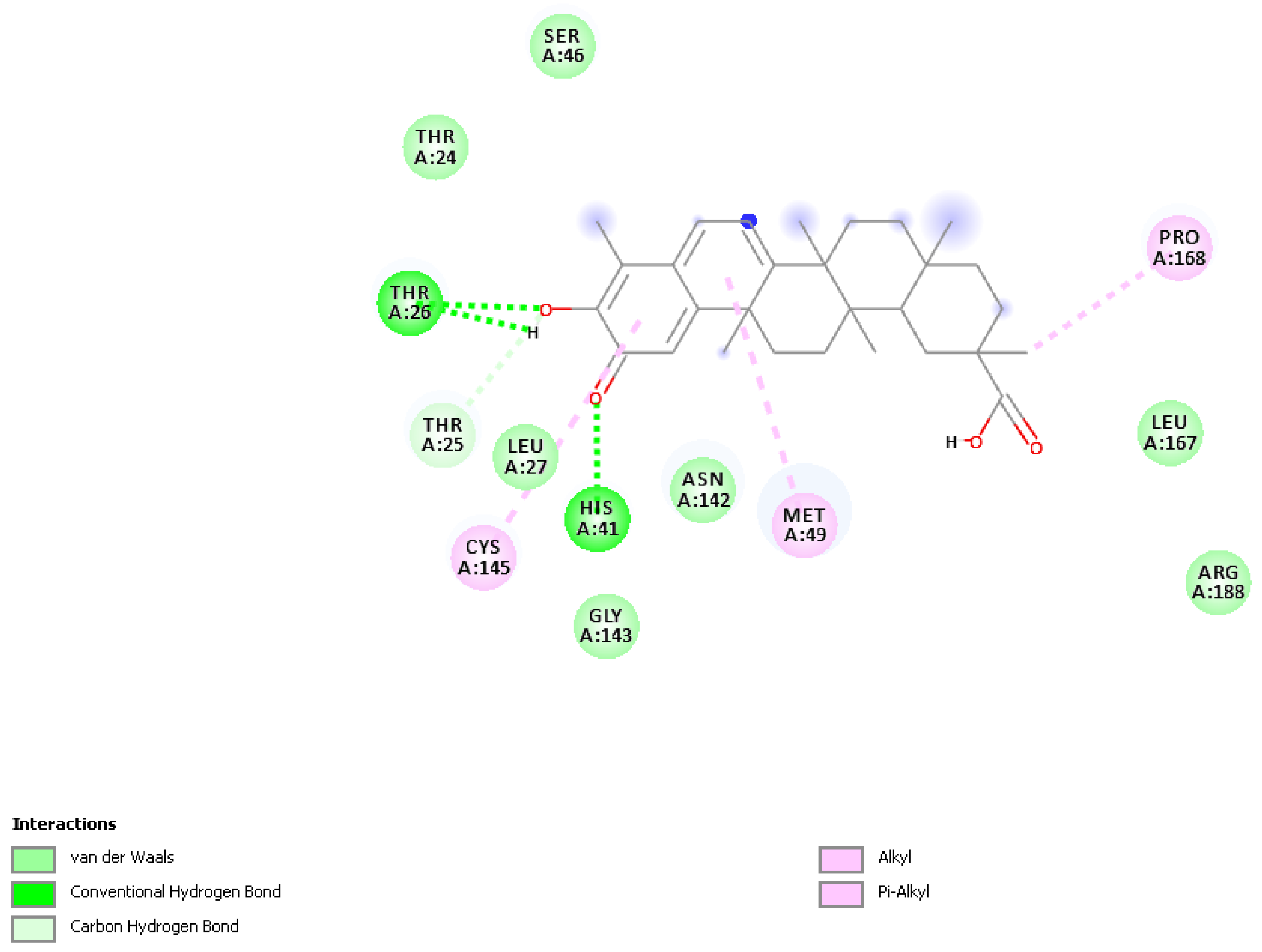
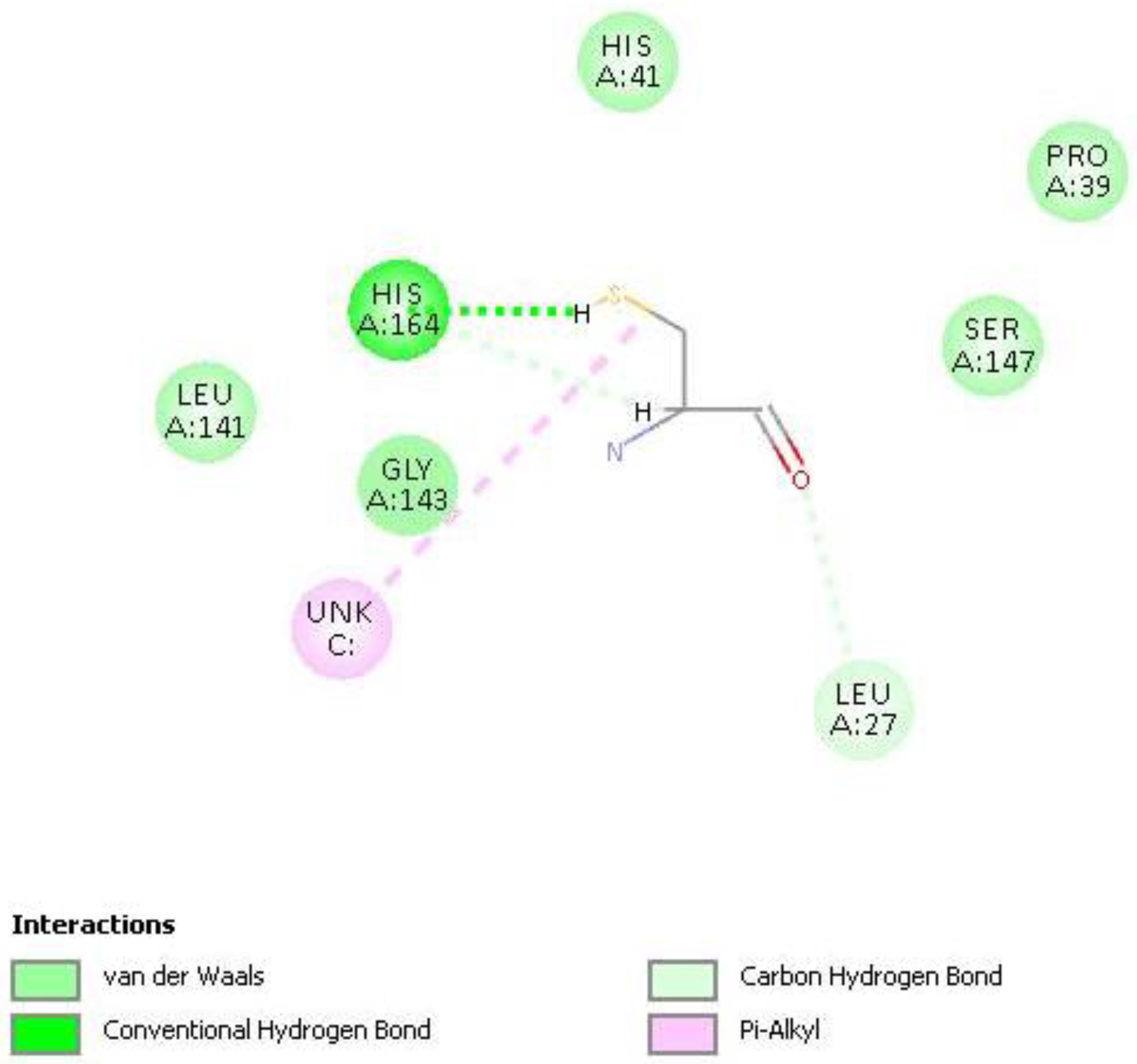
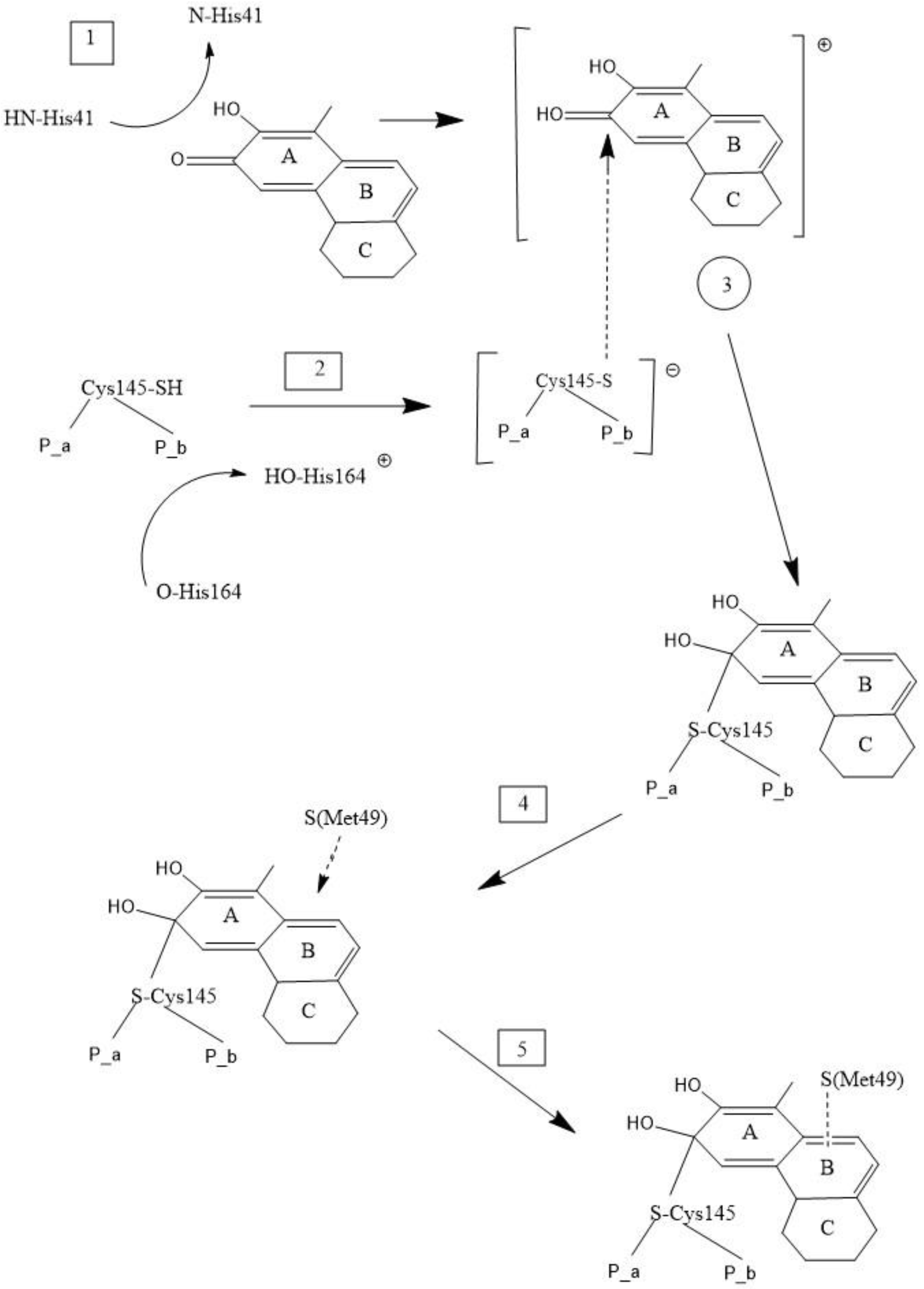
2.4. Docking the Protease COVID-19 3CLpro
3. Materials and Methods
3.1. Chemicals
3.2. Equipment
3.3. RRDE Study
3.4. Diffraction Study
3.5. Computational Study
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Setty, A.R.; Sigal, L.H. Herbal medications commonly used in the practice of rheumatology: Mechanisms of action, efficacy, and side effects. Semin. Arthritis Rheum. 2005, 34, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Chen, D.; Cui, Q.C.; Yuan, X.; Dou, Q.P. Celastrol, a Triterpene Extracted from the Chinese ‘Thunder of God Vine,’ Is a Potent Proteasome Inhibitor and Suppresses Human Prostate Cancer Growth in Nude Mice. Cancer Res. 2006, 66, 4758–4765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kannaiyan, R.; Muthu, K.S.; Gautam, S. Molecular Targets of Celastrol Derived from Thunder of God Vine: Potential Role in the Treatment of Inflammatory Disorders and Cancer. Cancer Lett. 2011, 303, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Sreeramulu, S.; Gande, S.L.; Goebel, M.; Schwalbe, H. Molecular mechanism of inhibition of the human protein complex hsp90–cdc37, a kinome chaperone–cochaperone, by triterpene celastrol. Angew. Chem. Int. Ed. 2009, 48, 5853–5855. [Google Scholar] [CrossRef]
- Hu, W.; Wang, L.; Du, G.; Guan, Q.; Dong, T.; Song, L.; Xia, Y.; Wang, X. Effects of Microbiota on the Treatment of Obesity with the Natural Product Celastrol in Rats. Diabetes Metab. J. 2020, 44, 747–763. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Liu, X.; Xiong, Q.; Shikano, S.; Li, M. Chronic inhibition of cardiac Kir2.1 and HERG potassium channels by celastrol with dual effects on both ion conductivity and protein trafficking. J. Biol. Chem. 2006, 281, 5877–5884. [Google Scholar] [CrossRef] [Green Version]
- Bai, J.P.; Shi, Y.L.; Fang, X.; Shi, Q.X. Effects of demethylzeylasteral and celastrol on spermatogenic cell Ca2+ channels and progesterone-induced sperm acrosome reaction. Eur. J. Pharmacol. 2003, 464, 9–15. [Google Scholar] [CrossRef]
- Aqil, F.; Kausar, H.; Agrawal, A.K.; Jeyabalan, J.; Kyakulaga, A.-H.; Munagala, R.; Gupta, R. Exosomal formulation enhances therapeutic response of celastrol against lung cancer. Exp. Mol. Pathol. 2016, 101, 12–21. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, T.; Zhou, X.; Liu, H.; Sun, H.; Ma, Z.; Wu, B. Enhancement of oral bioavailability of tripterine through lipid nanospheres: Preparation, characterization, and absorption evaluation. J. Pharm. Sci. 2014, 103, 1711–1719. [Google Scholar] [CrossRef]
