Defining Dry Eye from a Clinical Perspective
Abstract
1. Introduction and Existing Definitions of Dry Eye Disease (DED)
Clinical Implications
- The range of definitions has contributed to confusion and a lack of consistent diagnosis of DED across the globe.
- Tear film stability is a sensitive measure of tear dysfunction that can be easily measured, making it a clinically practical and reproducible marker of DED/tear dysfunction. It should be the key criterion in a clinical definition of DED.
2. A Proposed New Clinical Definition of Dry Eye Disease
Clinical Implications
- The proposed new definition of DED is based on a consensus of agreement between DED experts in different regions worldwide.
- The new definition is aimed at providing practitioners with a practical tool that helps diagnose DED in the clinic.
- Using this simple clinical definition, all practitioners can make a precise and consistent diagnosis of DED.
- As the diagnosis can be made using the DED questionnaire and a slit lamp examination with fluorescein, all practitioners can easily apply this definition in their clinics following the recommendations outlined in this article.
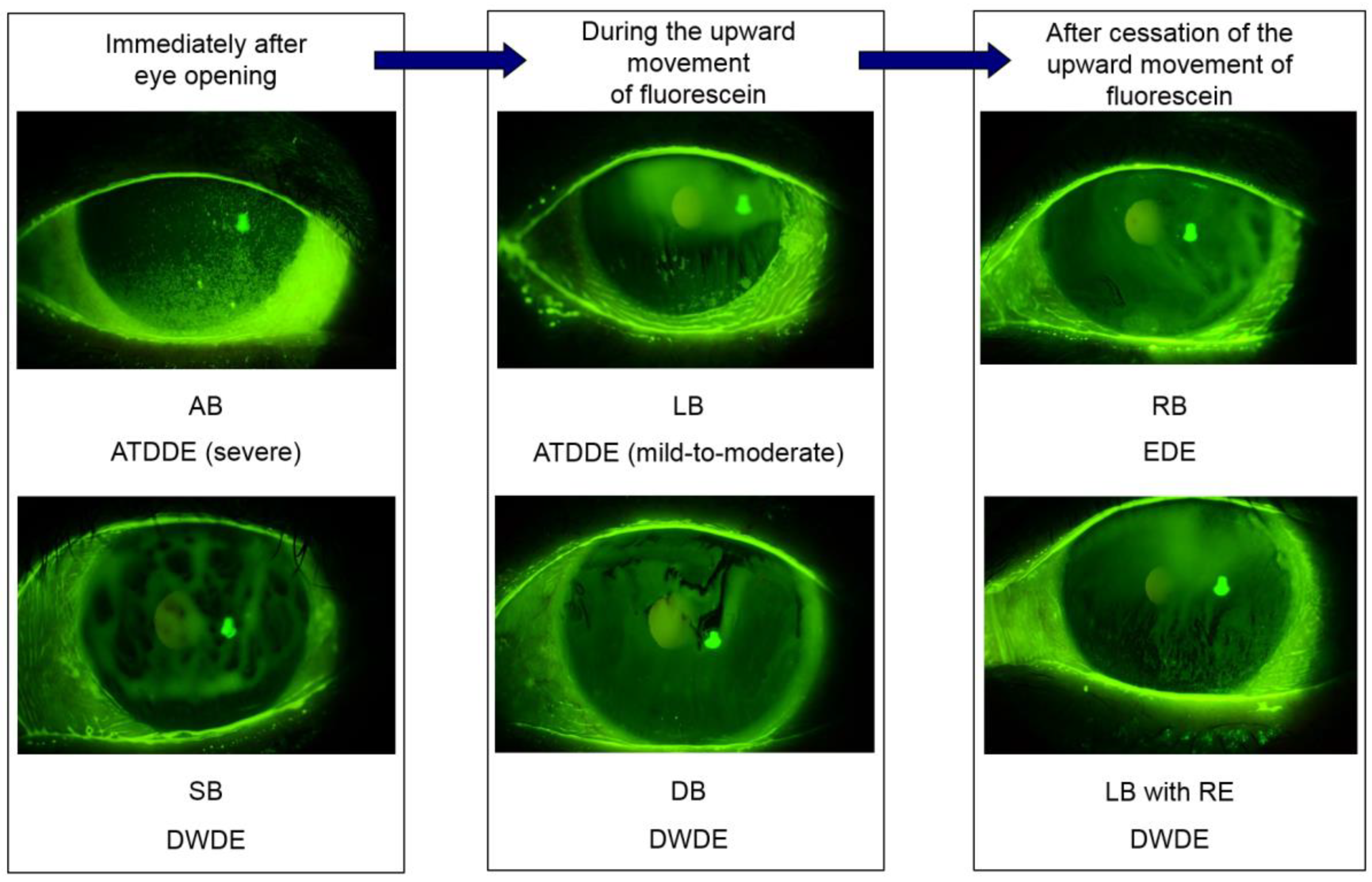
3. Unstable Tear Film: The Importance of Tear Film Breakup Patterns
3.1. Dynamic Process Involved in the Establishment of Tear Film to Understand Tear Film Breakup
3.2. Tear Film Breakup Patterns for Tear Film-Oriented Diagnosis and Their Interpretations
3.3. Clinical Implications
- Further advancement of this approach is expected through a deeper understanding of the function of each component of the ocular surface.
- TF stability is maintained by the components of the TF and ocular surface epithelium, and insufficiency of those components results in TFBU, a visible core manifestation of DED.
- Classification of TFBU patterns may elucidate the pathophysiology of DED.
- Clinicians can assess tear film-oriented diagnosis to learn more about the insufficient components responsible for TFBU, assess the specific DED subtype and, therefore, decide on the most appropriate treatment.
4. Ocular Discomfort: The Importance of Symptoms in Dry Eye Disease
4.1. Evaluation of Ocular Discomfort with Questionnaires
4.2. The Pathophysiology of Ocular Discomfort
4.3. Ocular Discomfort and Incongruity with Signs
4.4. Clinical Implications
- Ocular discomfort is common and has a range of symptoms. A careful symptom consultation is recommended for diagnosis.
- Ocular discomfort should be evaluated with a variety of patient-reported questionnaires.
- A thorough evaluation of both signs and symptoms is critical for a correct diagnosis of DED and to determine treatment decisions.
5. Visual Impairment Caused by Unstable Tear Film
Clinical Implications
- Given that DED can impair visual function, practitioners should properly evaluate DED symptoms and visual impairment.
- As simple visual acuity measurements cannot detect visual impairment associated with DED, it is possible that practitioners underestimate the extent of visual impairment.
- Assessments such as functional visual acuity can be used to assess visual function related to unstable TF.
- Simple measures such as reducing blue light emitted by devices can be used to improve visual function in those with unstable TF.
6. Epitheliopathy
6.1. Corneal Disease
6.2. Conjunctival Disease
6.3. Clinical Implications
- Epitheliopathy is frequently observed in DED but may be clinically undetectable in some patients.
- Epitheliopathy can contribute to instability of TF, blurred vision, altered permeability, nociceptor exposure and sensitization and pain in DED.
7. Dry Eye Disease and Inflammation
7.1. Innate Immune Responses in Dry Eye Disease
7.2. Adaptive Immune Responses in Dry Eye Disease
7.3. Neurogenic Inflammation
7.4. Clinical Implications
- Inflammation is a key aspect of DED pathogenesis and severity.
- Methods for identification of DED include the RPS InflammaDry Detector, an immunoassay test for the detection of elevated levels of the MMP-9.
- The control of inflammation, chronically or during clinical flares, is necessary for managing patient discomfort and ocular surface disease.
- While there can be a lack of correlation between patient symptoms and signs of ocular surface disease, managing inflammation can help attenuate both the signs and symptoms of DED.
8. Neurosensory Abnormalities
Clinical Implications
- The human cornea is densely innervated and corneal nerves participate in the stability of the ocular surface. Their continuous stimulation by a disrupted TF may cause symptoms.
- Neurosensory abnormalities may participate in three different but closely related mechanisms in DED: neurogenic inflammation, neuropathic pain and neurotrophic keratopathy. These may contribute to the lack of correlation often observed between DED signs and symptoms.
