Effects of Liposome and Cardiolipin on Folding and Function of Mitochondrial Erv1
Abstract
:1. Introduction
2. Results
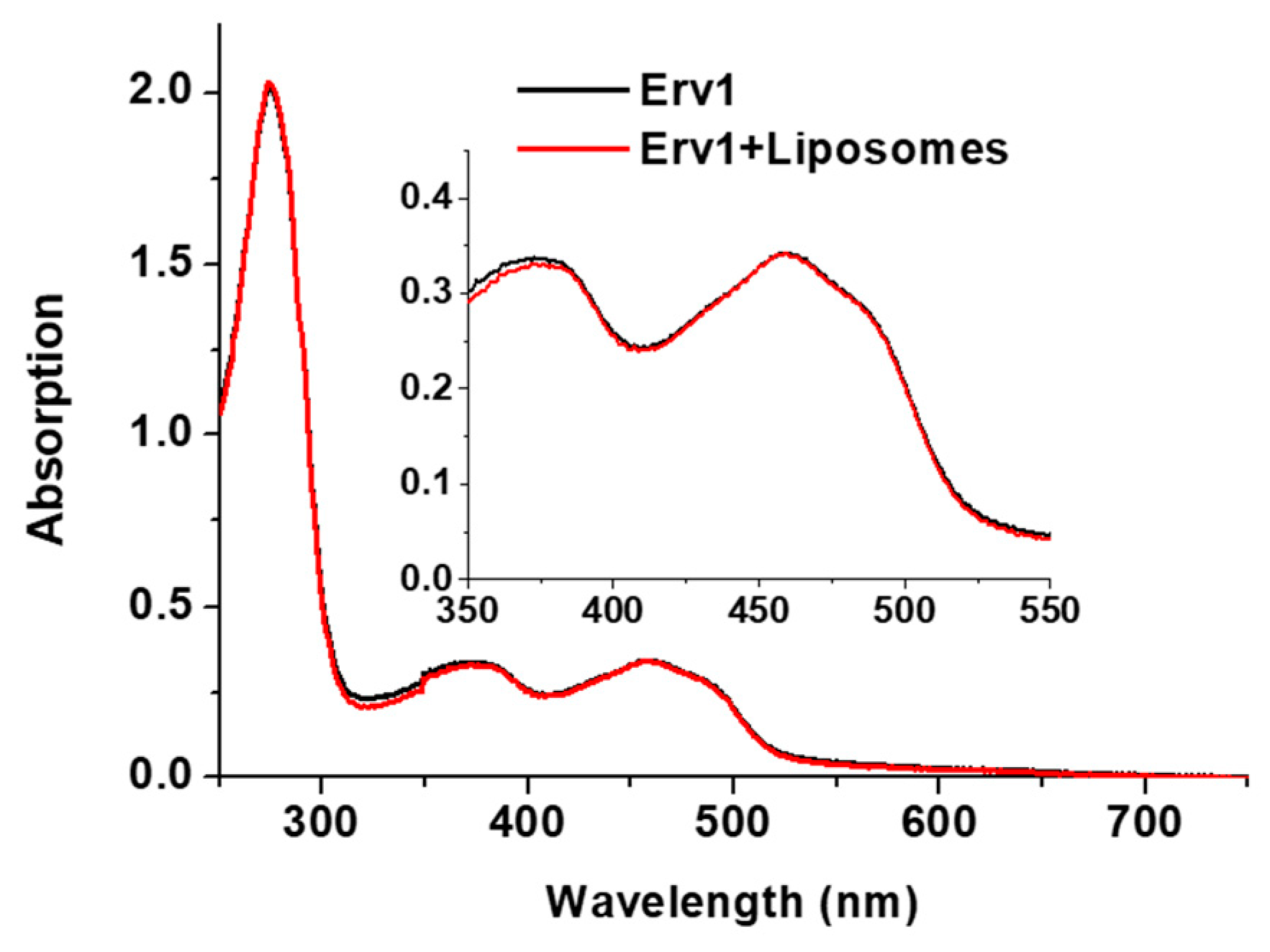
2.1. Preparation and Characterization of Liposomes
2.2. Effects of Liposomes on the Oxidase Activity of Erv1
2.3. Effects of Liposomes on Cytochrome c Reductase Activity of Erv1
3. Discussion
4. Materials and Methods
4.1. Protein Expression and Purification
4.2. Liposome Preparation
4.3. UV-Visible Spectroscopy
4.4. Oxygen Consumption Assays
4.5. Cytochrome c Reduction Assay
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ALR | Augmenter of liver regeneration |
| DTT | Dithiothreitol |
| DOPC | 1,2-dioleoyl-sn-glycero-3-phosphocholine |
| DOPCE | 1,2-dioleoyl-sn-glycero-3-phosphatidylethanolamine |
| Erv | Essential for respiration and viability |
| FAD | Flavin adenine dinucleotide |
| IMS | Intermembrane space |
| IM | Inner membrane |
| MIA | Mitochondrial import and assembly |
| OM | Outer membrane |
References
- Neupert, W. Protein import into mitochondria. Annu. Rev. Biochem. 1997, 66, 863–917. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, J.M.; Hell, K. Chopped, trapped or tacked–protein translocation into the IMS of mitochondria. Trends Biochem. Sci. 2005, 30, 205–212. [Google Scholar] [CrossRef]
- Stojanovski, D.; Bragoszewski, P.; Chacinska, A. The MIA pathway: A tight bond between protein transport and oxidative folding in mitochondria. Biochim. Biophys. Acta 2012, 1823, 1142–1150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hell, K. The Erv1-Mia40 disulfide relay system in the intermembrane space of mitochondria. Biochim. Biophys. Acta 2008, 1783, 601–609. [Google Scholar] [CrossRef] [Green Version]
- MacPherson, L.; Tokatlidis, K. Protein trafficking in the mitochondrial intermembrane space: Mechanisms and links to human disease. Biochem. J. 2017, 474, 2533–2545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, H.; Allen, S.; Wardleworth, L.; Savory, P.; Tokatlidis, K. Functional TIM10 chaperone assembly is redox-regulated in vivo. J. Biol. Chem. 2004, 279, 18952–18958. [Google Scholar] [CrossRef] [Green Version]
- Mesecke, N.; Terziyska, N.; Kozany, C.; Baumann, F.; Neupert, W.; Hell, K.; Herrmann, J.M. A disulfide relay system in the intermembrane space of mitochondria that mediates protein import. Cell 2005, 121, 1059–1069. [Google Scholar] [CrossRef] [Green Version]