- Sanna, V.; Chamcheu, J.C.; Pala, N.; Mukhtar, H.; Sechi, M.; Siddiqui, I.A. Nanoencapsulation of natural triterpenoid celastrol for prostate cancer treatment. Int. J. Nanomed. 2015, 10, 6835–6846. [Google Scholar] [CrossRef] [Green Version]
- Wolfram, J.; Suri, K.; Huang, Y.; Molinaro, R.; Borsoi, C.; Scott, B.; Boom, K.; Paolino, D.; Fresta, M.; Wang, J.; et al. Evaluation of anticancer activity of celastrol liposomes in prostate cancer cells. J. Microencapsul. 2014, 31, 501–507. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; Zhou, D.; Hang, T.; Wu, Z.; Liu, J.; Xu, Q.; Xie, X.; Zuo, J.; Wang, Z.; Zhou, Y. Preparation, characterization, and assessment of the antiglioma effects of liposomal celastrol. Anticancer Drugs 2012, 23, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Wang, J.; Song, H.; Cui, D.; Li, L.; Li, J.; Lin, L.; Zhou, J.; Liu, Y. Optimized preparation of celastrol-loaded polymeric nanomicelles using rotatable central composite design and response surface methodology. J. Biomed. Nanotechnol. 2012, 8, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wu, X.; Li, J.; Yao, L.; Sun, L.; Shi, Y.; Zhang, W.; Lin, J.; Liang, D.; Li, Y. Antitumor activity of celastrol nanoparticles in a xenograft retinoblastoma tumor model. Int. J. Nanomed. 2012, 7, 2389–2398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niemela, E.; Desai, D.; Nkizinkiko, Y.; Eriksson, J.E.; Rosenholm, J.M. Sugardecorated mesoporous silica nanoparticles as delivery vehicles for the poorly soluble drug celastrol enables targeted induction of apoptosis in cancer cells. Eur. J. Pharm. Biopharm. 2015, 96, 11–21. [Google Scholar] [CrossRef]
- Qi, X.; Qin, J.; Ma, N.; Chou, X.; Wu, Z. Solid self-microemulsifying dispersible tablets of celastrol: Formulation development, characterization and bioavailability evaluation. Int. J. Pharm. 2014, 472, 40–47. [Google Scholar] [CrossRef]
- Lawrence, T. The Nuclear Factor NF-κB Pathway in Inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.-H.; Koo, T.H.; Yoon, H.-J.; Haeng, S.; Jin, H.Z.; Lee, K.; Hong, Y.-S.; Lee, J.J. Inhibition of NF-κB activation through targeting iκb kinase by celastrol, a quinone methide triterpenoid. Biochem. Pharmacol. 2006, 72, 1311–1321. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, W.; Wang, M.; Xiao, Y.; Zhang, X.; Zhang, H.; Tong, X. Competitive profiling of celastrol targets in human cervical cancer HeLa cells via quantitative chemical proteomics. Mol. BioSyst. 2016, 13, 83–91. [Google Scholar] [CrossRef]