- Neurosensory abnormalities in DED must be studied further but should be considered when assessing signs, symptoms and treatment responsiveness in the clinic.
9. Implications for Regulatory Science
Clinical Implications
- Academic science tends to focus on efficacy; however, safety—a critical consideration for drug research—is key for regulatory agencies.
- The establishment of a well-organized surrogate endpoint is important for worldwide clinical research, as currently there is no global consensus on the measure of TF abnormalities in clinical trials.
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAO | American Academy of Ophthalmology |
| AP | activator protein |
| APC | antigen-presenting cell |
| AT | aqueous tear |
| ATD | aqueous tear deficiency |
| ATDDE | aqueous tear deficient dry eye |
| BU | breakup |
| CAM | cell adhesion molecules |
| DB | dimple break |
| DED | dry eye disease |
| DWDE | decreased wettability dry eye |
| EDE | evaporative dry eye |
| ICAM-1 | intercellular adhesion molecule |
| IFN | interferon |
| IL | interleukin |
| LB | line break |
| MGD | meibomian gland dysfunction |
| MHC | major histocompatibility complex |
| MMP | matrix metalloproteinase |
| NF-κ B | nuclear factor kappa-light chain-enhancer of activated B cells |
| NLRP3 | nucleotide-binding domain, leucine-rich containing family, pyrin domain containing-3 |
| RB | random break |
| RE | rapid expression |
| SB | spot break |
| TBU | tear breakup |
| TBUT | tear breakup time |
| TF | tear film |
| TFBU | tear film breakup |
| TFLL | tear film lipid layer |
| TGF | transforming growth factor |
| TLR | Toll-like receptor |
| TNF | tumor necrosis factor |
| Treg | regulatory T cell |
| VEGF | vascular endothelial growth factor |
Appendix A: Participant List
- Christophe Baudouin, Quinze-Vingts Hospital, Paris, France
- Reza Dana, Massachusetts Eye & Ear, Boston, MA, USA
- Hyo Myung Kim, Korea University Medical Center, Anam Hospital, Seoul, South Korea
- Shigeru Kinoshita, Kyoto Prefectural University of Medicine, Kyoto, Japan
- Friedrich Kruse, University of Erlangen-Nuremberg, Erlangen, Germany
- Zuguo Liu, Eye Institute of Xiamen University, Xiamen, China
- Elisabeth M. Messmer, Ludwig Maximilian University, Munich, Germany
- Stephen C. Pflugfelder, Baylor College of Medicine, Houston, TX, USA
- Maurizio Rolando, University of Genoa, Genoa, Italy
- Kazuo Tsubota, Keio University School of Medicine, Tokyo, Japan
- Norihiko Yokoi, Kyoto Prefectural University of Medicine, Kyoto, Japan
References
- Schaumberg, D.A.; Sullivan, D.A.; Buring, J.E.; Dana, M. Prevalence of dry eye syndrome among US women. Am. J. Ophthalmol. 2003, 136, 318–326. [Google Scholar] [CrossRef]
- Schaumberg, D.A.; Dana, R.; Buring, J.E.; Sullivan, D.A. Prevalence of dry eye disease among US men: Estimates from the Physicians Health Studies. Arch. Ophthalmol. 2009, 127, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Uchino, M.; Nishiwaki, Y.; Michikawa, T.; Shirakawa, K.; Kuwahara, E.; Yamada, M.; Dogru, M.; Schaumberg, D.A.; Kawakita, T.; Takebayashi, T.; et al. Prevalence and Risk Factors of Dry Eye Disease in Japan: Koumi Study. Ophthalmology 2011, 118, 2361–2367. [Google Scholar] [CrossRef] [PubMed]
- Jie, Y.; Xu, L.; Wu, Y.Y.; Jonas, J.B. Prevalence of dry eye among adult Chinese in the Beijing Eye Study. Eye 2008, 23, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Saw, S.-M.; Lamoureux, E.L.; Wang, J.J.; Rosman, M.; Tan, D.T.H.; Wong, T.Y. A Questionnaire-Based Assessment of Symptoms Associated with Tear Film Dysfunction and Lid Margin Disease in an Asian Population. Ophthal. Epidemiol. 2009, 16, 31–37. [Google Scholar] [CrossRef]
- Tan, L.L.; Morgan, P.; Cai, Z.Q.; Straughan, R.A. Prevalence of and risk factors for symptomatic dry eye disease in Singapore. Clin. Exp. Optom. 2015, 98, 45–53. [Google Scholar] [CrossRef]
- Tsubota, K.; Yokoi, N.; Shimazaki, J.; Watanabe, H.; Dogru, M.; Yamada, M.; Kinoshita, S.; Kim, H.-M.; Tchah, H.-W.; Hyon, J.Y.; et al. New Perspectives on Dry Eye Definition and Diagnosis: A Consensus Report by the Asia Dry Eye Society. Ocul. Surf. 2017, 15, 65–76. [Google Scholar] [CrossRef]
- Craig, J.P.; Nichols, K.K.; Akpek, E.K.; Caffery, B.; Dua, H.S.; Joo, C.K.; Liu, Z.; Nelson, J.D.; Nichols, J.J.; Tsubota, K.; et al. TFOS DEWS II Definition and Classification Report. Ocul. Surf. 2017, 15, 276–283. [Google Scholar] [CrossRef]
- Akpek, E.K.; Amescua, G.; Farid, M.; Garcia-Ferrer, F.J.; Lin, A.; Rhee, M.K.; Varu, D.M.; Musch, D.C.; Dunn, S.P.; Mah, F.S. Dry Eye Syndrome Preferred Practice Pattern®. Ophthalmology 2018, 126. [Google Scholar] [CrossRef]
- Lemp, M.A. Report of the National Eye Institute/Industry Workshop on Clinical Trials in Dry Eyes. CLAO J. 1995, 21, 221–232. [Google Scholar]
- Pflugfelder, S.C. Anti-inflammatory Therapy of Dry Eye. Ocul. Surf. 2003, 1, 31–36. [Google Scholar] [CrossRef]
- Lemp, M.A.; Foulks, G.N. The definition and classification of dry eye disease: Report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop. Ocul. Surf. 2007, 5, 75–92. [Google Scholar]
- Shimazaki, J. Definition and diagnosis of dry eye 2006. Atarashii Ganka 2007, 24, 181–184. [Google Scholar]
- Hyon, J.Y.; Kim, H.M.; Lee, D.; Chung, E.-S.; Song, J.-S.; Choi, C.Y.; Lee, J. Korean Corneal Disease Study Group Korean Guidelines for the Diagnosis and Management of Dry Eye: Development and Validation of Clinical Efficacy. Korean J. Ophthalmol. 2014, 28, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z. The preliminary recommendations on the name and classification of dry eye. Chin. J. Eye Otolaryngol. 2004, 3, 4–5. [Google Scholar]
- The Chinese Corneal Society. The consensus on clinical diagnosis and treatment of dry eye. Chin. J. Ophthalmol. 2013, 49, 73–75. [Google Scholar]
- Shimazaki, J.; Yokoi, N.; Watanabe, H. Definition and diagnosis of dry eye in Japan, 2016. Atarashii Ganka 2017, 34, 309–313. [Google Scholar]
- Uchino, M.; Yokoi, N.; Uchino, Y.; Dogru, M.; Kawashima, M.; Komuro, A.; Sonomura, Y.; Kato, H.; Kinoshita, S.; Schaumberg, D.A.; et al. Prevalence of Dry Eye Disease and its Risk Factors in Visual Display Terminal Users: The Osaka Study. Am. J. Ophthalmol. 2013, 156, 759–766.e1. [Google Scholar] [CrossRef]