- Ceh-Pavia, E.; Tang, X.; Liu, Y.; Heyes, D.J.; Zhao, B.; Xiao, P.; Lu, H. Redox characterisation of Erv1, a key component for protein import and folding in yeast mitochondria. FEBS J. 2020, 287, 2281–2291. [Google Scholar] [CrossRef]
- Allen, S.; Balabanidou, V.; Sideris, D.P.; Lisowsky, T.; Tokatlidis, K. Erv1 mediates the Mia40-dependent protein import pathway and provides a functional link to the respiratory chain by shuttling electrons to cytochrome c. J. Mol. Biol. 2005, 353, 937–944. [Google Scholar] [CrossRef]
- Herrmann, J.M.; Köhl, R. Catch me if you can! Oxidative protein trapping in the intermembrane space of mitochondria. J. Cell Biol. 2007, 176, 559–563. [Google Scholar] [CrossRef] [Green Version]
- Dabir, D.V.; Leverich, E.P.; Kim, S.K.; Tsai, F.D.; Hirasawa, M.; Knaff, D.B.; Koehler, C.M. A role for cytochrome c and cytochrome c peroxidase in electron shuttling from Erv1. EMBO J. 2007, 26, 4801–4811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hagiya, M.; Francavilla, A.; Polimeno, L.; Ihara, I.; Sakai, H.; Seki, T.; Shimonishi, M.; Porter, K.A.; Starzl, T.E. Cloning and sequence analysis of the rat augmenter of liver regeneration (ALR) gene: Expression of biologically active recombinant ALR and demonstration of tissue distribution. Proc. Natl. Acad. Sci. USA 1994, 91, 8142–8146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levitan, A.; Danon, A.; Lisowsky, T. Unique features of plant mitochondrial sulfhydryl oxidase. J. Biol. Chem. 2004, 279, 20002–20008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eckers, E.; Petrungaro, C.; Gross, D.; Riemer, J.; Hell, K.; Deponte, M. Divergent Molecular Evolution of the Mitochondrial Sulfhydryl: Cytochrome c Oxidoreductase Erv in Opisthokonts and Parasitic Protists. J. Biol. Chem. 2013, 288, 2676–2688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gross, E.; Sevier, C.S.; Vala, A.; Kaiser, C.A.; Fass, D. A new FAD-binding fold and intersubunit disulfide shuttle in the thiol oxidase Erv2p. Nat. Struct. Biol. 2002, 9, 61–67. [Google Scholar] [CrossRef]
- Guo, P.C.; Ma, J.D.; Jiang, Y.L.; Wang, S.J.; Bao, Z.Z.; Yu, X.J.; Chen, Y.X.; Zhou, C.Z. Structure of Yeast Sulfhydryl Oxidase Erv1 Reveals Electron Transfer of the Disulfide Relay System in the Mitochondrial Intermembrane Space. J. Biol. Chem. 2012, 287, 34961–34969. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.Z.; Winther, J.R.; Thorpe, C. Erv2p: Characterization of the redox behavior of a yeast sulfhydryl oxidase. Biochemistry 2007, 46, 3246–3254. [Google Scholar] [CrossRef] [Green Version]