- Salminen, A.; Lehtonen, M.; Paimela, T.; Kaarniranta, K. Celastrol: Molecular targets of Thunder God Vine. Biochem. Biophys. Res. Commun. 2010, 394, 439–442. [Google Scholar] [CrossRef]
- Peng, B.; Xu, L.; Cao, F.; Wei, T.; Yang, C.; Uzan, G.; Zhang, D. HSP90 inhibitor, celastrol, arrests human monocytic leukemia cell U937 at G0/G1 in thiol-containing agents reversible way. Mol. Cancer 2010, 9, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, H.R.; Seo, W.D.; Pyun, B.-J.; Lee, B.W.; Jin, Y.B.; Park, K.H.; Seo, E.-K.; Lee, Y.-J.; Lee, Y.-S. Radiosensitization by celastrol is mediated by modification of antioxidant thiol molecules. Chem. Biol. Interact. 2011, 193, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Boridy, S.; Le, P.U.; Petrecca, K.; Maysinger, D. Celastrol targets proteostasis and acts synergistically with a heat-shock protein 90 inhibitor to kill human glioblastoma cells. Cell Death Dis. 2014, 5, e1216. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Lee, B.H.; Kim, N.D.; Lee, J.Y. Celastrol blocks binding of lipopolysaccharides to a Toll-like receptor4/myeloid differentiation factor2 complex in a thiol-dependent manner. J. Ethnopharmacol. 2015, 172, 254–260. [Google Scholar] [CrossRef]
- Trott, A.; West, J.D.; Klaic, L.; Westerheide, S.D.; Silverman, R.B.; Morimoto, R.I.; Morano, K.A. Activation of heat shock and antioxidant responses by the natural product celastrol: Transcriptional signatures of a thiol-targeted molecule. Mol. Biol. Cell 2008, 19, 1104–1112. [Google Scholar] [CrossRef] [Green Version]
- Bittner, S. When quinones meet amino acids: Chemical, physical and biological consequences. Amino Acids 2006, 30, 205–224. [Google Scholar] [CrossRef]
- Caruso, F.; Rossi, M.; Pedersen, J.Z.; Incerpi, S. Computational studies reveal mechanism by which quinone derivatives can inhibit SARS-CoV-2. Study of embelin and two therapeutic compounds of interest, methyl prednisolone and dexamethasone. J. Infect. Public Health 2020, in press. Available online: https://www.sciencedirect.com/science/article/pii/S1876034120306717 (accessed on 3 December 2020). [CrossRef]