- Yokoi, N.; Uchino, M.; Uchino, Y.; Dogru, M.; Kawashima, M.; Komuro, A.; Sonomura, Y.; Kato, H.; Tsubota, K.; Kinoshita, S. Importance of Tear Film Instability in Dry Eye Disease in Office Workers Using Visual Display Terminals: The Osaka Study. Am. J. Ophthalmol. 2015, 159, 748–754. [Google Scholar] [CrossRef]
- Tong, L.; Chaurasia, S.S.; Mehta, J.S.; Beuerman, R.W. Screening for Meibomian Gland Disease: Its Relation to Dry Eye Subtypes and Symptoms in a Tertiary Referral Clinic in Singapore. Investig. Opthalmol. Vis. Sci. 2010, 51, 3449–3454. [Google Scholar] [CrossRef]
- Bron, A.J.; De Paiva, C.S.; Chauhan, S.K.; Bonini, S.; Gabison, E.E.; Jain, S.; Knop, E.; Markoulli, M.; Ogawa, Y.; Perez, V.; et al. TFOS DEWS II pathophysiology report. Ocul. Surf. 2017, 15, 438–510. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, N. Tear Film Oriented Diagnosis and Tear Film Oriented Therapy for Dry Eye Based on Tear Dynamics. Investig. Ophthalmol. Vis. Sci. 2018, 59, DES13–DES22. [Google Scholar] [CrossRef] [PubMed]
- King-Smith, P.E.; Fink, B.A.; Hill, R.M.; Koelling, K.W.; Tiffany, J.M. The thickness of the tear film. Curr. Eye Res. 2004, 29, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, N.; Georgiev, G.A. Tear-Film-Oriented Diagnosis and Therapy for Dry Eye. In Dry Eye Syndrome: Basic and Clinical Perspectives; Future Medicine Ltd.: London, UK, 2013; pp. 96–108. [Google Scholar]
- Yokoi, N.; Georgiev, G.A.; Kato, H.; Komuro, A.; Sonomura, Y.; Sotozono, C.; Tsubota, K.; Kinoshita, S. Classification of Fluorescein Breakup Patterns: A Novel Method of Differential Diagnosis for Dry Eye. Am. J. Ophthalmol. 2017, 180, 72–85. [Google Scholar] [CrossRef] [PubMed]
- Creech, J.; Do, L.T.; Fatt, I.; Radke, C. In vivo tear-film thickness determination and implications for tear-film stability. Curr. Eye Res. 1998, 17, 1058–1066. [Google Scholar] [CrossRef]
- Yokoi, N.; Georgiev, G.A. Tear-film-oriented diagnosis for dry eye. Jpn. J. Ophthalmol. 2019, 63, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Berger, R.E.; Corrsin, S. A surface tension gradient mechanism for driving the pre-corneal tear film after a blink. J. Biomech. 1974, 7, 225–238. [Google Scholar] [CrossRef]
- Yokoi, N.; Yamada, H.; Mizukusa, Y.; Bron, A.J.; Tiffany, J.M.; Kato, T.; Kinoshita, S. Rheology of Tear Film Lipid Layer Spread in Normal and Aqueous Tear-Deficient Dry Eyes. Investig. Opthalmol. Vis. Sci. 2008, 49, 5319–5324. [Google Scholar] [CrossRef]
- King-Smith, P.E.; Reuter, K.S.; Braun, R.J.; Nichols, J.J.; Nichols, K.K. Tear Film Breakup and Structure Studied by Simultaneous Video Recording of Fluorescence and Tear Film Lipid Layer Images. Investig. Opthalmol. Vis. Sci. 2013, 54, 4900–4909. [Google Scholar] [CrossRef]
- Bull, J.; Grotberg, J. Surfactant spreading on thin viscous films: Film thickness evolution and periodic wall stretch. Exp. Fluids 2003, 34, 1–15. [Google Scholar] [CrossRef]
- Zhang, Y.L.; Matar, O.K.; Craster, R.V. Analysis of tear film rupture: Effect of non-Newtonian rheology. J. Colloid Interface Sci. 2003, 262, 130–148. [Google Scholar] [CrossRef]
- Sharma, A.; Tiwari, S.; Khanna, R.; Tiffany, J.M. Hydrodynamics of meniscus-induced thinning of the tear film. Atherosclerosis 1998, 438, 425–431. [Google Scholar] [CrossRef]
- McDonald, J.E.; Brubaker, S. Meniscus-Induced Thinning of Tear Films. Am. J. Ophthalmol. 1971, 72, 139–146. [Google Scholar] [CrossRef]
- Yokoi, N.; Komuro, A. Non-invasive methods of assessing the tear film. Exp. Eye Res. 2004, 78, 399–407. [Google Scholar] [CrossRef]
- Shanker, R.M.; Ahmed, I.; Bourassa, P.A.; Carola, K.V. An in vitro technique for measuring contact angles on the corneal surface and its application to evaluate corneal wetting properties of water soluble polymers. Int. J. Pharm. 1995, 119, 149–163. [Google Scholar] [CrossRef]
- Sharma, A. Breakup and dewetting of the corneal mucus layer. An update. Single Mol. Single Cell Seq. 1998, 438, 273–280. [Google Scholar]
- Sharma, A. Surface-chemical pathways of the tear film breakup—Does corneal mucus have a role? Lacrimal Gland Tear Film Dry Eye Syndr. 1998, 438, 361–370. [Google Scholar]
- Lee, S.-H.; Tseng, S.C. Rose bengal staining and cytologic characteristics associated with lipid tear deficiency. Am. J. Ophthalmol. 1997, 124, 736–750. [Google Scholar] [CrossRef]
- Mcmonnies, C.W.; Ho, A. Patient history in screening for dry eye conditions. J. Am. Optom. Assoc. 1987, 58, 296–301. [Google Scholar]
- Schiffman, R.M.; Christianson, M.D.; Jacobsen, G.; Hirsch, J.D.; Reis, B.L. Reliability and Validity of the Ocular Surface Disease Index. Arch. Ophthalmol. 2000, 118, 615–621. [Google Scholar] [CrossRef]
- Mangione, C.M.; Lee, P.P.; Gutierrez, P.R.; Spritzer, K.; Berry, S.; Hays, R.D. National Eye Institute Visual Function Questionnaire Field Test, I., Development of the 25-item National Eye Institute Visual Function Questionnaire. Arch. Ophthalmol. 2001, 119, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Begley, C.; Caffery, B.; Chalmers, R.L.; Mitchell, G.L. Use of the Dry Eye Questionnaire to Measure Symptoms of Ocular Irritation in Patients With Aqueous Tear Deficient Dry Eye. Cornea 2002, 21, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.E.; Murphy, P.J. Measurement of Ocular Surface Irritation on a Linear Interval Scale with the Ocular Comfort Index. Investig. Opthalmol. Vis. Sci. 2007, 48, 4451–4458. [Google Scholar] [CrossRef] [PubMed]
- Schaumberg, D.A.; Gulati, A.; Mathers, W.D.; Clinch, T.; Lemp, M.A.; Nelson, J.D.; Foulks, G.N.; Dana, R. Development and Validation of a Short Global Dry Eye Symptom Index. Ocul. Surf. 2007, 5, 50–57. [Google Scholar] [CrossRef]
- Blackie, C.A.; Solomon, J.D.; Scaffidi, R.C.; Greiner, J.V.; Lemp, M.A.; Korb, D.R. The Relationship Between Dry Eye Symptoms and Lipid Layer Thickness. Cornea 2009, 28, 789–794. [Google Scholar] [CrossRef]
- Abetz-Webb, L.; Rajagopalan, K.; Mertzanis, P.; Begley, C.; Barnes, R.; Chalmers, R.L. Development and validation of the impact of dry eye on everyday life (IDEEL) questionnaire, a patient-reported outcomes (PRO) measure for the assessment of the burden of dry eye on patients. Health Qual. Life Outcomes 2011, 9, 111. [Google Scholar] [CrossRef]
- Sakane, Y.; Yamaguchi, M.; Yokoi, N.; Uchino, M.; Dogru, M.; Oishi, T.; Ohashi, Y.; Ohashi, Y. Development and Validation of the Dry Eye-Related Quality-of-Life Score Questionnaire. JAMA Ophthalmol. 2013, 131, 1331–1338. [Google Scholar] [CrossRef]