- Ang, S.K.; Lu, H. Deciphering Structural and Functional Roles of Individual Disulfide Bonds of the Mitochondrial Sulfhydryl Oxidase Erv1p. J. Biol. Chem. 2009, 284, 28754–28761. [Google Scholar] [CrossRef] [Green Version]
- Hofhaus, G.; Lee, J.E.; Tews, I.; Rosenberg, B.; Lisowsky, T. The N-terminal cysteine pair of yeast sulfhydryl oxidase Erv1p is essential for in vivo activity and interacts with the primary redox centre. Eur. J. Biochem. 2003, 270, 1528–1535. [Google Scholar] [CrossRef]
- Ceh-Pavia, E.; Ang, S.K.; Spiller, M.P.; Lu, H. The disease-associated mutation of the mitochondrial thiol oxidase Erv1 impairs cofactor binding during its catalytic reaction. Biochem. J. 2014, 464, 449–459. [Google Scholar] [CrossRef] [Green Version]
- Farrell, S.R.; Thorpe, C. Augmenter of Liver Regeneration: A Flavin-Dependent Sulfhydryl Oxidase with Cytochrome c Reductase Activity. Biochemistry 2005, 44, 1532–1541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoober, K.L.; Thorpe, C. Egg white sulfhydryl oxidase: Kinetic mechanism of the catalysis of disulfide bond formation. Biochemistry 1999, 38, 3211–3217. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Ang, S.K.; Ceh-Pavia, E.; Heyes, D.J.; Lu, H. Kinetic characterisation of Erv1, a key component for protein import and folding in yeast mitochondria. FEBS J. 2020, 287, 1220–1231. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.L. Phosphoglycerides of mitochondrial membranes. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1987; Volume 148, pp. 475–485. [Google Scholar]
- Scorrano, L.; Ashiya, M.; Buttle, K.; Weiler, S.; Oakes, S.A.; Mannella, C.A.; Korsmeyer, S.J. A Distinct Pathway Remodels Mitochondrial Cristae and Mobilizes Cytochrome c during Apoptosis. Dev. Cell 2002, 2, 55–67. [Google Scholar] [CrossRef] [Green Version]
- Bernardi, P.; Azzone, G.F. Cytochrome c as an electron shuttle between the outer and inner mitochondrial membranes. J. Biol. Chem. 1981, 256, 7187–7192. [Google Scholar]
- Suga, K.; Hamasaki, A.; Chinzaka, J.; Umakoshi, H. Liposomes modified with cardiolipin can act as a platform to regulate the potential flux of NADP+-dependent isocitrate dehydrogenase. Metab. Eng. Commun. 2016, 3, 8–14. [Google Scholar] [CrossRef]
- Rytömaa, M.; Kinnunen, P.K. Evidence for two distinct acidic phospholipid-binding sites in cytochrome c. J. Biol. Chem. 1994, 269, 1770–1774. [Google Scholar]