- Ryu, Y.B.; Park, S.-J.; Kim, Y.M.; Lee, J.-Y.; Seo, W.D.; Chang, J.S.; Park, K.H.; Rho, M.-C.; Lee, W.S. SARS-CoV 3CLpro inhibitory effects of quinone-methide triterpenes from Tripterygium regelii. Bioorg. Med. Chem. Lett. 2010, 20, 1873–1876. [Google Scholar] [CrossRef]
- Zhu, J.; Xu, S.; Li, X.; Wang, J.; Jiang, Y.; Hu, W.; Ruan, W. Infectious bronchitis virus inhibits activation of the TLR7 pathway, but not the TLR3 pathway. Arch. Virol. 2020, 165, 2037–2043. [Google Scholar] [CrossRef]
- Khalili, N.; Karimi, A.; Moradi, M.-T.; Shirzad, H. In vitro immunomodulatory activity of celastrol against influenza A virus infection. Immunopharmacol. Immunotoxicol. 2018, 40, 250–255. [Google Scholar] [CrossRef]
- Tseng, C.-K.; Hsu, S.-P.; Lin, C.-K.; Wu, Y.-H.; Lee, J.-C.; Young, K.-C. Celastrol inhibits hepatitis C virus replication by upregulating heme oxygenase-1 via the JNK MAPK/Nrf2 pathway in human hepatoma cells. Antivir. Res. 2017, 146, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-S.; Tseng, C.-K.; Lin, C.-K.; Hsu, Y.-C.; Wu, Y.-H.; Hsieh, C.-L.; Lee, J.-C. Celastrol inhibits dengue virus replication via up-regulating type I interferon and downstream interferon-stimulated responses. Antivir. Res. 2017, 137, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Narayan, V.; Ravindra, K.C.; Chiaro, C.; Cary, D.; Aggarwal, B.B.; Henderson, A.J.; Prabhu, K.S. Celastrol Inhibits Tat-Mediated Human Immunodeficiency virus (HIV) Transcription and Replication. J. Mol. Biol. 2011, 410, 972–983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caruso, F.; Rossi, M.; Kaur, S.; Garcia-Villar, E.; Molasky, N.; Belli, S.; Sitek, J.D.; Gionfra, F.; Pedersen, J.Z.; Incerpi, S. Antioxidant properties of embelin in cell culture. electrochemistry and theoretical mechanism of scavenging. Potential scavenging of superoxide radical through the membrane cell. Antioxidants 2020, 5, 382. [Google Scholar] [CrossRef] [PubMed]
- Caruso, F.; Paumier, S.; Rossi, M. X-ray Crystal Structure of Embelin and its DFT Scavenging of Superoxide Radical. J. Comput. Chem. 2018, 39, 1143–1148. [Google Scholar] [CrossRef] [PubMed]
- Ledford, H. Coronavirus breakthrough: Dexamethasone is first drug shown to save lives. Nature 2020, 582, 469. [Google Scholar] [CrossRef]
- Caruso, F.; Pedersen, J.Z.; Incerpi, S.; Rossi, M. Molecular mechanism for inhibition of the main Covid-19 protease by its inhibitor emodin and corticosteroids is mediated by Cys145. Molecules. Submitted.