- Chalmers, R.L.; Begley, C.G.; Caffery, B. Validation of the 5-Item Dry Eye Questionnaire (DEQ-5): Discrimination across self-assessed severity and aqueous tear deficient dry eye diagnoses. Contact Lens Anterior Eye 2010, 33, 55–60. [Google Scholar] [CrossRef]
- Zhao, H.; Liu, Z.; Yang, W.; Xiao, X.; Chen, J.; Li, Q.; Zhong, T. Development and assessment of a dry eye questionnaire applicable to the Chinese population. Chin. J. Ophthalmol. 2015, 51, 647–654. [Google Scholar]
- Seror, R.; Theander, E.; Bootsma, H.; Bowman, S.J.; Tzioufas, A.; Gottenberg, J.E.; Ramos-Casals, M.; Dorner, T.; Ravaud, P.; Mariette, X.; et al. Outcome measures for primary Sjogren’s syndrome: A comprehensive review. J. Autoimmun. 2014, 51, 51–56. [Google Scholar] [CrossRef]
- Belmonte, C.; Nichols, J.J.; Cox, S.M.; Brock, J.A.; Begley, C.G.; Bereiter, D.A.; Dartt, D.A.; Galor, A.; Hamrah, P.; Ivanusic, J.J.; et al. TFOS DEWS II pain and sensation report. Ocul. Surf. 2017, 15, 404–437. [Google Scholar] [CrossRef] [PubMed]
- Van Der Vaart, R.; Weaver, M.A.; Lefebvre, C.; Davis, R.M. The Association Between Dry Eye Disease and Depression and Anxiety in a Large Population-Based Study. Am. J. Ophthalmol. 2015, 159, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Toda, I.; Shimazaki, J.; Tsubota, K. Dry Eye with Only Decreased Tear Break-up Time is Sometimes Associated with Allergic Conjunctivitis. Ophthalmology 1995, 102, 302–309. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; Tseng, S.C.G.; Sanabria, O.; Kell, H.; Garcia, C.G.; Felix, C.; Feuer, W.; Reis, B.L. Evaluation of Subjective Assessments and Objective Diagnostic Tests for Diagnosing Tear-Film Disorders Known to Cause Ocular Irritation. Cornea 1998, 17, 38–56. [Google Scholar] [CrossRef]
- Rolando, M.; Iester, M.; Macrí, A.; Calabria, G. Low Spatial-Contrast Sensitivity in Dry Eyes. Cornea 1998, 17, 376–379. [Google Scholar] [CrossRef]
- Goto, E.; Yagi, Y.; Matsumoto, Y.; Tsubota, K. Impaired functional visual acuity of dry eye patients. Am. J. Ophthalmol. 2002, 133, 181–186. [Google Scholar] [CrossRef]
- Kaido, M.; Dogru, M.; Ishida, R.; Tsubota, K. Concept of Functional Visual Acuity and its Applications. Cornea 2007, 26, S29–S35. [Google Scholar] [CrossRef]
- Koh, S.; Naoyuki, M.; Hirohara, Y.; Mihashi, T.; Bessho, K.; Hori, Y.; Inoue, T.; Watanabe, H.; Fujikado, T.; Tano, Y. Serial Measurements of Higher-Order Aberrations after Blinking in Patients with Dry Eye. Investig. Opthalmol. Vis. Sci. 2008, 49, 133–138. [Google Scholar] [CrossRef]
- Kaido, M.; Kawashima, M.; Shigeno, Y.; Yamada, Y.; Tsubota, K. Relation of accommodative microfluctuation with dry eye symptoms in short tear break-up time dry eye. PLoS ONE 2017, 12, e0184296. [Google Scholar] [CrossRef]
- Kaido, M.; Kawashima, M.; Ishida, R.; Tsubota, K. Severe symptoms of short tear break-up time dry eye are associated with accommodative microfluctuations. Clin. Ophthalmol. 2017, 11, 861–869. [Google Scholar] [CrossRef]
- Kaido, M. Functional visual acuity. Invest. Ophthalmol. Vis. Sci. 2018, 59, DES29–DES35. [Google Scholar] [CrossRef] [PubMed]
- Kaido, M.; Ishida, R.; Dogru, M.; Tsubota, K. Visual Function Changes After Punctal Occlusion with the Treatment of Short BUT Type of Dry Eye. Cornea 2012, 31, 1009–1013. [Google Scholar] [CrossRef] [PubMed]
- Mun, Y.; Kwon, J.W.; Oh, J.Y. Therapeutic effects of 3% diquafosol ophthalmic solution in patients with short tear film break-up time-type dry eye disease. BMC Ophthalmol. 2018, 18, 237. [Google Scholar] [CrossRef] [PubMed]
- Kaido, M.; Kawashima, M.; Shigeno, Y.; Yamada, Y.; Tsubota, K. Randomized Controlled Study to Investigate the Effect of Topical Diquafosol Tetrasodium on Corneal Sensitivity in Short Tear Break-Up Time Dry Eye. Adv. Ther. 2018, 35, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Xia, S.; Chen, Y. Comparison of the efficacy between topical diquafosol and artificial tears in the treatment of dry eye following cataract surgery: A meta-analysis of randomized controlled trials. Medicine 2017, 96, e8174. [Google Scholar] [CrossRef] [PubMed]
- Koh, S.; Ikeda, C.; Takai, Y.; Watanabe, H.; Maeda, N.; Nishida, K. Long-term results of treatment with diquafosol ophthalmic solution for aqueous-deficient dry eye. Jpn. J. Ophthalmol. 2013, 57, 440–446. [Google Scholar] [CrossRef]
- Kaido, M.; Toda, I.; Oobayashi, T.; Kawashima, M.; Katada, Y.; Tsubota, K. Reducing Short-Wavelength Blue Light in Dry Eye Patients with Unstable Tear Film Improves Performance on Tests of Visual Acuity. PLoS ONE 2016, 11, e0152936. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; Tseng, S.C.; Yoshino, K.; Monroy, D.; Felix, C.; Reis, B.L. Correlation of Goblet Cell Density and Mucosal Epithelial Membrane Mucin Expression with Rose Bengal Staining in Patients with Ocular Irritation. Ophthalmology 1997, 104, 223–235. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; De Paiva, C.S.; Moore, Q.L.; Volpe, E.A.; Li, D.Q.; Gumus, K.; Zaheer, M.L.; Corrales, R.M. Aqueous Tear Deficiency Increases Conjunctival Interferon-gamma (IFN-gamma) Expression and Goblet Cell Loss. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7545–7550. [Google Scholar] [CrossRef]
- Tung, C.I.; Perin, A.F.; Gumus, K.; Pflugfelder, S.C. Tear Meniscus Dimensions in Tear Dysfunction and Their Correlation With Clinical Parameters. Am. J. Ophthalmol. 2014, 157, 301–310. [Google Scholar] [CrossRef]
- Göbbels, M.; Spitznas, M. Effects of artificial tears on corneal epithelial permeability in dry eyes. Graefe Arch. Clin. Exp. Ophthalmol. 1991, 229, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Wolffsohn, J.S.; Arita, R.; Chalmers, R.; Djalilian, A.; Dogru, M.; Dumbleton, K.; Gupta, P.K.; Karpecki, P.; Lazreg, S.; Pult, H.; et al. TFOS DEWS II Diagnostic Methodology report. Ocul. Surf. 2017, 15, 539–574. [Google Scholar] [CrossRef] [PubMed]
- Bron, A.J.; Argüeso, P.; Irkec, M.; Bright, F. Clinical staining of the ocular surface: Mechanisms and interpretations. Prog. Retin. Eye Res. 2015, 44, 36–61. [Google Scholar] [CrossRef] [PubMed]
- Pflugfelder, S.C.; Song, X.J.; Farley, W.; Li, D.-Q.; Stern, M.E.; Pflugfelder, S.C. Apoptosis of ocular surface cells in experimentally induced dry eye. Investig. Opthalmol. Vis. Sci. 2003, 44, 124–129. [Google Scholar] [CrossRef]