- Ascenzi, P.; Coletta, M.; Wilson, M.T.; Fiorucci, L.; Marino, M.; Polticelli, F.; Sinibaldi, F.; Santucci, R. Cardiolipin–cytochrome c complex: Switching cytochrome c from an electron-transfer shuttle to a myoglobin- and a peroxidase-like heme-protein. IUBMB Life 2015, 67, 98–109. [Google Scholar] [CrossRef]
- Mandal, A.; Hoop, C.L.; DeLucia, M.; Kodali, R.; Kagan, V.E.; Ahn, J.; van der Wel, P.C.A. Structural Changes and Proapoptotic Peroxidase Activity of Cardiolipin-Bound Mitochondrial Cytochrome c. Biophys. J. 2015, 109, 1873–1884. [Google Scholar] [CrossRef] [Green Version]
- Stojanovski, D.; Milenkovic, D.; Müller, J.M.; Gabriel, K.; Schulze-Specking, A.; Baker, M.J.; Ryan, M.T.; Guiard, B.; Pfanner, N.; Chacinska, A. Mitochondrial protein import: Precursor oxidation in a ternary complex with disulfide carrier and sulfhydryl oxidase. J. Cell Biol. 2008, 183, 195–202. [Google Scholar] [CrossRef] [Green Version]
- Sztolsztener, M.E.; Brewinska, A.; Guiard, B.; Chacinska, A. Disulfide Bond Formation: Sulfhydryl Oxidase ALR Controls Mitochondrial Biogenesis of Human MIA40. Traffic 2013, 14, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Daum, G.; Vance, J.E. Import of lipids into mitochondria. Prog. Lipid Res. 1997, 36, 103–130. [Google Scholar] [CrossRef]
- Wagner, S.; Paltauf, F. Generation of glycerophospholipid molecular species in the yeast Saccharomyces cerevisiae. Fatty acid pattern of phospholipid classes and selective acyl turnover at sn-1 and sn-2 positions. Yeast 1994, 10, 1429–1437. [Google Scholar] [CrossRef] [PubMed]
- Elsana, H.; Olusanya, T.O.B.; Carr-wilkinson, J.; Darby, S.; Faheem, A.; Elkordy, A.A. Evaluation of novel cationic gene based liposomes with cyclodextrin prepared by thin film hydration and microfluidic systems. Sci. Rep. 2019, 9, 15120. [Google Scholar] [CrossRef] [Green Version]
- Ang, S.K.; Zhang, M.; Lodi, T.; Lu, H. Mitochondrial thiol oxidase Erv1: Both shuttle cysteine residues are required for its function with distinct roles. Biochem. J. 2014, 460, 199–210. [Google Scholar] [CrossRef] [Green Version]
- Pinheiro, T.J.T.; Cheng, H.; Seeholzer, S.H.; Roder, H. Direct evidence for the cooperative unfolding of cytochrome c in lipid membranes from H-2H exchange kinetics. J. Mol. Biol. 2000, 303, 617–626. [Google Scholar] [CrossRef] [Green Version]