- Fadel, R.; Morrison, A.R.; Vahia, A.; Smith, Z.R.; Chaudhry, Z.; Bhargava, P.; Miller, J.; Kenney, R.M.; Alangaden, G.; Ramesh, M.S.; et al. Early short course corticosteroids in hospitalized patients with COVID-19. Clin. Infect. Dis. 2020, 71, 2114–2120. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Cryst. 2016, B72, 171–179. [Google Scholar] [CrossRef]
- Zha, J.; Zhang, Q.; Li, M.; Wang, J.R.; Mei, X. Improving Dissolution Properties by Polymers and Surfactants: A Case Study of Celastrol. J. Pharm. Sci. 2018, 107, 2860–2868. [Google Scholar] [CrossRef]
- Belli, S.; Rossi, M.; Molasky, N.; Middleton, L.; Caldwell, C.; Bartow-McKenney, C.; Duong, M.; Chiu, J.; Gibbs, E.; Caldwell, A.; et al. Effective and Novel Application of Hydrodynamic Voltammetry to the Study of Superoxide Radical Scavenging by Natural Phenolic Antioxidants. Antioxidants 2019, 8, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossi, M.; Wen, K.; Caruso, F.; Belli, S. Emodin scavenging of superoxide radical. X-ray crystal structure, hydrodynamic voltammetry and theoretical studies. Antioxidants 2020, 9, 194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, Z.; Du, X.; Xu, Y.; Deng, Y.; Liu, M.; Zhao, Y.; Zhang, B.; Li, X.; Zhang, L.; Peng, C.; et al. Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature 2020, 582, 289–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Habtemariam, S.; Nabavi, S.F.; Berindan-Neagoe, I.; Cismaru, C.A.; Izadi, M.; Sureda, A.; Nabavi, S.M. Should we try the antiinflammatory natural product, celastrol, for COVID-19? Phytother. Res. 2020, 34, 1189–1190. [Google Scholar] [CrossRef] [PubMed]
- Mayrhofer, K.; Strmcnik, D.; Blizanac, B.; Stamenkovic, V.; Arenz, M.; Markovic, N. Measurement of oxygen reduction activities via the rotating disc electrode method: From Pt model surfaces to carbon-supported high surface area catalysts. Electrochim. Acta 2008, 53, 3181–3188. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef]
- Delley, B.J. From molecules to solids with the DMol3 approach. J. Chem. Phys. 2000, 113, 7756–7764. [Google Scholar] [CrossRef]
- Perdew, J.P.; Chevary, J.A.; Vosko, S.H.; Jackson, K.A.; Pederson, M.R.; Singh, D.J.; Fiolhais, C. Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. 1992, 46, 6671–6687. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Perdew, J.P.; Wang, Y. Accurate and simple analytic representation of the electron-gas correlation energy. Phys. Rev. B 1992, 45, 13244–13249. [Google Scholar] [CrossRef]
- Wu, G.; Robertson, D.H.; Brooks, C.L.; Vieth, M.J. Detailed analysis of grid-based molecular docking: A case study of CDOCKER-A CHARMm-based MD docking algorithm. J. Comput. Chem. 2003, 24, 1549–1562. [Google Scholar] [CrossRef] [PubMed]