- Bandamwar, K.L.; Garrett, Q.; Papas, E.B. Mechanisms of superficial micropunctate corneal staining with sodium fluorescein: The contribution of pooling. Contact Lens Anterior Eye 2012, 35, 81–84. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; Farley, W.; Luo, L.; Chen, L.Z.; De Paiva, C.S.; Olmos, L.C.; Li, D.-Q.; Fini, M.E. Matrix Metalloproteinase-9 Knockout Confers Resistance to Corneal Epithelial Barrier Disruption in Experimental Dry Eye. Am. J. Pathol. 2005, 166, 61–71. [Google Scholar] [CrossRef]
- Fini, M.E.; Bauskar, A.; Jeong, S.; Wilson, M.R. Clusterin in the eye: An old dog with new tricks at the ocular surface. Exp. Eye Res. 2016, 147, 57–71. [Google Scholar] [CrossRef]
- Luo, L.; Li, D.-Q.; Doshi, A.; Farley, W.; Corrales, R.M.; Pflugfelder, S.C. Experimental Dry Eye Stimulates Production of Inflammatory Cytokines and MMP-9 and Activates MAPK Signaling Pathways on the Ocular Surface. Investig. Opthalmol. Vis. Sci. 2004, 45, 4293–4301. [Google Scholar] [CrossRef]
- De Paiva, C.S.; Corrales, R.M.; Villarreal, A.L.; Farley, W.; Li, D.-Q.; Stern, M.E.; Pflugfelder, S.C. Apical Corneal Barrier Disruption in Experimental Murine Dry Eye Is Abrogated by Methylprednisolone and Doxycycline. Investig. Opthalmol. Vis. Sci. 2006, 47, 2847–2856. [Google Scholar] [CrossRef]
- De Paiva, C.S.; Corrales, R.M.; Villarreal, A.L.; Farley, W.J.; Li, D.-Q.; Stern, M.E.; Pflugfelder, S.C. Corticosteroid and doxycycline suppress MMP-9 and inflammatory cytokine expression, MAPK activation in the corneal epithelium in experimental dry eye. Exp. Eye Res. 2006, 83, 526–535. [Google Scholar] [CrossRef]
- Pelegrino, F.; Pflugfelder, S.C.; De Paiva, C.S. Low humidity environmental challenge causes barrier disruption and cornification of the mouse corneal epithelium via a c-jun N-terminal kinase 2 (JNK2) pathway. Exp. Eye Res. 2012, 94, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Kohanim, S.; Palioura, S.; Saeed, H.N.; Akpek, E.K.; Amescua, G.; Basu, S.; Blomquist, P.H.; Bouchard, C.S.; Dart, J.K.; Gai, X.; et al. Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis—A Comprehensive Review and Guide to Therapy. I. Systemic Disease. Ocul. Surf. 2016, 14, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Pflugfelder, S.C.; De Paiva, C.S.; Villarreal, A.L.; Stern, M.E. Effects of sequential artificial tear and cyclosporine emulsion therapy on conjunctival goblet cell density and transforming growth factor-beta2 production. Cornea 2008, 27, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Ruiz, L.; Masli, S. Immunomodulatory Cross-Talk between Conjunctival Goblet Cells and Dendritic Cells. PLoS ONE 2015, 10, e0120284. [Google Scholar] [CrossRef]
- Xiao, Y.; De Paiva, C.S.; Yu, Z.; De Souza, R.G.; Li, D.-Q.; Pflugfelder, S.C. Goblet cell-produced retinoic acid suppresses CD86 expression and IL-12 production in bone marrow-derived cells. Int. Immunol. 2018, 30, 457–470. [Google Scholar] [CrossRef]
- De Paiva, C.S.; Villarreal, A.L.; Corrales, R.M.; Rahman, H.T.; Chang, V.Y.; Farley, W.J.; Stern, M.E.; Niederkorn, J.Y.; Li, D.Q.; Pflugfelder, S.C. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-gamma. Investig. Ophthalmol. Vis. Sci. 2007, 48, 2553–2560. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.S.; Raince, J.K.; McClellan, A.J.; Shanmugam, K.P.; Pangelinan, S.B.; Volpe, E.A.; Corrales, R.M.; Farley, W.J.; Corry, D.B.; Li, D.-Q.; et al. Homeostatic control of conjunctival mucosal goblet cells by NKT-derived IL-13. Mucosal Immunol. 2011, 4, 397–408. [Google Scholar] [CrossRef]
- McClellan, A.J.; Volpe, E.A.; Zhang, X.; Darlington, G.J.; Li, D.-Q.; Pflugfelder, S.C.; De Paiva, C.S. Ocular Surface Disease and Dacryoadenitis in Aging C57BL/6 Mice. Am. J. Pathol. 2014, 184, 631–643. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, W.; De Paiva, C.S.; Corrales, R.M.; Volpe, E.A.; McClellan, A.J.; Farley, W.J.; Li, D.Q.; Pflugfelder, S.C. Interferon-gamma exacerbates dry eye-induced apoptosis in conjunctiva through dual apoptotic pathways. Investig. Ophthalmol. Vis. Sci. 2011, 52, 6279–6285. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, W.; De Paiva, C.S.; Volpe, E.A.; Gandhi, N.B.; Farley, W.J.; Li, D.Q.; Niederkorn, J.Y.; Stern, M.E.; Pflugfelder, S.C. Desiccating stress induces CD4+ T-cell-mediated Sjogren’s syndrome-like corneal epithelial apoptosis via activation of the extrinsic apoptotic pathway by interferon-gamma. Am. J. Pathol. 2011, 179, 1807–1814. [Google Scholar] [CrossRef]
- Zhang, X.; De Paiva, C.S.; Su, Z.; Volpe, E.A.; Li, D.-Q.; Pflugfelder, S.C. Topical interferon-gamma neutralization prevents conjunctival goblet cell loss in experimental murine dry eye. Exp. Eye Res. 2014, 118, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Corrales, R.M.; De Paiva, C.S.; Li, D.-Q.; Farley, W.J.; Henriksson, J.T.; Bergmanson, J.P.G.; Pflugfelder, S.C. Entrapment of Conjunctival Goblet Cells by Desiccation-Induced Cornification. Investig. Opthalmol. Vis. Sci. 2011, 52, 3492–3499. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, J.T.; Coursey, T.G.; Corry, D.B.; De Paiva, C.S.; Pflugfelder, S.C. IL-13 Stimulates Proliferation and Expression of Mucin and Immunomodulatory Genes in Cultured Conjunctival Goblet Cells. Investig. Opthalmol. Vis. Sci. 2015, 56, 4186–4197. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Ruiz, L.; Ghosh-Mitra, A.; Shatos, M.A.; Dartt, D.A.; Masli, S. Modulation of Conjunctival Goblet Cell Function by Inflammatory Cytokines. Mediat. Inflamm. 2013, 2013, 1–11. [Google Scholar] [CrossRef]
- Tseng, S.C.; Hirst, L.W.; Maumenee, A.E.; Kenyon, K.R.; Sun, T.-T.; Green, W.R. Possible Mechanisms for the Loss of Goblet Cells in Mucin-deficient Disorders. Ophthalmology 1984, 91, 545–552. [Google Scholar] [CrossRef]
- Huang, A.J.; Tseng, S.C.; Kenyon, K.R. Morphogenesis of rat conjunctival goblet cells. Investig. Ophthalmol. Vis. Sci. 1988, 29, 969–975. [Google Scholar]
- Roat, M.I.; Ohji, M.; Hunt, L.E.; Thoft, R.A. Conjunctival Epithelial Cell Hypermitosis and Goblet Cell Hyperplasia in Atopic Keratoconjunctivitis. Am. J. Ophthalmol. 1993, 116, 456–463. [Google Scholar] [CrossRef]
- Tseng, S.C. Staging of conjunctival squamous metaplasia by impression cytology. Ophthalmology 1985, 92, 728–733. [Google Scholar] [CrossRef]
- Wittpenn, J.R.; Tseng, S.C.; Sommer, A. Detection of early xerophthalmia by impression cytology. Arch. Ophthalmol. 1986, 104, 237–239. [Google Scholar] [CrossRef]