- Butterfield, D.A.; Sultana, R. Redox proteomics: Understanding oxidative stress in the progression of age-related neurodegenerative disorders. Expert Rev. Proteom. 2008, 5, 157–160. [Google Scholar] [CrossRef] [Green Version]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef]
- Vogtle, F.N.; Burkhart, J.M.; Rao, S.; Gerbeth, C.; Hinrichs, J.; Martinou, J.C.; Chacinska, A.; Sickmann, A.; Zahedi, R.P.; Meisinger, C. Intermembrane space proteome of yeast mitochondria. Mol. Cell. Proteom. 2012, 11, 1840–1852. [Google Scholar] [CrossRef] [Green Version]
- Di Fonzo, A.; Ronchi, D.; Lodi, T.; Fassone, E.; Tigano, M.; Lamperti, C.; Corti, S.; Bordoni, A.; Fortunato, F.; Nizzardo, M.; et al. The mitochondrial disulfide relay system protein GFER is mutated in autosomal-recessive myopathy with cataract and combined respiratory-chain deficiency. Am. J. Hum. Genet. 2009, 84, 594–604. [Google Scholar] [CrossRef] [Green Version]
- Daithankar, V.N.; Schaefer, S.A.; Dong, M.; Bahnson, B.J.; Thorpe, C. Structure of the human sulfhydryl oxidase augmenter of liver regeneration and characterization of a human mutation causing an autosomal recessive myopathy. Biochemistry 2010, 49, 6737–6745. [Google Scholar] [CrossRef] [PubMed] [Green Version]


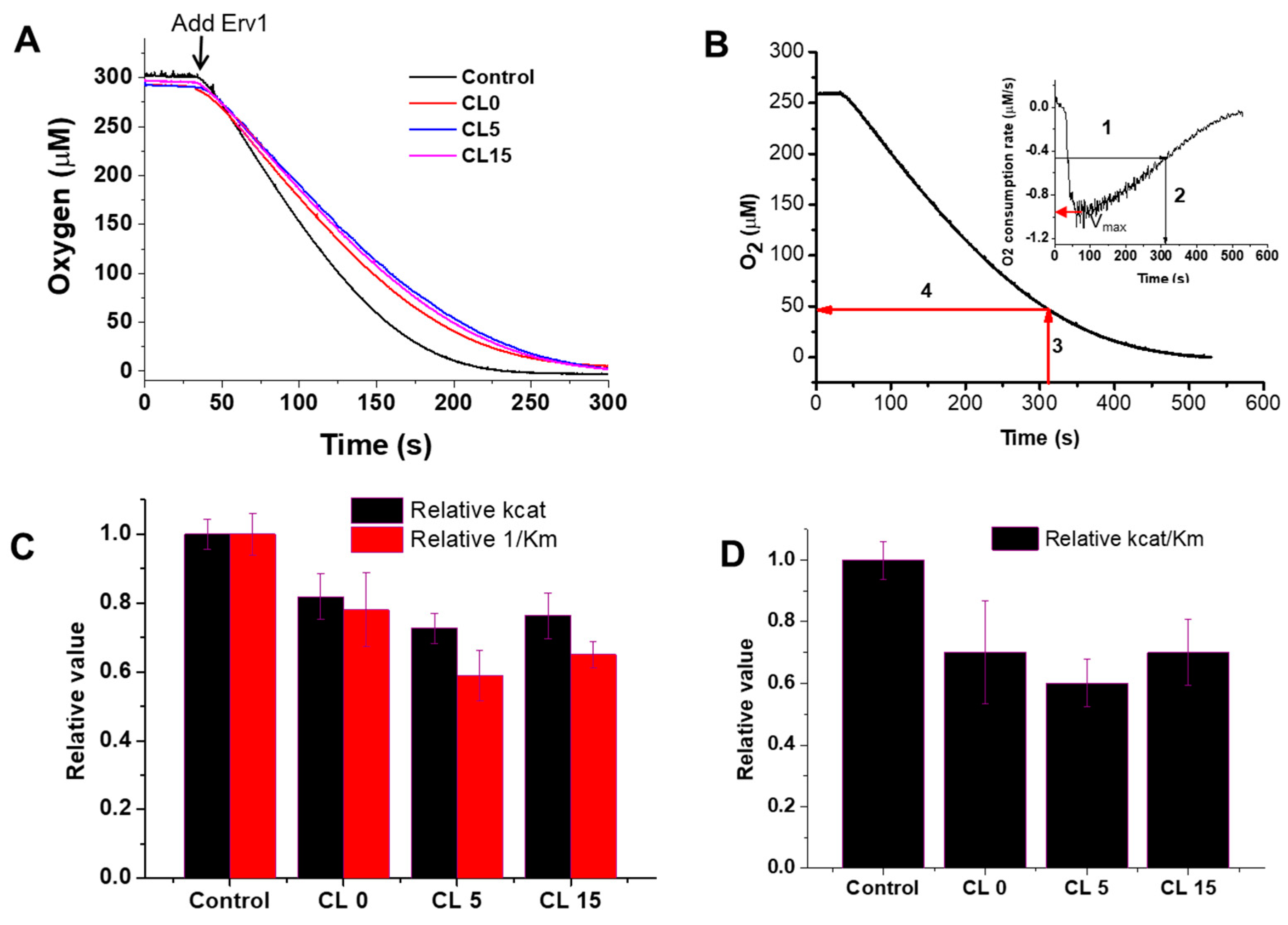

| Substrate | Liposomes | kcat (s−1) | Km (µM) | kcat/Km (M−1 s−1) |
|---|---|---|---|---|
| Oxygen | Control (No liposomes) | 1.0 ± 0.1 | 50 ± 10 | 2.0 × 104 (100%) |
| CL0 | 0.82 ± 0.02 | 60.0 ± 6 | 1.4 × 104 (70%) | |
| CL5 | 0.74 ± 0.01 | 58.8 ± 6 | 1.2 × 104 (60%) | |
| CL15 | 0.76 ± 0.02 | 53.2 ± 5 | 1.4 × 104 (70%) | |
| Cytochrome c | Control (No liposomes) | 0.82 ± 0.07 | 23 ± 4 | 3.6 × 104 (100%) |
| CL0 | 0.75 ± 0.03 | 52 ± 4 | 1.4 × 104 (39%) | |
| CL5 | 0.61 ± 0.04 | 74 ± 3 | 8.2 × 103 (23%) | |
| CL15 | 0.48 ± 0.02 | 105 ± 7 | 4.6 × 103 (13%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, X.; Harris, L.K.; Lu, H. Effects of Liposome and Cardiolipin on Folding and Function of Mitochondrial Erv1. Int. J. Mol. Sci. 2020, 21, 9402. https://doi.org/10.3390/ijms21249402
Tang X, Harris LK, Lu H. Effects of Liposome and Cardiolipin on Folding and Function of Mitochondrial Erv1. International Journal of Molecular Sciences. 2020; 21(24):9402. https://doi.org/10.3390/ijms21249402
Chicago/Turabian StyleTang, Xiaofan, Lynda K Harris, and Hui Lu. 2020. "Effects of Liposome and Cardiolipin on Folding and Function of Mitochondrial Erv1" International Journal of Molecular Sciences 21, no. 24: 9402. https://doi.org/10.3390/ijms21249402