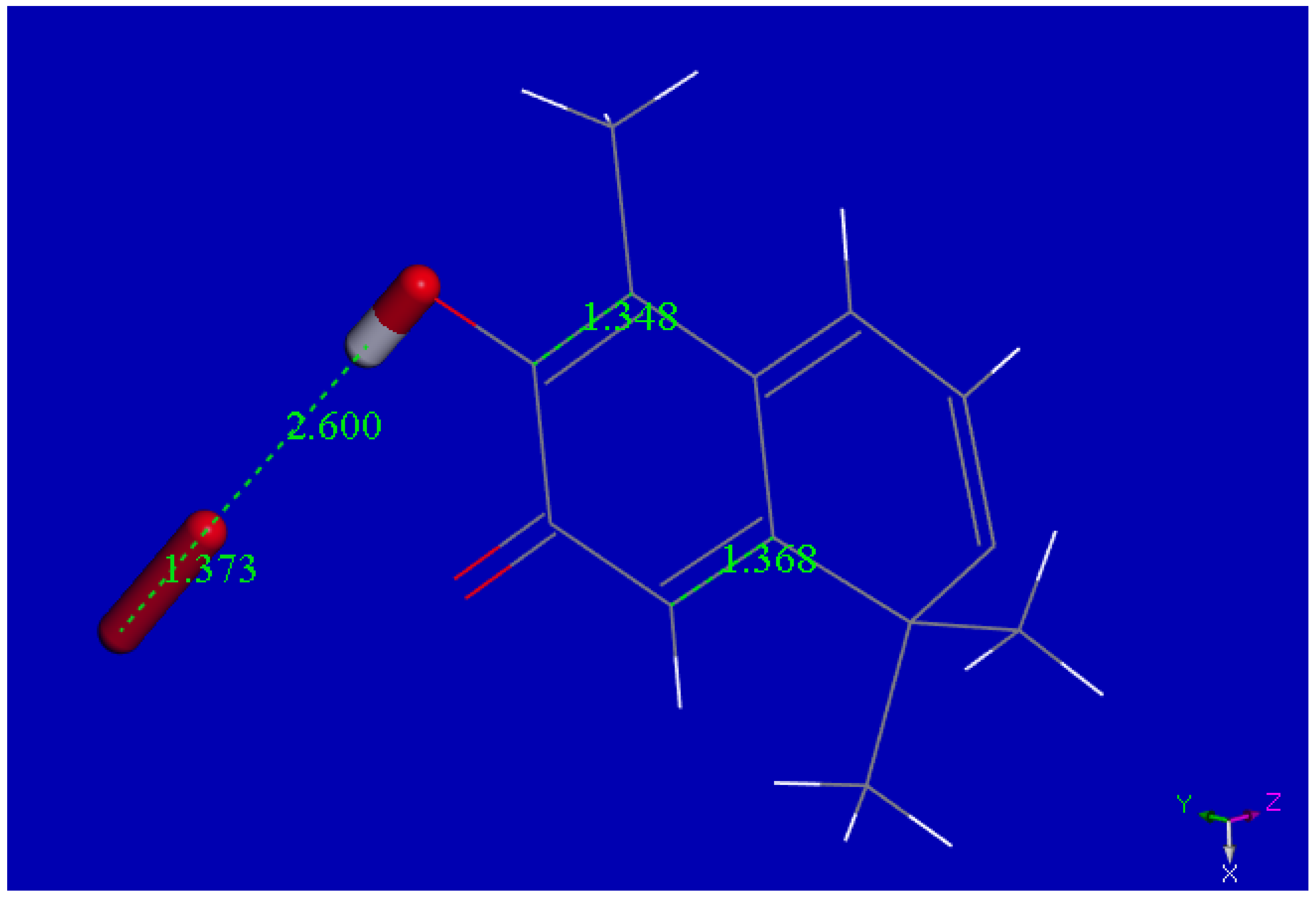

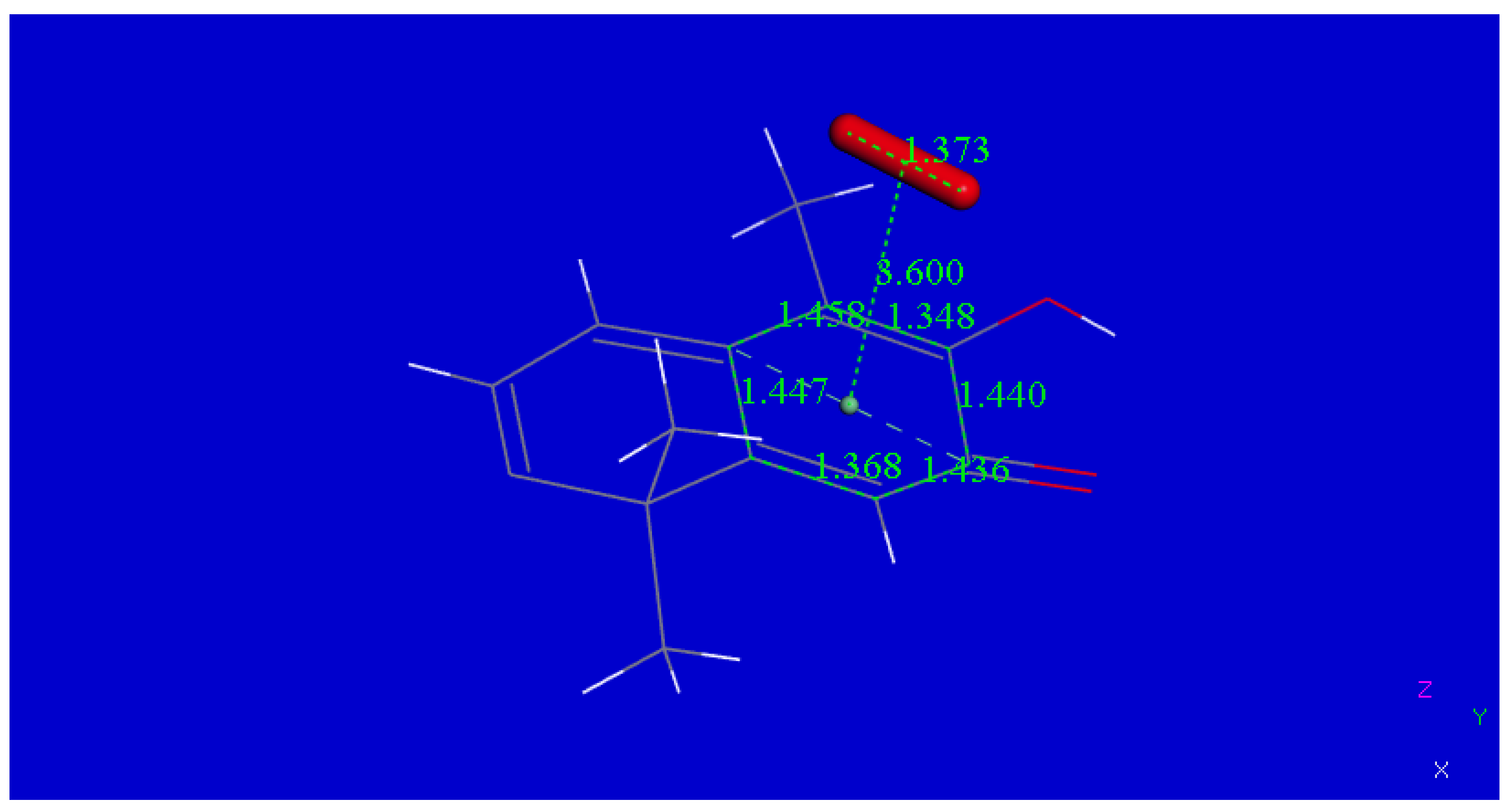
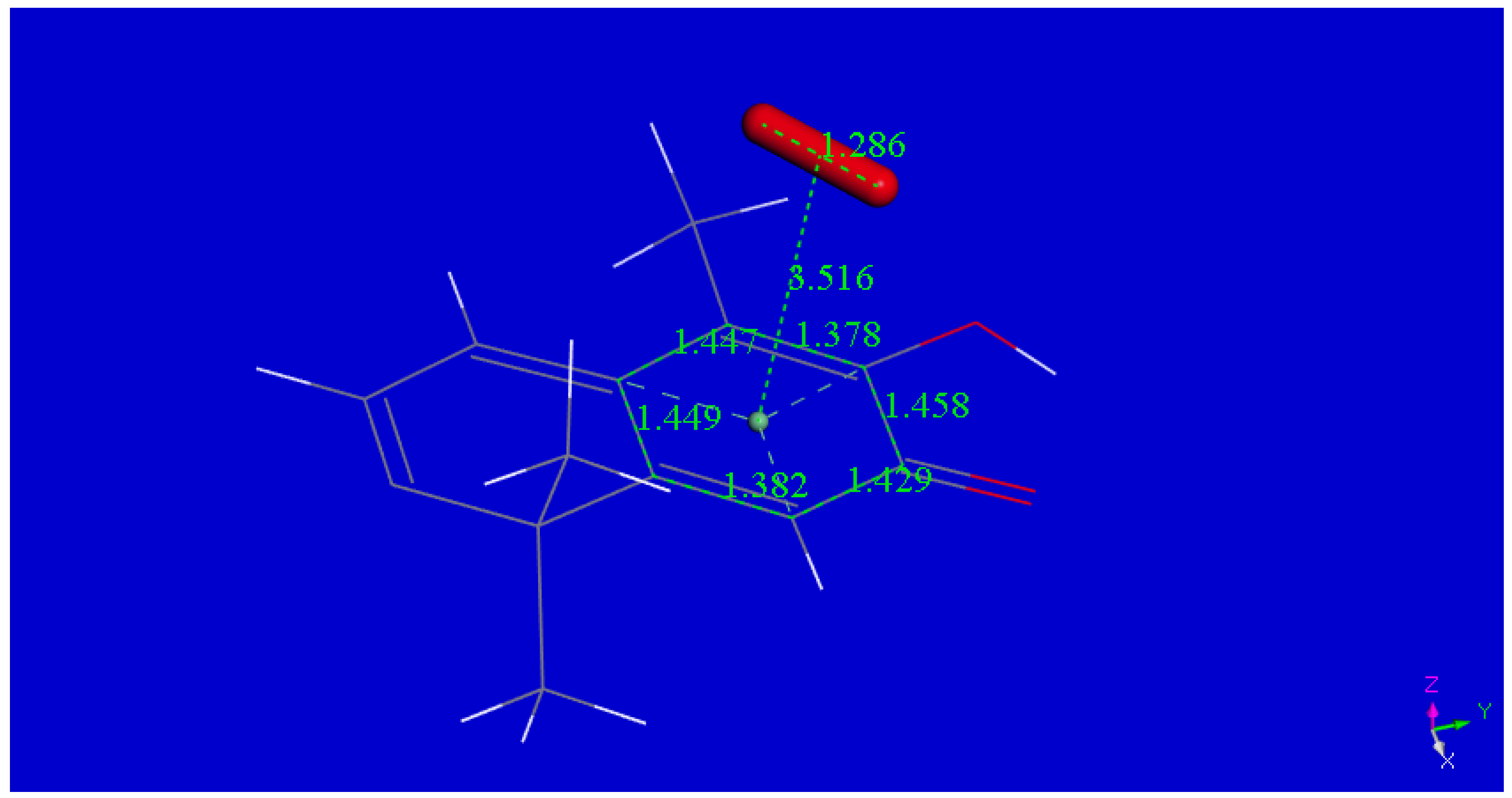
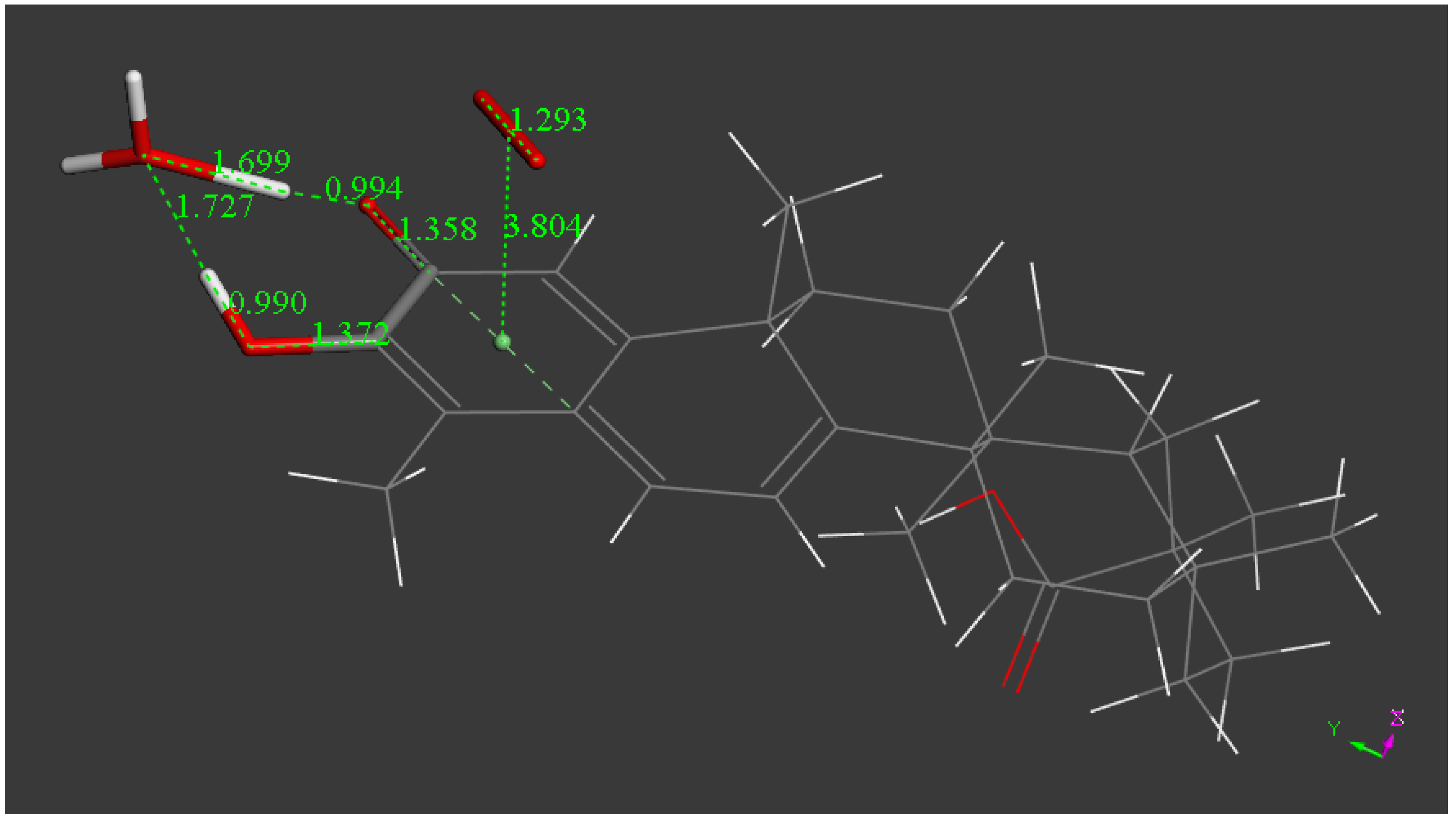

| Chemical formula | 2(C29H38O4) (CHCl3 solvent) | |
| Formula weight | 450.6 g/mol | |
| Temperature | 125(2) K | |
| Wavelength | 1.54178 Å | |
| Crystal system | orthorhombic | |
| Space group | P 21 21 21 | |
| Unit cell dimensions | a = 12.3174(19) Å | |
| b = 19.764(4) Å | α = 90° | |
| c = 22.017(3) Å | β = 90° | |
| Volume | 5359.8(17) Å3 | γ = 90° |
| Z | 8 | |
| Density (calculated) | 1.265 g/cm3 | |
| Absorption coefficient | 1.978 mm−1 | |
| F(000) | 2184 | |
| Theta range for data collection | 3.00 to 68.23° | |
| Index ranges | −14 ≤ h ≤ 14, −23 ≤ k ≤ 23, −25 ≤ l ≤ 26 | |
| Reflections collected | 60772 | |
| Independent reflections | 9745 [R(int) = 0.1955] | |
| Coverage of independent reflections | 99.7% | |
| Absorption correction | Multi-scan | |
| Refinement method | Full-matrix least-squares on F2 | |
| Refinement program | SHELXL-2014/6 (Sheldrick, 2014) | |
| Function minimized | Σ w(Fo2 − Fc2)2 | |
| Data/restraints/parameters | 9745/0/649 | |
| Goodness-of-fit on F2 | 1.059 | |
| Final R indices | 7424 data; I > 2σ(I) | R1 = 0.0641, wR2 = 0.1465 |
| all data | R1 = 0.0896, wR2 = 0.1653 | |