- Lemp, M.A.; Dohlman, C.H.; Kuwabara, T.; Holly, F.J.; Carroll, J.M. Dry eye secondary to mucus deficiency. Trans. Am. Acad. Ophthalmol. Otolaryngol. 1971, 75, 1223–1227. [Google Scholar]
- Ralph, R.A. Conjunctival goblet cell density in normal subjects and in dry eye syndromes. Investig. Ophthalmol. 1975, 14, 299–302. [Google Scholar]
- Nelson, J.D.; Wright, J.C. Conjunctival Goblet Cell Densities in Ocular Surface Disease. Arch. Ophthalmol. 1984, 102, 1049–1051. [Google Scholar] [CrossRef] [PubMed]
- Pflugfelder, S.C.; Huang, A.J.; Feuer, W.; Chuchovski, P.T.; Pereira, I.C.; Tseng, S.C. Conjunctival cytologic features of primary Sjogren’s syndrome. Ophthalmology 1990, 97, 985–991. [Google Scholar] [CrossRef]
- Murube, J.; Rivas, L. Impression Cytology on Conjunctiva and Cornea in Dry Eye Patients Establishes a Correlation between Squamous Metaplasia and Dry Eye Clinical Severity. Eur. J. Ophthalmol. 2003, 13, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Uchino, Y.; Uchino, M.; Yokoi, N.; Dogru, M.; Kawashima, M.; Komuro, A.; Sonomura, Y.; Kato, H.; Argüeso, P.; Kinoshita, S.; et al. Impact of Cigarette Smoking on Tear Function and Correlation between Conjunctival Goblet Cells and Tear MUC5AC Concentration in Office Workers. Sci. Rep. 2016, 6, 27699. [Google Scholar] [CrossRef]
- Yeo, A.C.H.; Carkeet, A.; Carney, L.G.; Yap, M.K.H. Relationship between goblet cell density and tear function tests. Ophthalmic Physiol. Opt. 2003, 23, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Bhargava, R.; Kumar, M.; Ranjan, S.; Kumar, M.; Verma, P. The Correlation of Routine Tear Function Tests and Conjunctival Impression Cytology in Dry Eye Syndrome. Korean J. Ophthalmol. 2014, 28, 122–129. [Google Scholar] [CrossRef]
- Sapkota, K.; Franco, S.; Sampaio, P.; Lira, M. Goblet cell density association with tear function and ocular surface physiology. Contact Lens Anterior Eye 2015, 38, 240–244. [Google Scholar] [CrossRef]
- Chi, W.; Hua, X.; Chen, X.; Bian, F.; Yuan, X.; Zhang, L.; Wang, X.; Chen, D.; Deng, R.; Li, Z.; et al. Mitochondrial DNA oxidation induces imbalanced activity of NLRP3/NLRP6 inflammasomes by activation of caspase-8 and BRCC36 in dry eye. J. Autoimmun. 2017, 80, 65–76. [Google Scholar] [CrossRef]
- Deng, R.; Hua, X.; Li, J.; Chi, W.; Zhang, Z.; Lu, F.; Zhang, L.; Pflugfelder, S.C.; Li, D.-Q. Oxidative Stress Markers Induced by Hyperosmolarity in Primary Human Corneal Epithelial Cells. PLoS ONE 2015, 10, e0126561. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. TLR signaling. Cell Death Differ. 2006, 13, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Enríquez-De-Salamanca, A.; Castellanos, E.; Stern, M.E.; Fernández, I.; Carreño, E.; García-Vázquez, C.; Herreras, J.M.; Calonge, M. Tear cytokine and chemokine analysis and clinical correlations in evaporative-type dry eye disease. Mol. Vis. 2010, 16, 862–873. [Google Scholar] [PubMed]
- Li, D.-Q.; Chen, Z.; Song, X.J.; Luo, L.; Pflugfelder, S.C. Stimulation of Matrix Metalloproteinases by Hyperosmolarity via a JNK Pathway in Human Corneal Epithelial Cells. Investig. Opthalmol. Vis. Sci. 2004, 45, 4302–4311. [Google Scholar] [CrossRef] [PubMed]
- Li, D.Q.; Chen, Z.; Song, X.J.; Farley, W.; Pflugfelder, S.C. Hyperosmolarity Stimulates Production of MMP-9, IL-1ß and TNF- by Human Corneal Epithelial Cells Via a c-Jun NH2-terminal Kinase Pathway. Investig. Ophthalmol. Vis. Sci. 2002, 43, 1981. [Google Scholar]
- Li, D.Q.; Shang, T.Y.; Kim, H.S.; Solomon, A.; Lokeshwar, B.L.; Pflugfelder, S.C. Regulated expression of collagenases MMP-1, -8, and -13 and stromelysins MMP-3, -10, and -11 by human corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2003, 44, 2928–2936. [Google Scholar] [CrossRef]
- Chen, Y.T.; Nikulina, K.; Lazarev, S.; Bahrami, A.F.; Noble, L.B.; Gallup, M.; McNamara, N.A. Interleukin-1 as a phenotypic immunomodulator in keratinizing squamous metaplasia of the ocular surface in Sjogren’s syndrome. Am. J. Pathol. 2010, 177, 1333–1343. [Google Scholar] [CrossRef]
- Epstein, S.P.; Gadaria-Rathod, N.; Wei, Y.; Maguire, M.G.; Asbell, P.A. HLA-DR expression as a biomarker of inflammation for multicenter clinical trials of ocular surface disease. Exp. Eye Res. 2013, 111, 95–104. [Google Scholar] [CrossRef]
- Barabino, S.; Montaldo, E.; Solignani, F.; Valente, C.; Mingari, M.C.; Rolando, M. Immune response in the conjunctival epithelium of patients with dry eye. Exp. Eye Res. 2010, 91, 524–529. [Google Scholar] [CrossRef]
- Baudouin, C.; Brignole, F.; Pisella, P.-J.; Jean, M.D.S.; Goguel, A. Flow Cytometric Analysis of the Inflammatory Marker HLA DR in Dry Eye Syndrome: Results from 12 Months of Randomized Treatment with Topical Cyclosporin A. In Advances in Experimental Medicine and Biology; Springer: Boston, MA, USA, 2002; Volume 506, pp. 761–769. [Google Scholar]
- Stern, M.E.; Gao, J.; Schwalb, T.A.; Ngo, M.; Tieu, D.D.; Chan, C.C.; Reis, B.L.; Whitcup, S.M.; Thompson, D.; Smith, J.A. Conjunctival T-cell subpopulations in Sjogren’s and non-Sjogren’s patients with dry eye. Investig. Ophthalmol. Vis. Sci. 2002, 43, 2609–2614. [Google Scholar]
- Tsubota, K.; Fujita, H.; Tsuzaka, K.; Takeuchi, T. Quantitative analysis of lacrimal gland function, apoptotic figures, Fas and Fas ligand expression of lacrimal glands in dry eye patients. Exp. Eye Res. 2003, 76, 233–240. [Google Scholar] [CrossRef]
- Chauhan, S.K.; El Annan, J.; Ecoiffier, T.; Goyal, S.; Zhang, Q.; Saban, D.R.; Dana, R. Autoimmunity in Dry Eye is due to Resistance of Th17 to Treg Suppression1. J. Immunol. 2009, 182, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- El Annan, J.; Chauhan, S.K.; Ecoiffier, T.; Zhang, Q.; Saban, D.R.; Dana, R. Characterization of Effector T Cells in Dry Eye Disease. Investig. Opthalmol. Vis. Sci. 2009, 50, 3802–3807. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.C.; Park, C.-S.; You, I.-C.; Choi, H.-J.; Lee, K.-H.; Im, S.-K.; Park, H.-Y.; Pflugfelder, S.C. Expression of CXCL9, -10, -11, and CXCR3 in the Tear Film and Ocular Surface of Patients with Dry Eye Syndrome. Investig. Opthalmol. Vis. Sci. 2010, 51, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Gulati, A.; Sacchetti, M.; Bonini, S.; Dana, M.R. Chemokine Receptor CCR5 Expression in Conjunctival Epithelium of Patients With Dry Eye Syndrome. Arch. Ophthalmol. 2006, 124, 710–716. [Google Scholar] [CrossRef][Green Version]
- Mills, K.H.G. Induction, function and regulation of IL-17-producing T cells. Eur. J. Immunol. 2008, 38, 2636–2649. [Google Scholar] [CrossRef]
- De Paiva, C.S.; Chotikavanich, S.; Pangelinan, S.B.; Pitcher, J.D.; Fang, B.; Zheng, X.; Ma, P.; Farley, W.J.; Siemasko, K.F.; Niederkorn, J.Y.; et al. IL-17 disrupts corneal barrier following desiccating stress. Mucosal Immunol. 2009, 2, 243–253. [Google Scholar] [CrossRef]
- Kang, M.H.; Kim, M.K.; Lee, H.J.; Lee, H.I.; Wee, W.R.; Lee, J.H. Interleukin-17 in Various Ocular Surface Inflammatory Diseases. J. Korean Med. Sci. 2011, 26, 938–944. [Google Scholar] [CrossRef]
- Goyal, S.; Chauhan, S.K.; El Annan, J.; Nallasamy, N.; Zhang, Q.; Dana, R. Evidence of corneal lymphangiogenesis in dry eye disease: A potential link to adaptive immunity? Arch. Ophthalmol. 2010, 128, 819–824. [Google Scholar] [CrossRef]
- Chauhan, S.K.; Jin, Y.; Goyal, S.; Lee, H.S.; Fuchsluger, T.A.; Lee, H.K.; Dana, R. A novel pro-lymphangiogenic function for Th17/IL-17. Blood 2011, 118, 4630–4634. [Google Scholar] [CrossRef]
- Chen, Y.; Chauhan, S.K.; Shao, C.; Omoto, M.; Inomata, T.; Dana, R. IFN-gamma-Expressing Th17 Cells Are Required for Development of Severe Ocular Surface Autoimmunity. J. Immunol. 2017, 199, 1163–1169. [Google Scholar] [CrossRef]
- Chen, Y.; Chauhan, S.K.; Lee, H.S.; Saban, D.R.; Dana, R. Chronic dry eye disease is principally mediated by effector memory Th17 cells. Mucosal Immunol. 2014, 7, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chauhan, S.K.; Tan, X.; Dana, R. Interleukin-7 and -15 maintain pathogenic memory Th17 cells in autoimmunity. J. Autoimmun. 2016, 77, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Siemasko, K.F.; Gao, J.; Calder, V.L.; Hanna, R.; Calonge, M.; Pflugfelder, S.C.; Niederkorn, J.Y.; Stern, M.E. In Vitro Expanded CD4+CD25+Foxp3+Regulatory T Cells Maintain a Normal Phenotype and Suppress Immune-Mediated Ocular Surface Inflammation. Investig. Opthalmol. Vis. Sci. 2008, 49, 5434–5440. [Google Scholar] [CrossRef] [PubMed]
- McDermott, A.M.; Perez, V.; Huang, A.J.W.; Pflugfelder, S.C.; Stern, M.E.; Baudouin, C.; Beuerman, R.W.; Burns, A.R.; Calder, V.L.; Calonge, M.; et al. Pathways of corneal and ocular surface inflammation: A perspective from the cullen symposium. Ocul. Surf. 2005, 3, S131. [Google Scholar] [CrossRef]
- Barabino, S.; Chen, Y.; Chauhan, S.; Dana, R. Ocular surface immunity: Homeostatic mechanisms and their disruption in dry eye disease. Prog. Retin. Eye Res. 2012, 31, 271–285. [Google Scholar] [CrossRef]
- Baudouin, C.; Aragona, P.; Messmer, E.M.; Tomlinson, A.; Calonge, M.; Boboridis, K.G.; Akova, Y.A.; Geerling, G.; Labetoulle, M.; Rolando, M. Role of Hyperosmolarity in the Pathogenesis and Management of Dry Eye Disease: Proceedings of the OCEAN Group Meeting. Ocul. Surf. 2013, 11, 246–258. [Google Scholar] [CrossRef]
- Labbé, A.; Liang, Q.; Wang, Z.; Zhang, Y.; Xu, L.; Baudouin, C.; Sun, X. Corneal Nerve Structure and Function in Patients With Non-Sjögren Dry Eye: Clinical Correlations. Investig. Opthalmol. Vis. Sci. 2013, 54, 5144–5150. [Google Scholar] [CrossRef]
- Hamrah, Y.Q.P.; Qazi, Y.; Shahatit, B.; Dastjerdi, M.H.; Pavan-Langston, D.; Jacobs, D.S.; Rosenthal, P. Corneal Nerve and Epithelial Cell Alterations in Corneal Allodynia: An In Vivo Confocal Microscopy Case Series. Ocul. Surf. 2017, 15, 139–151. [Google Scholar] [CrossRef]
- Tepelus, T.C.; Chiu, G.B.; Huang, J.; Huang, P.; Sadda, S.R.; Irvine, J.; Lee, O.L. Correlation between corneal innervation and inflammation evaluated with confocal microscopy and symptomatology in patients with dry eye syndromes: A preliminary study. Graefe Arch. Clin. Exp. Ophthalmol. 2017, 255, 1771–1778. [Google Scholar] [CrossRef]
- Wei, Y.; Gadaria-Rathod, N.; Epstein, S.; Asbell, P.A. Tear Cytokine Profile as a Noninvasive Biomarker of Inflammation for Ocular Surface Diseases: Standard Operating Procedures. Investig. Opthalmol. Vis. Sci. 2013, 54, 8327–8336. [Google Scholar] [CrossRef]
- Brignole-Baudouin, F.; Riancho, L.; Ismail, D.; Deniaud, M.; Amrane, M.; Baudouin, C. Correlation Between the Inflammatory Marker HLA-DR and Signs and Symptoms in Moderate to Severe Dry Eye Disease. Investig. Opthalmol. Vis. Sci. 2017, 58, 2438–2448. [Google Scholar] [CrossRef] [PubMed]
- Galor, A.; Moein, H.-R.; Lee, C.; Rodriguez, A.; Felix, E.R.; Sarantopoulos, K.D.; Levitt, R.C. Neuropathic pain and dry eye. Ocul. Surf. 2018, 16, 31–44. [Google Scholar] [CrossRef]
- Galor, A.; Levitt, R.C.; Felix, E.R.; Martin, E.R.; Sarantopoulos, C. Neuropathic ocular pain: An important yet underevaluated feature of dry eye. Eye 2015, 29, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Launay, P.-S.; Reboussin, E.; Liang, H.; Kessal, K.; Godefroy, D.; Rostene, W.; Sahel, J.-A.; Baudouin, C.; Parsadaniantz, S.M.; Goazigo, A.R.-L. Ocular inflammation induces trigeminal pain, peripheral and central neuroinflammatory mechanisms. Neurobiol. Dis. 2016, 88, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Indart, S.; Hugon, J.; Guillausseau, P.J.; Gilbert, A.; Dumurgier, J.; Paquet, C.; Sene, D. Impact of pain on cognitive functions in primary Sjogren syndrome with small fiber neuropathy: 10 cases and a literature review. Medicine 2017, 96, e6384. [Google Scholar] [CrossRef]
- Nepp, J.; Wirth, M. Fluctuations of Corneal Sensitivity in Dry Eye Syndromes—A Longitudinal Pilot Study. Cornea 2015, 34, 1221–1226. [Google Scholar] [CrossRef]
- Mackie, I. Neuroparalytic Keratitis. In Current Ocular Therapy; W.B. Saunders: Philadelphia, PA, USA, 1995; pp. 452–454. [Google Scholar]
- Dua, H.S.; Said, D.G.; Messmer, E.M.; Rolando, M.; Del Castillo, J.M.B.; Hossain, P.; Shortt, A.J.; Geerling, G.; Nubile, M.; Figueiredo, F.C.; et al. Neurotrophic keratopathy. Prog. Retin. Eye Res. 2018, 66, 107–131. [Google Scholar] [CrossRef]
- EMA. EU Regulatory Workshop—Ophthalmology—Summary and Report. Clinical Development, Scientific Advice and Paediatric Investigation Plans; EMA/450332/2012; EMA: London, UK, 2012. [Google Scholar]
- PMDA. Diquas Eye Drops; PMDA: Tokyo, Japan, 2010.
- PMDA. Mucosta Eye Drops; PMDA: Tokyo, Japan, 2011.
- CDER. Summary Review Xiidra Eye Drops (Lifitegrast Ophthalmic Solution) 5%; Center for Drug Evaluation and Research: Silver Spring, MD, USA, 2016.