| Weighting scheme | w = 1/[σ2(Fo2)] where P = (Fo2 + 2Fc2)/3 | |
| Extinction coefficient | 0.0018(3) | |
| Largest diff. peak and hole | 0.421 and −0.484 eÅ−3 | |
| R.M.S. deviation from mean | 0.101 eÅ−3 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caruso, F.; Singh, M.; Belli, S.; Berinato, M.; Rossi, M. Interrelated Mechanism by Which the Methide Quinone Celastrol, Obtained from the Roots of Tripterygium wilfordii, Inhibits Main Protease 3CLpro of COVID-19 and Acts as Superoxide Radical Scavenger. Int. J. Mol. Sci. 2020, 21, 9266. https://doi.org/10.3390/ijms21239266
Caruso F, Singh M, Belli S, Berinato M, Rossi M. Interrelated Mechanism by Which the Methide Quinone Celastrol, Obtained from the Roots of Tripterygium wilfordii, Inhibits Main Protease 3CLpro of COVID-19 and Acts as Superoxide Radical Scavenger. International Journal of Molecular Sciences. 2020; 21(23):9266. https://doi.org/10.3390/ijms21239266
Chicago/Turabian StyleCaruso, Francesco, Manrose Singh, Stuart Belli, Molly Berinato, and Miriam Rossi. 2020. "Interrelated Mechanism by Which the Methide Quinone Celastrol, Obtained from the Roots of Tripterygium wilfordii, Inhibits Main Protease 3CLpro of COVID-19 and Acts as Superoxide Radical Scavenger" International Journal of Molecular Sciences 21, no. 23: 9266. https://doi.org/10.3390/ijms21239266