- Kondo, T. Rational Medicine Initiative; PMDA: Tokyo, Japan, 2017. [Google Scholar]
- WHO. The Pursuit of Responsible Use of Medicines: Sharing and Learning from Country Experiences; WHO/EMP/March/2012.3; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Uchino, M.; Dogru, M.; Yagi, Y.; Goto, E.; Tomita, M.; Kon, T.; Saiki, M.; Matsumoto, Y.; Uchino, Y.; Yokoi, N.; et al. The Features of Dry Eye Disease in a Japanese Elderly Population. Optom. Vis. Sci. 2006, 83, 797–802. [Google Scholar] [CrossRef]
- Kawashima, M.; Sano, K.; Takechi, S.; Tsubota, K. Impact of lifestyle intervention on dry eye disease in office workers: A randomized controlled trial. J. Occup. Health 2018, 60, 281–288. [Google Scholar] [CrossRef]
- Kawashima, M.; Uchino, M.; Yokoi, N.; Uchino, Y.; Dogru, M.; Komuro, A.; Sonomura, Y.; Kato, H.; Nishiwaki, Y.; Kinoshita, S.; et al. The Association between Dry Eye Disease and Physical Activity as well as Sedentary Behavior: Results from the Osaka Study. J. Ophthalmol. 2014, 2014, 1–6. [Google Scholar] [CrossRef]
- Kawashima, M.; Yamatsuji, M.; Yokoi, N.; Fukui, M.; Ichihashi, Y.; Kato, H.; Nishida, M.; Uchino, M.; Kinoshita, S.; Tsubota, K. Screening of dry eye disease in visual display terminal workers during occupational health examinations: The Moriguchi study. J. Occup. Health 2015, 57, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Ayaki, M.; Kawashima, M.; Negishi, K.; Tsubota, K. High prevalence of sleep and mood disorders in dry eye patients: Survey of 1000 eye clinic visitors. Neuropsychiatr. Dis. Treat. 2015, 11, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Coursey, T.G.; Henriksson, J.T.; Marcano, D.C.; Shin, C.S.; Isenhart, L.C.; Ahmed, F.; De Paiva, C.S.; Pflugfelder, S.C.; Acharya, G. Dexamethasone nanowafer as an effective therapy for dry eye disease. J. Control. Release 2015, 213, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Bucolo, C.; Fidilio, A.; Fresta, C.G.; Lazzara, F.; Platania, C.B.M.; Cantarella, G.; Di Benedetto, G.; Burgaletto, C.; Bernardini, R.; Piazza, C.; et al. Ocular Pharmacological Profile of Hydrocortisone in Dry Eye Disease. Front. Pharmacol. 2019, 10, 1240. [Google Scholar] [CrossRef]
- Holland, E.J.; Luchs, J.; Karpecki, P.M.; Nichols, K.K.; Jackson, M.A.; Sall, K.; Tauber, J.; Roy, M.; Raychaudhuri, A.; Shojaei, A. Lifitegrast for the Treatment of Dry Eye Disease: Results of a Phase III, Randomized, Double-Masked, Placebo-Controlled Trial (OPUS-3). Ophthalmology 2017, 124, 53–60. [Google Scholar] [CrossRef]

| Group | Instability of Tear Film | Global Consensus |
|---|---|---|
| NEI/Industry Workshop on Clinical Trials in Dry Eye (1995) [10] | − | − |
| Pflugfelder (2003) [11] | P | − |
| TFOS DEWS I (2007) [12] | S | + |
| TFOS DEWS II (2017) [8] | S | + |
| AAO PPP (2018) [9] | P | − |
| Japanese Dry Eye Research Group (2006) [13] | − | − |
| Korean Corneal Disease Study Group [14] | P | − |
| Chinese Medical Association Ophthalmic Branch Corneal Group [15,16] | P | − |
| Asia Dry Eye Society 2017 [7] | P | + |
| Japanese Dry Eye Research Group (2016) [17] | P | − |
| New Global Consensus Definition | P | − |
| Questionnaire | Initial Launch | No. of Questions | Screening Criteria for DED | Recommendations for Use |
|---|---|---|---|---|
| McMonnies [40] | 1987 | 12 | >14.5 | |
| Ocular Surface Disease Index (OSDI) [41] | 2000 | 12 | Mild: 13–22 Moderate: 23–32 Severe: ≥33 | Suitable for research and ATD |
| National Eye Institute’s Visual Function Questionnaire (NEI-VFQ 25) [42] | 2001 | 25 | Adapted for moderate-to-severe dry eye Strongly correlated with OSDI | |
| Dry Eye Questionnaire (DEQ) [43] | 2002 | 23 | ||
| Ocular Comfort Index (OCI) [44] | 2007 | 12 | The only questionnaire that provides valid measurement based on Rasch analysis | |
| Symptom Assessment in Dry Eye (SANDE) [45] | 2009 | 2 | Suitable for clinical assessment | |
| Standard Patient Evaluation of Eye Dryness (SPEED) [46] | 2009 | 12 | No symptoms: 0 Mild-to-moderate: 1–9 Severe: ≥10 | Suitable for Meibomian gland dysfunction |
| Impact of Dry Eye on Everyday Living (IDEEL) [47] | 2011 | 57 | Mild 40–50 Moderate 51–63 Severe > 64 | |
| Dry Eye-related Quality-of-life Score (DEQS) [48] | 2013 | 15 | ||
| 5-Item Dry Eye Questionnaire (DEQ-5) [49] | 2013 | 5 | keratoconjunctivitis sicca: > 6 Suspected Sjögren’s syndrome: >12 | |
| Chinese Dry Eye Questionnaire [50] | 2015 | 12 | Questionnaire validated in the Chinese population |
| Location | Tear Breakup Time (Seconds) | Schirmer I Test (mm) |
|---|---|---|
| USA | Not determined | Not determined |
| Germany | Abnormal < 10 Severe abnormal < 5 | Abnormal < 10 Severe abnormal < 5 |
| France | ≤5 | ≤5 |
| Italy | ≤7 | ≤5 |
| Japan | ≤5 | ≤5 |
| China | 5 < Dry eye disease suspected ≤ 10 Dry eye ≤ 5 | 5 < Dry eye disease suspected ≤ 10 Dry eye ≤ 5 |
| South Korea | 5 < Dry eye suspected ≤ 10 Dry eye ≤ 5 | 5 < Dry eye suspected ≤ 10 Dry eye ≤ 5 |
| Location | Sign | Symptom | Duration of Clinical Study (Months) |
|---|---|---|---|
| US | Primary Any * | Primary Any * | 3 |
| EU | Primary Any * | Primary Any * | 6 |
| Japan | Primary Staining | Secondary Any * | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsubota, K.; Pflugfelder, S.C.; Liu, Z.; Baudouin, C.; Kim, H.M.; Messmer, E.M.; Kruse, F.; Liang, L.; Carreno-Galeano, J.T.; Rolando, M.; et al. Defining Dry Eye from a Clinical Perspective. Int. J. Mol. Sci. 2020, 21, 9271. https://doi.org/10.3390/ijms21239271
Tsubota K, Pflugfelder SC, Liu Z, Baudouin C, Kim HM, Messmer EM, Kruse F, Liang L, Carreno-Galeano JT, Rolando M, et al. Defining Dry Eye from a Clinical Perspective. International Journal of Molecular Sciences. 2020; 21(23):9271. https://doi.org/10.3390/ijms21239271
Chicago/Turabian StyleTsubota, Kazuo, Stephen C. Pflugfelder, Zuguo Liu, Christophe Baudouin, Hyo Myung Kim, Elisabeth M. Messmer, Friedrich Kruse, Lingyi Liang, Jimena Tatiana Carreno-Galeano, Maurizio Rolando, and et al. 2020. "Defining Dry Eye from a Clinical Perspective" International Journal of Molecular Sciences 21, no. 23: 9271. https://doi.org/10.3390/ijms21239271
APA StyleTsubota, K., Pflugfelder, S. C., Liu, Z., Baudouin, C., Kim, H. M., Messmer, E. M., Kruse, F., Liang, L., Carreno-Galeano, J. T., Rolando, M., Yokoi, N., Kinoshita, S., & Dana, R. (2020). Defining Dry Eye from a Clinical Perspective. International Journal of Molecular Sciences, 21(23), 9271. https://doi.org/10.3390/ijms21239271







